 International Journal of Clinical Medicine, 2011, 2, 224-230 doi:10.4236/ijcm.2011.23037 Published Online July 2011 (http://www.SciRP.org/journal/ijcm) Copyright © 2011 SciRes. IJCM Management of Oral Iron Chelator Deferasirox for Transfusion-Dependent Patients with Hematological Disorders: 2-Year Experience at a Single Institution in Japan Michihide Tokuhira, Morihiko Sagawa, Reiko Watanabe, Tomoe Nemoto, Takeshi Tomikawa, Shigehisa Mori, Masahiro Kizaki Department of Hematology, Saitama Medical Center, Saitama Medical University, Kawagoe, Japan. Email: tokuhira@saitama-med.ac.jp Received February 3rh, 2011; revised March 16th, 2011; accepted April 11th, 2011. ABSTRACT Introduction: Deferasirox is an oral iron chelator, approved worldwide for the treatment of chronic iron overload due to transfusion. Deferasirox was permitted two years ago in Japan, but there is little known regarding its efficacy and tolerability in clinical practice. Methods: We conducted a retrospective study of 18 patients with transfusion-dependent anemias treated by deferasirox at our institution. The starting dose was individualized and ranged from 6.4 to 26.3 mg/kg/day. Routine clinical laboratory data were followed, and serum ferritin was assessed every 4 weeks. Results: The mean serum ferritin level of 18 patients at the time of deferasirox induction was 3162 ng/ml. 10 of 18 patients could sustain deferasirox treatment for at least 6 months, at an average maintenance dose of 10.8 mg/kg/day. Serum ferritin reduction was observed in 4 patients, at doses less than 20 mg/kg/day. Eighty-nine percent of the patients had adverse events and 13 of them in all ultimately discontinued. Myelodysplastic syndrome (MDS) patients showed poor tolerabil- ity. Severe infections of grade 3 or more were documented in 6 patients, and 2 of them were fatal. Conclusions: The potential for beneficial iron chelation of deferasirox at less than the recommended 20-mg/kg dose was demonstrated. On the contrary, poor tolerability was documented, with adverse events such as severe infections, especially in MDS patients. Although it was not clearly demonstrated that deferasirox was responsible for impaired immunity, careful watching is required to administrate deferasirox. Keywords: Omponent, Formatting, Style, Styling, Insert 1. Introduction Patients with hematological disorders such as myelo- dysplastic syndrome (MDS) and aplastic anemia (AA) frequently require ongoing red blood cell (RBC) trans- fusion support. The frequent transfusions result in iron overload, leading to increased non-transferrin-bound iron, which generates toxic oxygen free-radicals and damage to multiple organs such as skin, liver, heart, pancreas, and other organs [1-3]. As previous observa- tions have demonstrated that severe iron overload is associated with increased morbidity and mortality, iron chelation therapy is important [4-6]. Deferoxamine (DFO) is a chelation medication that has been effective at achieving a negative iron balance when applied to patients with transfusion-related iron overload [7,8]. On the other hand, DFO requires intravenous injection or subcutaneous administration every day, resulting in im- paired quality of life. By contrast, deferasirox is a once-daily, oral iron chelator approved for the treatment of chronic iron overload due to blood transfusions [9-13]. The EPIC (Evaluation of Patients’ Iron Chelation with Exjade™) study indicated that the overall med ian serum ferritin level decreased significantly with deferasirox administration, both in chelation-naïve patients and those previously receiving chelation