Open Journal of Medical Microbiology
Vol.05 No.04(2015), Article ID:61699,9 pages
10.4236/ojmm.2015.54024
Batumin―A Selective Inhibitor of Staphylococci―Reduces Biofilm Formation in Methicillin Resistant Staphylococcus aureus
Larisa Churkina1*, Mario Vaneechoutte2, Elena Kiprianova1, Natalia Perunova3, Lilja Avdeeva1, Oleg Bukharin3
1Department of Antibiotics, Institute of Microbiology and Virology, National Academy of Sciences of Ukraine, Kyiv, Ukraine
2Laboratory for Bacteriology Research, Faculty of Medicine & Health Sciences, Ghent University, Ghent, Belgium
3Institute of Cellular and Intracellular Symbiosis, Ural Branch of Russian Academy of Sciences, Orenburg, Russian Federation

Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).


Received 15 October 2015; accepted 1 December 2015; published 4 December 2015

ABSTRACT
The antibiotic batumin, produced by Pseudomonas batumici, has been shown to be highly active against 123 type and reference strains and clinical isolates of 30 Staphylococcus species (including MRSA and small colony variants―(SSCVs) of S. aureus, S. epidermidis and S. haemolyticus). Batumin activity against these bacteria did not depend on the species, origin or resistance to other antibiotics and its MIC was 0.0625 - 0.5 mg/ml. Batumin influence on biofilm formation was studied in clinical isolates of S. aureus, S. epidermidis and S. intermedius. Addition of batumin at a concentration of half of the MIC in the broth, i.e. 0.125 µg/ml, decreased the biofilm of 16 out of 20 S. aureus strains to varying degrees. Batumin was more effective against Staphylococcus strains with strong biofilm formation. Using atomic-force microscopy, it could be shown that batumin reduced the number of S. aureus ATCC 25923 adherent cells more than fourfold. The adherent cells of staphylococci were visualized as monolayers of separate islets. A detailed study of the surface of bacterial cells treated with batumin allowed to establish significant reduction of their roughness values. Observed values were typical for planktonic S. aureus cells. The obtained data explain one of the mechanisms of the antimicrobial activity of batumin, which is based оn preventing the formation of S. aureus biofilm. As such, batumin could be considered as an agent offering opportunities for the treatment of staphylococcal biofilm-associated infections.
Keywords:
Batumin, Staphylococci, SSCVs, Biofilm

1. Introduction
Batumin, a polyketide antibiotic, produced by Pseudomonas batumici, has high and selective activity against staphylococci [1] [2] . This predetermined its use not only for treatment of infections and nasal carriage of staphylococci but also for diagnosis of staphylocococcal infection, using the “Diastaph” preparation, which consists of batumin-impregnated disks [3] . The high susceptibility of staphylococci to batumin distinguishes them from representatives of other taxons, as was demonstrated using “Diastaph” in several thousands of clinical isolates of bacteria [4] .
Bacteria growing as biofilm are highly resistant to antibiotics [5] . However, our previous studies have shown that addition of half the concentration of the batumin MIC (0.125 μg/ml) to the media considerably reduced biofilm formation in 80% of nasal staphylococcal strains, which had initially high levels of biofilm formation [6] .
The objective of this work was to broaden our knowledge about batumin activity against Staphylococcus species, against atypical forms of this pathogen, i.e. small staphylococcal colony variants (SSCVs), and to obtain more detailed insights into the influence of batumin on biofilm formation by using atomic-force microscopy.
2. Materials and Methods
Batumin, obtained by fermentation of Pseudomonas batumici and purified by silica gel preparative chromatography to 85% of purity, was used.
Batumin is commercially available from Santa Cruz Biotechnology (Santa Cruz, CA) or Enzo Life Sciences (Antwerp, Belgium).
Type and collection strains of 30 different species belonging to the genus Staphylococcus (Table 1) and 50 methicillin resistant S. aureus (MRSA) strains isolated in the
Identification of MRSA was carried out according to methods described in [7] . Susceptibility of staphylococcal clinical strains to a wide spectrum of antibiotics was tested by the Kirby-Bauer method. NCCLS criteria [8] were used to interpret the susceptibility to antibiotics.
Thirty strains of SSCVs were isolated as subpopulations on Columbia agar with 5% sheep blood, as pinpoint colonies (0.1 - 0.3 mm) after 48 hours of aerobic incubation at 37˚C, among the more numerous colonies (2 - 3 mm) with normal Staphylococcus morphology, considered as the parental isolates of the SSCVs. Identification of the staphylococci was carried out according to standard methods [9] .
