Advances in Microbiology
Vol.4 No.5(2014), Article ID:45251,9 pages DOI:10.4236/aim.2014.45034
Cellulase Production by Endophytic Strains of Trichoderma reesei from Baccharis dracunculifolia D. C. (Asteraceae)
Sideney Becker Onofre1, Taismara Bonfante2, Zípora Morgana Quinteiro dos Santos2, Marielly Coradin de Moura2, Aline Filakowski Cardoso2
1Laboratório de Microbiologia, Departamento de Ciências Biológicas da União de Ensino do Sudoeste do Paraná, UNISEP, Campus de Francisco Beltrão, Francisco Beltrão, Paraná, Brasil
2Departamento de Ciências Biológicas e da Saúde, Universidade Paranaense, UNIPAR, Unidade Universitária de Francisco Beltrão, Francisco Beltrão, Paraná, Brasil
Email: becker@unisep.edu.br
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/


Received 1 March 2014; revised 3 April 2014; accepted 13 April 2014
ABSTRACT
Cellulases are enzymes responsible for the degradation of cellulose, the major compound in plant cells. Cellulose is a polysaccharide composed of several glucose units linked together by chemical bonds. Cellulases, such as endoglucanases, beta-glucosidase and exoglucanases, break the chemical bonds between the glucose units. Fungi, including the endophytic species, can be great cellulase producers. This study aimed to evaluate cellulase production by four endophytic strains of Trichoderma reesei in semi-solid media containing sugarcane bagasse, supplemented or not with salts. Two fermentations were carried out for 43 days. Samples were taken every seven days to obtain production peaks. The enzymes were characterized by their optimum pH and temperature of activity and stability upon incubation in the presence of ions, pH and temperature variations. The results showed that the endophytic strains FB1, FB2, FB3 and FB4 of Trichoderma reesei produce cellulases in a sugarcane bagasse medium, supplemented or not with salts, at pH 5.5 and 30˚C. The supplemented medium proved to be more appropriate to induce cellulase production after 29 days of fermentation, with FB4 having the best yield: 16.32 ± 2.65 IU/gram of fermented substrate.
Keywords:Substrate, Fermentation Processes, Enzymes, Cellulases

1. Introduction
A constant increase in agricultural activities has led to a growing need to find an application for agro-industrial residues, as they accumulate in the environment, changing their characteristics and losing their resources. Numerous technologies have been created for utilizing these materials, which became high value added products. Sugarcane bagasse, generated by sugar refineries and ethanol plants, is one of them [1] [2] .
The study of industrial fermentation media for obtaining biotechnological products has received great attention in recent years. Several byproducts and raw materials from the food industry and agro-industry have been employed to obtain biotechnological products, such as enzymes, due to high availability and low commercial value [3] [4] .
Enzyme production is often limited by the cost of the substrates used for microorganism cultivation. It is estimated that approximately 30% - 40% of the cost involved in the production of enzymes is due to the culture medium, making its optimization important to reduce costs [5] .
Lignocellulosic residues are considered a great alternative source of energy due to their high availability in nature. They are frequently used in technological means of feeding, as protein for animal feed and in the paper industry [6] .
Biodegradation of lignocellulosic materials can be employed in technological processes, besides corresponding to an important stage of the carbon cycle in nature. Enzymes produced by fungal decomposers of lignocellulosic materials have also been in demand: xylanases and laccases for pulp bleaching and cellulases for the enzymatic hydrolysis of cellulose in the production of second-generation ethanol [7] .
Enzymatic hydrolysis of cellulose includes the synergistic activity of a cellulolytic complex, usually from fungi, consisting of endoglucanases, exoglucanases and β-D-glucosidase. However, such a technique has low productivity and high cost [8] .
Cellulases form a complex able of acting on cellulosic materials, causing their hydrolysis. Cellulases are highly specific biocatalysts, which operate in synergy to release sugars. Glucose is the sugar with the highest industrial importance, as it can be converted into ethanol [6] [9] [10] .
A wide variety of microorganisms have been used in fermentation processes with sugarcane bagasse as substrate, in order to obtain cellulases. The filamentous fungi are the most used, especially Trichoderma reesei, which is known for producing various extracellular enzyme systems involved in the hydrolysis of polysaccharides [11] -[14] .