treatment [10]. The current treatment guidelines, based on these clinical studies and expert consensus, recommends chelation therapy for transfusion-dependent patients with serum ferritin levels > 1000 ng/mL, depending on the transfu- sion rate [14-16]. In Japan, deferasirox was approved  Management of Oral Iron Chelator Deferasirox for Transfusion-Dependent Patients with Hematological Disorders: 225 2-year Experience at a Single Institution in Japan about two years ago, and data such as efficacy and tol- erability have accumulated; however, there have been few publications regarding its use in clinical practice. In this retrospective study, we share our 2-year experience of deferasirox regarding clinical outcomes including advers e ev en ts (A Es ). 2. Methods Deferasirox was given to 18 hematological disorder pa- tients with serum ferritin levels of >1000 ng/mL due to frequent RBC transfusion. The starting dose was indi- vidualized according to blood transfusion frequency or age. Routine clinical laboratory data regarding CBC, liver, renal, and other functions were followed, and se- rum ferritin was assessed every 4 weeks. Efficacy was evaluated as a change in the serum ferritin level and clinical indicators (frequency of transfusion, skin pig- mentation, liver function, cardiac function, glycoalbu- min, etc., according to the organ damage in each patient). Safety was evaluated by monitoring of laboratory data and a medical physical exam. Dose adjustment was per- formed according to the physician’s discretion based on each patient’s clinical indicators. In the present study, there were no patients in whom the serum ferritin values fell to less than 500 ng/mL during the clinical course. All patients had received previous DFO administra- tion for iron chelation, but had not experienced success with it. In Japan, subcutaneous DFO injection is not permitted, and it is difficult to use intravenous infection every day for chelation. Instead, the patients in this study were given DFO in every transfusion, but the cu- mulative effect of the transfusions had outweighed its effect, resulting in gradually increased serum ferritin. AEs were graded according to the National Cancer In- stitute Common Toxicity Criteria (version 3). The inter- action between total RBC transfusion units and serum ferritin level in all patients was calculated by the Pear- son product-moment correlation coefficient using JMP software (Tokyo, Japan). The data of basal serum ferritin was collected just before deferasirox induction. Changes in serum ferritin levels and overall clinical improvement were analyzed in the 10 patients who could sustain de- ferasirox administration for at least 6 months. 3. Results 3.1. Patient Characteristics Data from 18 patients who received deferasirox at our institution were collected (Table 1). The sex ratio was 11:7 (male:female), and the median age at deferasirox initiation was 67 years (range: 44 - 82 years). The un- derlying diseases in patients were MDS (n = 10), AA (n = 4), myeloproliferative neoplasm (MPN; n = 3) and pure red cell aplasia (n = 1). The subtypes of MDS were refractory anemia (RA; n = 5), refractory anemia with excess blast (RAEB; n = 4), and refractory anemia with ring sideroblasts (RARS; n = 1). The median duration from diagnosis to deferasirox induction was 3.9 years (range: 0.7 - 17.7 years). The average total RBC transfu- sion already received before deferasirox initiation was Table 1. Patients’ characteristics. No Age Sex Diagnosis*1 Disease duratiion (years)*2 Total T/F dose (Units)*3 Serum Ferritin (ng/dl)*4 T/F require- ment*5 Complication*6 