Open Journal of Ecology
Vol.3 No.6(2013), Article ID:38380,8 pages DOI:10.4236/oje.2013.36048
Floss release, seed fall and germination of kapok seeds (Ceiba pentandra) in an urban environment
![]()
1Department of Wildlife Management, Faculty of Forestry and Nature Conservation, Sokoine University of Agriculture, Morogoro, Tanzania; *Corresponding Author: al.rija10@gmail.com
2Range Management Section, Department of Animal Science and Production, Sokoine University of Agriculture, Morogoro, Tanzania
3Department of Fish and Wildlife Conservation, Virginia Polytechnic and State University, Blacksburg, USA
Copyright © 2013 Alfan A. Rija et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received 20 September 2013; revised 18 October 2013; accepted 22 October 2013
Keywords: Ceiba Pentandra; Floss Release and Seed Fall; Human Impacts; Plant Recruitment; Urban Environments
ABSTRACT
The tropical forest kapok tree occurs widely in urban environments, dispersing thousands of floss during the fruiting season. However, its seedlings are rarely seen around human settlements, raising questions on what factors might be limiting its recruitment. We examined the pattern of floss release, seed fall post-dispersal and influences of watering frequency on germination of Ceiba pentandra seeds. Evening, overall, was associated with a significantly higher rate and quantity of floss release (p = 0.0001) than were mid-day and morning, correlating well with the speed of wind across the day (rh = 0.666, p = 0.001). Mean dispersal quantity differed significantly between the individual sampled trees, in which the highest dispersal was recorded from trees located in relatively open areas and the lowest was recorded in closed stand trees. Seed fall was higher within 100 m distances than seed fall farther away from dispersing trees, depending on the microsites and weather conditions. Germination started on day 5 onwards and only 15.67% of sown seeds germinated. Watering frequency strongly influenced seed germination while local human activities determined fate of dispersed seeds. We conclude that recruitment of C. pentandra species in the study area may have been limited by environmental and human factors operating at various local and spatial scales.
1. INTRODUCTION
The silk-cotton tree (also Kapok) C. pentandra (Bombacaceae) is a fast-growing and emergent tropical forest tree species that can grow up to 60 m height [1]. On the continent of Africa, this species occurs naturally in West Africa and has expanded its range in other African regions, largely due to introduction by humans. Several studies show that C. pentandra pollinates through a range of mechanisms that vary across local and spatial scales. For example, in West Africa [2], some Pacific islands [3,4], Costa Rica [5] and Amazonia [1], bats have been documented to pollinate the silk-cotton tree. Further, self-fertilization of C. pentandra has also been widely documented in West Africa [6], Samoan Islands [4] Barro Colorado Island, Panama [7] and Amazonia [1]. A considerable number of studies also document that the silk-cotton tree in some other areas is pollinated by hummingbirds [1,8] and non-flying primate mammals, particularly Saimiri sciureus, Cebus apella, and Ateles paniscus [9]. Despite having a range of reproductive strategies, recruitment in this species appears to be limited by several factors. For example, Gribel et al. [1] and Murawski and Hamrick [7] observed that trees’ originality (whether wild or cultivated), seasonal and local conditions within which a tree grows, as well as the mating system influence its flowering and fruiting phenology and potentially population recruitment. Local and spatial patterns of seed dispersal have generally been reported to have a greater influence on tree population performance. For example, Nathan and Muller-Landau [10] reviews that distance traversed by seeds after dispersal and seed density have a great impact on tree recruitment in plant populations. The local conditions caused by both biotic and abiotic factors have consistently been observed to have greater influence on seed dispersal patterns. For example, wind-dispersed seeds may end up being deposited on unsuitable substrate such as rough surfaces [11] while animal dispersed seeds often end up being deposited in nesting and roosting sites [12], thus reducing chances for such seeds to germinate. Post-dispersal seed movement and seed predation also explain variation in most seed dispersal patterns and seedling recruitment observed across many plant species [1,13-16]. These factors may interactively limit seed germination and negatively impact plant population performance in the long-term. Besides dispersal mechanisms and seed predation, human activities play a central role in the influence of plant population dynamics in various ways. First, deforestation through extraction of timber has often put C. pentandra under intense pressure across its distribution range [17]. Second, local medicinal use of this species [18] and use for livestock fodder [19] increase pressure on this species.
The urban city of Morogoro has localized populations of C. pentandra and flowering takes place yearly, releasing several hundred thousand seeds on the ground (Rija & Said pers.obs 2012). Despite the fact that Ceiba species have been widely studied in terms of breeding boilogy and socio-economic use, the information on certain ecological aspects such as seed release and germination patterns, especially in urban landscapes, is lacking. Coupled with increasing pressures from human activities in urban areas, this species is threatened with severe population decline and local extinction. Despite the high flowering capacity and fruiting phenology of C. pentandra, it is not clear why its seedlings are rarely seen around urban landscapes, thus raising questions on what factors might be limiting its recruitment. The present study documents on the floss release, seed fall and germination of the C. pentandra in an urban area in order to broaden ecological understanding of this species in human-dominated landscapes. The aim of this study was to understand the seed dispersal patterns as well as ecological factors affecting seed germination and, potentially, plant recruitment of this species in the Morogoro municipality. The objectives were to: 1) understand the patterns of floss release across the day and dispersal of floss from some selected Ceiba trees, 2) test whether or not micro-sites conditions (shade and sunny) on which seeds land have any impact on subsequent seed germination and 3) examine the optimal watering frequency required for germination of C. pentandra seeds in the area as well as local human activities impacting on C. pentandra in the area.