Trichoderma reesei is an endophytic filamentous fungus, which is distinguished by its ability to generate the cellulolytic enzyme complex [15] . It is found in various environments, such as soil and saprophytic, epiphytic and endophytic plants [16] .
The search for new fungal species with potential to produce the cellulolytic complex justifies this work, which aims to evaluate cellulase production in four endophytic strains of Trichoderma reesei in a semi-solid medium containing sugarcane bagasse, supplemented or not with salts.
2. Materials and Methods
2.1. Microorganisms
Strains FB1, FB2, FB3 and FB4 of the endophytic fungus Trichoderma reesei, isolated from Baccharis dracunculifolia DC (Asteraceae), were used. These strains can potentially produce cellulases in a semi-solid medium containing sugarcane bagasse and supplemented with salts. They were isolated and kept in the mycology collection at União de Ensino do Sudoeste do Paraná (UNISEP), Francisco Beltrão Campus, Paraná, Brazil.
2.2. Determination of Cellulolytic Activity
Sugarcane bagasse, washed successively in running water to completely remove the sugars, was used as the basic support for determination of cellulolytic activity. The bagasse was dried in a drying oven until constant weight was obtained. It was subsequently ground in a mill, packaged and stored in a dry place for further testing.
2.3. Fermentation in Supplemented Medium
A semi-solid culture medium consisting of 10 g of dehydrated sugarcane bagasse, and supplemented with 0.5 g/L carboxymethylcellulose, 1 g/L glucose, 3 g/L NaNO3, 1 g/L KH2PO4, 0.5 g/L MgSO4, 0.5 g/L KCl, 10 mg/L FeSO4∙7H2O, 5.0 mg/L ZnSO4 and 3 g/L of urea was used to assess the cellulolytic activity. All medium components were added to 120 mL of distilled water and then autoclaved for 15 min. A total of 30 mL of a spore suspension (±108 mL) was used for inoculation. The material was incubated at 28˚C for 21 days, when the first analysis of enzyme activity occurred. Afterwards, further analyses were carried out every three days until completing 43 days of incubation.
2.4. Fermentation in Unsupplemented Medium
A semi-solid culture medium containing 10 g of dried and ground sugarcane bagasse was used. To it was added 0.5% carboxymethylcellulose and 0.1% glucose in 120 mL of distilled water. Afterwards, the medium was autoclaved for 15 min. A spore suspension (±108 mL) was used for inoculation. A total of 30 mL of this inoculum suspension was added to each vial containing the culture medium. The material remained in an incubator at 30˚C for 22 days, when the first enzyme quantification occurred. The cellulolytic activity was measured every three days until day 43 of fermentation.
2.5. Substrate Fermentation Analysis
Aliquots of 5 g of the medium of each strain were weighed and placed in Erlenmeyer flasks. A total of 50 mL of deionized sterile water was added to each flask, which were stirred for 30 min at 30˚C. The solution was filtered to remove the solids and the pH was determined, after adding 2 mL of a 7.0 phosphate buffer. The extract was centrifuged at 3000 rpm for 15 min and the supernatant was the enzyme source to determine the reducing sugars by the indirect spectrophotometric method. This method was used to determine enzyme activity based on the release of glucose molecules by the action of the cellulolytic enzyme complex [16] [17] .
2.6. Determination of Reducing Sugars
Reducing sugars were determined by reaction with 3,5-dinitrosalicylic acid (DNS) [18] . In a basic medium and at high temperature, this acid becomes 3-amino-5-nitrosalicylic acid, developing a red color and absorbing light at 540 nm. One unit of cellulase was defined as the amount of enzyme released on the substrate that liberate 1 μmol of reducing sugar (expressed as glucose) per minute, under test conditions [19] [20] .
2.7. pH Optimization
Strain enzymatic activity was evaluated under different pH values (4.0, 5.0, 5.5, 6.0 and 6.5). These tests were conducted to determine the optimum pH for subsequent assays.
2.8. Effect of Temperature
Tests were carried out at different temperatures (25˚C, 30˚C, 35˚C, 40˚C and 45˚C) and under optimal pH conditions, to verify the optimum temperature for the production of the cellulolytic complex.
