Advances in Biological Chemistry
Vol.4 No.1(2014), Article ID:43292,5 pages DOI:10.4236/abc.2014.41006
Phytochemical screening, hepatoprotective and antioxidant effects of leaf extracts of Zapoteca portoricensis
![]()
1Biochemistry Department, Ebonyi State University, Abakaliki, Nigeria
2Biochemistry Department, Abia State University, Uturu, Nigeria
Email: *kagbafor@yahoo.co.uk
Copyright © 2014 Kingsley Nwonu Agbafor et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. In accordance of the Creative Commons Attribution License all Copyrights © 2014 are reserved for SCIRP and the owner of the intellectual property Kingsley Nwonu Agbafor et al. All Copyright © 2014 are guarded by law and by SCIRP as a guardian.
Received 26 December 2013; revised 29 January 2014; accepted 7 February 2014
KEYWORDS:Albino Rats; Carbon Tetrachloride; Zapoteca portoricensis; Phytochemicals; Liver Enzymes; Antioxidant Activity
ABSTRACT
Fresh leaves of Zapoteca portoricensis are used in Eastern Nigeria for management/treatment of various disorders without any scientific basis. Hepatoprotective and antioxidant properties in albino rats, and phytochemical composition of distilled water and ethanol leaf extracts were studied. Fifty-five animals were placed in eleven groups (A-K) of five in each. Different doses (100, 200, 400 and 600 mg/kg body weight) of the extracts, 20 mg/kg body weight of vitamin C (standard antioxidant) and distilled water were orally administered to groups A-H, I and J-K respectively for six consecutive days. On the seventh day, 2.5 ml/kg body weight of carbon tetrachloride (CCl4) was given intraperitoneally to groups A-J, while group K received distilling. Serum levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST) and gamma glutamyltransferase (GGT) were used to study hepatoprotective effect. Malondialdehyde (MDA), superoxide dismutase (SOD) and catalase (CAT) levels in the liver were monitored to assess antioxidant activity. Phytochemical analysis revealed the presence of alkaloids, tannins, saponins, flavonoids, anthraquinones, terpenoids and cardiac glycosides in the extracts. Pretreatment of the rats with the extracts produced a significant decrease (p < 0.05) in ALT, AST, GGT and MDA, while the activity of SOD and CAT increased significantly (p < 0.05) relative to the positive control. These results, which were dose-dependent, are indicative of hepatoprotective and antioxidants potentials of the extracts, and may be due to their phytoconstituents.
1. INTRODUCTION
All plants produce chemical compounds as part of their normal metabolic activities. These can be divided into primary metabolites such as sugars, and fats found in all plants and secondary metabolites, which include alkaloids, tannins, saponins, phenols, and glycosides. These secondary metabolites and other chemical constituents of medicinal plants account for their medicinal value [1].
Zapoteca portoricensis, commonly called white stick pea, is a member of the family Fabaceae [2]. It is a perennial shrub with small oval green leaves. It is mostly found in Eastern part of Nigeria. Traditional medicine practitioners in Eastern Nigeria use shoot system of the plant in the management/treatment of diseases such as convulsions, constipation, madness, external wounds, skin infections, and so on.
Phytochemicals are bioactive compounds found in plants that work with nutrients and dietary fibre to protect against diseases. They are non-nutritive compounds (secondary metabolites) that contribute to flavour colour [3]. Many phytochemicals have antioxidant activity and reduce the risk of many diseases, such as alkyl sulfide (found in onions, garlic, etc.), carotenoids (from carrots) and flavonoids (present in fruits and vegetables) [3]. Reactive oxygen free radicals (ROS) have been implicated in many diseases and in aging process. These free radicals, which cause tissue damage via oxidative stress, are generated by aerobic respiration inflammation, lipid peroxidation, etc. Antioxidant systems minimize or prevent deleterious effects of the ROS [4].
Lipid peroxidation is an established mechanism of cellular injury, and is used as an indicator of oxidative stress. Polyunsaturated fatty acids peroxides generate malondialdehyde (MDA) and 4-hydroxyalkanals upon decomposition [5].
