Modern Plastic Surgery
Vol.3 No.3(2013), Article ID:33959,4 pages DOI:10.4236/mps.2013.33021
Use of the LigaSureTM in Cervicofacial Venous Malformation Excision
![]()
Department of Plastic and Reconstructive Surgery, Keio University School of Medicine, Tokyo, Japan.
Email: *kkishi@a7.keio.jp
Copyright © 2013 Eri Konno et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received April 11th, 2013; revised May 12th, 2013; accepted May 19th, 2013
Keywords: Venous Malformations (VMs); LigaSureTM; Vessel Sealing System; Postoperative Bleeding
ABSTRACT
Complete surgical excision of venous malformations (VMs) is frequently complicated by massive bleeding and nerve damage, particularly in the cervicofacial area. This report describes a novel technique for the resection of cervicofacial VMs using the LigaSureTM (Valleylab, Tyco International Healthcare, Boulder, CO) vessel sealing system. We report three cases of VM excision using this vessel sealing system, and recorded intraoperative and postoperative blood loss, operative time, and postoperative complications. In all cases employing the LigasureTM, perioperative blood loss was less than 600 ml, no patients developed postoperative hematomas or other bleeding complications, and no patients developed a facial palsy even if the VM was located close to the facial nerve. LigaSureTM provides excellent hemostasis with few complications when used for cervicofacial VMs.
1. Introduction
Venous malformations (VMs) are low flow vascular malformations. They can present as single or multiple lesions in any part of the body. They never regress and may expand over a patient’s life as venous pressure increases. Symptoms are related to the size and location of the lesion and can include discomfort, pain, neuropathy, ulceration, bleeding, and a direct or indirect mass effect. The treatment of VMs is surgical resection, sclerotherapy, embolisation, laser therapy, or a combination of these modalities [1-6]. Complete surgical excision can cure the disease, but may not be possible in all cases, secondary to concerns for massive bleeding or nerve damage, especially when they are located in the face and neck [1,3]. Hemorrhage and the possibility of nerve damage frequently preclude an extensive resection. This report describes the use of the LigaSureTM device (Valleylab, Tyco International Healthcare, Boulder, CO) for VM excision and its efficacy in controlling perioperative bleeding and avoiding damage to the facial nerve or other adjacent organs. We used this vessel sealing system in three cases, and blood loss, operating time, postoperative complications were recorded.
2. Patients and Methods
We used a LigaSureTM in three patients with VMs (two male, one female). The patient age ranged from 18, 52, and 67 years old. VM sites included the cheek and submandibula. Sizes ranged from 6 × 4, 10 × 7, and 15 × 8 cm. We recorded the periand postoperative blood loss, the need for blood transfusion, size of the resected tumor, operative time, and postoperative complications in all three patients.
Operative Technique
After incising the skin overlying the VM with a surgical knife and electrocautery, the tumors were grasped with Kocher clamps. The LigaSureTM was then used for the majority of coagulation, sealing, and cutting. Around the facial nerve, a nerve stimulator was used to confirm that the facial nerve did not cross the sealing area. Care was taken to overlap the newly sealed areas with those already sealed in order to minimize areas that would bleed in the postoperative period (Figure 1). Following tumor resection, the subdermal space was re-approximated with absorbable suture. Fluid collections were prevented by placing a drain beneath the dermis.
3. Results
In all cases using the LigaSureTM, perioperative blood loss was less than 600 ml (Table 1). No patients required blood transfusions (auto-transfusion was done in two cases). Drains were placed in all patients (open drainage: 2 cases, closed drainage: 1 case). We chose open drainage if the excision defect was small and closed drainage for large defects. No patients developed postoperative hematomas or other bleeding complications nor did any patient suffer nerve damage.