2. MATERIAL AND METHODS
2.1. The Study Area and the Species
The study area is located in Morogoro (37˚39'E, 6˚51'S) on the northern foot of the Uluguru Mountains, Tanzania. The vegetation type comprised of both fruit and non-fruit trees planted for various purposes including research, teaching and shelter grounds [20]. The original vegetation was miombo woodland [21]; however, due to human activities only a few patches of natural vegetation remain near the lower slopes of the Uluguru Mountains. The annual rainfall ranges from 600 mm to 1000 mm with a bimodal pattern characterized by short rains from November to January and long rains during March to May. The mean monthly temperature varies between 21˚C and 27˚C. The urban C. pentandra population is atypical, as only isolated individuals are seen around the city. Within the main campus of Sokoine University of Agriculture (SUA) where this study was done, Ceiba trees are localized and appear to have been introduced a few decades ago by humans (Rija et al. in review). Other tree species found within the study site include planted trees such as Leucaena leucocephala, Ficus species, Senna spp, Melicia spp, Albizia spp, Khaya spp, Azadirachta indica, Mangifera indica, Citrus spp and Persea americana. We conducted this study during the fruiting season from September-October 2012 when C. pentandra releases hundreds of thousands of seed wings, whitening the landscape with scattered flosses. This tree frequently experiences patterns of supra-annual flowering with different individuals flowering during different years [1]. However, within any particular year, the individuals that flower are highly synchronized and do so within a relatively short period of six weeks [1,22,23]. Detailed accounts of the flowering phenology and breeding biology of this species have been well-covered [1]. In the study area, C. pentandra exhibited synchronized flowering that lasted between five and six weeks.
2.2. Sampling of Floss Release and Wind Speed
Field observations were made on purposefully selected study trees (n = 10) within the SUA main campus. These trees were chosen because they were located between 100 and 800 m of each other, which was easier for researchers to walk during the sampling. On each sampling tree, observations of floss dispersal (release bout) were done every five minutes, followed by another five minutes of resting during two hours in the morning (8:00 - 10:00), two hours in the afternoon (12:00 - 2:00) and two hours in the evening (4:00 - 6:00). Sampling was conducted by two people each sampling one tree during any sampling day. Each selected study tree was sampled for two days (12 hrs) and the number of events of fruit burst (release bout) was recorded and seed wings (floss) dispersed were estimated within the five minute intervals. To understand how wind speed influenced the pattern of floss release and dispersal, we recorded wind speed data from a weather station located within 100 m of the study trees. Wind direction was nearly consistent across the sampling period and blew in a south-north direction. No evidence of significant change of wind direction was apparent, as floss dispersed by wind exhibited a nearly linear pattern following a south-north wind direction.
2.3. Sampling Seed Fall and Floss Distribution
To understand the patterns of seed fall across a dispersal gradient from any dispersing tree, we surveyed plots along transects (100 m length) radiating from a base tree northward along the direction of floss movement. Transects were conducted from isolated trees because three trees had overlapping floss distribution, thus they were not considered in the sampling. Seven trees were used as starting points of these transects. On each transect, five systematically placed plots each measuring 4 × 4 m were made with inter-plot distances of 16 m. Within each plot all flosses were recorded, separating seeded and nonseeded floss. This sampling resulted in 37 total sampling plots. Moreover, we collected seeds from bare soil or grass litter under sunny or shady conditions for subsequent examination of whether or not these microsite conditions affect germination of C. pentandra seeds in the area. About 2.5 kg of seeds were also collected in order to examine pattern of post-dispersal seed predations in the area to ascertain whether or not predation limits seed germination and plant recruitment (results not reported here).
