Journal of Biosciences and Medicines
Vol.06 No.03(2018), Article ID:83018,9 pages
10.4236/jbm.2018.63006
Comparison of Chondrogenic Ability between Mesenchymal Stem Cells and Buffy Coat in Vitro
Longhao Jin1*, Zhongye Zhang2*, Yanqun Liu1, Kumar Gurung1, Yongjun Jin1, Yuanming He1, Yu Jin3#, Zhehu Jin4#
1Department of Orthopedic Surgery, Yanbian University Hospital, Yanji City, China
2Department of Orthopedic Surgery, Songyuan Central Hospital, Songyuan City, China
3Department of Anatomy, School of Medicine, Yanbian University, Yanji City, China
4Department of Dermatology, Yanbian University Hospital, Yanji City, China
Copyright © 2018 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: February 8, 2018; Accepted: March 11, 2018; Published: March 14, 2018
ABSTRACT
Objective: In vitro comparison of chondrogenic differentiation ability of bone marrow extracted mesenchymal stem cells (MSCs) and buffy coat. Methods: MSCs of New Zealand white rabbits were cultured in vitro and adherent cells were passaged. The cells were inoculated on polyglycolic acid (PGA) scaffold (3 mm in diameter and 2 mm in height) liquid for 21 days. Under the same conditions, without subculture, buffy coat was directly inoculated into the cell scaffold. The degree of chondrogenic differentiation was compared by Safranin-O staining, histological scoring and biochemical functional detection. Results: The chondrogenic differentiation ability of the buffy coat group was better than that of the MSC group. Safranin-O stain was stronger in buffy coat group than in MSC group. The Bern Score was also higher in the buffy coat group than in the MSC group and the total amount of sulfated glycosaminoglycans (GAGs) in buffy coat group was higher than that in MSC group. Conclusion: The chondrogenic differentiation ability of buffy coat is higher than that of MSC. Through this result, it was found buffy coat can also differentiate into sub-cartilage without stimulating growth factor. Thus, buffy coat can make a great source in artificial cartilage engineering.
Keywords:
Chondrogenic, Buffy Coat, Mesenchymal Stem Cells

1. Introduction
According to World Health Organization statistics, there are currently more than 50 million patients with osteoarthritis in China, a figure that will continue to increase as the population ages. Cartilage injury is an important cause of knee osteoarthritis. However, there is currently no effective method of articular cartilage regeneration. Therefore, the exploration and development of a new cartilage regeneration method have important value.
With the development of tissue engineering, repairing damaged cartilage tissue has become a hot topic. The main factors of tissue engineered cartilage are seed cells, scaffold materials, and cell growth factors. Currently used cells include chondrocytes, mesenchymal stem cells (MSCs), and induced pluripotent stem cells [1] . Among them, MSCs are convenient, widely available, and useful for autologous transplantation; plus, they have the potential for multi-directional differentiation and are easy to isolate and amplify in vitro. MSC differentiation into chondrocytes involves a complex control process consisting of biological, physical, genetic, and other factors. Transforming growth factor-β (TGF-β), insulin-like growth factor (IGF), bone morphogenic proteins, and fibroblast growth factor are widely used in chondrocyte differentiation [2] . However, the mechanisms of action of these cytokines and regulatory factors are not yet fully clear, their clinical application is still a doubt.
Numerous studies have demonstrated the therapeutic potential of mononuclear cells (MNCs) extracted from bone marrow for tissue regeneration after ischemic femoral head necrosis, myocardial infarction, and cartilage injury [3] . Jin et al. (2011) first observed that the buffy coat containing monocytes, lymphocytes, macrophages, platelets, and various cytokines displayed good cartilage regeneration [4] . Therefore, in vitro comparison of cartilage differentiation ability of MSCs and bone marrow extract buffy coat has been done in this study.
2. Materials and Methods
2.1. Ethical Approval Statement
Healthy 16-week-old New Zealand white rabbits were purchased from the experimental animal center of Yanbian University. All of the rabbits were housed during the experimental study under 12:12 hour light/dark conditions at approximately 27˚C. The experimental procedure was approved by the Institutional Animal Care and Use Committee of Yanbian University Hospital (2015036).
