Open Journal of Obstetrics and Gynecology
Vol.3 No.8(2013), Article ID:38788,3 pages DOI:10.4236/ojog.2013.38113
Salmonella typhimurium—A threat for a newborn
![]()
Department of Intensive Care and Congenital Malformations of the Neonates and Infants, Polish Mother’s Memorial Hospital Research Institute, Lodz, Poland
Email: *imaroszynska@wp.pl
Copyright © 2013 Iwona Maroszyńska et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received 6 August 2013; revised 5 September 2013; accepted 13 September 2013
Keywords: Salmonella; Typhimurium; Newborn; Infections
ABSTRACT
The most common clinical manifestation of gram negative Salmonella typhimurium infection is gastroenteritis. In Poland extraintestinal infections occur in about 0.4% of cases of reported salmonellosis. Poor personal hygiene practices of parents are the most common cause of Salmonella spp. infections in the neonatal period. The neonatal salmonellosis is significantly more frequently reported in endemic areas such as Central Africa or India, than in Poland. The authors present a case of a newborn with Salmonella typhimurium sepsis, who was treated in the Department of Intensive Care and Congenital Malformations of the Neonates and Infants, Polish Mother’s Memorial Research Institute.
1. INTRODUCTION
Salmonella spp. is the most frequent cause of bacterial food-borne infectious diseases. However, recently there is an increasing incidence of infections resulting from the direct contact with carriers and animals. Salmonella bacilli and Campylobacter are the bacterial pathogens that are most often cultured in stool of patients with severe gastroenteritis or diarrhea [1].
Salmonella spp. infections are characterized by vast spectrum of clinical symptoms, from gastroenterocolitis to sepsis or meningitis. The course of the infection depends on virulence of bacteria and immune response of the host. After the infection, carrier-state may persist for several months [3].
2. CASE REPORT
The authors present a male neonate born at 40 weeks of gestation by spontaneous vaginal delivery in good general condition with the birth weight of 3380 g. His Apgar score was 10. The boy was discharged from the primary neonatal care division on the 5 day of life. He was fed with breast milk and with formula. He did not vomit and his stools were normal. On the 8 day of life his parents noticed that the boy was less active and did not wake up for feeding. They sought advice of the primary care physician who decided to refer the child to the local hospital urgently. In the emergency room the neonate presented symptoms of shock with deep metabolic acidosos. Suspicion of sepsis was made and the boy was transferred to the Department of Intensive Care and Congenital Malformations of the Neonates and Infants of Polish Mother’s Memorial Hospital Research Institute.
At the admission the neonate’s condition was severe. The clinical examination revealed signs of significant dehydration, tachypnoe, increased muscle tone, reduced reaction to pain, numerous erosions in the oral cavity and papular skin lesions on the trunk and limbs.
The laboratory abnormalities included signs of metabolic acidosis (pH 7.14, Base Excess-19 mmol/L, lactic acid 40 mg/dL; normal value: 6.3 - 18.9 mg/dL), increased biochemical markers of infection (C-reactive protein 3.47 mg/dL; normal value: < 1.0 mg/dL) and signs of acute prerenal renal failure (creatinine 2.6 mg/dL; normal value: 0.3 - 0.7 mg/dL, urea 80 mg/dL; normal value: 4.3 - 34.2 mg/dL). After 48 hours Salmonella typhimurium was cultured in the stool and blood, but cerebrospinal fluid culture was negative. The result was verified by the Regional Sanitary and Epidemiological Station and members of the family were tested for carrier state.
On the first day of hospitalization intensive intravenous hydration was performed. Broad spectrum antibiotic therapy and total parenteral nutrition were administered. Based on the result of the antibiogram, antibiotic therapy was adjusted and it was continued for 21 days. Antibiotic therapy was ended after obtaining negative blood and stool culture and after normalization of biochemical signs of infection. Parenteral nutrition was continued for 9 days.
As a result of the treatment applied, the infant’s condition improved. The neurological symptoms (increased muscle tone, disturbances concerning social interactions) were the clinical symptoms observed for the longest period of time. The boy was referred to the outpatient neurodevelopmental care facility. Now his development is normal.
3. DISCUSSION
Over 2500 serotypes of Salmonella spp. identified to date belong to the two species: Salmonella enterica and Salmonella bongori [1]. Before introducing modern genetic diagnostic methods, many serotypes were thought to be separate species. The old names-typhi, paratyphi, typhimurium, enteritidis and choleraesuis, are still being used in case of the serotypes that are most frequently diagnosed in humans [2]. In this paper the authors present the neonate with the severe Salmonella typhimurium infection.
Today the estimated incidence of Salmonella spp. infections in the developed countries, where the most often source of infection is contaminated food, is 500/100,000 with only 12 - 14/100,000 confirmed with laboratory tests. 20% of infected patients require hospitalization and 0.6 percent dies. The highest mortality rates can be observed among neonates, infants and immunocompromised patients. The estimated mortality associated with Salmonella spp. infections in people infected with HIV in Africa is 60% [3-5]. People younger than 20 and older than 70 years of age are the most prone to Salmonella spp. infections. The highest incidence of this type of infection is observed in the first years of life. 25% to 30% of pediatric patients are infants. Factors that make patients susceptible to Salmonella spp. infections are: reduced immunity, antibiotic therapy, malnutrition, neoplasms and reduced gastric acidity [6].
