Journal of Cosmetics, Dermatological Sciences and Applications
Vol.4 No.3(2014), Article ID:46383,7 pages DOI:10.4236/jcdsa.2014.43023
Treatment of Edematofibrosclerotic Panniculitis (Cellulite) and of Localised Adiposity with Extracts of Birch, Melilotus, Orthosiphon, Bromelin and Red Vine Leukocyanidine: Results from a Single-Centre Observational Prospective Study
Concettina Elio1*, Filippo Intilangelo1, Attilio Cavezzi2, Galya Dimitrova2, Stefania Paccasassi2, Gianni Sigismondi1
1Vascular Unit, Poliambulatorio Hippocrates, San Benedetto del Tronto, Italy
2Eurocenter Venalinfa, San Benedetto del Tronto, Italy
Email: *concyelio@alice.it
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/


Received 24 March 2014; revised 20 April 2014; accepted 28 April 2014
ABSTRACT
The Edematofibrosclerotic Panniculitis (EFP) and the Localised Adiposity (LA) have controversial pathogenesis and therapy. A single-centre observational study has been performed to assess the potential efficacy, the limitations and the side effects of the medical treatment of EFP and AL by means of a multicomponent nutraceutical product. Fifty female subjects have been enrolled, mean age and BMI of the patients were 46 years and 24.18 respectively. The subjects have been submitted to a two-month treatment with two tablets of a nutraceutical based on extracts of Birch (100 mg), Orthosiphon (100 mg), Red Vine Leukocyanidines (100 mg), Melilotus (5 mg) and Bromelin (150 mg). The patients have been investigated before the treatment and during the treatment (at 30 and 60 days) by means of: a) volumetry of the lower limbs with tape measurement; b) visual analogue scale assessment (VAS) of symptoms such as pain paresthesia/dysesthesia, heaviness, and of the daily diuresis; c) duplex ultrasound evaluation of the thickness of the adipose tissue at the peri-trochanteric level; d) multifrequency segmental bioimpedance (MSB) analysis for the tissue liquid component. Forty-five of the 50 enrolled subjects have completed the study and five patients have discontinued the treatment, four of which due to unexpected personal problems or health problems not related to the treatment itself and one for reporting gastrointestinal disorders (such as constipation/diarrhea) with the nutraceutical intake. At the end of the therapeutical cycle, volumetry of the limbs decreased of 7% (right) and of 6% (left). Pre/post treatment VAS assessment of symptoms showed the following mean figures: heaviness from 5.10 to 0.88; paresthesia/dysesthesia from 0.90 to 0.00; pain from 2.03 to 0.12. Diuresis increased of 31%. Duplex- ultrasound assessment of the subcutaneous adipose layers at the trochanteric level showed a reduction of slightly more than 20% at the end of the treatment in both limbs. L-Dex bioimpedance values have been as follows: 3.97 at the enrolment; 1.17 at the end of the treatment (reduction of the subcutaneous tissue fluid content). The present single-centre observational study proved that the tested nutraceutical multicomponent is effective and free of relevant side effects in the treatment of EFP and AL of the lower limbs. Larger cohort studies may possibly confirm the results of this preliminary experience.
Keywords:Edematofibrosclerotic Panniculitis, Localised Adiposity, Nutraceutical, Bioimpedance Spectroscopy

1. Introduction
The Edematofibrosclerotic Panniculitis (EFP) and the Localised Adiposity (LA) nowadays represent one of the most relevant causes of medical consulting. A large number of studies focused on the research of the causes of such disorder [1] , on the possible risk factors which influence EFP and its most effective treatment methods [2] . Currently, in addition to the various possible surgical treatments (liposuction-liposculpture, laser-lipolysis, etc.) and complementary to the numerous, more or less effective and invasive medical therapies (mesotherapy, electrolipolysis, carboxytherapy, intralipotherapy with various pharmacological solutions, homeopathy, electromedical equipment, etc.), the pharmacological treatments or with nutraceuticals represent however a possible option for cellulite treatment, though relative validation is still suboptimal.