2.9. Statistical Analysis
Results were submitted to analysis of variance using the statistical analysis system Assistat [21] . The comparison of means was done by the Tukey’s test at 5% probability.
3. Results and Discussion
Data on cellulase production by four endophytic strains of T. reesei are shown in Tables 1-4, and Figure 1 and Figure 2. Table 1 relates to enzymatic volumes produced by the strains in different pH values.
Among the physical parameters, the medium pH plays an important role because it induces morphological changes in the microorganism and interferes in enzyme secretion. The pH change observed during microorganism growth also affects product stability in the medium [22] [23] .
The pH of a culture varies according to reactions that occur during the metabolic activities of the microorganism. The secretion of organic acids, such as acetic or lactic, causes a decrease in pH.
The use of a nitrogen source causes pH variation; for example, with ammonium salts, the pH usually decreases throughout cell growth, due to hydrogen ion formation during the consumption of ammonia [24] .
The highest yields of cellulolytic complex were observed at pH 5.5 for the four tested strains, a decrease occurring at pH 4.0 and higher pH values (6.0 and 6.5).
Solid state fermentations (SSF) are exothermic. The large amounts of heat released are directly proportional to the metabolic activity of the microorganism. In filamentous fungi, the temperature directly influences spore germination, growth and product formation. The temperature is a critical factor in virtually all SSF, due to the accumulation of metabolic heat generated [25] .
Table 1. Cellulase production by four endophytic strains of Trichoderma reesei in different pH values.
*Means followed by lowercase letters in the columns and uppercase letters horizontally do not differ at 5% significance level by the Tukey’s test.
Table 2. Cellulase production by four endophytic strains of Trichoderma reesei in different temperatures and pH 5.5.
*Means followed by lowercase letters in the columns and uppercase letters horizontally do not differ at 5% significance level by the Tukey’s test.
Table 3. Cellulase activity (U/g substrate) by endophytic strains of Trichoderma reesei in a supplemented medium.
*Means followed by lowercase letters in the columns and uppercase letters horizontally do not differ at 5% significance level by the Tukey’s test.
Table 4. Cellulase activity (U/g substrate) by endophytic strains of Trichoderma reesei in unsupplemented media.
*Means followed by lowercase letters in the columns and uppercase letters horizontally do not differ at 5% significance level by the Tukey’s test.
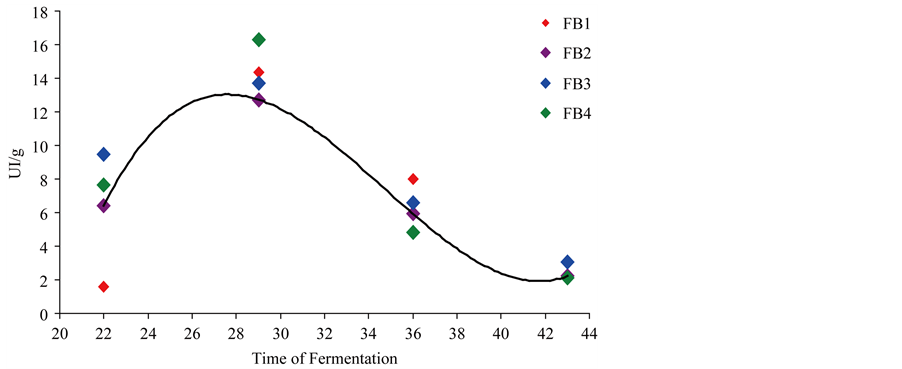
Figure 1. Cellulase production by four endophytic strains of Trichoderma reesei in supplemented media.