Although more than 90% of infections are caused by consumption of infected food (food-borne infections), there is an increasing incidence of infections resulting from the direct contact with carriers and animals recently [7].
According to the data of The Polish National Institute of Hygiene, in 2012 in Poland 8451 cases of salmonellosis were reported (morbidity 21.94/100,000) with only 185 cases of extraintestinal infections (Table 1). Sepsis was diagnosed in 85 patients and there was only one neonate in this group.
In the developed countries the incidence of neonatal infections caused by Salmonella spp. is low and such infections are usually caused by inappropriate food stor-
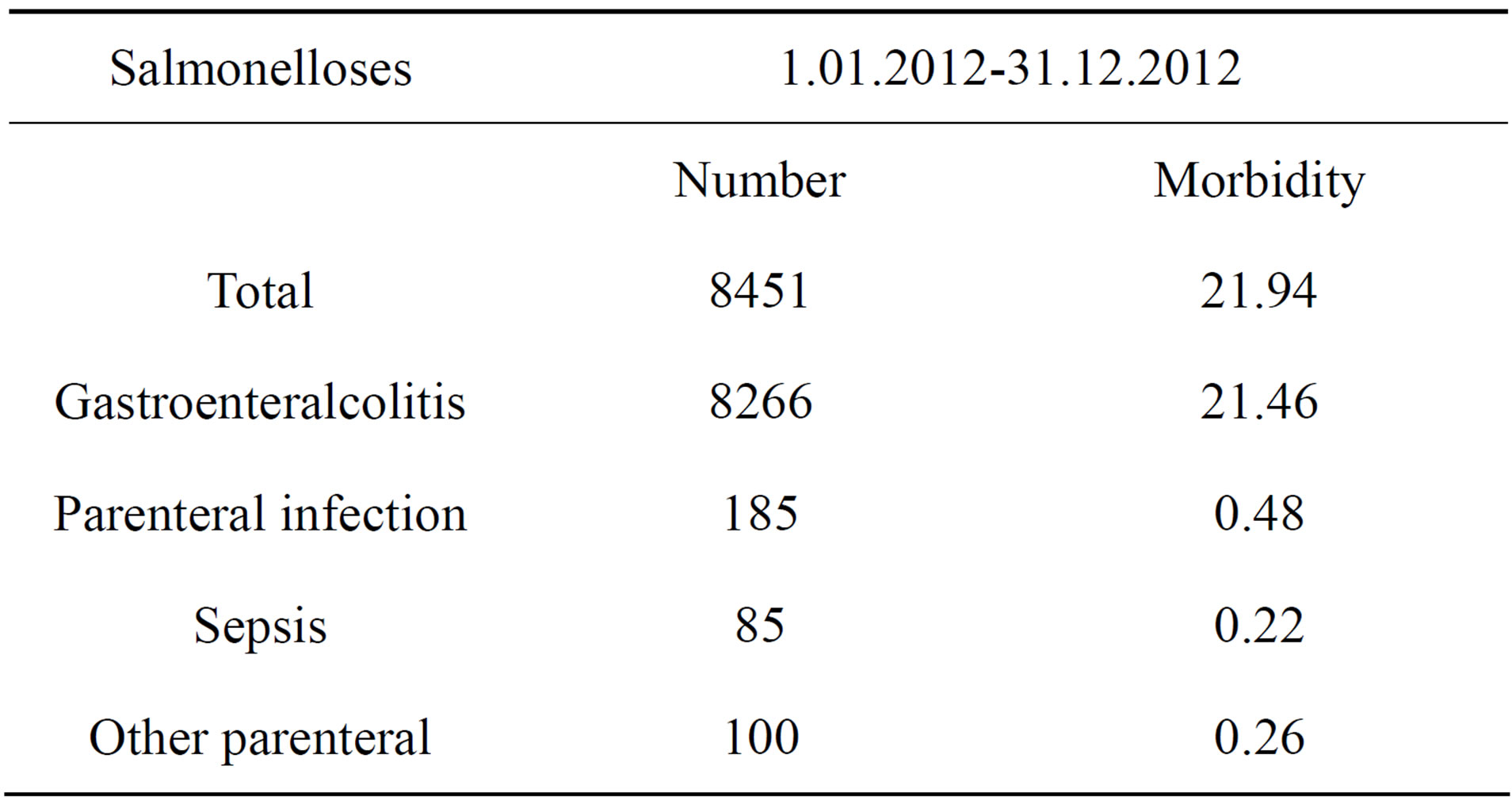
Table 1. Salmonelloses in Poland in 2012 r.—according to data of The Polish National Institute of Hygiene.
age, poor hygienic practices (improper hand washing) especially, when there are people with gastroenterocolitis or carriers in the child’s environment [8]. Rarely Salmonella spp. infection in perinatal period develops by an ascending route in pregnant women with symptoms of gastroenterocolitis. In such cases neonates develop congenital infection, often presenting as sepsis or meningitis [9,10].