2.2. Preparation of MSCs, Buffy Coat, and Polyglycolic Acid Scaffold
Rabbit bone marrow-derived buffy coat and MSCs were obtained from 16-week-old male (n = 12) New Zealand white rabbits. Two milliliters of bone marrow blood was harvested from the iliac crest. The samples were diluted in phosphate buffered saline (PBS) and the buffy coat was isolated by centrifugation using a Ficoll density gradient (17-1440-02; Amersham Biosciences, Sweden) at 1000 × g for 30 min [4] . A total of 0.5 mL of buffy coat in the serum was isolated from this suspension of which 6 buffy coat were resuspended in α-minimum essential medium supplemented with 1% antibiotic-antimycotic and 10% fetal bovine serum. The buffy coat was cultured at 37˚C in a 5% CO2 incubator. After 6 days, non-adherent cells were removed and adherent cells were further cultured. At day 12 after cell seeding, the adherent MSCs were retrieved by trypsin treatment and re-plated at 3 × 105 cells/plate (100 mm) for expansion. The second passage seed cells were collected and re-suspended in a polyglycolic acid (PGA) scaffold (5 × 106 MNCs/each) and incubated in a CO2 incubator for 90 minutes and then placed in chondrogenic medium.
The bone marrow (2 mL) aspirates were obtained aseptically from the iliac crest, while the buffy coat was obtained by Ficoll gradient centrifugation at 1000 × g for 30 min. The buffy coat (0.5 mL) was dispensed with a pipette and placed in a 1.5-mL tube. The concentrated buffy coat was centrifuged at 1000 × g for 5 min. After supernatant removal, 400 μL was collected and 100 μL of concentrated buffy coat seeding was collected onto a PGA scaffold (5 × 106 MNCs/scaffold) and incubated in a CO2 incubator for 90 minutes and then placed in chondrogenic medium.
The PGA scaffold in sheet form was purchased from Albany International Inc. (Mansfield, MA, USA). The PGA scaffold was cut by a biopsy punch to 2 mm thick and 3 mm in diameter. The PGA scaffolds were submerged in ethylene oxide for sterilization.
2.3. Inducing Chondrogenic Differentiation
Chondrogenic medium (Dulbecco’s modified Eagle’s medium supplemented with ITS mixture, 50 mg/mL ascorbate 2-phosphate, 100 nM dexamethasone, 40 mg/mL proline, and 1.25 mg/mL bovine serum albumin), without TGF-β, a typical chondrogenic inducer. The cell-seeded scaffolds were placed in 24-well plates and incubated until 4 weeks at 37˚C under 5% CO2 to induce chondrogenic differentiation (n = 6/scaffold).
2.4. Live/Dead Cytotoxicity Assay
The percentage of viable and dead cells was measured using a live/dead viability/ cytotoxicity assay (Molecular Probes, Eugene, OR, USA) according to the manufacturer’s instructions. One-day samples were incubated in a solution containing 2 mM ethidium homodimer-1 and 4 mM calcein AM for 30 min. After being washed in PBS, the construct was placed on a glass slide and carefully pressed flat using a cover glass. The fluorescence image was visualized using a laser canning confocal microscope (Zeiss LSM510 Meta; Carl Zeiss, Jena, Germany). The number of viable (green fluorescence) or dead (red fluorescence) cells was quantified by Image-Proplus 4.0 image analysis software (Media Cybernetics, Silver Spring, MD, USA). Cell viability was determined by dividing the number of green cells (viable cells) by the total cells (green cells + red cells) [5] .
2.5. Histological Evaluations
The PGA scaffolds were fixed with 4% paraformaldehyde for 4 days, embedded in paraffin, cut into ultrathin sections, and stained with Safranin O/solid green staining in accordance with the literature. The prepared paraffin sections were dewaxed to water; stained with hematoxylin for 3 minutes; washed with 1% hydrochloric acid for 15 seconds and water for 3 minutes, a 0.02% solid green water solution for 10 seconds, and 1% acetic acid to remove residual solid green; and stained with 0.1% Safranin O for 6 minutes. They were then dried in air, dehydrated, xylene transparent, and finally sealed with neutral gum for microscopic observation.
2.6. Histological Scoring (Bern Score)
To minimize the effects of subjective bias, three researchers independently evaluated the quality of the engineered cartilage and graded the Safranin O-fast green-stained slides using the Bern Score, which consists of seven categories and assigns a score of 0 - 9 [6] .