In recent years, the potential of the active ingredients derived from herbal remedies or from drugs, has been emphasised in terms of improvement of the micro-circulation and the trophism of the skin tissues and of the subcutaneous tissue [3] .
Through a single-centre observational study, the authors wanted to assess the potential effectiveness, the limits and the possible side effects of medical treatment of EFP and/or LA, by means of a nutraceutical product that combines the action of more components.
2. Patients and Methods
A clinical-instrumental observational study has been performed, in order to evaluate the efficacy of an oral treatment for patients affected by Edematofibrosclerotic Panniculitis and/or Localised Adiposity of the lower limbs. Diagnosis has been based on history taking, clinical consultation and colour-duplex ultrasound.
Fifty female subjects with a mean age of 46 (±10 S.D.) years, with an average BMI of 24.18 (±2.07 DS) have been enrolled, according to the following inclusion and exclusion criteria:
Inclusion criteria:
Edematofibrosclerotic Panniculitis (EFP) and/or Localised Adiposity (LA) of the lower limbs.
Exclusion criteria:
Age <20 years old, or >50 years old.
Cardiac, or renal, or liver insufficiency.
Intake of calcium antagonists, alpha-lytics, corticosteroids.
Acute skin diseases.
Ongoing pregnancy.
Acute venous thromboses.
The subjects have been submitted to a cycle of two months therapy, after an informative talk concerning the type of the treatment and the provided protocol; twice a day patients took one tablet of a multicomponent nutraceutic based on extracts of Birch (100 mg), Orthosiphon (100 mg), Red Vine Leukocyanide (100 mg), Melilotus (5 mg) and Bromelin (150 mg).
Throughout the treatment period the patients did not have any compression therapy with elastic stockings or bandages, or other phlebolymphotropic drugs, or anti-edema therapies. The patients have been instructed on the need to maintain, throughout the whole study period, a constant lifestyle regimen, similar to the one before the beginning of the study. The patients finally have been required to report any side effects elapsed during the undertaken treatment.
At the enrolment a medical record has been filled with the patient’s personal data and with the following pre-treatment data:
2.1. Volume of the Lower Limbs, Derived from a Centimetre Tape Measurement
Circumference measurements of the lower limbs were taken starting from the ankle (just above the point of maximum salience of the medial malleolus) every 8 cm for a total of 8 circumferences, with calculation of the volume of the limb by the following formula adopted for the quantification of the truncated cone:
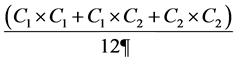
where C1 is the most proximal circumference (e.g. immediately above the malleolus) and C2 is the circumference upstream, while ¶ = 3.14.
For each truncated cone the relative C1 and C2 and the volume have been calculated, therefore the segmental volumes have been summed in order to calculate the volume of the entire limb.
2.2. Visual Analogue Scale (VAS) Assessment
(Numerical value from 1 to 10, while 1 is the value of lower intensity and 10 is of maximum (worse) intensity) of the following symptoms reported by the patient:
• Heaviness.
• Dysesthesia/paresthesia.
• Pain.
Each patient has then given a numerical value, always based on the VAS, of the extent of her daily diuresis.
2.3. Ultrasound Evaluation of the Thickness of the Adipose Tissue at the Peri-Trochanteric Region Level
Through a B/W ultrasound evaluation (13 MHz linear probe), measurements of the distance between epidermis and muscular fascia have been taken. Prior to this measurement an identification of precise skin reference points (e.g. Telangiectasia or nevus) and at the muscle fascia level, have been identified to get better reproducibility of ultrasound scans. In order to reduce the inherent variability of each measurement a quantity of ultrasound gel has been interposed such as to prevent the contact between the probe and the patient’s skin, while not exerting any pressure on the probe itself. Two ultrasound scans (transverse and longitudinal) for each single examine point have been performed, calculating then the mean value (and standard deviation) for each measurement.