1 74 Female AA 3.4 170 6425 5 Skin, Herat 2 79 Female MDS(RA) 1.7 84 1806 4 Skin 3 65 Female MDS(RA) 2 60 1105 5 - 4 61 Male MDS(RARS) 5.6 146 4838 6 - 5 61 Female AA 0.7 66 1811 8 - 6 44 Male MDS(RA) 2 18 1247 2 - 7 82 Female MPN 4 191 6693 6 - 8 61 Male MDS(RAEB) 5.1 130 2561 9 Skin, Herat 9 69 Male AA 10.5 160 9423 6 Skin, Herat, DM, Liver 10 67 Male MPN 2.2 180 1440 14 Skin 11 67 Female AA 2.2 132 2327 4 Skin 12 67 Female MDS(RA) 3.4 102 2645 4 - 13 72 Male MDS(RAEB) 1 44 1265 7 - 14 71 Male PRCA 2.8 84 3980 6 - 15 63 Male MPN 17.7 206 1563 4 - 16 69 Male MDS(RAEB) 3.3 132 3328 6 - 17 75 Male MDS(RAEB) 1.8 116 2612 8 - 18 60 Male MDS(RA) 1 20 1851 6 - Mean 67 3.9 113.8 3162 6.1 *1. AA, aplastic anemia; MDS, myelodysplastic syndrome; RA, refractory an emia; RARS, refracto ry anemia with ring sideroblas ts; MPN, myeloproliferative neoplasm; RAEB, r ef ract or y ane mia wi th exces s blas ts ; PRCA, pu re red cell apl asi a. *2 . Dur ation from di agno sis t o th e time of d eferasirox induction. *3. Total recieving RBC dose until deferasirox. Copyright © 2011 SciRes. IJCM 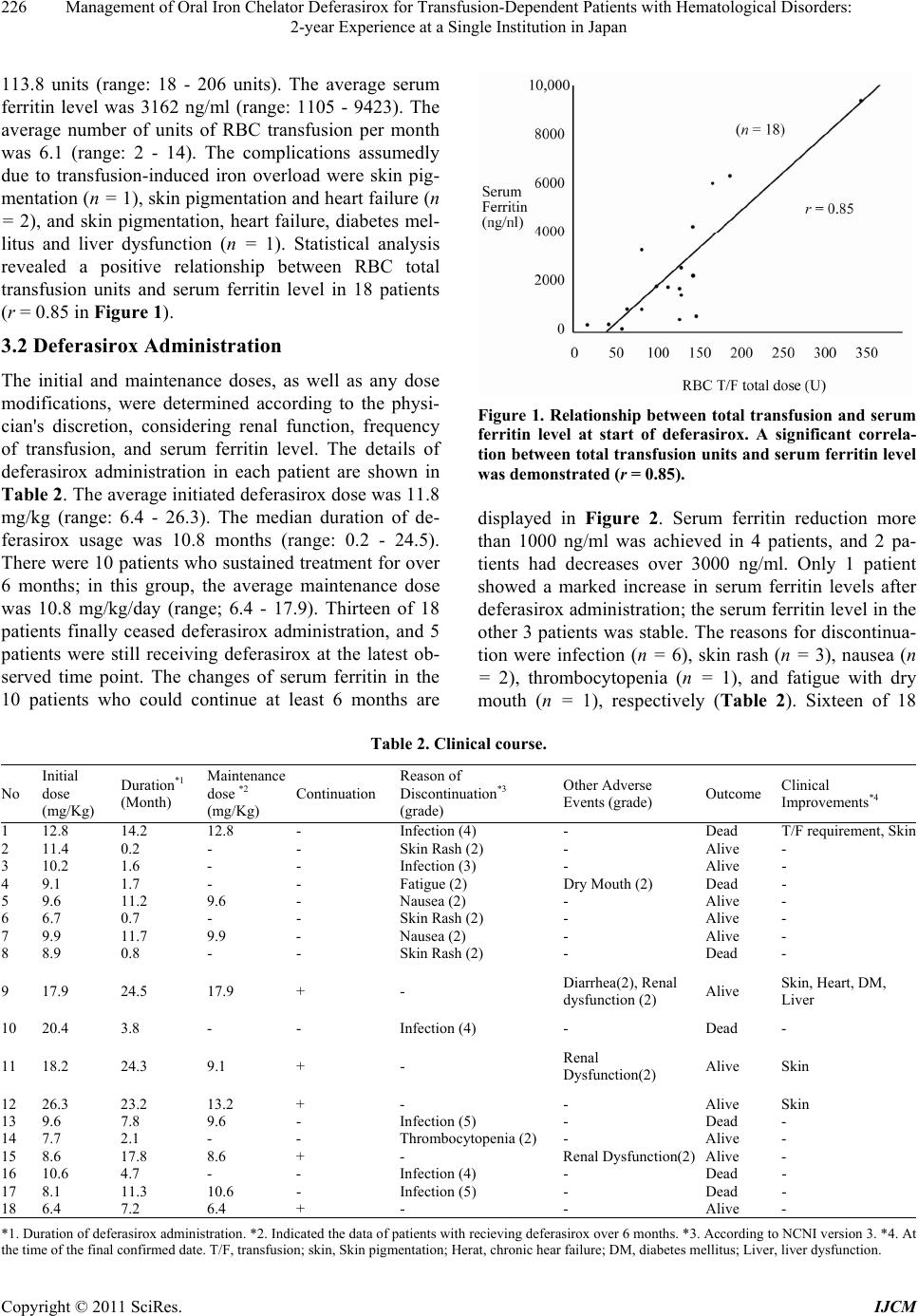 Management of Oral Iron Chelator Deferasirox for Transfusion-Dependent Patients with Hematological Disorders: 226 2-year Experience at a Single Institution in Japan 113.8 units (range: 18 - 206 units). The average serum ferritin level was 3162 ng/ml (range: 1105 - 9423). The average number of units of RBC transfusion per month was 6.1 (range: 2 - 14). The complications assumedly due to transfusion-induced iron overload were skin pig- mentation (n = 1), skin pigmentation and heart failure (n = 2), and skin pigmentation, heart failure, diabetes mel- litus and liver dysfunction (n = 1). Statistical analysis revealed a positive relationship between RBC total transfusion units and serum ferritin level in 18 patients (r = 0.85 in Figure 1). 3.2 Deferasirox Administration The initial and maintenance doses, as well as any dose modifications, were determined according to the physi- cian's discretion, considering renal function, frequency of transfusion, and serum ferritin level. The details of deferasirox administration in each patient are shown in Table 2. Th e av erag e in itiated d eferasirox do se was 11 .8 mg/kg (range: 6.4 - 26.3). The median duration of de- ferasirox usage was 10.8 months (range: 0.2 - 24.5). There were 10 patients who sustained treatment for over 6 months; in this group, the average maintenance dose was 10.8 mg/kg/day (range; 6.4 - 17.9). Thirteen of 18 patients finally ceased deferasirox administration, and 5 patients were still receiving deferasirox at the latest ob- served time point. The changes of serum ferritin in the 10 patients who could continue at least 6 months are Figure 1. Relationship between total transfusi on and serum ferritin level at start of deferasirox. A significant correla- tion between total transfusion units and serum ferritin level was demonstrated (r = 0.85). displayed in Figure 2. Serum ferritin reduction more than 1000 ng/ml was achieved in 4 patients, and 2 pa- tients had decreases over 3000 ng/ml. Only 1 patient showed a marked increase in serum ferritin levels after deferasirox administration; the serum ferritin level in the other 3 patients was stable. The reasons for discontinua- tion were infection (n = 6), skin rash (n = 3), nausea (n = 2), thrombocytopenia (n = 1), and fatigue with dry mouth (n = 1), respectively (Table 2). Sixteen of 18 Table 2. Clinical course. No Initial dose (mg/Kg) Duration*1 (Month) Maintenance dose *2 (mg/Kg) Continuation Reason of Discontinuation*3 (grade) Other Adverse