Superoxide dismutase (SOD) decomposes superoxide anion into hydrogen peroxide and oxygen at very high rates. Superoxide radical is involved in diverse physiological and pathophysiological processes [6]. Catalase (CAT) is an antioxidant enzyme ubiquitously present in aerobic cells. It catalyses the decomposition of hydrogen peroxide to water and oxygen. High concentration of hydrogen peroxide is deleterious to cells, and its accumulation causes oxidation of cellular targets such as DNA, proteins and lipids, leading to mutagenesis and cell death [7].
The liver is a central organ in biochemical homeostasis [8]. Aspartate aminotransferase (AST), alanine aminotransferase and gamma-glutamyltransferase are important in diagnosis of liver damage by chemical toxicity or infection. These enzymes are located intracellularly, but leak out into the blood stream when the tissue is damaged [8,9].
In spite of the tremendous strides in modern medicine, there are hardly any drugs that can stimulate liver function or offer protection to the hepatocytes from damage. Thus, many folk remedies from plants are studied for their possible hepatoprotective effect against chemically induced liver damage in experimental animals [10]. The present study was designed to examine the phytochemical constituents, hepatoprotective and antioxidant activities of fresh leaves of Zapoteca portoricensis in carbon tetrachloride (CCl4)-induced hepatotoxicity.
2. Materials and Methods
2.1. Collection of Fresh Leaves of Zapoteca portoricensis
The leaves were obtained in May, 2010 from a bush in Abakaliki, Ebonyi State of Nigeria. They were identified by Prof. S. C. Onyekwelu of Biology Department, Ebonyi State University in Abakaliki, washed with distilled water and used immediately for the research, which lasted fifteen days.
2.2. Preparation of Leaf Extracts
The methods of extraction used by Agbafor [11] were adopted, utilizing distilled water and ethanol (separately) as solvents. The extracts were prepared by pounding the fresh leaves into a paste and soaking the paste in the respective solvents for 24 hours, followed by decantation. The extracts were concentrated using rotor evaporator to get gel-like dark brown extracts.
2.3. Phytochemical Analysis of the Extracts
The methods of Harbone [12] and Trease and Evans [13] were used to identify the following phytochemicals in the extracts: alkaloids, saponins, tannins, anthraquinones, flavornoids, terpenoids and cardiac glycosides. Dried samples of the extracts were used in each of the specific methods for testing for the presence of the afore-mentioned phytochemicals as described by Harbone [12] and Trease and Evans [13].
2.4. Assessment of Hepatoprotective and Antioxidant Properties
Animals and handling: Fifty-five adult male albino rats, weighing 115 - 124 g were brought from the animal house of Biochemistry Department, University of Nigeria, Nsukka. They were placed in eleven groups (A-K) of five rats in each group and kept in animals’ house of Biochemistry Department, Ebonyi State University Abakaliki for seven days to acclimatize. All the rats were allowed free access to feed (rat chaw) and water before and throughout the experiment.
Ethical approval was obtained from Ebonyi State University Research and Ethics Committee.
Animal groups and treatments: Solutions of the extracts were made with distilled water. Different doses of the extracts and 20 mg/kg body weight of vitamin C (Emzor Pharmaceutical Industries, Nigeria) were given orally to groups A-H and I respectively, while J and K received distilled water for six consecutive days.
2.5. Inducement of Liver Damage
On the seventh day, groups A-J treated with a single dose of 2.5 ml/kg body weight of CCl4 and olive oil (1:1) intraperitoneally. Group K was given distilled water/ olive oil (1:1).
Summary:
Group A—100 mg/kg b. wt water extract + CCl4
Group B—200 mg/kg b. wt water extract + CCl4
Group C—400 mg/kg b. wt water extract + CCl4
Group D—600 mg/kg b. wt water extract + CCl4
Group E—100 mg/kg b. wt ethanol extract + CCl4
Group F—200 mg/kg b. wt ethanol extract + CCl4
Group G—400 mg/kg b. wt ethanol extract + CCl4
Group H—600 mg/kg b. wt ethanol extract + CCl4
Group I—20 mg/kg b. wt vitamin C + CCl4
Group J—Distilled water + CCl4
Group K—Distilled water.