2.4. Multifrequency Segmental Bioimpedance (MSB)
All patients have been submitted to a MSB measurement, with L-Dex U400 device (Impedimed, Australia) [4] [5] , that measures the variation of the impedance of the limbs in which a small alternating current of 200 µA at a frequency of approximately 3 kHz to 1000 kHz is passed between two current electrodes spanning the body. The low MSB frequency is only passing through the extracellular/interstitial spaces, therefore the resistance is lower in presence of a higher content of fluids and vice-versa. The reliability of MSB has been demonstrated in several studies and in recent years the above-mentioned method has been used to complement other diagnostic venous and lymphatic investigations [4] -[8] . The L-Dex value is derived from the ratio between the impedance values of the two limbs, so it is an expression of all possible differences in the content of the fluids between both limbs.
The flow chart at the enrolment has comprised volumetry both limbs, interviewing by the VAS, ultrasound assessment and MSB measurement, then the patients have been reviewed at the end of the first and of the second month of therapy in order to perform the same examinations accomplished at the enrolment.
The collected data have been processed by Microsoft Excel 2003® program and the statistical analysis provided the calculation of mean value, standard deviations and the changes in percentage of the same mean value for the numerical values of the volumes, the VAS for symptoms and diuresis, the thickness of the subcutaneous adipose tissue on the ultrasound examination, the resistance with MSB.
3. Results
45 of the 50 subjects enrolled have completed the study and five patients have discontinued the treatment, four of which due to unexpected personal problems or health problems, independent of the treatment itself, and one for reporting gastrointestinal disorders (such as constipation/diarrhea) with the nutraceutical intake.
3.1. Volumes
(Formulated as mean value and SD):
The following Table 1 summarises the results as to lower limb volume measurements at the enrolment time and the variations at 30 days and 60 days. A significant variation was highlighted at 30 and especially 60 days follow-up. As the overall (right + left limbs) assessment, the main results were the following:
• Mean volume of both limbs at the enrolment: 6005.60 cm3 ± 567.13.
• Mean volume of both limbs after 30 days treatment: 5729.23 cm3 ± 492.65.
(5% reduction compared to the mean volume at the enrolment.)
• Mean volume of both limbs after 60 days treatment: 5669.00 cm3 ± 410.73.
(6% reduction compared to the mean volume at the enrolment.)
3.2. VAS Parameters
The evaluated parameters have been calculated according to the scale from 1 to 10 as to the VAS, therefore the increase (worsening) and the decrease (improvement) of the value of each parameter has been calculated in percentage. Conversely diuresis was scored as increasing with higher numbers and vice-versa.
As far as the examined parameters concern, the collected data (mean value and SD) are summarised in Table 2, where noticeable changes in all symptoms and in diuresis are highlighted.
3.3. Ultrasound Evaluation of the Thickness of the Adipose Tissue at the Peri-Trochanteric Region Level (Mean Values and SD)
Table 3 below summarises the ultrasound-based results which confirm the reduction of the adipose layer in the two limbs.
Table 1. Variation of the lower limb volume.
Table 2. Variation of the symptoms and of diuresis (VAS parameters).
Table 3. Variation of the adipose layer (ultrasound evaluation).
Regarding the ultrasound assessment of the two limbs taken as an overall, all parameters had a significant improvement. More in detail:
Mean thickness value of both limbs at the enrolment: 47.49 mm ± 11.07.
Mean thickness value of both limbs after 30 days treatment examination: 42.20 mm ± 11.05.
(13% reduction compared to the enrolment.)
Mean thickness value of both limbs after 60 days treatment examination: 39.13 mm ± 6.42.
(21% reduction compared to the enrolment.)
3.4. Multifrequency Segmental Bioimpedance with L-Dex Calculation
Mean L-Dex value (and SD) (taking in consideration both limbs as altogether) at the enrolment: 3.97 ± 4.27.
L-Dex after 30 days treatment examination: 2.16 ± 4.38.
L-Dex after 60 days treatment examination: 1.17 ± 3.86. (Reduction of the liquid content of the subcutaneous tissue in both limbs.)