4. Case Reports
Patient 1
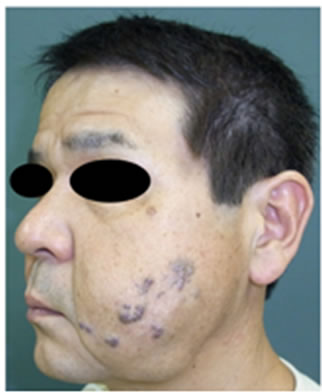
The first patient is a 52-year-old male who had VMs on his left cheek to the level of his mandible (Figure 2(a)). We planned excision of an area measuring 15 × 18 cm including the skin lesion. As MRI image indicate that the masseter muscle was involved with the VMs (Figure 2(b)), VMs were partially removed. We used the LS1200 handpiece (LigaSure PreciseTM disposable handpiece) (Figure 2(c)). The thermal spread was less than 1 mm beyond the seal site, and thus this handpiece was ideal for this case and others with VMs close to the facial nerve or muscle. Because this handpiece could not cut this particular VM after sealing, we used scissors to divide the sealed area (Figures 2(d) and (e)). After resection, bleeding was less with facial nerve intact (Figure 2(f)). An open drain was placed prior to closing. Total perioperative blood loss was 425 ml, and operative time was 4 hours. On postoperative day 2 the open drain was removed, and the patient was discharged without complications. After 8 months, the scar was inconspicuous with high patient satisfaction in regards to cosmesis and no evidence of nerve damage (Figure 2(g)).
5. Discussion
Although the treatment of VMs frequently involves surgical excision as part of the curative plan, complete surgical eradication of extensive cervicofacial VMs is rarely possible without jeopardizing nerve function or risking disfigurement. These surgeries can also be fraught with recalcitrant intraoperative hemorrhage that can prolong surgical time and lead to failed resection [1,3,4].
Several non-operative or preoperative options have been developed to prevent perioperative hemorrhage. Options include percutaneous and transcatheter embolisation, sclerotherapy, laser therapy, use of electrocautery (monopolar, bipolar, or ultrasonic coagulators), use of topical hemostatic agents (thrombin, Gelfoam, etc.), and in some cases, packing with pressure or ligation of the surrounded tissue. Recently, the use of recombinant factor VIIa during excision has also been reported [1].
Embolisation is recommended immediately prior to surgical intervention. But the precise vessel to embolize cannot always be identified. Additionally, embolisation with glue can result in various complications, such as skin necrosis, non-targeted embolisation, and ischemia of the adjacent normal tissue [4,5].
Sclerotherapy with various agents is effective for small or medium-sized lesions. It has a complication rate of 10% - 15%, with complications including facial nerve palsy, bleeding, skin ulceration, hemoglobinuria, transient pulmonary hypertension, and deep vein thrombosis. [2-5].
Laser therapy is costly and mostly inadequate for all except the thinnest lesions [6].
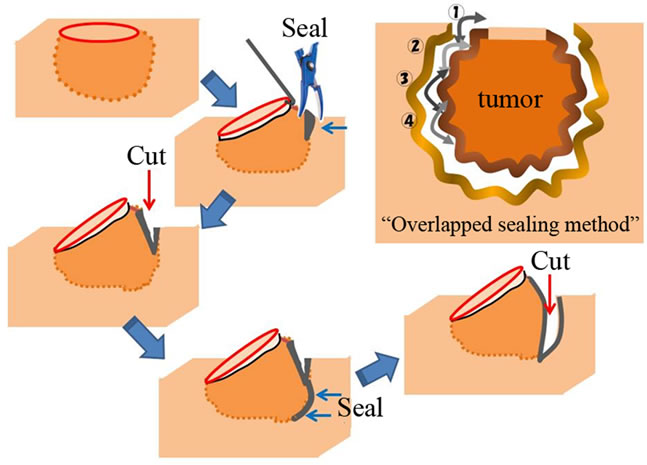
Figure 1. Schematic views of “overlapped sealing method” using LigaSureTM. After sealing the venous malformation with LigaSureTM, the mid-line of the sealed areas are cut with scissors. The next area of sealing and cutting proceeds from the previously sealed area in an overlapping manner.