Events (grade) Outcome Clinical Improvements*4 1 12.8 14.2 12.8 - Infection (4) - Dead T/F requirement, Skin 2 11.4 0.2 - - Skin Rash (2) - Alive - 3 10.2 1.6 - - Infection (3) - Alive - 4 9.1 1.7 - - Fatigue (2) Dry Mouth (2) Dead - 5 9.6 11.2 9.6 - Nausea (2) - Alive - 6 6.7 0.7 - - Skin Rash (2) - Alive - 7 9.9 11.7 9.9 - Nausea (2) - Alive - 8 8.9 0.8 - - Skin Rash (2) - Dead - 9 17.9 24.5 17.9 + - Diarrhea(2), Renal dysfunction (2) Alive Skin, Heart, DM, Liver 10 20.4 3.8 - - Infection (4) - Dead - 11 18.2 24.3 9.1 + - Renal Dysfunction(2) Alive Skin 12 26.3 23.2 13.2 + - - Alive Skin 13 9.6 7.8 9.6 - Infection (5) - Dead - 14 7.7 2.1 - - Thrombocytopenia (2) - Alive - 15 8.6 17.8 8.6 + - Renal Dysfunction(2) Alive - 16 10.6 4.7 - - Infection (4) - Dead - 17 8.1 11.3 10.6 - Infection (5) - Dead - 18 6.4 7.2 6.4 + - - Alive - *1. Duration of deferasirox administration. *2. Indicated the data of patients with recieving deferasirox over 6 months. *3. According to NCNI version 3. *4. At the time of the final confirme d date. T/F, transfusion; s kin, Skin pigmentation; Herat, chronic hear failure; DM, diabetes mellitus; Liver, liver dysfuncti on. Copyright © 2011 SciRes. IJCM 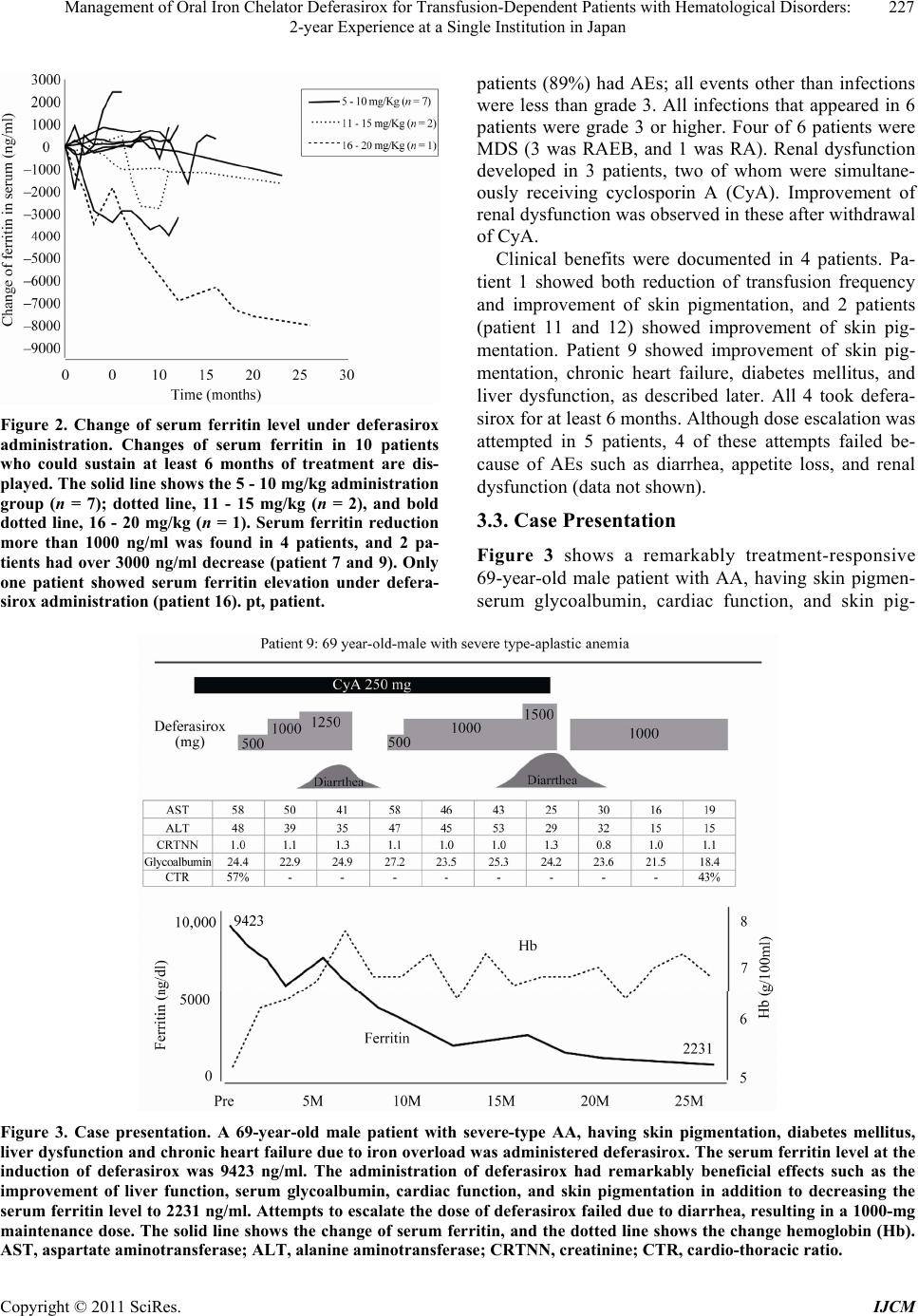 Management of Oral Iron Chelator Deferasirox for Transfusion-Dependent Patients with Hematological Disorders: 227 2-year Experience at a Single Institution in Japan Figure 2. Change of serum ferritin level under deferasirox administration. Changes of serum ferritin in 10 patients who could sustain at least 6 months of treatment are dis- played. The solid line shows the 5 - 10 mg/kg administration group (n = 7); dotted line, 11 - 15 mg/kg (n = 2), and bold dotted line, 16 - 20 mg/kg (n = 1). Serum ferritin reduction more than 1000 ng/ml was found in 4 patients, and 2 pa- tients had over 3000 ng/ml decrease (patient 7 and 9). Only one patient showed serum ferritin elevation under defera- sirox administration (patient 16). pt, patient. patients (89%) had AEs; all events other than infections were less than grade 3. All infections that appeared in 6 patients were grade 3 or higher. Four of 6 patients were MDS (3 was RAEB, and 1 was RA). Renal dysfunction developed in 3 patients, two of whom were simultane- ously receiving cyclosporin A (CyA). Improvement of renal dysfunction was observed in these after withdrawal of CyA. Clinical benefits were documented in 4 patients. Pa- tient 1 showed both reduction of transfusion frequency and improvement of skin pigmentation, and 2 patients (patient 11 and 12) showed improvement of skin pig- mentation. Patient 9 showed improvement of skin pig- mentation, chronic heart failure, diabetes mellitus, and liver dysfunction, as described later. All 4 took defera- sirox for at least 6 months. Although dose escalation was attempted in 5 patients, 4 of these attempts failed be- cause of AEs such as diarrhea, appetite loss, and renal dysfunction (data not shown). 3.3. Case Presentation Figure 3 shows a remarkably treatment-responsive 69-year-old male patient with AA, having skin pigmen- serum glycoalbumin, cardiac function, and skin pig- Figure 3. Case presentation. A 69-year-old male patient with severe-type AA, having skin pigmentation, diabetes mellitus, liver dysfunction and chronic heart failure due to iron overload was administered deferasirox. The serum ferritin level at the induction of deferasirox was 9423 ng/ml. The administration of deferasirox had remarkably beneficial effects such as the improvement of liver function, serum glycoalbumin, cardiac function, and skin pigmentation in addition to decreasing the serum