Journal of Materials Science and Chemical Engineering
Vol.03 No.07(2015), Article ID:58045,6 pages
10.4236/msce.2015.37026
Breaking the Adhesive Bond between Dialyll Phthlate, Barco Bond 185 and PBX 9501
Matt Jackson1, Benton Allen1, Trent Kelly1, Courtney Waddell1, Emily M. Hunt1*, Stephanie Steelman2, Neil Koone2
1West Texas A & M University, Canyon, Texas, USA
2CNS Pantex, Amarillo, Texas, USA
Email: *ehunt@wtamu.edu


Received 6 May 2015; accepted 9 July 2015; published 16 July 2015

ABSTRACT
Use of epoxy as an adhesive is a common practice. The most common applications are permanent sealants. Epoxies have a wide range of operating temperatures, and are very resistance to adhesive failure. When a need to remove this adhesive arises, it is not always easily accomplished especially if the part has excessive adhesive. To maintain fidelity of the parts attached by epoxy, a project evaluating several methods of epoxy removal was conducted. Methods evaluated included low wavelength, near-ultraviolet radiation, solvent dissolution, and thermal cycling. The UV method failed to demonstrate a repeatable dissociation. The solvent study did result in dissociation of bonds, but introduced chemicals that could make subsequent chemical analysis of parts suspect. Thermal cycling showed a high repeatability for dissociation of bonds and may prove to be relatively inexpensive to implement.
Keywords:
Barco Bond, Bond Dissociation, Cyrogenic Cycling, Dialyll Phthlate, Epoxy Bond, Epoxy Degredation, Epoxy Dissociation, Thermal Cycling, PBX 9501, Plastic Bonded Explosive, Thermal Cycling