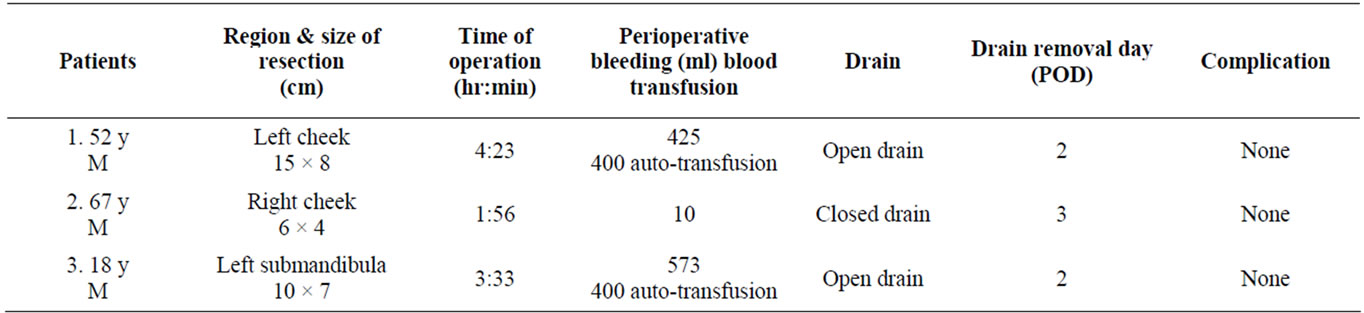
Table 1. Background and outcome of patients treated in the present study.
 (a)
(a)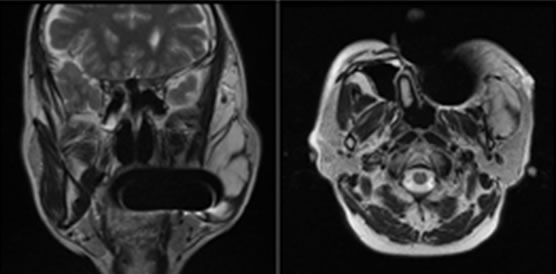 (b)
(b) (c)
(c)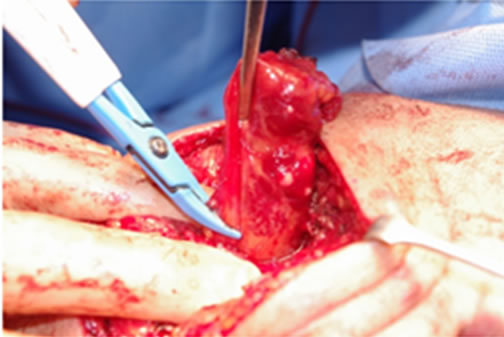 (d)
(d)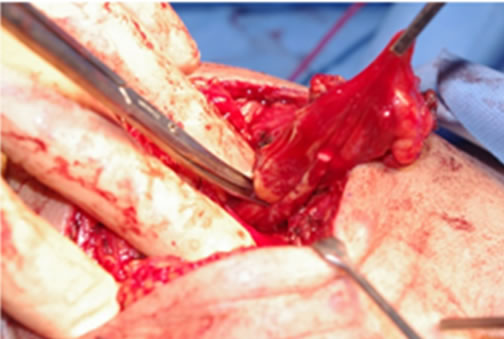 (e)
(e)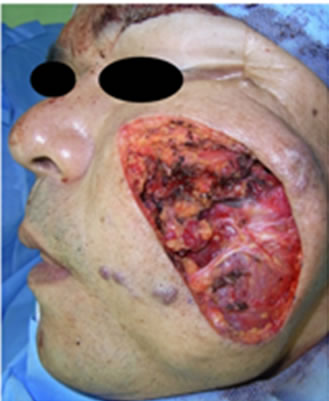 (f)
(f)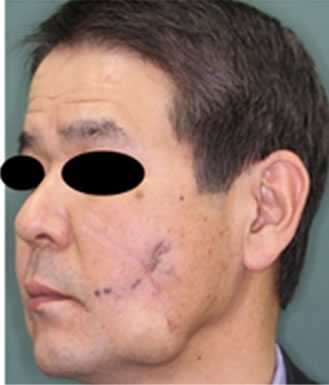 (g)
(g)
Figure 2. A 52-year-old male with VMs on the left cheek (a). Axial and coronal view of the MRI shows that the masseter muscle was involved with the VMs (b). Using the LigaSureTM (c), elevation of the edge of the VM with minimal undermining. VMs were sealed (d) and cut (e). The cut edges are effectively sealed, and safely dissected from the facial nerve (f). The scar using this technique is inconspicuous 8 months after operation (g).
LigaSureTM is a vessel sealing system that uses an enhanced form of bipolar current to achieve permanent vessel wall fusion by denaturing the collagen and elastin in vessel walls and reforming them into a hemostatic seal [7]. LigaSureTM measures the appropriate voltage and current automatically for the target vessels, and can seal vessels with surrounding soft tissue ranging in thickness from 1 to 7 mm [7-9].