2.4. Germination Experiment and Water Treatment
A germination experiment was set up with seeds collected under sunny and shady conditions. In this experiment, we assumed other conditions for germination such as temperature, oxygen and light [24] to be unlimited and thus only controlled for water quantity and frequency of watering, based on prior local information on germination of Ceiba seeds around the study area (Local peoplepers.com. 2013). Seeds were sown in sixty polythene bags (each 5 cm diameter and 10 cm height) filled with soil from the same location and collected within areas where seeds fall after release. The soil, collected at a 5 cm depth, was assumed to have approximate similar conditions in terms of minerals (not measured), as it was collected from a single location within 10 - 20 m from the base of C. pentandra. The seed bags were placed in a shady location by the wall of the zoology laboratory to minimize external environmental conditions such as rainfall. Twenty bags each were sown with fifty seeds collected from shady conditions, and twenty others were sown with fifty seeds from sunny conditions. The last twenty bags were each sown with eighty seeds collected from shady and sunny conditions. The sown polythene bags were randomly subjected to five watering treatments. Water (100 ml) was applied once per day in the first treatment group, which contained a mixture of bags with seeds from shady and sunny conditions. The second group was watered twice per day (morning and evening) with a similar volume of water. The third group received water thrice per day, in the morning, afternoon and evening, while the fourth group was watered only once at the start of experiment and left so until the end of the experiment. The fifth group constituted a control and thus was not given water throughout the experimental period. These treatments were applied on the sown seeds and monitored for any emerging seeds until no emerging seedling was observed. Observations began from the start of the experiment and recorded any seedling emerging from each treatment. A total of 3600 seeds from shady and sunny conditions were sown and monitored for a period of 15 days.
2.5. Data Analysis
Floss release rate (events/time) was computed as a ratio of release events recorded within the five-minute observation time. Release events (bouts) within the sampling intervals were obtained by summing the release events for each individual sampled tree. To understand how wind speed influenced release bouts at any given time and for each sampled tree, we used Spearman’s rank correlation coefficient test (p = 0.01). Further, a Kruskal Wallis test was used to examine the difference of release rates of wings between the sampling times and trees. The pattern of seed fall and floss dispersal was examined by comparing the number of winged and non-winged seeds at a plot level using the student t-test. Post-dispersal floss movement was examined through comparison between winged and non-winged seeds from the sampling plots along the dispersal gradient using the Wilcoxon signed rank test. To compare germination of the seeds collected from shady and sunny conditions, we used the independent t-test after conforming to Levene’s test for equality of variance (F = 0.259, p = 0.625). A general linear model (GLM) was used to examine the interacting effects of watering frequency (treatments) and seed source conditions (shady/sunny) on germination of C. pentandra seeds. Data were tested for homogeneity of variance and transformation was applied where appropriate before analysis under SPSS v16.0.
3. RESULTS
3.1. Patterns of Floss Release and Seed Fall
C. pentandra species exhibited a wide variation in the pattern of release of flosses across the time of the day (Figure 1). The number of seeds released from the C. pentandra fruit was significantly different across the sampling time of the day (Kruskal-Wallis χ2 = 899.43; DF = 2; p = 0.0001, Table 1). Evening had a higher median dispersal quantity than afternoon or morning (χ2 = 723.47, median = 1.82, p = 0.0001). Dispersal rates (number of floss per unit time) were significantly different between individual trees (Kruskal-Wallis χ2 = 374.44; DF = 9; p = 0.0001) as well as sampling time (KruskalWallis χ2 = 899.44; DF = 2; p = 0.0001). Overall, evening had higher rates of seed dispersal from C. pentandra fruit, correlating well with the speed of wind across the day (rh = 0.666, p = 0.001). Further, the mean of seed dispersal bouts was 7.85 ± 5.82 events. Dispersal events recorded within the five minute intervals were different between the sampling periods (Kruskal-Wallis χ2 = 120.78; DF = 2; p = 0.0001), being higher in the evening than in the afternoon or morning. Similarly a significant difference in dispersal events was evident between individual sampled trees (Kruskal-Wallis χ2 = 82.38; DF = 9; p = 0.0001). The pattern of seed fall during post-dispersal floss movement was variable across distances from the seeddispersing tree. On plots closer (within 40 m) to the dispersing trees, the numbers of flosses with and without
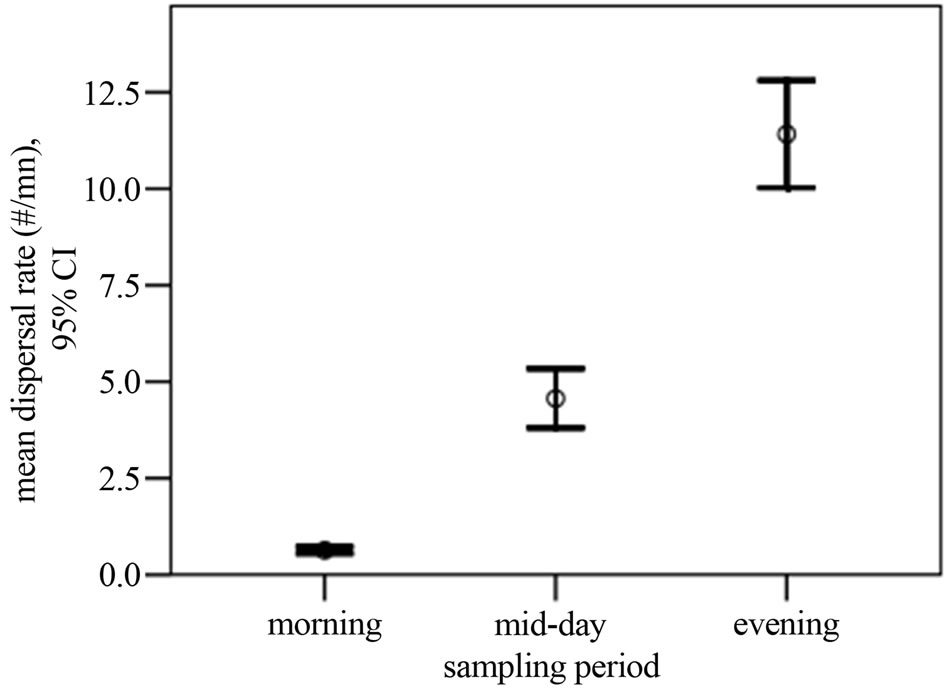
Figure 1. Mean dispersal rate for the Kapok species across the sampling time in the study area showing significant differences between different times of the day (see text for statistics).