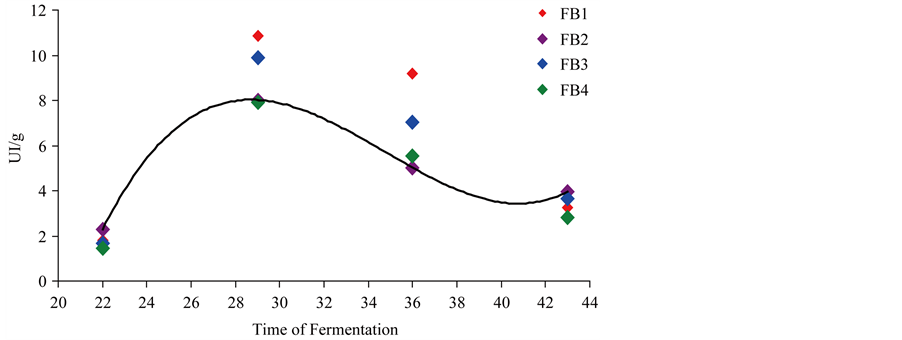
Figure 2. Cellulase production by four endophytic strains of Trichoderma reesei in unsupplemented media.
Sato and Sudo [26] reported that temperature affects both the growth and the production of enzymes, thus an efficient heat removal method is necessary. According to Bianchi et al. [27] , a large amount of heat may be produced during SSF due to the metabolic activity of the microorganisms, and also depending on the substrate. The heat produced must be immediately dissipated to avoid hindering the process, because temperature directly affects spore germination, microorganism growth and sporulation, and product formation.
Table 2 shows that the optimum temperature for growth and production of cellulolytic enzymes for four endophytic strains of Trichoderma reesei is 30˚C. Enzyme volumes decreased for all tested strains at temperatures below and above 30˚C. Thus, with data referring to the optimum temperature and pH, it was possible to proceed to the other steps of this work.
It is noteworthy that 30˚C is a suitable temperature for mesophilic bacteria to develop. This corroborates Buswell et al. [28] , who reported 30˚C as the optimum temperature for the production of cellulolytic enzyme complex by Trichoderma spp. This was also observed by Chen and Jan [29] , Dalsenter et al. [30] , Mitchell et al. [31] , Gomes et al. [32] , Carvalho et al. [33] and Taneda et al. [34] .
According to Camassola et al. [35] , 5.5 is the optimum pH for the production of cellulolytic enzymes by Penicillium echinulatum. Alam et al. [36] optimized the process of cellulase production by Trichoderma reesei through the bioconversion of the liquid sewage sludge, finding maximal activity at pH 5.5, thus corroborating Qin et al. [37] , Zang et al. [38] , Bendig and Weuster-Botz [39] and Puglisi et al. [40] .
Table 3 and Figure 1 show the same behavior for the four tested strains during the production of cellulolytic complex, with enzyme production occurring from 22 days of fermentation to reach maximum yields at day 29. Afterwards, a decrease in enzyme production was observed.
At day 29, enzyme production was 14.33 ± 2.56, 12.73 ± 2.34, 13.73 ± 1.86 and 16.32 ± 2.65 IU/g of substrate for FB1, FB2, FB3 and FB4, respectively, showing no significant differences at 5% by the Tukey’s test.
During the 42 days of fermentation, enzyme production in media unsupplemented with salts was lower when compared to the salt-supplemented media (Table 3).
Enzymatic production at 22 days was: 1.79 ± 0.23, 2.27 ± 0.46, 1.69 ± 0.32 and 1.43 ± 0.78 for FB1, FB2, FB3 and FB4, respectively. These enzymatic indices do not differ at 5% significance level.
These volumes are lower than those produced by the same strains at the same period of time, but in media supplemented with salts. It is noteworthy that FB2 and FB3 produced 6.42 ± 1.23 and 9.50 ± 2.20, respectively, during the same time, values higher than those observed for unsupplemented media.
Trichoderma reesei produced 18 IU/g of cellulolytic complex in supplemented medium and 6.0 IU/g in unsupplemented medium, at 30 days of fermentation, corroborating Aguiar [1] .
In 29 days, the strain FB4 produced 16.32 and 7.93 in media supplemented and unsupplemented with salts, respectively. Basso, Gallo and Basso [13] (2010), when evaluating the cellulolytic activity of T. reesei strains, isolated from sugarcane bagasse and decaying wood, observed values of 12.58 IU/g of unsupplemented substrate at 28˚C in 35 days. Once more, the data are similar to those observed in this work, but considering the behavior in unsupplemented medium, the values obtained in this study are lower, reaching 7.93 IU/g in 29 days.