Collection of samples from the animals: Blood samples were collected from the animals following an overnight fast through cardiac puncture under mild anaesthesia using diethylether. The samples were put into specimen bottles without anticoagulant. Liver was also quickly excised, perfused with cold normal saline and homogenized in 0.25 M sucrose in phosphate buffer (0.2 M, pH 7.4).
2.6. Hepatoprotective Property
Serum levels of ALT, AST and GGT were used to study hepatoprotective effect of the extracts. Their levels were determined by the methods described by Tietz [14].
Determination of AST activity: AST catalyses the transfer of the α-amino group from L-aspartate to α-ketoglutarate resulting in the formation of oxaloacetate and L-gultamate. The oxalocetate reacts with 2,4-dinitrophenylhydrazine (DNPH) to form 2,4-dinitrophenylhydrazone which in an alkaline medium gives a red-brown colour. The absorbance of the colour produced is measured using a spectrophotometer at 510 nm.
Determination of ALT activity: ALT catalyses the transfer of the amino group from L-alanine to α-ketoglutarate resulting in the formation of pyruvate and Lglutamate. ALT is measured by monitoring the concentration of pyruvate hydrazone formed with 2,4-dinitrophenylhydrazine.
Measurement of GGT activity: The substrate L-γ-glutamyl-3-carboxy-4-nitroanilide, in the presence of glycylglycine is converted by GGT in the sample to 5-amino- 2-nitrobenzoate which can be measured at 405 nm.
2.7. Antioxidant Activity
This was assessed by the concentration of the lipid peroxidation product, MDA and antioxidant oxidant enzymes, SOD and CAT in the liver homogenate.
Catalase activity was measured by the method of Aebi [15]. Catalase reduces the hydro peroxide which can induce damage in the membrane by initiating lipid peroxidiation. The rate of decomposition of H2O2 to water and oxygen was measured spectrophotometrically at 240 nm. The absorbance of H2O2 at 240 nm is measured directly to calculate the reaction since water and oxygen do not absorb at this wavelength.
Lipid peroxidation levels were measured by the thiobarbituric acid (TBA) reaction using the method of Ohkawa et al. [16]. This method was used to measure spectrophotometrically the color produced by the reaction of TBA with malondialdehyde (MDA) at 532 nm.
Estimation of Superoxide Dismutase (SOD): The activity of superoxide dismutase (SOD) was assayed by the method of Kakkar, et al. [17] based on the oxidation of epinephrine adrenochrome transition by enzyme. Change in absorbance is read at 470 nm.
3. Data Analysis
Statistical analysis was done using analysis of variance (ANOVA). Means were compared for significance using Duncan’s Multiple Range test (p < 0.05) [18].
4. Results and Discussion
The reviews cited in this section are in agreement with the major findings of this research.
The phytochemical constituents of the extracts are presented in Table 1. The extracts contained alkaloids, tannins, saponins, flavonoids, anthraquinones, terpenoids and cardiac glycosides in varied proportions. These phytochemicals contribute to the various medicinal applications of leaves of zapoteca portoricensis. In line with Varadarajan et al. [1], the secondary metabolites (phytochemicals) and other chemical constituents of medicinal plants are responsible for their medicinal values.
Table 2 shows the hepatoprotective effect of the extracts. The levels of ALT, AST and GGT in the group treated with CCl4 only (group J) were significantly higher (p < 0.05) than those in the untreated group K. Thus, CCl4 induced liver damage in group J. Kartik et al. [19] reported a similar observation. However, pretreated of animals with the extracts or vitamin C resulted to a significant (p < 0.05) and dose-dependent decrease in the levels of these enzymes. This is also consistent with the findings of Kartik et al. [19]. The ability of the extracts to reduce the levels of these enzymes points to hepatoprotective property. Clinical observations and experimental studies have shown that subtle changes in the membranes of hepatocytes are sufficient to allow passage of intracellular enzymes into the extracellular space. Cell damage increases membrane permeability, causing cytosolic enzymes to spill into circulation [8]. For instance, in liver disease associated with hepatic necrosis, serum ALT and AST levels are elevated even before the clinical signs and symptoms of the disease appear [8]. From our results, this hepatoprotective effect of the extracts was comparable with that of vitamin C, and did not show any significant difference (p > 0.05) between the extracts.