2.7. Chemical Assay
The specimens were dried and digested with papain. The glycosaminoglycan (GAG) content was detected using the 1,9-dimethyl methylene blue (DMB) color method as follows: 0, 0.00625, 0.0125, 0.025, 0.05, 0.1 mg/mL chondroitin sulfate standard solution (50 µL) added to 200 µL of DMB reagent mixture, left to react in the dark for 30 minutes and then examined at a 530-nm wavelength colorimetric absorbance to create the standard GAG curve. Aliquots of the supernatant of the experimental and control groups (1 mL and 50 µL, respectively) were collected after the centrifugation, and 200 µL of DMB was added for 30 minutes. The supernatant absorbance was determined by the same method, and the supernatant GAG was determined by the standard curve. DNA content was detected by Qubit Fluorometer in papain-digested specimens as described previously [7] .
2.8. Statistical Method
Student’s t test in Microsoft Excel 2007 was used to analyze the data. The data are expressed as mean ± standard deviation. Group comparisons were made using single factor analysis of variance, and values of p < 0.05 were considered statistically significant (*p < 0.05, **p < 0.01, and ***p < 0.001).
3. Results
3.1. Live/Dead Cytotoxicity Assay
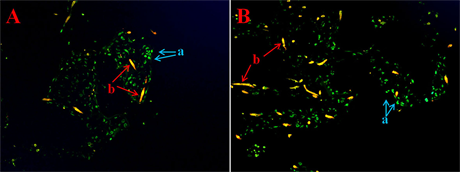
The MSCs and buffy coat were inoculated into PGA cell stents for 1 day and a live/dead cytotoxicity assay (Figure 1) was performed to observe cell viability and survival. The inoculation and survival rates were high in both groups; we observed virtually no apoptotic cells. These results indicate that the number of seed cells of these two groups were almost similar.
Figure 1. Live/dead cytotoxicity assay images after 1 day of mesenchymal stem cell (MSC) and buffy coat inoculation into polyglycolic acid (PGA) cell scaffolds. (A), viable cells; (B), PGA cell scaffold. No red apoptotic cells are visible.
Figure 2. Histological observation of artificial cartilage cultured for 21 days (Safranin O/solid green staining). (A)-(C) are tissue sections of the mesenchymal stem cell (MSC) group, and (B) and (C) are enlarged images of regions of interest (ROI) 1 and 2 in the Figures 2(D)-(F) are tissue sections of the buffy coat group, while D and E are enlarged images of ROI 1 and ROI 2 in Figure 2(A) and Figure 2(D), ×40 magnification; (B), (C), (E), and (F), ×100 magnification.
3.2. Histological Analysis
Cells in the experimental and control groups were cultured on the PGA scaffolds for 21 days and were stained with Safranin O stain (Figure 2), and a significant intergroup difference was noted. On microscopic observation, cells in the outer region of the MSC group displayed differentiation into chondrocytes and GAG secretion; however, the central cells did not differentiate into chondrocytes and apoptosis was noted. On the contrary, in the experimental group, cells in the center and surrounding areas displayed active differentiation into chondrocytes and GAG secretion.
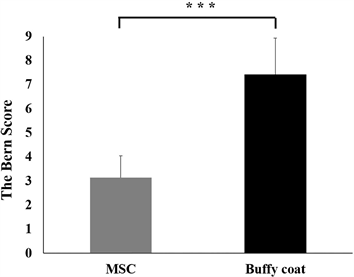
Analysis of these sections using the Bern Score supports this observation. The cells in the constructs with high cell densities showed increased Safranin-O staining in the buffy coat group, which demonstrated a rounded cell shape and higher extracellular matrix (ECM) production (Figure 2). That buffy coat and MSC groups showed a difference in Bern Score (number of engineered cartilages, n = 7; p < 0.001; Figure 3).
3.3. Chemical Assay
DMB colorimetry was performed in the experimental and control groups after 21 days of culture on the PGA scaffold for the quantitative detection of GAG content. The results showed that the GAG content of the buffy coat group was greater than that of the MSC group (n = 7; p < 0.001) (Figure 4(A)). The results met the histological analysis in contrast to GAG content, DNA content was significantly
Figure 3. Histology rating assessed by the Bern Score. The higher seeding concentrations showed higher rates (n = 7; p < 0.001).
Figure 4. The glycosaminoglycan (GAG) content of the experimental and control groups after 21 days of culture on the polyglycolic acid (PGA) scaffold. The GAG content in the buffy coat group was significantly higher than that in the mesenchymal stem cell (MSC) group. There was a significant intergroup difference (n = 7; p < 0.001). (B), DNA content of the test results in which the MSC group had significantly higher content than the buffy coat group (n = 7; p < 0.001).
higher in the MSC group than in the buffy coat group (n = 7; p < 0.001; Figure 4(B)). Cells in a cartilage are not so dense, and the buffy coat group of cells had an ECM ratio close to that of normal cartilage.