4. Discussion
The Edematofibrosclerotic Panniculitis (EFP) and the Localized Adiposity (LA) indicate an altered state of the subcutaneous tissue which is rich of fat cells. The EFP de facto is more related to the microcirculation disorder, to the systemic hormonal metabolism, lifestyle and nourishing habits, while the localized fat deposits are the result of a transformed deposit of the fat tissue for a localized lipodystrophy. The presence of such “changes” represent a cosmetic, mainly, and functional disorder which has been reported in the Caucasian and female populations mostly [2] (85% of the lipodystrophy cases of the lower extremities affect women and 15% men [9] ).
From a pathological point of view the genesis appears to be multifactorial and its etiology substantially is still not well defined. As a matter of fact the scientific literature in some cases reports an alteration of the intracellular matrix of the subcutaneous connective tissue with water retention, a causal mechanism that lipodystrophy is based on. This edematous tissue would evolve gradually to fibrosclerosis [10] -[12] over time. For some authors, at the basis of the problem “cellulite”, there would be an alteration of the microcirculation (conditioned by the action of the estrogens whose receptors are localized in the vascular endothelium), in particular of the venous capillaries and lymph vessels, with increased intraluminal pressure and consequently water retention and protein filtration in the interstice. The mechanism cascade would lead to the fibrotic evolution of the tissue, which compromises the micro-vascular flow (with a mechanism that is “self-renewing” in time) [13] -[15] . Finally, other authors sustain that once an adipose accumulation has been generated, the adipose lobules tend to form “rhombus-like nodules” thus giving the orange-skin look (and the subsequent nodularity) [2] [16] -[18] .
Probably, on a constitutional basis (e.g. structural subcutaneous differences, microcirculation differences and estrogen component differences) additional precipitating factors (e.g. diet, hormonal therapies, pregnancies, sedentary lifestyle, smoking, overweight) contribute to the evolution of the microcirculatory problems and/or to tissue degeneration.
In fact, the therapeutic approach to the EFP and to LA is always of multi-faceted nature, [2] with the purpose of an improvement of the esthetic appearance and of the related symptoms and signs.
The multicomponent nutraceutic which has been an object of this study, contains active ingredients with various actions on the venous-lymphatic microcirculation and on the interstitial matrix. More specifically: a) melilotus extract (titrated at 20% coumarin) has a prolymphokinetic action and a proteolytic action, hence favouring lymphatic drainage [19] [20] ; b) bromelin is known for its anti-inflammatory [21] and proteolytic effect; c) birch has a draining and anti-inflammatory [22] -[24] action; d) leucocyanidine extracted from red vine have an anti-oxidant effect [24] and e) orthosiphon has been shown to induce diuresis plus being characterised by anti-oxidant and anti-inflammatory [25] proprieties. This single-center observational prospective study has shown an improvement of the symptoms after one month of treatment with the nutraceutical multicomponent, with an additional, less relevant improvement in the second month. An analogous positive therapeutic effect, especially at 30 days follow-up, has been documented in the context of the limb volume [26] , which decreased from 5984.40 cm3 ± 556.49 (right limb) and 6026.80 cm3 ± 509.98 (left limb) to 5575.95 cm3 ± 346.04 and 5705.50 cm3 ± 402.65 respectively (7% and 6% reduction for right and left limb respectively, compared to the enrolment).
Tissue fluids decreased as well, as shown by MSB assessment, with a reduction of the impedance value both at 30 days and at 60 days follow-up.
The significant increase in diuresis within the two months of treatment (VAS enrolment value of 5.65 ± 1.92, after 60 days increased to 8.15 ± 1.59 corresponding to a final 31% increase) probably confirms the fluid decrease in the adipose tissues.
The duplex-ultrasound evaluation of the subcutaneous adipose layers at the trochanteric level testify the reduction of the adipose tissue of slightly more than 20% at the end of the treatment in both limbs.
Among the limits of this study it is to be mentioned the 10% rate of drop-out of the enrolled patients and the lack of a control group (placebo or other therapy). Finally the limited sample of patients and the operator-dependance of volume measurements have to be taken into account.