ferritin level to 2231 ng/ml. Attempts to escalate the dose of deferasirox failed due to diarrhea, resulting in a 1000-mg maintenance dose. The solid line shows the change of serum ferritin, and the dotted line shows the change hemoglobin (Hb). AST, aspartate aminotransferase; ALT, alanine aminotransferase; CRTNN, creatinine; CTR, cardio-thoracic ratio. Copyright © 2011 SciRes. IJCM  Management of Oral Iron Chelator Deferasirox for Transfusion-Dependent Patients with Hematological Disorders: 228 2-year Experience at a Single Institution in Japan mentation in addition to decreasing the serum ferritin level to 2231 ng/ml. Dose escalation was attempted two times, but 1250 or 1500 mg/day administration failed, tation, diabetes mellitus, liver dysfunction and chronic heart failure due to iron overload. His serum ferritin level at the induction of deferasirox was 9423 ng/ml, and the administration of deferasirox had numerous benefi- cial effects such as the improvement of liver function, and the maintenance dose was defined as 1000 mg (17.9 mg/kg). Though the frequency of transfusion was not changed, the hemoglobin was gradually elevated from its baseline value, showing the effectiveness of defera- sirox. CyA was not effective and renal impairment was found, so that CyA had to be eventually stopped. Liver dysfunction, renal impairment, and diabetes mellitus were improved as indicated in Figure 3. Although he had a chronic heart failure leading to a wide cardio- thoracic ratio (CTR), deferasirox administration seemed to ameliorate the CTR, from 57% to 43%. 4. Discussion Deferasirox, a once-daily oral iron chelator, has been demonstrated in various studies worldwide involving large numbers of patients with a variety of transfu- sion-dependent anemias, with similar efficacy to DFO at comparable doses. A phase I and extended study in Ja- pan was developed with similar potential efficacy and safety as those seen in several western clinical trials [17]. However, there have been few publications regarding the outcome of its usage in clinical practice in Japan. To discuss this point, we collected and analyzed data from 18 patients who have been treated with deferasirox at our institution. In this retrospective study, there were several impor- tant findings regarding the usage of deferasirox in clini- cal practice. First, this study showed that lower doses (<20 mg/kg) had some extent of clinical efficacy. Pre- vious clinical trials have clearly demonstrated the dose-dependent iron-chelation effects of deferasirox administration, and have implied that at least 20 mg/kg is required for efficacy [18]. It was hard to evaluate this effect because there were no patients in the present study who could tolerate a dose greater than 20mg/kg; how- ever, 4 patients who received a dose lower than 20mg/kg showed decreased serum ferritin levels. In particular, the ferritin reductions in 2 patients (patient 7 and 9, receiv- ing 9.9 mg/kg and 17.9 mg/kg, respectively) were more than 3000 ng/ml as shown in Figure 2. Considering that these patients receive 6 units of RBC transfusion per month, deferasirox achieved sustained iron chelation even at lower-than-recommended doses. The clinical course of the latter patient was remarkable (Figure 3), showing improvement of multiple organs with iron che- lation, and 7000 ng/dl serum ferritin reduction. Second, the maintenance dose of deferasirox in 10 pa- tients who sustained at least 6 months of treatment was 10.8 mg/kg on averag e, less than those of the previously reported clinical studies. The 1-year EPI C study enrolled 1744 patients with