The data obtained in this study corroborate Juwaied et al. [10] on culture media optimization for the production of total cellulase, using different culture media. T. reesei yielded 2.6 IU/gram of substrate at pH 5.5 after 20 days in media containing sugarcane bagasse. This volume of produced enzyme is similar to that obtained at 22 days in this work, but much lower than the volumes obtained after 29 days of fermentation (up to 10.85 IU/g in unsupplemented media and 16.32 IU/g in supplemented media), thus showing potential to produce the cellulolytic complex.
No influence on enzymes production was observed by the sugarcane bagasse and glucose contained in the media; changes are related to the added salts. These results corroborate Castro [41] , who compared the production of endoglucanase and β-glucosidase by Trichoderma reesei and Humicola grisea, when grown on insoluble (cellulignin from sugarcane bagasse and Avicel) and soluble substrates (carboxymethylcellulose and cellobiose). Slower kinetics of enzyme production were observed when the insoluble sources had been used, especially of lignocellulosic origin, due to an initial acclimation period of the cells to this raw material. Yet, when carboxymethylcellulose and cellobiose were considered, the times corresponding to the maximum enzyme production were anticipated in about 50 h. It has been concluded that there is no direct correlation with the carbon source used for induction in the synthesis of the cellulolytic complex enzymes by T. reesei and Humicola grisea var. thermoidea. Cellulases were also produced by strains grown on a substrate containing only cellobiose, and this synthesis had not a constitutive character.
The results obtained here also confirm Rodríguez-Zúñiga et al. [42] who studied Aspergillus niger and found that the largest amounts of cellulases were obtained in medium supplemented with salts, a modified Mandels and Weber medium, added of carboxymethylcellulose inductor. Under these conditions, cellulase and endoglucanase activities were 0.4 and 21.0 IU/g of substrate, respectively, values representing 2.6 and 4-fold increases in their enzyme activities compared to the unsupplemented medium. The same behavior has been observed by Buswell et al. [28] , Camassola et al. [43] , Spier [44] , Zhang et al. [45] , Chandra et al. [46] , Sanchez [47] , Ahamed and Vermette [48] and Gao et al. [49] .
4. Conclusions
The endophytic strains FB1, FB2, FB3 and FB4 of Trichoderma reesei can produce cellulases in media containing sugarcane bagasse, supplemented and unsupplemented, at pH 5.5 and 30˚C.
The medium supplemented with salts proved to be more appropriate to induce cellulase production after 29 days of fermentation, with the strain FB4 having the best yield: 16.32 ± 2.65 IU/gram of fermented substrate.
Acknowledgements
We would like to thank the União de Ensino do Sudoeste do Paraná for the financial support and donation of material, which enabled the development of this study.
Conflict of Interest
The authors declare no conflict of interest.
References
- Aguiar, J.M.M. (2008.) Análise enzimática dos fungos lignocelulolíticos cultivados em vinhaça e bagaço-de-cana-deacucar. 80f. Dissertação em Agronomia. Escola Superior de Agricultura “Luiz Queiroz”. Universidade de Sao Paulo, São Paulo.
- Menezes, C.M. and Silva, I.S. (2009) Bagaço de cana: Fonte para produção de enzimas ligninocelulolíticas. Estudos Tecnológicos, 5, 68-78. http://dx.doi.org/10.4013/ete.2009.51.05
- Silva, G.A.B., Almeida, W.E.S., Cortes, M.S. and Martins, E.S. (2009) Produção e caracterização de protease obtida por Gliocladium verticilloides através da fermentação em estado sólido de subprodutos agroindustriais. Revista Brasileira de Tecnologia Agroindustrial, 3, 28-41. http://dx.doi.org/10.3895/S1981-36862009000100003
- Ernandes, F.M.P.G., Boscolo, M. and Cruz, C.H.G. (2010) Influência da composição do meio para a produção de Zimomonas mobilis. Acta Scientiarum. Technology, 32, 21-26. http://dx.doi.org/10.4025/actascitechnol.v32i1.7454
- Joo, H.S. and Chang, C.S. (2005) Production of Protease from a New Alkalophilic Bacillus sp. I-312 Grown on Soybean Meal: Optimization and Some Properties. Process Biochemistry, 40, 1263-1270. http://dx.doi.org/10.1016/j.procbio.2004.05.010
- Santos, T.T. and Varavallo, M.A. (2011) Aplicação de microrganismos endofíticos na agricultura e na produção de substâncias de interesse econômico. Semina, 32, 199-212.