The atypical forms of staphylococci were previously identified by standard methods and using tDNA-PCR analysis and were assigned to three species: S. aureus, S. epidermidis and S. haemolyticus. Their detailed description was presented previously [10] .
The minimal inhibitory concentration (MIC) of batumin was studied according to CLSI Standards (2005) in Mueller-Hinton agar or broth [11] . The microbial load of SSCVs was 0.5 × 108 cfu/ml and the Petri dishes were incubated at 37°С for 48 hours.
Half the MIC of batumin was used to study its effect upon biofilm formation in staphylococci. Biofilm formation was studied by a photometric method determining the bacterial capacity to adhere onto the 96-hole polystyrole plane-table surface with subsequent crystal violet colouring [12] . Optical density measurement was done using a photometer ELx808 (
For the study of batumin effect on biofilm formation we used S. aureus B-904 (UCM) as test-culture. For biofilm production, glass coverslips were immersed into Luria-Bertani broth with 0.125 μg/ml of batumin and incubated for 48 h at 37˚C.
Visualization of the biofilms was done by atomic force microscopy using the SMM-2000 microscope (Proton- MIET Closed JOINT Stock Company, Russia), in contact mode in air environment [13] [18] .
3. Results and Discussion
The susceptibility to batumin of Staphylococcus species reference strains of some collection strains and of methicillin-resistant S. aureus strains are presented in (Table 1). It is of interest to note the high uniformity of the susceptibility to batumin for the different Staphylococcus species. Batumin inhibited most of the studied strains and species, including MRSA, at concentrations between 0.25 and 0.5 μg/ml, and rarely 1.0 μg/ml was needed.
Table 1. Susceptibility to batumin of species belonging to different 16S RNA complexes of the genus Staphylococcus.
The strain numbers of the Ukrainian Collection of Microorganisms (UCM) are presented. Numbers from АТСС or from other collections between brackets.
Our data show that the most susceptible species to batumin were species of the S. epidermidis complex (МIC 0.0625 - 0.125 μg/ml). Strains of this complex demonstrated the highest batumin susceptibility also during previous clinical trials (unpublished data).
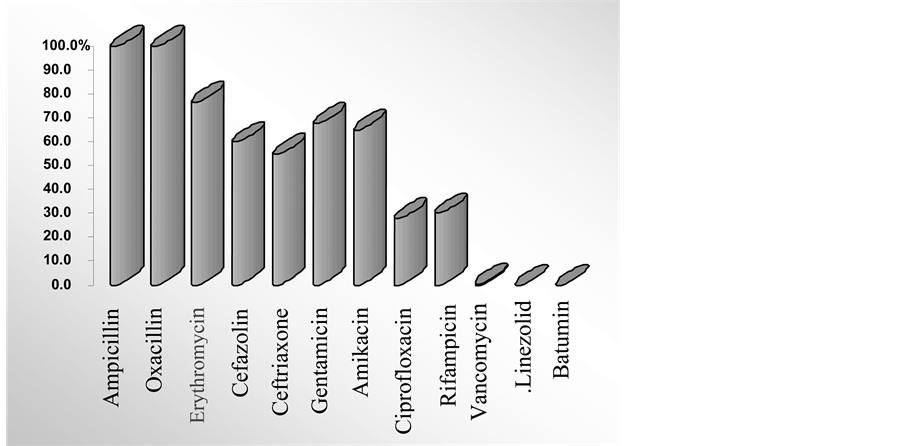
Figure 1 shows the antibiotic-resistance profile of the studied MRSA strains, indicating that these strains are resistant to all commonly used antibiotics. At the same time, all 50 strains―regardless their origin and their susceptibility to different antibiotics―were inhibited by 0.5 μg/ml of batumin.
Batumin is also effective against macrococci which were set separately from the genus Staphylococcus and included in the new genus Macrococcus. On the other hand, Gemella morbillorum and G. haemolysans, isolated from osteomyelitis patients, did not show growth inhibition zones around batumin disks [14] , despite their relationship to staphylococci. Batumin was also not effective against bacilli, Listeria, planococci and other bacteria which belong to the order of the Bacillales, comprising the family of the Staphylococcaceaе [2] [4] .
Data presented here, as well as studies of the past years, demonstrate high activity of batumin against metabolically normal members of the genus Staphylococcus [2] [3] , as well as against atypical forms of staphylococci, the so-called small colony-variants (SSCVs). SSCVs are formed as a result of mutations and characterized by a complex pleiotropic phenotype which can be largely explained by defective electron transport [15] [16] .