4. Discussion
The results showed that the cartilage differentiation of the experimental group and the buffy coat group was much more than that of the single MSCs in vitro. The bone marrow cells and cytokines were cultured in vitro with different growth factors.
The extracted concentration of bone marrow MSC and MNC from the iliac crest of the rabbit was 607.8/mL and 2.3 × 106/mL, respectively [4] . Thus, there are approximately 1216 MSCs and 4.6 × 106 MNCs in 2 mL of bone marrow. The MSC concentration in bone marrow is very low. Therefore, the same amount of MSCs cannot be inoculated, although the same number of MSCs and MNCs must be inoculated.
The same number of cells was inoculated into the same cell scaffold, and we found no difference in the initial cell coverage or viability (Figure 1). However, we know that the MSC content in the buffy coat group was significantly less than that in the MSC group. However, there were significant intergroup differences after 21 days of differentiation in terms of MSC differentiation into cartilage cells around the scaffold, GAG secretion, and the appearance of undifferentiated and apoptotic cells in the central part of the culture plate. The buffy coat group was homogeneous with respect to the surrounding and central differentiation (Figure 2) and displayed much higher GAG secretion than the MSC group (Figure 4(A)). The DNA content of the buffy coat group as well as the ECM ratio of the artificial cartilage was close to that of normal cartilage (Figure 4(B)).
The MSC concentration in the buffy coat extracted from the bone marrow of the rabbit from Ficoll was 2.34 × 104 MNCs [4] , while the MSC concentration in the buffy coat extracted from human bone marrow was 1 × 103 - 105 MNCs [8] . However, the cell differentiation efficiency of the buffy coat group was higher, probably because the cells were not sub-cultured but were relatively healthy and energetic. Cytokines and hormones are important factors in the differentiation of MSCs into chondrocytes, but the growth factors currently used cannot be used clinically, presenting the largest obstacle to the clinical use of MSCs for cell therapy and artificial tissue culture. The buffy coat group also improves MSC differentiation efficiency without the use of growth factors, which is a good condition for clinical application. The reason for the high efficiency of buffy coat is unclear; we suspect that the components of the buffy coat―hematopoietic stem cells, lymphocytes, macrophages, and cytokines―play an active role in MSC differentiation of MSC, but additional in-depth research is needed to further explore this hypothesis.
In this experiment, buffy coat extraction was related to cartilage formation when the red blood cell (RBC) content was high as previously described by Horn et al. [9] . RBC hinders MSC adhesion and proliferation. Bone marrow concentrate (BMC) is currently extracted by the centrifugation of bone marrow and widely used in the clinical setting. BMC contains MSCs, including the various differentiation stages of many bioactive molecules and cells such as lymphocytes, neutrophils, monocytes, and platelets. Some researchers believe that BMC is the product of platelet-rich plasma (PRP) plus MSCs when, in fact, BMC and PRP contain different cytokines and growth factors. In an orthopedic study, there is little cytological analysis of BMC. Another study showed that the BMC contains many platelets, monocytes, and CD34-positive cells. Cassano et al. [10] demonstrated that BMC is rich in interleukin-1 receptor antagonist, a receptor antagonist of natural interleukin-1, which can be used to treat autoimmune diseases such as rheumatoid arthritis. This is a meaningful finding that can be used clinically. The authors think that the buffy coat is BMC that lacks RBC; that is, there is no obstruction of MSC adhesion and RBC proliferation, and the buffy coat can easily induce chondrogenic differentiation and produce tissue engineered cartilage. However, because of the toxicity of Ficoll medium, the buffy coat extracted by the Ficoll gradient centrifugation method cannot be used clinically. It is necessary to develop a method by which the buffy coat can be harvested without using the Ficoll gradient centrifugation method and then applied clinically.
5. Conclusion
The ultimate goal of tissue engineering is its clinical application, and the ability to resolve MSC differentiation without the application of interventional conditions is a key factor in achieving such clinical goals. The buffy coat addresses this problem and can become the best source of cells for tissue engineering.
Acknowledgements
This study was supported by the grant of the National Natural Science Foundation of China (81560353); China Postdoctoral Science Foundation (2016M601394); Jilin Postdoctoral Science Foundation, China; The Science and Technology Research Project of the Department of Education, Jilin, China.