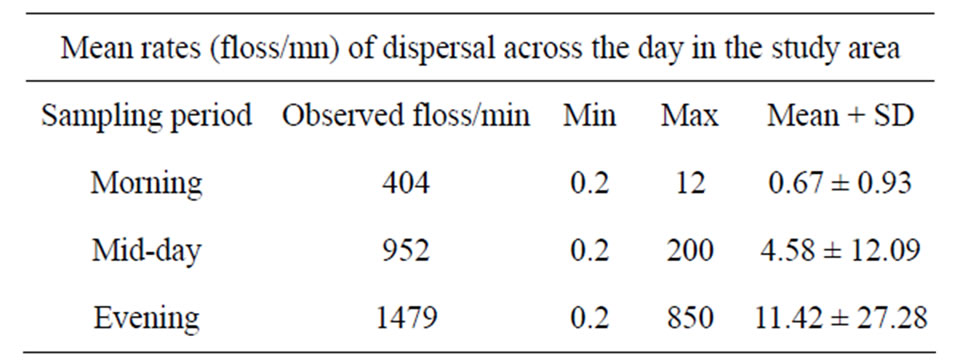
Table 1. Mean dispersal rates of floss for the morning, afternoon and evening based on pooled data from all the sampled trees.
seeds were not different (t = −0.629, DF = 57.94, p = 0.532). This pattern changed when the same was compared with plots further away (>40 m). At a distance between 50 - 100 m onwards from the dispersing trees, a significantly greater proportion of floss without seeds was found as compared to plots relatively closer to the dispersing tree (Wilcoxon signed rank test Z = 5.39, p = 0.001).
3.2. Influence of Water Treatments and Seed Source on Germination of C. pentandra Seeds
The first seed emerged on day five of sawing, with more seeds emerging on each subsequent day of the experiment. Seed germination ceased on day 15 of the experiment. Of the 3600 seeds sown, only 15.67% germinated. About 53.72% of germinated seeds were collected from sunny conditions while the remainder were from shady conditions (t = −0.978, DF = 8, p = 0.375). The seeds collected from both shady and sunny conditions showed variation in germination abilities following different watering levels. For example, 23.75% of seeds collected from shady conditions (N = 261) germinated upon watering once/day, 20.68% when watered twice/ day and 22.98% seeds germinated when watered thrice/ day. Further, 21.07% of seeds germinated when watered only once and 14.9% of seeds when not watered at all. On the other hand, 26.81% of seeds collected from sunny conditions germinated upon watering once/day, 26.05% of seeds when watered twice/day and 27.58% of seeds when watered thrice/day. Twenty-one percent of seeds collected from sunny conditions germinated when water was applied only once and 14.55% of seeds when no water was applied at all. Overall, water treatments were shown to have a significant influence on seed germination of C. pentandra species (F = 3.031, DF = 4, p = 0.021, Figure 2), but the main effect was not due to seed source (shady or sunny) (F = 0.611, DF = 4, p = 0.436). The interactions between water treatment and seed source shown not significant effect on germination (F = 0.283, DF = 4, p = 0.888).
3.3. Perception of Humans on Floss Dispersal in the Study Area
Most respondents (87.9%, n = 15) claimed that they had unpleasant experience with regard to floss dispersal when questioned on how they perceived C. pentandra during flowering season. The reasons mentioned included pollution of the air and environment, people inhaling floss, and eye problems upon encounters with airing flosses. The respondents further said that flowering by C. pentandra has always been associated with dirtying of the environment with scattered flosses.