Results of the examination of the antioxidant activity of the extracts are presented in Table 3. There was a significant (p < 0.05) increase in MDA levels and decrease in SOD and CAT activities of group J, treated with CCl4 only relative to the untreated control group. This also reflects hepatotoxicity of CCl4, as observed by Singh et al. [10]. The results were reversed on pretreatment with the leaf extracts or vitamin C. The MDA concentration of the pretreated groups was significantly lower (p < 0.05) than the untreated. On the hand, the activities of SOD and CAT were significantly higher (p < 0.05) in the pretreated groups than in the positive control. These observations are indicative of antioxidant property of the extracts.
Free radical damage and oxidative stress are the major reasons for liver tissue damage. The antioxidant enzymes

Table 1. Phytochemical constituents of the extracts.

Table 2. Serum activities of ALT, AST and GGT of the animals after treatment.
are the first-line defense against such damage and thus provide protection against the deteriorating outcome [20]. Oxidative injury and lipid peroxidation can be monitored by measuring liver MDA. Lipid peroxidation is regarded as one of the basic mechanisms of tissue damage caused by free radicals [19,21].
It is probable that the various phytoconstituents of the extracts are involved in scavenging free radicals from tissues, thus, reducing oxidative stress. For example, flavonoids and tannins are phenolic compounds, and plant phenolics are a major group of compounds that act as primary antioxidants or free radical scavengers. Similarly, terpenoids, as vitamins, act as regulators of metabolism and play a protective role as antioxidants [22].