All 30 SSCVs included in this study were isolated on solid growth media as a subpopulation among the larger number of colonies with normal for staphylococci morphology. They were characterized by altered colony morphology, delayed growth and lack of pigmentation. Moreover, many of them lacked lecithinase, phosphatase, coagulase and haemolytic activity and the metabolism of carbohydrates such as sucrose, lactose and fructose was altered, which is consistent with the literature and with our previous results [10] [17] .
Coming from our previous data on batumin antimicrobial action against SSCVs we first estimated this effect studying the growth inhibiting concentrations.
Most of the SSCVs were inhibited by a concentration of 0.25 μg batumin/ml. Strain S. aureus 71 was most resistant, with an MIC of 0.5 μg/ml.
So the data presented in this work and first obtained using strains of practically all known staphylococci species, multiresistant clinical isolates and atypical forms of this pathogen give evidence that batumin is highly active selective inhibitor of representatives of the genus Staphylococcus.
Susceptibility of nasal and skin clinical isolates of staphylococci to batumin was the same as of type and collection strains of different species of the genus Staphylococcus studied before [6] . At the same time, the antibiotic effect against biofilm formation was different in different strains and species. Their biofilm formation values varied from 2.6 - 3.1 CU for S. aureus to 1.8 - 2.3 for S. epidermidis and 1.7 - 1.9 for S. intermedius strains (Table 2). Presence of half of batumin MIC in the broth reduced the biofilm formation in 85% of studied
Figure 1. The antibiotic resistance profiles of 50 methicillin-resistant Staphylococcus aureus strains. Legend: x-axis: studied antibiotics; y-axis: % of MRSA strains, resistant to these antibiotics.
Table 2. Batumin effect against biofilm formation by staphylococci.
*The ratio of the optical density of the samples in the experiment and control. Expressed as conventional unit (CU) = OD630 in experimental samples/OD630 in control samples. Optic density of control samples is 0.05, which is the nutrient broth density.
S. aureus strains. It should be noted that batumin is more effective against Staphylococcus strains with strong biofilm formation (CU values between 2.6 and 3.1).
Analysis of experimental data on batumin effect on the stages of biofilm formation in S. aureus demonstrated that the effectiveness of batumin depended on the stage of biofilm formation (Table 3). Simultaneous batumin addition with S. aureus into culture medium promoted reduction of biofilm formation values in all studied staphylococci strains, at that already formed film was more resistant to studied preparation.
More detailed study of batumin action upon S. aureus biofilm formation was carried out using atomic force microscopy. The object of this study was the reference strain S. aureus UCM B-904. This strain formed biofilm on the surface of the glass, as can be seen from Figure 2(a). Surface biofilm was formed by exopolymeric matrix with cells of round shape immersed in it (Figure 2(b)).
For strain S. aureus B-904, the effect of batumin with regard to biofilm formation disturbance corresponded to a more than fourfold decrease of the number of adherent cells. Moreover, in the presence of batumin, the cells of staphylococci were observed in the form of monolayer of separate islets (Figure 2(c)). Particles of exopolymeric matrix were determined with average values of thickness of 63 - 62 nm, located between the cells and on their surface (Figure 2(d)). In this case the transverse dimension of cells was 0.64 ± 0.08 mm, not significantly different from control values. A detailed study of the surface of bacterial cells treated with batumin allows to establish significant reduction of their roughness values (Table 4). Observed values were typical for planktonic S. aureus cells [19] .
Atomic force microscopy revealed qualitative and quantitative changes in the exopolymeric matrix due to batumin treatment, as well as a significant reduction in the number of cells adhered to the coverslip, preventing formation of S. aureus biofilm.
It is known that some biofilms are covered by a surface film composed of lipid components similar to those in bacterial membranes which are a barrier for the penetration of antibiotics [20] . We have previously shown that batumin has significant effects on lipid metabolism of S. aureus [21] . Based on the similarity of a biofilm matrix lipid on the one hand and membrane of bacterial cell on the other hand, it can be assumed that batumin penetrates well into staphylococcal biofilm, explaining the significant changes of exopolymeric matrix that can be observed.
4. Conclusions
The data presented in this work show that batumin is a highly active antimicrobial agent, inhibiting all species of staphylococci and macrococci, regardless their origin or antibiotic susceptibility. The MIC-values for batumin
Table 3. Influence of batumin (0.125 mg/ml) on the steps of biofilm formation by Staphylococcus aureus.