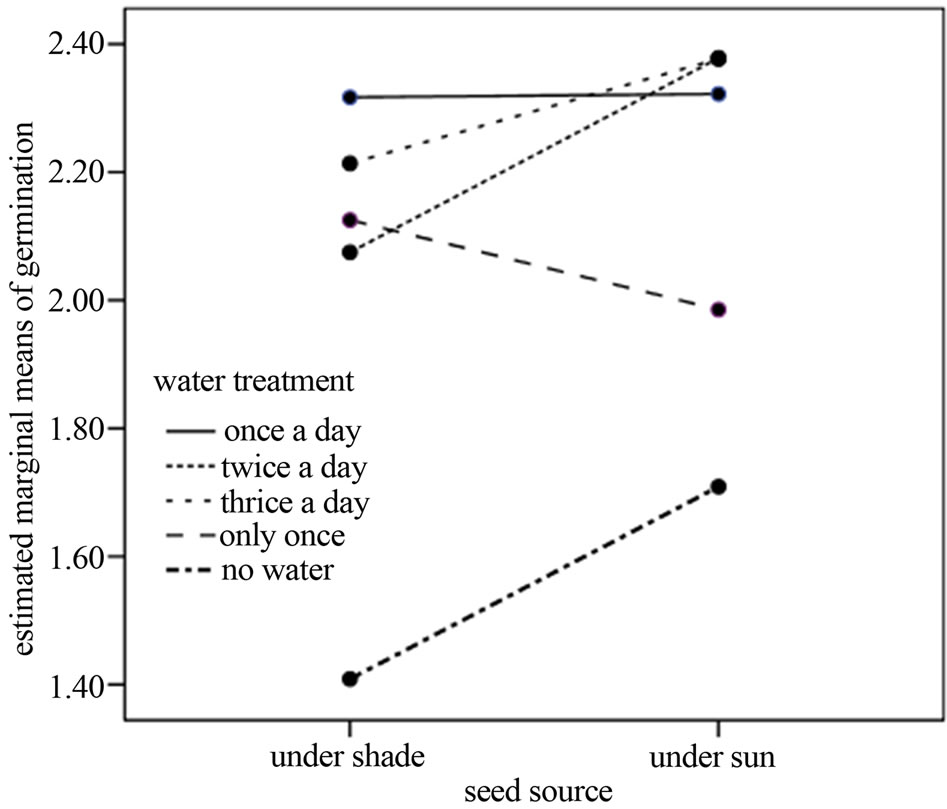
Figure 2. Water treatments and seed source effects on germination of the C. pentandra seeds. Frequent watering showed significant influence on seed germination. Seed source alone or in interaction with watering frequency produced no effects on seed germination, however (see text for statistics).
Cleaning of scattered floss and burning of the areas whitened by floss was mentioned as a widespread activity, particularly in premises around human habitation.
4. DISCUSSION
Seed dispersal is an important phenomenon in the life history of plants. In this study the C. pentandra demonstrated highly variable seed dispersal ability among the individual trees studied. This was expected given that the sampled trees had grown in localities influenced by various local conditions such as wind and also due to individual traits such as canopy openness, which enhanced various seed dispersion patterns. Although we did not record data on the canopy structure, trees in the study area were highly scattered with open canopy that most likely enhanced seed dispersal. Increasing evidence [e.g. 25,26] shows that tree canopy heterogeneity influences the turbulence characteristics inside and above the canopy, thus affecting seed dispersal patterns. The higher rate of seed dispersal from mature fruits in the evening rather than other times of the day may be due to the fact that fruits dehisce more often in late afternoon through evening after being subjected to considerable heating by the sun. This observation is consistent with a study on another plant species, Pinus halepensis, (a Mediterranean serotinous wind dispersed tree) where Nathan et al. [27] recorded substantial seed releases (up to 60%) during dry and hot weather. Moreover, higher rate of seed dispersal in the evening may be due to high wind speed observed in the evening that probably facilitated dispersion of dried floss. These findings are also consistent with the findings by Greene and Johnson [28], who observed seeds typically released in higher-than-average speedy winds. There was high variation in the rates of seed dispersal between individual trees probably because of the local positioning of the trees and plant traits [10]. Furthermore, field observations of post-dispersal floss movement showed a rather linear pattern towards the direction (south-north) of the winds. This is consistent with the suppositions that a seed flies in a straight line along certain wind direction [29]. Such movements and distances were also influenced by the local positioning of individual kapok trees as well as wind speed. Trees located in relatively open areas had flosses moving as far as 0.8 km, while in closed stand trees floss movements were observed within 50 - 100 m distances from the source tree. This thus depicts the effects of such wind breakers in influencing distribution of floss in the area. In comparing effects of landscape structure on flight distances of dispersed seeds, Nathan and others [29] found shorter dispersal distances in dense forests than in open landscape, corroborating our findings. Moreover, field observations indicated that floss distribution decreases with increased distance from the source tree, consistent with the seed shadow that peaks at distances near the source, with an extended tail further away [30].