- Aguiar, A. and Ferraz, A. (2011) Mecanismos envolvidos na biodegradação de materiais lignocelulósicos e aplicações tecnológicas correlatas. Química Nova, 34, 1729-1738.
- Silva, L.A.D. (2008) Produção e caracterização de enzimas celulásicas por Aspergillus phoenicis. f 119. Dissertação em Microbiologia Agrícola e do Meio Ambiente. Instituto de Ciências Básicas e da Saúde, Universidade Federal do Rio Grande do Sul, Porto Alegre.
- Castro, A.M. and Pereira Jr., N. (2010) Produção, propriedades e aplicação de celulases na hidrólise de resíduos agroindustriais. Química Nova, 33, 181-188. http://dx.doi.org/10.1590/S0100-40422010000100031
- Juwaied, A.A., Al-Amiery, A.A.H., Abdumuniem, Z. and Anaam, U. (2011) Optimization of Cellulase Production by Aspergillus niger and Tricoderma viride Using Sugar Cane Waste. Journal of Yeast and Fungal Research, 2, 19-23.
- Ruegger, M.J.S. and Tauk-Tornisielo, S.M. (2004) Atividade da celulase de fungos isolados do solo da Estação Ecológica de Juréia-Itatins, São Paulo, Brasil. Revista Brasileira de Botanica, 27, 205-211. http://dx.doi.org/10.1590/S0100-84042004000200001
- Martinez, D. and Murray, R.W. (2008) A sequenciação do genoma e análise da biomassa degradantes fungo Trichoderma reesei (syn. Hypocrea jecorina). Nature Biotechnology, 26, 553-5560. http://dx.doi.org/10.1038/nbt1403
- Basso, T.P., Gallo, C.R. and Basso, L.C. (2010) Atividade celulolítica de fungos isolados de bagaço de cana de acucar e madeira em decomposição. Pesquisa Agropecuária Brasileira, 45, 1282-1289.
- Alnoch, R.C. (2010) Seleção qualitativa de fungos filamentosos com atividade celulolítica isolados em solos de áreas de floresta na região de Tangará da Serra—MT. Anais: Congresso de Iniciação Científica, Cáceres/MT, Brasil, 6, 20- 24.
- Cuzzi, C., Link, S., Vilani, A., Sartori, C. and Onofre, S.B. (2012) Endophytic fungi of the “vassourinha” (Baccharis dracunculifolia D. C.—Asteraceae). Revista Brasileira de Biociências, 10, 135-139.
- Santos, Z.M.Q., Bonfante, T., Silva, G.C., Fidriszewski, J., Bergamin, R. and Onofre, S.B. (2012) Comportamento de linhagens endofíticas de Trichoderma reesei em produzir celulases em meio não suplementado. Anais 11o Encontro de Iniciação Científica e 11o Fórum de Pesquisa da Unipar.
- Moura, A.C., Cardoso, A.F., Bergamim, R. and Onofre, S.B. (2012) Capacidade de linhagens endofíticas de Trichoderma reesei em produzir celulases em meio suplementado. Anais do 11o Encontro de Iniciação Científica E 11o Fórum de Pesquisa da Unipar.
- Onofre, S.B., Groff, S.A., Sartori, A., Bertolini, J., Kagimura, F.Y., Rotta, D., Mazzali, L. and Steilmann, P. (2012) Production of α-Amylase and Amyloglucosidase by the Fungus Cylindrocladium sp. in Semi-Solid State Fermentation. Journal of Microbiology Research, 2, 123-126. http://dx.doi.org/10.5923/j.microbiology.20120205.02
- Alazard, D. and Raimbault, M. (1981) Comparative Study of Amylolytic Enzymes Production by Aspergillus niger in Liquid and Solid-State Cultivation. European Journal of Applied Microbiology and Biotechnology, 12, 113-117.