Peterson et al. demonstrated the effectiveness of the seals created by the LigaSureTM system. The seal quality, as reflected in burst strength, was determined to be comparable to existing mechanical-based techniques such as clips and sutures. Mean burst pressures in excess of 900 mmHg were obtained for vessels of 3 to 7 mm diameter, comparable to results obtained using surgical clips and suture on vessels in the same size range [7]. Moreover, thermal spread was measured histologically and noted to extend on average less than 2 mm beyond the seal site.
We previously used the LigaSureTM system in the resection of NF-1 tumors (write out full name of tumor), and successfully reduced perioperative bleeding [10]. There are currently no reports describing the use of a LigaSureTM for the resection of VMs or other vascular anomalies. As indicated in the present report, using LigaSureTM for resection during VM excision achieves excellent hemostasis and safe undermining near the facial nerve with few complications in the cervicofacial area.
REFERENCES
- L. M. Buckmiller, G. T. Richter, M. Waner and J. Y. Suen, “Use of Recombinant Factor VIIa during Excision of Vascular Anomalies,” Laryngoscope, Vol. 117, No. 4, 2007, pp. 604-609. doi:10.1097/MLG.0b013e318030450e
- C. A. James, L. E. Braswell, L. B. Wright, et al., “Preoperative Sclerotherapy of Facial Venous Malformations: Impact on Surgical Parameters and Long-Term FollowUp,” Journal of Vascular and Interventional Radiology, Vol. 22, No. 7, 2011, pp. 953-960. doi:10.1016/j.jvir.2011.03.004
- L. Su, X. Fan, L. Zheng and J. Zheng, “Absolute Ethanol Sclerotherapy for Venous Malformations in the Face and Neck,” Journal of Oral and Maxillofacial Surgery, Vol. 68, No. 7, 2010, pp. 1622-1627. doi:10.1016/j.joms.2009.07.094
- S. Hoque and B. K. Das, “Treatment of Venous Malformations with Ethanolamine Oleate: A Descriptive Study of 83 Cases,” Pediatric Surgery International, Vol. 27, No. 5, 2011, pp. 527-531. doi:10.1007/s00383-010-2824-x
- B. E. Cil, I. Vargel, S. Geyik, B. Peynircioglu and T. Cavusoglu, “Venous Vascular Malformations of the Craniofacial Region: Pre-Operative Embolisation with Direct Percutaneous Puncture and N-Butyl Cyanoacrylate,” British Journal of Radiology, Vol. 81, No. 972, 2008, pp. 935-939. doi:10.1259/bjr/66893325
- K. Scherer and M. Waner, “Nd:YAG Lasers (1,064 nm) in the Treatment of Venous Malformations of the Face and Neck: Challenges and Benefits,” Lasers in Medical Science, Vol. 22, No. 2, 2007, pp. 119-126. doi:10.1007/s10103-007-0443-0
- S. L. Peterson, P. L. Stranahan, D. Schmaltz, C. Mihaichuk and N. Cosgriff, “Comparison of Healing Process Following Ligation with Sutures and Bipolar Vessel Sealing,” Surgical Technology International, Vol. 10, 2002, pp. 55-60.
- G. R. Lamberton, R. S. Hsi, D. H. Jin, T. U. Lindler, F. C. Jellison and D. D. Baldwin, “Prospective Comparison of Four Laparoscopic Vessel Ligation Devices,” Journal of Endourology, Vol. 22, No. 10, 2008, pp. 2307-2312. doi:10.1089/end.2008.9715
- F. J. Kim, M. F. Chammas Jr., E. Gewehr, et al., “Temperature Safety Profile of Laparoscopic Devices: Harmonic ACE (ACE), Ligasure V (LV), and Plasma Trisector (PT),” Surgical Endoscopy, Vol. 22, No. 6, 2008, pp. 1464-1469. doi:10.1007/s00464-007-9650-9
- E. Konno and K. Kishi, “Use of the LigaSure (TM) Vessel Sealing System in Neurofibroma Excision to Control Postoperative Bleeding,” Journal of Plastic, Reconstructive & Aesthetic Surgery, Vol. 65, No. 6, 2012, pp. 814- 817.
NOTES
*Corresponding author.