Seed distribution was higher in plots near the dispersing trees (within 50 m) than further away, presumably because of floss’ leathery structure that stacks onto grasses and debris after landing. Thus, floss structure may have contributed to limiting long distance dispersal by C. pentandra seeds, consequently increasing the risk of seed fatality by natural and human induced causes. During low-speed winds, flosses were observed landing in proximity to the source tree, resulting in “whitening” of the surrounding areas. While dispersal pattern (whether long or short distances) has been widely recognized as crucially important for recruitment and population dynamics of various plant species [10], Ceiba plants growing in the urban environment may have been disadvantaged by their dispersal patterns. In the study area, scattered floss has always been perceived by locals as mucking the environment, consequently resulting in routine collection of the floss with brooms and ultimately burning, a practice that has always followed seed dispersal. This action has been decimating considerable amount of C. pentandra seeds in the area. Seed fall from the dispersed floss differed with distances from the base tree perhaps because of weather conditions and microsite structure on which particular floss lands after release. Flosses that were examined in plots relatively close to the source tree exhibited a rather different pattern of seed fall across different substrates. On the bare ground, a majority of the floss had very few seeds still attached, as opposed to floss stacking on grasses. This pattern may have been attributed to greater heat irradiance from bare ground, which resulted in the seed stalk rapidly drying out, thereby quickening seed detachment from the floss.
Watering frequency had a significant influence on the germination of Kapok seeds. Microsite conditions (sunny or shady) where seeds were collected were less important factors in the germination of Kapok seeds. On the other hand, watering of the seeds once per day resulted in the highest germination over no watering, or watering more than once per day. This result implies that optimum water (100 ml), when applied to the seeds once per day, can effect highest germination in C. pentandra. The study area receives relatively high rainfall total per year. Heavy and frequent rain showers in the area may inhibit germination of Kapok seeds buried in the soil seed bank. Further, Pearson et al. [31] observed an inverse relationship between seedling emergence and soil depth influencing a species’ capability for seed dormancy. Despite C. pentandra seed having a relatively large seed mass (70 mg, [31]) its seed coat is negligible and unlikely to withstand high irradiant temperatures. Thus, seed dormancy may not be effective in carrying this particular species through a dry and hot season with viable seed. The germination percentage observed in this study is within a germination range (11% - 15%) reported for C. pentandra in India [32]. Furthermore, our results on germination are especially useful when considering species restoration or afforestation programs for this particular plant species where establishment of nursery beds for seedling development may be a necessary option. The reported proportion of germination of the C. pentandra seeds could be improved by using germination enhancers under controlled laboratory conditions [31]. Watering frequency had a great role in the germination of the seeds, thus may have been a limiting factor for germination of C. pentandra seeds in the area. This may suggest that the variable rainfall pattern in the study area, characterized by heavy and sometimes frequent rains, may be limiting germination of C. pentandra seeds that may have survived a harsh dry season.
The simple design adopted by this study in the germination experiment was underlain by some important assumptions. As we wanted to mimic the natural conditions in the field, we assumed temperature to be at par in the surrounding area where this experiment was set up. To effect this assumption, germination bags were placed at a single shady location so that no variation in ambient temperature (mean monthly temperature between 21˚C and 27˚C) would be due to the locality. This is because temperature has been observed to strongly influence seed germination of tropical forest trees, including C. pentandra [31]. The volume of water used was also assumed to mimic the rainfall that seeds in the soil bank would receive at a given point in time in the study area. However, the 100 ml water may perhaps not reflect the true situation, given that rainfall varies at different times, years (between 600 mm and 1000 mm) and locations. Our field experience shows that the study area experiences seasonal variation in rainfall patterns, often with erratic heavy and light down pours. This situation may influence germination of seeds in the soil. To control for this effect, our study used various treatment levels of watering frequency to mimic existing field situations. However, it is worth noting that the conditions leading to the germination of seeds may be complex given that several factors including environmental (e.g. temperature, water), seed attributes (e.g. size, weight and shape [32]), physiological [33,34] and seed predation [14,16] singly or together may affect seed viability and subsequent seed germination ability. Therefore, we believe that our treatment levels captured possible situations that seeds in the soil bank may face in the study area.
The Kapok tree has been under increasing pressure from humans. The residents expressed concerns over their experience living around the study species, subsequently discouraging the presence of such species in places where people live. Cleaning and burning of areas whitened with flosses have been widespread and common practices to avoid perceived potential negative effects. This situation is especially critical to the long-term population persistence of C. pentandra in urban environments. These results suggest that germination and recruitment of C. pentandra in the area may have been limited by several factors (water limitation, human activities and local conditions) operating at local and spatial scales. Apparently, the species provides important roosting sites for some mammal species (bats), insects and pollinator bees as well as several bird species [1,35]. Also, seeds of C. pentandra provide important food resources to small mammalian wildlife (rodents) inhabiting the urban and study area (Rija pers. obs. 2013) and it is an important source of local human medicines [18]. Despite these ecosystem services it offers, C. pentandra trees, including the studied individuals, have been clear felled to avoid its perceived negative effects and for timber extraction.