Figure 2. Аtomic force microscopy-topography images of Staphylococcus aureus B-904. ((а) (b)) S. aureus B-904 without batumin; ((c) (d)) S. aureus B-904 grown in the presence of 0.125 μg/ml of batumin. Arrows: 1: particles of exopolymeric matrix; 2 bacterial cells; Scale―2 μm ((a) (b)); 1 μm ((c) (d)).
Table 4. Morphological characteristics of cells and adherence to glass of S. aureus B-904 in the presence of 0.125 µg/ml batumin.
*p < 0.05 (Mann-Whitney U-test). **To determine bacterial cell length and width, cells that were at least immersed into exopolymeric matrix were selected. However, height determination of the cells in this case would have been incorrect and therefore was not implemented.
range between 0.25 and 0.5 µg/ml, rarely reaching 1.0 μg/ml. It should be noted that these results were obtained with 85% pure batumin, whereas, according to [22] , the MIC of 95% pure batumin against S. aureus was much higher, i.e. 0.05 μg/ml.
Probably batumin differs in its mechanism of antimicrobial action from all antibiotic substances used at present in clinical practice, and the nature of its selectivity is connected with some peculiarity of staphylococcal metabolism which sets them apart from other genetically related bacteria. Taking into account, the polyketide nature of batumin and our earlier data about batumin activity upon Staphylococcus lipids [21] , we may suppose that these distinctions are determined by specific characteristics of the staphylococcal fatty acids metabolism.
The obtained data on atomic force microscopy explain one of the mechanisms of the antimicrobial action of batumin, based оn preventing formation of S. aureus biofilm that allows considering it as a promising agent in treatment of staphylococcal biofilm-associated infections.
Cite this paper
LarisaChurkina,MarioVaneechoutte,ElenaKiprianova,NataliaPerunova,LiljaAvdeeva,OlegBukharin, (2015) Batumin—A Selective Inhibitor of Staphylococci—Reduces Biofilm Formation in Methicillin Resistant Staphylococcus aureus. Open Journal of Medical Microbiology,05,193-201. doi: 10.4236/ojmm.2015.54024
References
- 1. Kiprianova, E.A., Klochko, V.V., Zelena, L.B., Churkina, L.N. and Avdeeva, L.V. (2011) Pseudomonas batumici sp. nov., the Antibiotic-Producing Bacteria Isolated from Soil of the Caucasus Black Sea Coast. Mikrobiologichny Zhurnal, 73, 3-8.
- 2. Klochko, V.V., Kiprianova, E.A., Churkina, L.N. and Avdeeva, L.V. (2008) Antimicrobial Spectrum of Antibiotic Batumin. Mikrobiologichny Zhurnal, 70, 41-46.
- 3. Churkina, L.N., Kiprianova, E.A., Bidnenko, S.I., Marchenko, K.P. and Artysyuk, E.I. (2009) Antibiotic Batumin for Diagnostics of Staphylococci and Treatment of Staphylococcus aureus Nasal Carriage. Likarska Sprava, 1-2, 61-67.
- 4. Smirnov, V.V., Churkina, L.N., Nosenko, G.A., Bidnenko, S.I., Artysiuk, E.I., Pustovalova, L.I., Kiprianova, E.A. and Garagulya, A.D. (2002) Efficacy of Diagnostic Disks with Batumin in Identification and Indication of Staphylococci. Likarska Sprava, 5-6, 27-31.
- 5. Frank, K.L., Reichert, E.J., Patel, R. and Piper, K.E. (2007) In Vitro Effects of Antimicrobial Agents on Planktonic and Biofilm Forms of Staphylococcus lugdunensis Clinical Isolates. Antimicrobial Agents Chemotherapy, 51, 888-895.
http://dx.doi.org/10.1128/AAC.01052-06 - 6. Bukharin, O.V., Churkina, L.N., Perunova, N.B., Ivanova, E.V., Novikova, I.V., Avdeeva, L.V. and Yaroshenko, L.V. (2012) Influence of Antistaphylococcal Antibiotic Batumin on Microorganisms Biofilm Formation. Journal of Microbiology Epidemiology and Immunobiology, 2, 8-12. (In Russian)
- 7. Boutiba-Ben Boubaker, I., Ben Abbes, R., Ben Abdallah, H., Mamlouk, K., Mahjoubi, F., Kammoun, A., Hammami, A. and Ben Redjeb, S. (2004) Evaluation of a Cefoxitin Disk Diffusion Test for the Routine Detection of Methicillin-Resistant Staphylococcus aureus. Clinical Microbiology Infection, 10, 762-765.
http://dx.doi.org/10.1111/j.1469-0691.2004.00919.x - 8. Vandepitte, J., Engback, K., Piot, P. and Heuck, C.C. (1991) Basic Laboratory Procedures in Clinical Bacteriology. WHO Library, Geneva.