transfusion-dependent anemias, of whom 79.6% completed 1 year of treatment; over 90% of these took doses more than 20 mg/kg [19]. In this study, we tried with 4 patients to escalate the deferasirox dose over 20 mg/kg, but these escalations failed due to AEs; for some reason, it was difficult to maintain the recommenced deferasirox dose in this study. The phase I clinical study of deferasirox on Japanese patients with transfusion-dependent anemias indicated that the phar- macokinetics–dose relationship was similar in Japanese and Caucasians [17]; hence, this phenomenon was not likely due to race. Third, the discontinuation rate (72%: 13 of 18 patients) was higher than those of previous studies. For instance, the phase I and extended study in Japan had a discon- tinuation rate of only 19% (4 of 21 patients) [17]. To understand this discrepancy, it is important to focus on the underlying disease. The EPIC study demonstrated that the incidence of AEs and discontinuation in MDS patient groups was higher than those in other anemia patient groups [19], and this study included 10 MDS patients (55 %), of whom 8 ultimately discontinued the medication because of AEs. In addition, with regard to subtypes of MDS, 2 of 5 MDS-RA patients could sustain deferasirox administration at the time of writing, and improvement of skin pigmentation and the reduction of serum ferritin was observed in patient 12; however, all 4 MDS-RAEB patients interrupted the medication because of AEs. Moreover, 3 of them developed severe infec- tions, resulting in fatality in 2 patients. Our data con- firmed current consensus-driven guidelines in deferasi- rox usage, recommending deferasirox usage for low-risk MDS patients, not fo r high-risk g roups su ch as RAEB or leukemic patients [20]. Meanwhile, it was surprising that the leading AE type in this study was infection, and that all of them were severe, i.e., grade 3 or more. It has been suggested that iron chelation therapies enhance immu- nity against infections, based on the fact that iron over- load promotes free radical tissue damage and organ fail- ure, undermines immune protection and facilitates pathogen invasion [21]. On the contrary, DFO, an iron chelator, is also a siderophore, which binds iron and transports it into microorganisms to help maintain their activity and grow, and thus exacerbates infections in various immunosuppressive settings [22]. Indeed, 46 of 59 dialysis patients who received DFO developed mu- Copyright © 2011 SciRes. IJCM  Management of Oral Iron Chelator Deferasirox for Transfusion-Dependent Patients with Hematological Disorders: 229 2-year Experience at a Single Institution in Japan cormycosis [23]. Of 1774 patients receiving deferasirox in the EPIC study, serious pyrexia (n = 51; 2.9%), pneumonia (n = 23; 1.3%), and sepsis (n = 20; 1.1%) were observed [17]. Furthermore, 26 deaths occurred in 341 MDS patients including 3 cases of septic shock [18]. Although deferasirox is not a siderophore so it is not clear-cut whether deferasirox might impair immunity against bacteria and fungus as DFO does, the results of the present study cannot rule out the possibility that de- ferasirox impairs immunity, given the fact that 2 non- MDS anemias developed (patient 1-AA and patient 9-MPN). Further clinical investigation and analysis of this issue will be required. In summary, the potential for beneficial iron chelation of deferasirox was demonstrated in patients receiving less than recommended 20-mg/kg dose in clinical prac- tice. On the other hand, deferasirox did not show clinical efficacy in high-risk MDS such as RAEB, as in other