Although higher doses or longer treatment period have not been taken in consideration, basically this study confirms the potential efficacy and safety of the nutraceutical multicomponent based on melilotus, birch, orthosiphon, pineapple and red vine leucocyanidine within the treatment of EFP or LA.
5. Conclusions
The subjects in this observational study have shown an overall improvement of the clinical and instrumental parameters after a 2-month treatment with the tested nutraceutical product. A good compliance to the treatment with a substantial absence of side effects was recorded as well. Within the due multi-faceted therapy of lipodystrophy of the lower limbs, a complementary oral supplementation with the above-mentioned nutraceutic, may ultimately constitute an interesting option in the still perfectible therapy of so-called “cellulite”.
Further clinical studies, possibly with larger numbers of patients and/or with a control group may potentially confirm the results of this preliminary observational study.
References
- Terranova, F., Berardesca, E. and Maibach, H. (2006) Cellulite: Nature and Aetiopathogenesis. International Journal of Cosmetic Science, 28, 175-190. http://dx.doi.org/10.1111/j.1467-2494.2006.00316.x
- Rawlings, A.V. (2006) Cellulite and Its Treatment. International Journal of Cosmetic Science, 28, 175-190. http://dx.doi.org/10.1111/j.1467-2494.2006.00318.x
- Hexsel, D., Orlandi, C. and Zechmeister do Prado, D. (2005) Botanical Extracts Used in Treatment of Cellulite. Dermatologic Surgery, 31, 866-872. http://dx.doi.org/10.1111/j.1524-4725.2005.31733
- Cornish, B.H., Chapman, M., Hirst, C., Mirolo, B.R., Bunce, H.I., Ward, L.C., et al. (2001) Early Diagnosis of Lymphoedema Using Multiple Frequency Bioimpedance. Lymphology, 34, 2-11.
- Cornish, B.H., Thomas, B.J., Mard, L.C., Hirst, C. and Bunce, I.H. (2002) A New Technique for the Quantification of Peripheral Edema with Application in both Unilateral and Bilateral Cases. Angiology, 53, 41-47. http://dx.doi.org/10.1177/000331970205300106
- Dittmar, M. (2003) Reliability and Variability of Bioimpedance Measures in Normal Adults: Effect of Age, Gender and Body Mass. American Journal of Physical Anthropology, 122, 361-370. http://dx.doi.org/10.1002/ajpa.10301
- Czerniec, S.A., Ward, L.C., Refshauge, K.M., Beith, J., Lee, M.J., York, S., et al. (2010) Assessment of Breast CancerRelated Arm Lymphedema—Comparison of Physical Measurement Methods and Self Report. Cancer Investigation, 28, 54-62. http://dx.doi.org/10.3109/07357900902918494
- Cavezzi, A., Shingale, F. and Elio, C. (2010) Limb Volume Measurement: From the Past Methods to Optoelectronic Technologies, Bioimpedance Analysis and Laser Devices. International Angiology, 29, 392-394.
- Bassas Grau, E. and Bassas Grau, M. (1964) Consideraciones clinicas etipopatogenicas y terapeuticas sobre la mal nomada “cellulitis”. Annals of Medicine, 16, 2-17.
- Lotti, T., Ghersetich, I., Grappone, C. and Dini, G. (1990) Proteoglycans in So Called Cellulite. International Journal of Dermatology, 29, 272-274. http://dx.doi.org/10.1111/j.1365-4362.1990.tb02560.x
- Ghersetich, I., Brazzini, B. and Lotti, T. (2002) La cellulite. Kosmè, 1, 27-33.
- Ryan, T.J. and Curri, S.B. (1989) The Development of Adipose Tissue and Its Relationship to the Vascular System. Clinics in Dermatology, 7, 1-8. http://dx.doi.org/10.1016/0738-081X(89)90038-2
- Merlen, J.F., Curri, S.B. and Sarteel, A.M. (1979) Cellulite, a Conjunctive Microvascular Disease. Phlebologie, 32, 279-282.