This antioxidant property of the extracts was found to be dose-dependent. The effect increased to at 400 mg/kg body weight and decreased afterwards (Table 3). The
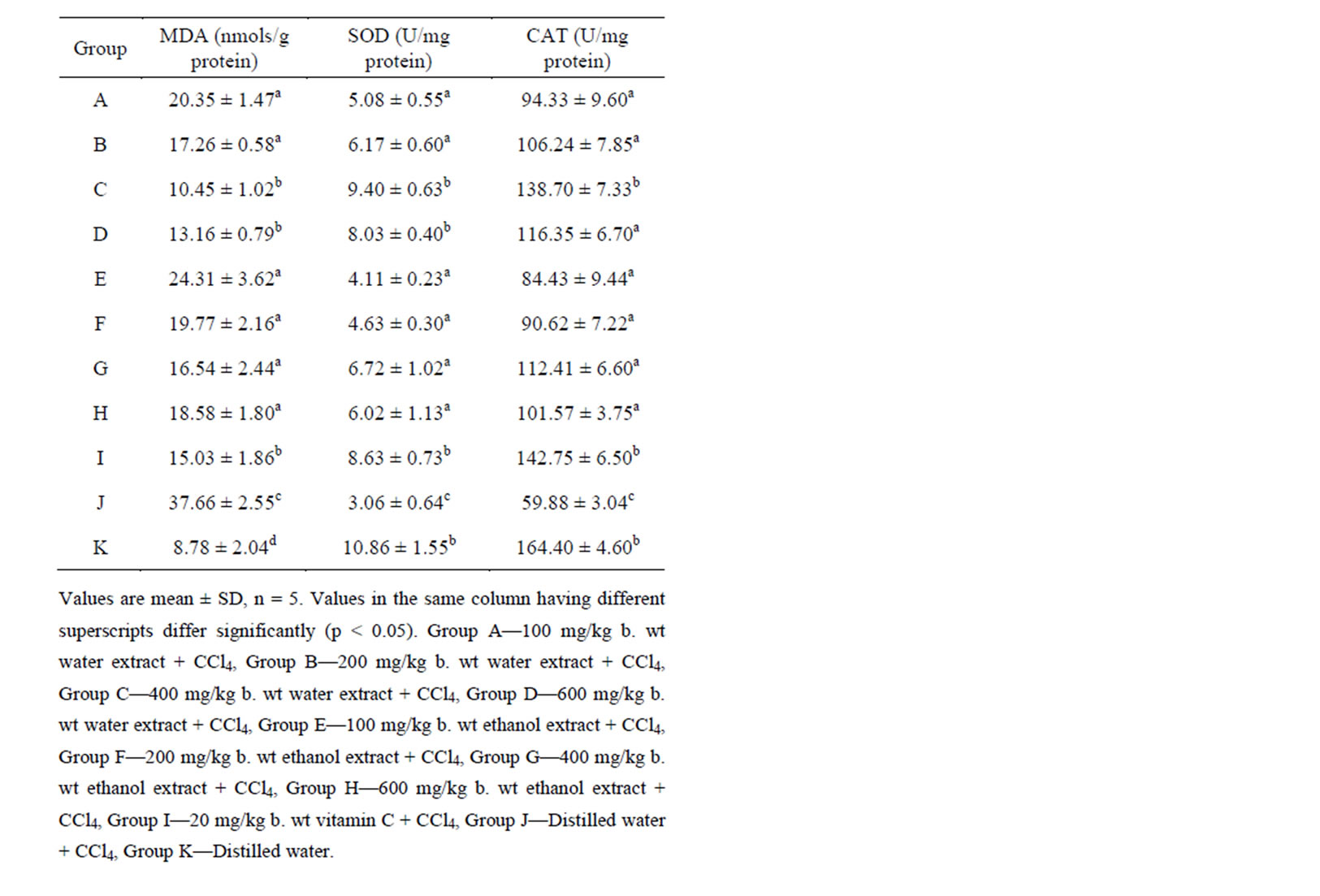
Table 3. Liver levels of MDA, SOD and CAT of the animals.
actual reason for this observation. However, the higher doses of the extracts may produce concentrations higher than the therapeutic level and non-pharmacologic or toxic effects. This observation is consistent with that reported by Agbafor [11] on leaf extracts of Baphia nitida.
The effects in the groups treated with water extracts were not significantly different (p > 0.05) from those in ethanol extract treated groups. The antioxidant potential of 400 mg/kg was insignificantly higher (p > 0.05) than that of vitamin C. Nair et al. [23] got a similar result between vitamin E and some Indian foods.
5. Conclusion
The presence of the identified phytochemicals in the extracts partly explains the medicinal applications of leaves of Zapoteca portoricensis. The exert compounds responsible for the pharmacological activities are currently being investigated. The hepatoprptective potential may not be unrelated to the antioxidant property of the phytoconstituents. However, this is subject to further examination. Our findings may be useful in explaining the medicinal values of fresh leaves of Zapotca portoricensis.
REFERENCES
- Varadarajan, P., Rathinaswamy, G. and Asirvatahm, D. (2008) Antimicrobial properties and phytochemical constituents of rheo discolor. Ethnobotanical Leaflet, 12, 841-845.
- Izo, A.A., Cario, G.D. and Briscard, D. (1995) Biological screening of Italian plants for antibacterial activity. Plant Medicine, 9, 281-284.
- Craig, W.J. (1999) Health-promoting properties of common Herbs. American Journal of Clinical Nutrition, 70, 491-499.