recent clinical studies. Poor tolerability was indicated by AEs, especially infections, 2 of which became fatal. Considering the fact that 2 non-MDS patients developed severe infection in this study, the possibility of defera- sirox-mediated impaired immunity cannot be ruled out. As this was a small study, further investigations in clinical practice are very important to analyze strategies focusing on the real efficacy of deferasirox among transfusion-dependent anemia patients, including ways of addressing the infection issue. REFERENCES [1] N. C. Andrews, “Disorders of Iron Metabolism,” The New England Journal of Medicine, Vol. 341, 1999, pp. 1986-1995. doi:10.1056/NEJM199912233412607 [2] J. P. Kushner, J. P. Porter and N. F. Olivieri, “Secondary Iron Overload,” The American Society of Hematology Education Program Book, Vol. 1, 2001, pp. 47-61. doi:10.1182/asheducation-2001.1.47 [3] M. Takatoku, T. Uchiyama, S. Okamoto, Y. Kanakura, et al., “Retrospective Nationwide Survey of Japanese Pa- tients with Transfusion-Dependent MDS and Aplastic Anemia Highlights the Negative Impact of Iron Overload on Morbidity/Mortality,” European Journal of Haema- tology, Vol. 78, No. 6, 2007, pp. 487-494. doi:10.1111/j.1600-0609.2007.00842.x [4] A. Shander and K. Sazama, “Clinical Consequences of Iron Overload from Chronic Red Blood Cell Transfusions, Its Diagnosis, and Its Management by Chelation Ther- apy,” Transfusion, Vol. 50, No. 5, 2010, pp. 1144-1155. doi:10.1111/j.1537-2995.2009.02551.x [5] L. Malcovati, “Impact of Transfusion Dependency and Secondary Iron Overload on the Survival of Patients with Myelodysplastic Syndromes,” Leukemia Research, Vol. 31, No. S3, 2007, pp. S2-S6. doi:10.1016/S0145-2126(07)70459-9 [6] E. N. Oliva, F. Ronco, A. Marino, C. Alati, G. Praticò and F. Nobile, “Iron Chelation Therapy Associated with Im- provement of Hematopoiesis in Transfusion-Dependent Patients,” Transfusion, Vol. 50, No. 7, 2010, pp. 1568- 1570. doi:10.1111/j.1537-2995.2010.02617.x [7] P. J. Giardina and R. W. Grady, “Chelation Therapy in Beta-Thalassemia: The Benefits and Limitations of Des- ferrioxamine,” Seminars in Hematology, Vol. 32, No. 4, 1995, pp. 304-312. [8] V. Gabutti and A. Piga, “Results of Long-Term Iron- Chelating Therapy,” Acta Haematologica, Vol. 95, No. 1, 1996, pp. 26-36. doi:10.1159/000203853 [9] M. D. Cappellini, A. Cohen, A. Piga, M. Bejaoui, et al., “A Phase 3 Study of Deferasirox (ICL670), a Once-Daily Oral Iron Chelator, in Patients with β-Thalassemia,” Blood, Vol. 107, No. 9, 2006, pp. 3455-3462. doi:10.1182/blood-2005-08-3430 [10] J. Porter, R. Galanello, G. Saglio, E. J. Neufeld, et al., “Relative Response of Patients with Myelodysplastic Syndromes and Other Transfusion-Dependent Anaemias to Deferasirox (ICL670): A 1-yr Prospective Study,” Eu- ropean Journal of Haematology, Vol. 80, No. 2, 2008, pp. 168-176. doi:10.1111/j.1600-0609.2007.00985.x [11] N. Gattermann, C. Finelli, M. D. Porta, P. Fenaux, A. Ganser, A. Guerci-Bresler, M. Schmid, K. Taylor, D. Vassilieff, D. Habr, G. Domokos, B. Roubert and C. Rose, “Deferasirox in Iron-Overloaded Patients with Transfu- sion-Dependent Myelodysplastic Syndromes: Results from the Large 1-Year EPIC Study,” Leukemia Research, Vol. 34, No. 9, 2010, pp. 1143-1150. doi:10.1016/j.leukres.2010.03.009 [12] F. Wimazal, T. Nosslinger, C. Baumgartner, W. R. Sperr, M. Pfeilstocker and P. Valent, “Deferasirox Induces Re- gression of Iron Overload in Patients with Myelodysplas- tic Syndromes,” European Journal of Clinical Investiga- tion, Vol. 39, No. 5, 2009, pp. 406-411. doi:10.1111/j.1365-2362.2009.02108.x [13] D. Chirnomas, A. L. Smith, J. Braunstein, Y. Finkelstein, L. Pereira, A. K. Bergmann, F. D. Grant, C. Paley, M. Shannon and E. J. Neufeld, “Deferasirox Pharmacokinet- ics in Patients with Adequate versus Inadequate Re- sponse,” Blood, Vol. 114, No. 19, 2009, pp. 4009-4013. doi:10.1182/blood-2009-05-222729 [14] J. M. Bennett, “Consensus Statement on Iron Overload in Myelodysplastic Syndromes,” American Journal of He- matology, Vol. 83, No. 11, 2008, pp. 858-861. doi:10.1002/ajh.21269 [15] T. Suzuki, M. Tomonaga, Y. Miyazaki, S. Nakao, K. Ohyashiki, I. Matsumura, Y. Kohgo, Y. Niitsu, S. Kojima and K. Ozawa, “Japanese Epidemiological Survey with Consensus Statement on Japanese Guidelines for Treat- ment of Iron Overload in Bone Marrow Failure Syn- dromes,” International Journal of Hematology, Vol. 88, No. 1, 2008, pp. 30-35. doi:10.1007/s12185-008-0119-y Copyright © 2011 SciRes. IJCM  Management of Oral Iron Chelator Deferasirox for Transfusion-Dependent Patients with Hematological Disorders: 2-year Experience at a Single Institution in Japan Copyright © 2011 SciRes. IJCM 230 [16] National Comprehensive Cancer Network (NCCN) Clini- cal Practice Guidelines in Oncology Version 1, “Myelo- dysplastic Syndromes; 2009,” 2009. http://www.nccn.org/professionals/physician gls/pdf/mds.pdf. [17] K. Miyazawa, K. Ohyashiki, A. Urabe, T. Hata, S. Nakao, K. Ozawa, T. Ishikawa, J. Kato, Y. Tatsumi, H. Mori, M. Kondo, J. Taniguchi, H. Tanii, L. Rojkjaer and M. Omine, “A Safety, Pharmacokinetic and Pharmacodynamic In- vestigation of Deferasirox (Exjade, ICL670) in Patients with Transfusion-Dependent Anemias and Iron-Overload: A Phase I Study in Japan,” International Journal of He- matology, Vol. 88, No. 1, 2008, pp. 73-81. doi:10.1007/s12185-008-0115-2 [18] A. Taher, A. El-Beshlawy, M. S. Elalfy, K. Al Zir, S. Daar, D. Habr, U. Kriemler-Krahn, A. Hmissi and A. Al Jefri, “Efficacy and Safety of Deferasirox, an Oral Iron Chelator, in Heavily Iron-Overloaded Patients with Beta-Thalassaemia: The ESCALATOR Study,” European Journal of Haematology, Vol. 82, No. 6, 2009, pp. 458-465. doi:10.1111/j.1600-0609.2009.01228.x [19] M. D. Cappellini, J. Porter, A. El-Beshlawy, C. K. Li, J. F. Seymour, M. Elalfy, N. Gattermann, S. Giraudier, J. W. Lee, L. L. Chan, K. H. Lin, C. Ros e, A . Taher, S . L. Thein, V. Viprakasit, D. Habr, G. Domokos, B. Roubert and A. Kattamis, “Tailoring Iron Chelation by Iron Intake and Serum Ferritin: The Prospective EPIC Study of Deferasi- rox in 1744 Patients with Transfusion-Dependent Ane- mias,” Haematologica, Vol. 95, No. 4, 2010, pp. 557-566. doi:10.3324/haematol.2009.014696 [20] J. M. Bennett, “Consensus Statement on Iron Overload in Myelodysplastic Syndromes,” American Journal of He- matology, Vol. 83, No. 11, 2008, pp. 858-861. doi:10.1002/ajh.21269 [21] J. R. Boelaert, J. Van Cutsem, M. de Locht, Y. J. Schnei- der and R. R. Crichton, “Deferoxamine Augments Growt h and Pathogenicity of Rhizopus, While Hydroxypyridi- none Chelators Have No Effect,” Kidney International, Vol. 45, No. 45, 1994, pp. 667-671. doi:10.1038/ki.1994.89 [22] H. Keberle, “The Biochemistry of Desferrioxamine and Its Relation to Iron Metabolism,” Annals of the New York Academy of Sciences, Vol. 119, 1964, pp. 758-768. doi:10.1111/j.1749-6632.1965.tb54077.x [23] J. R. Boelaert, M. de Locht, J. Van Cutsem, V. Kerrels, B. Cantinieaux, A. Verdonck, H. W. Van Landuyt and Y. J. Schneider, “Mucormycosis during Deferoxamine The- rapy is a Siderophore-Mediated Infection. in vitro and in vivo Animal Studies,” The Journal of Clinical Investiga- tion, Vol. 91, No. 5, 1993, pp. 1979-1986. doi:10.1172/JCI116419
|