- Sato, A., Miura, H., Liu, Y., et al. (2002) Effect of Gender on Endothelium-Dependent Dilation to Bradykinin in Human Adipose Microvessels. American Journal of Physiology—Heart and Circulatory Physiology, 283, H845-H852.
- Orshal, J.M. and Khalil, R.A. (2004) Gender, Sex Hormones and Vascular Tone. American Journal of Physiology. Regulatory, Integrative and Comparative Physiology, 286, R233-R249. http://dx.doi.org/10.1152/ajpregu.00338.2003
- Querleux, B., Cornillon, C., Jolivet, O. and Bittoun, J. (2002) Anatomy and Physiology of Subcutaneous Adipose Tissue by in Vivo Magnetic Resonance Imaging and Spectroscopy: Relationship with Sex and Presence of Cellulite. Skin Research and Technology, 8, 118-124. http://dx.doi.org/10.1034/j.1600-0846.2002.00331.x
- Mirrashed, F., Sharp, J.C., Krause, V., Morgan, J. and Tomanek, B. (2004) Pilot Study of Dermal and Subcutaneous Structure by MRI in Individuals Who Differ in Gender, BMI and Cellulite Grading. Skin Research and Technology, 10, 161-168. http://dx.doi.org/10.1111/j.1600-0846.2004.00072.x
- Dodke, M.K., Di Bernardo, B., Thompson, C. and Usai, H. (2002) Assessment of Biomechanical Skin Properties: Is Cellulitic Skin Different? Aesthetic Surgery Journal, 22, 260-266. http://dx.doi.org/10.1067/maj.2002.124711
- Földi-Börcsök, E., Bedall, F. and Rahlfs, V.W. (1971) The Anti-Inflammatory and Anti-Edematous Effects of Coumarins from Melilotus officinalis. Arzneimittelforschung, 21, 2025-2030.
- Pastura, G., Mesiti, M., Saitta, M., Romeo, D., Settineri, N., Maisano, R., Petix, M. and Giudice, A. (1999) Lymphedema of the Upper Extremity in Patients Operated for Carcinoma of the Breast: Clinical Experience with Coumarinic Extract from Melilotus officinalis. Clinical Therapeutics, 150, 403-408.
- Pavan, R., Jain, S. and Kumar, A. (2012) Biotechnol Properties and Therapeutic Application of Bromelain: A Review. Biotechnology Research International, 2012, 1-6. http://dx.doi.org/10.1155/2012/976203
- Gründemann, C., Gruber, C.W., Hertrampf, A., Zehl, M., Kopp, B. and Huber, R. (2011) An Aqueous Birch Leaf Extract of Betula Pendula Inhibits the Growth and Cell Division of Inflammatory Lymphocytes. Journal of Ethnopharmacology, 136, 444-451. http://dx.doi.org/10.1016/j.jep.2011.05.018
- Germanò, M.P., Cacciola, F., Donato, P., Dugo, P., Certo, G., D’Angelo, V., Mondello, L. and Rapisarda, A. (2012) Betula Pendula Leaves: Polyphenolic Characterization and Potential Innovative Use in Skin Whitening Products. Fitoterapia, 83, 877-882. http://dx.doi.org/10.1016/j.fitote.2012.03.021
- Apostolou, A., Stagos, D., Galitsiou, E., Spyrou, A., Haroutounian, S., Portesis, N., Trizoglou, I., Wallace Hayes, A., Tsatsakis, A.M. and Kouretas, D. (2013) Assessment of Polyphenolic Content, Antioxidant Activity, Protection against ROS-Induced DNA Damage and Anticancer Activity of Vitis vinifera Stem Extracts. Food and Chemical Toxicology, 61, 60-68. http://dx.doi.org/10.1016/j.fct.2013.01.029
- Ameer, O.Z., Salman, I.M., Asmawi, M.Z., Ibraheem, Z.O. and Yam, M.F. (2012) Orthosiphon Stamineus: Traditional Uses, Phytochemistry, Pharmacology, and Toxicology. Journal of Medicinal Food, 15, 678-690. http://dx.doi.org/10.1089/jmf.2011.1973
NOTES

*Corresponding author.