Conflicts of Interest Statement
The authors who have contributed certify that they have NO affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical Approval Statement
Healthy 16-week-old New Zealand white rabbits were purchased from the experimental animal center of Yanbian University. All of the rabbits were housed during the experimental study under 12:12 hour light/dark conditions at approximately 27˚C. The experimental procedure was approved by the Institutional Animal Care and Use Committee of Yanbian University Hospital (2015036).
Cite this paper
Jin, L.H., Zhang, Z.Y., Liu, Y.Q., Gurung, K., Jin, Y.J., He, Y.M., Jin, Y. and Jin, Z.H. (2018) Comparison of Chondrogenic Ability between Mesenchymal Stem Cells and Buffy Coat in Vitro. Journal of Biosciences and Medicines, 6, 86-94. https://doi.org/10.4236/jbm.2018.63006
References
- 1. Tallheden, T., Bengtsson, C., Brantsing, C., Sjogren-Jansson, E., Carlsson, L., Peterson, L., et al. (2005) Proliferation and Differentiation Potential of Chondrocytes from Osteoarthritic Patients. Arthritis Research & Therapy, 7, R560-R568. https://doi.org/10.1186/ar1709
- 2. Matsumoto, T., Okabe, T., Ikawa, T., Iida, T., Yasuda, H., Nakamura, H., et al. (2010) Articular Cartilage Repair with Autologous Bone Marrow Mesenchymal Cells. Journal of Cell Physiology, 225, 291-295. https://doi.org/10.1002/jcp.22223
- 3. Azizi, S.A., Stokes, D., Augelli, B.J., Di Girolamo, C. and Prockop, D.J. (1998) Engraftment and Migration of Human Bone Marrow Stromal Cells Implanted in the Brains of Albino Rats—Similarities to Astrocyte Grafts. Proceedings of National Academy of Sciences USA, 95, 3908-3913. https://doi.org/10.1073/pnas.95.7.3908
- 4. Jin, L.H., Choi, B.H., Kim, Y.J., Park, S.R., Jin, C.Z. and Min, B.H. (2011) Implantation of Bone Marrow-Derived Buffy Coat Can Supplement Bone Marrow Stimulation for Articular Cartilage Repair. Osteoarthritis Cartilage, 19, 1440-1448. https://doi.org/10.1016/j.joca.2011.07.012
- 5. Lee, H.J., Choi, B.H., Min, B.H. and Park, S.R. (2007) Low-Intensity Ultrasound Inhibits Apoptosis and Enhances Viability of Human Mesenchymal Stem Cells in Three-Dimensional Alginate Culture during Chondrogenic Differentiation. Tissue Engineering, 13, 1049-1057. https://doi.org/10.1089/ten.2006.0346
- 6. Grogan, S.P., Barbero, A., Winkelmann, V., Rieser, F., Fitzsimmons, J.S., O’Driscoll, S., et al. (2006) Visual Histological Grading System for the Evaluation of in Vitro-Generated Neocartilage. Tissue Engineering, 12, 2141-2149.
- 7. Jin, C.Z., Park, S.R., Choi, B.H., Lee, K.Y., Kang, C.K. and Min, B.H. (2007) Human Amniotic Membrane as a Delivery Matrix for Articular Cartilage Repair. Tissue Engineering, 13, 693-702. https://doi.org/10.1089/ten.2006.0184
- 8. Zhang, Z., Teoh, S.-H., Choolani, M. and Chan, J. (2010) Development of Human Fetal Mesenchymal Stem Cell Mediated Tissue Engineering Bone Grafts Tissue Engineering.
- 9. Horn, P., Bork, S. and Wagner, W. (2011) Standardized Isolation of Human Mesenchymal Stromal Cells with Red Blood Cell Lysis. Methods in Molecular Biology, 698, 23-35.https://doi.org/10.1007/978-1-60761-999-4_3
- 10. Cassano, J.M., Kennedy, J.G., Ross, K.A., Fraser, E.J., Goodale, M.B. and Fortier, L.A. (2016) Bone Marrow Concentrate and Platelet-Rich Plasma Differ in Cell Distribution and Interleukin 1 Receptor Antagonist Protein Concentration. Knee Surgery, Sports Traumatology, Arthroscopy, 26, 333-342. https://doi.org/10.1007/s00167-016-3981-9
NOTES
*These authors contributed equally to this work.