- Onofre, S.B., Groff, S.A., Sartori, A., Bertolini, J., Kagimura, F.Y., Rotta, D., Mazzali, L. and Steilmann, P. (2011) Amylolytic Enzymes Produced by the Fungus Colletotrichum gloeosporioides in Rice Semi-Solid Fermentation. Journal of Yeast and Fungal Research, 2, 28-32.
- Silva, F.A.S.E. and Azevedo, C.A.V. (2002) Versão do programa computacional Assistat para o sistema operacional Windows. Revista Brasileira de Produtos Agroindustriais, 4, 71-78.
- Pandey, A. (2003) Solid State Fermentation. Biochemical Engineering Journal, 13, 81-84. http://dx.doi.org/10.1016/S1369-703X(02)00121-3
- Sales, M.R., Moura, R.B., Porto, G.R. and Porto, A.L.F. (2010) Variáveis que influenciam a produção de celulases e xilanase por espécies de Aspergillus. Pesquisa Agropecuária Brasileira, 45, 1290-1296. http://dx.doi.org/10.1590/S0100-204X2010001100009
- Doelle, D.W., Mitchell, D.A. and Rolz, C.E. (1992) Solid Substrate Cultivation. Elsevier Science Publishers Ltd., Amsterdam.
- Pinto, G.A.S. (2003) Produção de tanase por Aspergillus niger. Tese (Doutorado em Tecnologia de Processos Químicos e Bioquímicos)-Escola de Química, Universidade Federal do Rio de Janeiro, Rio de Janeiro.
- Sato, K. and Sudo, S. (1999) Small-Scale Solid-State Fermentations. Manual of Industrial Microbiology and Biotechnology, 6, 60-79.
- Bianchi, V.L.D., Moraes, I.O. and Capalbo, D.M.F. (2001) Fermentação em estado sólido. In: Schmidell, W.I., Lima, V.A., Aquarone, E. and Borzani, W., Eds., Biotecnologia Industrial, Vol. 3, Edgard Blucher Ltda, São Paulo, 247-276.
- Buswell, J.A., Cai, Y.J., Changs, S.T., Peberdy, J.F., Fu, S.Y. and Yu, H.S. (1998) Lignocellulolytic Enzyme Profiles of Edible Mushroom Fungi. World Journal of Microbiology and Biotecnology, 12, 537-542.
- Jan, H.D. and Chen, K.S. (2003) Production and Characterization of Thermostable Cellulases from Streptomyces Transformant T3-1. World Journal Microbiology and Biotechnology, 19, 263-268. http://dx.doi.org/10.1023/A:1023641806194
- Dalsenter, F.D.H., Viccini, G., Barga, M.C., Mitchell, D.A. and Krieger, N. (2005) A Mathematical Model Describing the Effect of Temperature Variations on the Kinetics of Microbial Growth in Solid-State Culture. Process Biochemistry, 40, 801-807. http://dx.doi.org/10.1016/j.procbio.2004.02.007
- Mitchell, D.A., Berovic, M. and Krieger, N. (2006) Solid-State Fermentation Bioreactors. In: Mitchell, D.A., Krieger, N. and Berovic, M., Eds., Springer, Heidelberg, 19.
- Gomes, E., Guez, M.A.U., Martin, N. and da Silva, R. (2007) Enzimas termoestáveis: Fontes, produção e aplicação industrial. Química Nova, 30, 136-145. http://dx.doi.org/10.1590/S0100-40422007000100025
- Carvalho, R.V., Côrrea, T.L.R., Silva, J.C.M., Mansur, L.R.C.O. and Martins, M.L.L. (2008) Properties of an Amylase from Thermophilic Bacillus sp. Brazilian Journal of Microbiology, 39, 102-107. http://dx.doi.org/10.1590/S1517-83822008000100023
- Taneda, D., Ueno, Y., Ikeo, M. and Okino, S. (2012) Characteristics of Enzyme Hydrolysis of Cellulose under Static Condition. Bioresource Technology, 121, 154-160. http://dx.doi.org/10.1016/j.biortech.2012.06.104
- Camassola, M., Bittencourt, L.R., Shenem, N.T., Andreaus, J. and Dillon, A.J.P. (2004) Characterization of the Cellulase Complex of Penicillium echinulatum. Biocatalysis and Biotransformation, 22, 391-396. http://dx.doi.org/10.1080/10242420400024532
- Alam, M.Z., Muyibi, S.A. and Wahid, R. (2008) Statistical Optimization of Process Conditions for Cellulase Production by Liquid State Bioconversion of Domestic Wastewater Sludge. Bioresource Technology, 99, 4709-4716. http://dx.doi.org/10.1016/j.biortech.2007.09.072
- Qin, Y., Wei, X., Song, X. and Qu, Y. (2008) Engineering Endoglucanase II from Trichoderma reesei to Improve the Catalytic Efficiency at a Higher pH Optimum. Journal of Biotechnology, 135, 190-195.