- 9. Kloos, W.E. and Schleifer, K.H. (1986) Genus IV. Staphylococcus. In: Sneath, P.H.A., Mair, N.S., Sharpe, M.E. and Holt, J.G., Eds., Bergey’s Manual of Systematic Bacteriology, Volume 2, Williams & Wilkins, Baltimore.
- 10. Churkina, L.N., Bidnenko, S.I., dos Santos Santiago, G.L., Vaneechoutte, M., Avdeeva, L.V., Lutko, O.B. and Oserjanskaja, N.M. (2012) Application of the Antibiotic Batumin for Accurate and Rapid Identification of Staphylococcal Small Colony Variants. BMC Research Notes, 5, 374-378.
http://dx.doi.org/10.1186/1756-0500-5-374 - 11. Clinical and Laboratory Standards NCCLS (2005) Performance Standards for Antimicrobial Susceptibility Testing; Fifteenths Informational Supplement. CLSI/NCCLS Document M100-S 15, 165 p.
- 12. O’Tool, G.A. and Kolter, R. (1998) Initiation of Bioilm Formation in Pseudomonas fluorescens WCS365 Proceeds via Multiple, Convergent Signaling Pathways: A Genetic Analysis. Molecular Microbiology, 28, 449-461.
- 13. Megan, E.N., Martin, M.O. and Chan, P.H. (2005) Atomic Force Microscopy of Bacterial Communities. Methods Enzymology, 397, 256-258.
http://dx.doi.org/10.1016/S0076-6879(05)97015-8 - 14. Bidnenko, S.I., Lutko, O.B., Oserjanskaja, N.M. and Churkina, L.N. (2010) Microflora of Periprosthetic Tissues According to Aseptic Instability of Hip Endoprosthesis and Features of Its Sensitivity to Antibiotics. Biomedical Biosocial Anthropology, 15, 87-91.
- 15. Looney, W.J. (2000) Small Colony Variants of Staphylococcus aureus. British Journal Biomedical Sciences, 57, 317-322.
- 16. McNamara, P. and Proctor, R. (2000) Staphylococcus aureus Small Colony Variants, Electron Transport and Persistent Infections. International Journal Antimicrobial Agents, 14, 117-122.
http://dx.doi.org/10.1016/S0924-8579(99)00170-3 - 17. Churkina, L.N., Bidnenko, S.I., Avdeeva, L.V., Vaneechoutte, M., Makushenko, A.S., Lutko, O.B. and Oserjanskaja, N.M. (2011) Characteristics of Atypical Forms of Staphylococci (SCVs), Isolated from Patients with Osteomielitis. Antibiotics and Chemotherapy, 55, 36-40. (In Russian)
- 18. Eaton, P., Fernandes, J.C., Pereira, E., Pintado, M.E. and Xavier Malcata, F. (2008) Atomic Force Microscopy Study of the Antibacterial Effects of Chitosans on Escherichia coli and Staphylococcus aureus. Ultramicroscopy, 108, 1128-1134.
http://dx.doi.org/10.1016/j.ultramic.2008.04.015 - 19. Francius, G., Domenec, O., Mingeot-Leclercq, M.P. and Dufrêne, Y.F. (2008) Direct Observation of Staphylococcus aureus Cell Wall Digestion by Lysostaphin. Journal of Bacteriology, 109, 7904-7909.
http://dx.doi.org/10.1128/JB.01116-08 - 20. Tetz, V.V., Korobov, V.P., Artemenko, N.K., Lenikina, L.M., Panjkova, N.V. and Tetz, G.V. (2004) Extracellular Phospholipids of Isolated Bacterial Communities. Biofilms, 1, 149-155.
http://dx.doi.org/10.1017/S147905050400136X - 21. Smirnov, V.V., Churkina, L.N. and Vasiurenko, Z.P. (1988) Lipids. In: Nikitina, I.I., Ed., Staphylococci, Naukova Dumka, Kiev, 35-63.
- 22. Mattheus, W., Gao, L.-J., Herdewijn, P., Landuyt, B., Verhaegen, J., Masschelein, J., Volckaert, G. and Lavigne, R. (2010) Isolation and Purification of a New Kalimantacin/Batumin-Related Polyketide Antibiotic and Elucidation of Its Biosynthesis Gene Cluster. Chemistry & Biology, 17, 149-159.
http://dx.doi.org/10.1016/j.chembiol.2010.01.014
NOTES

*Corresponding author.