Clinical symptoms of neonatal Salmonella spp. infections are not specific and do not differ from infections caused by other Gram negative bacilli. The most common are: fever, rash, leucopenia, splenomegaly and abdominal distention. In the literature there are reports of hypotonia, anorexia, dehydration, hypersensitivity, jaundice, diarrhea, respiratory distress, hypothermia, seizures, pneumonia, meningitis, cerebral abscesses, cholecystitis. Salmonella spp. bacilli are sensitive to the third-generation cephalosporins, carbapenems, ampicillin, quinolones and co-trimoxazole [11].
In the presented case of the neonate the course of the infection was typical for Salmonella spp. infections described in the literature and included: diarrhea, anorexia, fever and dehydration. The diarrhea and anorexia probably caused dehydration, shock and prerenal renal failure. The neurological disorders observed, included: increased muscle tone, reduced pain reaction and disturbances of social interactions. Despite the evident neurological symptoms, the culture of the cerebrospinal fluid was negative and the result of its analysis was normal (cytosis 6, protein 75.0 mg/dL, glucose 49 mg/dL).
Neither splenomegaly, nor leukopenia (white blood cell count was 11.84 × 103/mm3) was observed in this case.
The presented neonate comes from the rural environment of low economic status. Laboratory tests performed by the Regional Sanitary and Epidemiological Station excluded carrier-state of the mother and other members of the family. The mother denied symptoms of gastroenterocolitis during pregnancy as well. So the most probable route of infection was poor hygienic practices of the caregivers that could result in transmission of bacteria from animals in the farm. However, regarding the onset of symptoms (8 day of life), a congenital infection is also probable.
4. CONCLUSION
The presented case shows that Salmonella spp. infections should be taken into consideration in neonates, especially from rural environment with non specific clinical symptoms including reduced activity or refusal to feed. It should be remembered as well, that children of mothers with gastroenterocolitis in the perinatal period may develop Salmonella spp. congenital infection. Loose stools in a neonate in the first days of life, particularly in babies fed with breast milk, may stay unnoticed, which may delay a medical intervention. In case of Salmonella bacilli infection this may result in development of a severe infection, shock, serious complications after meningitis or/and death [8].
REFERENCES
- Martin, R.J., Fanaroff, A.A. and Walsh, M.C. (2010) Neonatal-perinatal medicine. 9th Edition, Elsevier, Amsterdam.
- Popoff, M.Y. and Le Minor, L. (1997) Antigenic formulas of the Salmonella serovars. WHO collaborating centre for reference and research on Salmonella. Institute Pasteur, Paris.
- Preziosi, M.J., Kandel, S.M., Guiney, D.G. and Browne, S.H. (2012) Microbiological analysis of nontyphoidal Salmonella strains causing distinct syndromes of bacteremia or enteritis in HIV/AIDS patients in San Diego, Clinical Microbiology, 11, 3598-3603.
- De Jong, W.H., De Jonge, R., Garssen, J., Takumi, K. and Havelaar, A.H. (2005) Salmonella, assessment of infection risk. In: Vohr, H.-W., Ed., Encyclopedic Reference of Immunotoxicology, Springer, Berlin, 573-578.
- Sauteur, P.M., Relly, C., Hug, M., Wittenbrink, M.M. and Berger, C. (2013) Risk factors for invasive reptile-as-sociated salmonellosis in children. Vector-Borne and Zoonotic Diseases, 6, 419-421.
- Sadkowska-Todys, M., Furman, S. and Czarkowski, M. (2012) Salmonelozy w Polsce w 2010 roku. Przegląd Epidemiologiczny, 66, 259-266.
- Rennels, M.B. and Levine, M.M. (1986) Classical bacterial diarrhea: Perspectives and update: Salmonella, Shigella, Escherichia coli, aeromonas and plesiomonas. Journal of Pediatric Infectious Diseases, 5, 91.
- Cooke, F.J., Ginwalla, S., Hampton, M.D., Wein, J., et al. (2009) Report of neonatal meningitis due to Salmonella enterica serotype agona and review of breast milk-associated neonatal Salmonella infections. Journal of Clinical Microbiology, 9, 3045-3049.
- Pessao-Silva, C.L., Toscano, C.M., Moreira, B.M., Santos, A.L., Frota, A.C., Solari, C.A. et al. (2002) Infection due to extended spectrum beta-lactamase-producing Salmonella enterica subsp. enterica serotype infants in a neonatal unit. Journal of Pediatrics, 141, 381-387.
- Singh, A., Wilkins, T. and Schade, R.R. (2011) Salmonella newport bacteremia in a 12-day-old infant. The Journal of American Board of Family Medicine, 24, 214-217.
- Dzierżanowska, D., (2000) Antybiotykoterapia praktyczna, Medica Press, Bielsko Biała.
NOTES
*Corresponding author.