- Zhang, J., Tang, M. and Viikari, L. (2012) Xylans Inhibit Enzymatic Hydrolysis of Lignocellulosic Materials by Cellulases. Bioresource Technology, 121, 8-12.
- Bendig, C. and Weuster-Botz, D. (2012) Reaction Engineering Analysis of Cellulase Production with Trichoderma reesei RUT-C30 with Intermittent Substrate Supply. Bioprocess and Biosystems Engineering, 132, 121-128.
- Puglisi, I., Faedda, R., Sanzaro, V., Lo Piero, A.R., Petrone, G. and Cacciola, S.O. (2012) Identification of Differentially Expressed Genes in Response to Mercury I and II Stress in Trichoderma harzianum. Gene, 506, 325-330.
- Castro, A.M. (2006) Produção e Propriedades de Celulases de Fungos Filamentosos, obtidas a partir de Celulignina de Bagaço de cana-de-açucar (Saccharum spp.). Dissertação de Mestrado, Universidade Federal do Rio de Janeiro, Rio de Janeiro.
- Rodríguez-Zúñiga, U.F., Farinas, C.S., Bertucci Neto, V., Couri, S. and Crestana, S. (2011) Produção de celulases por Aspergillus niger por fermentação em estado sólido. Pesquisa Agropecuária Brasileira, 46, 912-919. http://dx.doi.org/10.1590/S0100-204X2011000800018
- Camassola, M., Bittencourt, L.R., Shenem, N.T., Andreaus, J. and Dillon, A.J.P. (2004) Characterization of the Cellulase Complex of Penicillium echinulatum. Biocatalysis and Biotransformation, 22, 391-396. http://dx.doi.org/10.1080/10242420400024532
- Spier, M.R. (2005) Produção de enzimas amilolíticas fúngicas α-amilase e amiloglucosidase por fermentação no estado sólido. Dissertação (Mestrado em Tecnologia de Alimentos), Universidade Federal do Paraná, Curitiba.
- Zhang, Y.H.P., Himmel, M.E. and Mielenz, J.R. (2006) Outlook for Cellulase Improvement: Screening and Selection Strategies. Biotechnology Advances, 24, 452-481. http://dx.doi.org/10.1016/j.biotechadv.2006.03.003
- Chandra, M.S., Viswanath, B. and Reddy, B.R. (2007) Cellulolytic Enzymes on Lignocellulosic Substrates in Solid Sate Fermentation by Aspergillus niger. Indian Journal of Microbiology, 47, 323-328. http://dx.doi.org/10.1007/s12088-007-0059-x
- Sanchez, C. (2009) Lignocellulosic Residues: Biodegradation and Bioconversion by Fungi. Biotechnology Advances, 27, 185-194. http://dx.doi.org/10.1016/j.biotechadv.2008.11.001
- Ahamed, A. and Vermette, P. (2010) Effect of Mechanical Agitation on the Production of Cellulases by Trichoderma reesei RUT-C30 in a Draft-Tube Airlift Bioreactor. Biochemical Engineering Journal, 49, 379-387. http://dx.doi.org/10.1016/j.bej.2010.01.014
- Gao, D., Chundawat, S.P.S., Uppugundla, N., Balan, V. and Dale, B.E. (2011) Binding Characteristics of Trichoderma reesei Cellulases on Untreated, Ammonia Fiber Expansion (AFEX), and Dilute-Acid Pretreated Lignocellulosic Biomass. Biotechnology and Bioengineering, 108, 1788-1800. http://dx.doi.org/10.1002/bit.23140