- Valko, M., Moncol, J., Mazur, M. and Telser, J. (2007) Free radicals and antioxidants in Normal physiological functions and human disease. International Journal of Biochemistry & Cell Biology, 39, 44-84. http://dx.doi.org/10.1016/j.biocel.2006.07.001
- Esterbauer, H., Schaur, R.J. and Zollner, H. (1991) Chemistry and Biochemistry of 4-hydroxyalkanals, malondialdehyde and related aldehydes. Free Radical Biology & Medicine, 11, 81-128. http://dx.doi.org/10.1016/0891-5849(91)90192-6
- Johnson, F. and Giulivi, C. (2005) Superoxide dismutases and their impact on human health. Molecular Aspects of Medicine, 26, 340-352. http://dx.doi.org/10.1016/j.mam.2005.07.006
- Chelikani, P., Fita, I. and Loewen, P. (2004) Diversity of structures and properties among catalases. Cellular and Molecular Life Sciences, 61, 192-208. http://dx.doi.org/10.1007/s00018-003-3206-5
- Burtis, C.A. and Ashwood, E.R. (2003) Tietz fundamentals of clinical chemistry. 5th Edition, Elsevier Publishing, India.
- Akubugwo, I.E. and Agbafor, K.N. (2007) Hepatotoxical evaluation of water and salt from Okposi and Uburu salt lakes, Nigeria. Estudos de Biologia, 29, 99-104.
- Singh, J., Bagla, A. and Pahal, V. (2010) Hepatoprotective activity of herbal extracts in CCl4 intoxicated albino rats by measuring antioxidant enzymes. International Journal of PharmTech Research, 2, 2112-2115.
- Agbafor, K.N. (2004) The effect of aqueous and organic extracts of fresh leaves of Baphia nitida on tissue acetylcholinesteraase in guinea pigs. Journal of Science and Technology, 10, 1-8.
- Harborne, I.B. (1973) Phytochemical methods: A guide to modern techniques of plant analysis. 2nd Edition, Chapman and Hall, New York, 88-185.
- Trease, G.E. and Evans, W.C. (1983) Textbook of pharmacognosy. 12th Edition, Tindall and Co., London, 343- 383.
- Tietz, N. (2000) Fundamentals of clinical chemistry. 4th Edition, W.B. Saunders, Philadelphia, 984-1002.
- Aebi, H. (1974) Catalase. In: Bergmeyer, H.V., Eds., Methods in Enzymatic Analysis, Academic Press Inc., New York, 673-686. http://dx.doi.org/10.1016/B978-0-12-091302-2.50032-3
- Ohkawa, H., Ohishi, N. and Yagi, K. (1979) Assay for lipid peroxide in animal tissues by thiobarbituric acid reaction. Annal Biochem. 95, 51-55. http://dx.doi.org/10.1016/0003-2697(79)90738-3
- Kakkar, P., Das, B. and Viswanathan, P. N. (1984) Modified spectrophotometric assay of SOD. Indian Journal of Biochemistry & Biophysics, 2, 130-132.
- Sokal, R.R. and Rholf, F.J. (1969) Practice of statistic in research. Freeman Press, San Francisco, 21-26.
- Kartik, R., Rao, V., Trivedi, S.P. and Reddy, G.D. (2010) Amelioration effects against N-nitrosodiethylamine and CCl4-induced hepatocarcinogenesis in rats by extracts of Achyranthes aspera. Indian Journal of Pharmacology, 42, 370-375. http://dx.doi.org/10.4103/0253-7613.71921
- Gaurav, M., Apminder, S.B. and Giridhar, S. (2008) Nnitrosodiethylamine-induced toxicity in relation to oxidative stress and development of atherosclerosis in hypercholesterolemic diet-fed rabbits. Experimental and Toxicologic Pathology, 59, 409-414. http://dx.doi.org/10.1016/j.etp.2007.10.009
- El-Segacy, O., Ab-Allah, A. and Al-Nooman, S.A. (2007) Experimental study of antioxidant and hepatoprotective effects of Clove and Cardamon in ethanol-induced hepatotoxicity. Tanta Medical Sciences Journal, 2, 27-36.
- Soetan, K.O. (2008) Pharmacological and other benefits of antinutritional factors in plants. A. Review, African Journal of Biotechnology, 7, 4713-4721.
- Nair, S., Nagar, R. and Gupta, R. (1998) Antioxidant phenolics and flavonoids in common Indian foods. Journal of the Association of Physicians of India, 46, 708-710.
NOTES
*Corresponding author.