5. CONCLUSION
This study has revealed some important insights into the ecology of the species, particularly documenting the patterns of seed release, post-dispersal movements, seed fall and germination of C. pentandra seeds in an urban environment. Seed release rate and events were variable across the sampling times of the day. The pattern of seed fall showed increased frequency of seed fall away from the source tree with the microsites and local weather conditions highly influencing this pattern. Clear felling of trees of C. pentandra has the potential to negatively affect its population in urban areas and the animal species dependent on it. Thus, this has further negative consequences to the biodiversity and ecosystem services. It is recommended that local residents and responsible authorities explore possible ways to utilize this species, by harvesting mature fruits before floss dispersal. Floss can be sold to the local industries to make pillows and clothes. Floss harvesting not only will improve the local incomes of humans in the area but it will also minimize anthropogenic threats due to the perceived negative effects from floss dispersal, and therefore improve conservation of C. pentandra in urban areas and the ecosystem services it provides to the human population and urban biodiversity [36].
6. ACKNOWLEDGEMENTS
Authors wish to thank Ismail S. Selemani for providing critical comments on earlier drafts of this paper, and Bonnie Tighe for language correction on the manuscript. We thank staff working at the SUA Meteorological Station for sharing data on wind speed and the residents in the study area for sharing their experience with regard to the study plant species.
REFERENCES
- Gribel, R., Gibbs, P. E. and Queiroâz, A.L. (1999) Flowering phenology and pollination biology of Ceiba pentandra (Bombacaceae) in Central Amazonia. Journal of Tropical Ecology, 15, 247-263. http://dx.doi.org/10.1017/S0266467499000796
- Baker, H.G. and Harris, B.J. (1959) Bat-pollination of the silk-cotton tree, Ceiba pentandra (L.) Gaertn. (sensu lato), in Ghana. Journal of the West African Science Association, 4, 1-9.
- Cox, P.A. (1983) Observations on the natural history of Samoan bats. Mammalia, 47, 519-521. http://dx.doi.org/10.1515/mamm.1983.47.4.519
- Elmqvist, T., Cox, P.A., Rainey, W.E. and Pierson, E.D. (1992) Restricted pollination on oceanic islands: Pollination of Ceiba pentandra by flying foxes in Samoa. Biotropica, 24, 15-23. http://dx.doi.org/10.2307/2388469
- Heithaus, E.R., Fleming, T.H. and Opler P.A. (1975) Foraging patterns and resource utilization in seven species of bats in a seasonal tropical forest. Ecology, 56, 841-854. http://dx.doi.org/10.2307/1936295
- Baker, H.G. (1955) Self-compatibility and establishment after “long-distance” dispersal. Evolution, 9, 347-349. http://dx.doi.org/10.2307/2405656
- Murawski, D.A. and Hamrick, J.L. (1992) Mating system and phenology of Ceiba pentandra (Bombacaceae) in Central Panama. Journal of Heredity, 83, 401-404.
- Toledo, V.M. (1977) Pollination of some rain forest plants by non-hovering birds in Veracruz, Mexico. Biotropica, 9, 262-267. http://dx.doi.org/10.2307/2388144
- Janson, C.H., Terborgh, J. and Emmons L.H. (1981) Nonflying mammals as pollinating agents in the Amazonian forest. Biotropica, 13, 1-6. http://dx.doi.org/10.2307/2388065
- Nathan, R. and Muller-Landau, H.C. (2000) Spatial patterns of seed dispersal, their determinants and conesquences for recruitment. Trends in Ecology & Evolution, 15, 278-285. http://dx.doi.org/10.1016/S0169-5347(00)01874-7
- Vander Wall, S.B. and Joyner, J.W. (1998) Secondary dispersal by the wind of winged pine seeds across the ground surface. The American Midland Naturalist Journal, 139, 365-373. http://dx.doi.org/10.1674/0003-0031(1998)139[0365:SDBTWO]2.0.CO;2
- Voysey, B.C., McDonald, K.E., Rogers, M.E., Tutin, C.E. G. and Parnell R. J. (1999) Gorillas and seed dispersal in the Lopé Reserve, Gabon. II: Survival and growth of seedlings. Journal of Tropical Ecology, 15, 23-38. http://dx.doi.org/10.1017/S0266467499000656
- Schupp, E.W. and Fuentes, M. (1995) Spatial patterns of seed dispersal and the unification of plant population ecology. Ecoscience, 2, 267-275.
- Vander Wall, S.B., Kuhn, K.M. and Beck, M.J. (2005) Seed removal, seed predation, and secondary dispersal. Ecology, 86, 801-806. http://dx.doi.org/10.1890/04-0847
- Orrock, J.L., Levey, D.J., Danielson, B.J. and Damschen, E.I. (2006) Seed predation, not seed dispersal, explains the landscape-level abundance of an early-successional plant. Journal of Ecology, 94, 838-845. http://dx.doi.org/10.1111/j.1365-2745.2006.01125.x
- VazFerreira, A., Bruna, E.M. and Vasconcelos, H.L. (2011) Seed predators limit plant recruitment in Neotropical savannas. Oikos, 120, 1013-1022. http://dx.doi.org/10.1111/j.1600-0706.2010.19052.x
- Gentry, A.H. and Vasquez, R. (1988) Where have all the Ceibas gone? A case history of mismanagement of a tropical forest resource. Forest Ecology and Management, 23, 73-76. http://dx.doi.org/10.1016/0378-1127(88)90015-1
- Friday, E.T., James, O., Olusegun, O. and Gabriel, A. (2011) Investigations on the nutritional and medicinal potentials of Ceiba pentandra leaf: A common vegetable in Nigeria. International Journal of Plant Physiology and Biochemistry, 3, 95-101.
- Theng, K., Preston, T.R. and Ly, J. (2003) Studies on utilisation of trees and shrubs as the sole feedstuff by growing goats; foliage preferences and nutrients utilization. Livestocks Research & Rural Development, 15, 7.
- Rija, A. A. (2003) Feeding habits, abundance and distribution of pied crow Corvus albus around the Uluguru Mountains, Morogoro Tanzania. BSc. Thesis, Library of Sokoine University of Agriculture, Tanzania.
- Morrison, M.E.S. and Lind, E.M. (1974) East Africa vegetation. Longman, London, 257 p.
- Frankie, G.W., Baker, H.G. and Opler, P.A. (1974) Comparative phenological studies of trees in tropical wet and dry forests in the lowlands of Costa Rica. Journal of Ecology, 62, 881-913. http://dx.doi.org/10.2307/2258961
- Lobo, J.A., Quesada, M., Stoner, K.E., Fuchs, E.J., Herrerias-Diego, Y., Rojas, J. and Saborio G. (2003) Factors affecting phenological patterns of bombacaceous trees in seasonal forests in Costa Rica and Mexico. American Journal of Botany, 90, 1054-1063. http://dx.doi.org/10.3732/ajb.90.7.1054
- Khurana, E. and Singh, J.S. (2001) Ecology of seed and seedling growth for conservation and restoration of tropical dry forest: A review. Environmental Conservation, 28, 39-52. http://dx.doi.org/10.1017/S0376892901000042
- Horn, H.S., Nathan, R. and Kaplan, S.R. (2001) Longdistance dispersal of tree seeds by wind. Ecological Research, 16, 877-885. http://dx.doi.org/10.1046/j.1440-1703.2001.00456.x
- Bohrer, G., Katul, G.G., Nathan, R., Walko, R.L. and Avissar, R. (2008) Effects of canopy heterogeneity, seed abscission and inertia on wind-driven dispersal kernels of treeseeds. Journal of Ecology, 96, 569-580. http://dx.doi.org/10.1111/j.1365-2745.2008.01368.x
- Nathan, R., Safrie, U.N., Noy-Meir, I. and Schiller, G. (1999) Seed release without fire in Pinus halepensis, a Mediterranean serotinous wind-dispersed tree. Journal of Ecology, 87, 659-669. http://dx.doi.org/10.1046/j.1365-2745.1999.00382.x
- Greene, D.F. and Johnson, E.A. (1992) Fruit abscission in Acer saccharinum with reference to seed dispersal. Canadian Journal of Botany, 70, 2277-2283. http://dx.doi.org/10.1139/b92-283
- Nathan, R., Horn, H.S., Chave, J. and Levin, S.A. (2002) Mechanistic models for tree seed dispersal by wind in dense forests and open landscapes. In: Levey, D.J., Silva, W.R. and Galetti, M., Eds., Seed Dispersal and Frugivory: Ecology, Evolution and Conservation, CABI International, Oxfordshire, 69-82.
- Bullock, J.M. and Clarke, R.T. (2000) Long distance seed dispersal by wind: Measuring and modelling the tail of the curve. Oecologia, 124, 506-521. http://dx.doi.org/10.1007/PL00008876
- Pearson, T.R.H., Burslem, D.F.R. Mullins, P.C.E. and Dalling, J.W. (2002) Germination ecology of neotropical pioneers: Interacting effects of environmental conditions and seed size. Ecology, 83, 2798-2807. http://dx.doi.org/10.1890/0012-9658(2002)083[2798:GEONPI]2.0.CO;2
- Dileep, M., Sudhakara, K., Santosh Kumar, A.V., Nazeema, K.K. and Ashokan, P.K. (1994) Effect of seed size, rooting medium and fertilizers on the growth of seedlings of Ceiba pentandra (Linn) Gaertn. Indian Journal of Forestry, 17, 293-300.
- Khan, A.A. (1980) The physiology and biochemistry of seed dormancy and germination. North Holland, New York.
- Kandya, S. (1990) Studies on the biochemical basis of seed dormancy in some forest tree species used in social forestry programmes. Ph.D. Thesis, Dr H.S. Gour University Sagar, India.
- Singaravelan, N. and Marimuthu, G. (2004) Nectar feeding and pollen carrying from Ceiba pentandra by pteropodid bats. Journal of Mammalogy, 85, 1-7. http://dx.doi.org/10.1644/1545-1542(2004)085<0001:NFAPCF>2.0.CO;2
- Rija, A.A. (2010) Incentives for conserving and greening our cities. Tanzania Wildlife Magazine-Kakakuona, 57, 58-61.