1. Introduction
Adhesive bonding refers to the practice of applying a thin coat of material between material surfaces to create either a soluble or an insoluble bond. Epoxy adhesives are typically a two-part product with an epoxy group at both ends (Figure 1). When activated, epoxies form a high strength bond that is very hard to break. Epoxies are commonly used in weapon assembly to produce lasting, high strength bonds. However, when these weapons must be disassembled and refurbished, the epoxy bonds can be hard to break without damaging high fidelity parts. This problem is especially prevalent in parts that have aged for long periods of time.
Figure 1. Epoxy group.
In July 2013, B & W Pantex initiated a rapid response Plant Directed Research and Development project in collaboration with the West Texas A & M University (WTAMU) Engineering and Computer Science Department. The study focused on designing a three-prong approach project to identify a method to cause adhesive failure of the epoxy, BARCO BOND 185. A successful project would yield a method for Pantex Plant production technicians to separate the explosive PBX 9501 from a glass-reinforced polymer called diallyl phthalate (DAP) at B & W Pantexfacilities in a safe and efficient manner. The project identified three possible methods to cause the failure of Astro Chemical’s BARCO BOND MB-185 epoxy and subsequent separation of the DAP and PBX 9501 material. The three methods included exposure to solvents, exposure to radiation in the ultraviolet and near visible range, end exposure to thermal cycles.
1.1. Background
Plastic bonded explosives are comprised of a high explosive and a binder. PBX 9501 is made from 95% by weight high explosive, cyclotetramethylene-tetranitramine (HMX); 2.5% by weight binder, Estane; and 2.5% by weight nitroplasticizer [1]. The nature of plastic bonded explosives lends their reliability, or safety, to be highly dependent on their environment. Plastic bonded explosives are susceptible to decomposition at high temperatures and fracturing due to mechanical shock [2]. Due to safety concerns regarding the sensitivity of the explosives, many common practices used to remove epoxy from ordinary surfaces. Because of safety concerns, techniques such as heat gun treatment, infrared radiation, grinding, and prying require detailed study before incorporating into disassembly procedures. While the introduction of high temperatures for thermal cycling of PBX 9501, DAP, and epoxy assemblies result in heat flow into the plastic bonded explosive, low temperature thermal cycling results in the removal of thermal energy, and is able to significantly affect the mechanical integrity of plastic bonded explosives. This makes epoxy-breaking methods that utilize low temperature thermal cycling ideal for this application.
1.2. Theory
As a material undergoes changes in temperature, it expands or contracts according to its coefficient of thermal expansion (CTE), which is a material’s tendency to change in volume or length based on the material’s change in temperature. Different materials have different CTEs, causing them to expand or contract by a different proportion while undergoing the same temperature change. When materials with different CTE are bonded and subjected to temperature changes, the differential expansion or contraction of the materials causes mechanical stresses in the region of the bond. The stresses caused by the expansion or contraction of the materials cause bonds to fatigue and or one or more of the materials in the bonding region to fail as a function of repeated exposure to temperature gradients [2]. This principal forms the basis for the thermal cycling method employed in these experiments to weaken the epoxy and ultimately break the epoxy bonds. Several low temperature thermal cycling methods were tested, but cryogenic thermal cycling by exposure to liquid nitrogen was the most effective method. To control application of the liquid nitrogen to the fabricated substrates, tests utilized a Brymill CRY-Ac® typically used by physicians to freeze tissue on patients (Figure 2). Experiments showed that composite assemblies regularly dissociated upon thermal cycling using the Brymill CRY-Ac® to apply liquid nitrogen.
To model temperature gradients within materials, tests performed by WTAMU determined the efficiency of the Brymill CRY-Ac® at transferring heat from an assembly and measured temperature gradients within a slab of material. Analytical and numerical analyses described the mechanism responsible for thermal cycling dissociation within assemblies, efficiency of heat removal by the Brymill CRY-Ac® and differing temperature gradients within the materials in a composite assembly.
The coefficient of thermal expansion (CTE) describes thermal expansion (or contraction) of a material due to
Figure 2. Brymill CRY-Ac®.
a temperature change, this calculation is show in Equation (1).
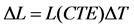 (1)
(1)
where DL is the change in length,
L is the original length,
CTE is the coefficient of linear expansion, and
DT is the change in temperature.
Liquid nitrogen can be applied to objects to create a large change in temperature. This temperature gradient induced change in volume creates a strain in the interface of the materials, represented by Equation (2).
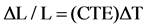 (2)
(2)
where DL/L is strain.
If known, the coefficient of linear expansion in Equation (2) yields the material strain for the −216 K temperature change. Table 1 lists the coefficients of linear expansion and strains for a temperature change of −216 K.
When materials experience a reduction in temperature, a negative expansion occurs (the material contracts). When an assembly consisting of DAP epoxied to PBX 9501 experiences a temperature drop of 216 K, all three materials contract. This results in all materials experiencing tension. PBX 9501 typically fails under tension at a small fraction of the strain associated with a temperature drop of 216 K [7]. Since a portion of the PBX 9501 shrinks by 1.2% when exposed to a sharp temperature change, adjacent PBX 9501 [7] undergoes significant strain. A tensile failure near the temperature gradient provides a repeatable mechanism for dissociating epoxy bonds between DAP and PBX 9501 [7]. Cellular PVC was selected as a surrogate material for PBX 9501 because cellular PVC has a comparable CTE to PBX 9501 (Table 1); thus, it is reasoned that successful dissociation of epoxy bonds between PVC and DAP behave comparably to epoxy bonds between PBX 9501 and DAP.
2. Materials and Setup
BARCO BOND MB-185 two-part epoxy; (Part A composed of 4.4'-Isopropylidenediphenol-Epichlorohydrin Copolymer and Silica, Amorphous, Fumed 7; and Part B composed of ditetrahydrofurfuryl phthalate and two proprietary catalysts), was supplied by Astro Chemical. Dialyll phthalate (DAP) was provided in granular form by Sumitomo Bakelite. Polyvinyl chloride (PVC) polymer board was provided by Royal Mouldings as a surrogate for PBX 9501. PVC possesses a coefficient of thermal expansion, or CTE, of 5.4 × 10−4 length/length K [6], [6] that is very close to the CTE of 5.5 × 10−4 length/length K for PBX 9501, [8].
A Brymill Cry-Ac-3® liquid nitrogen-dispensing unit (Cry-Ac) was used to apply liquid nitrogen. The Cry-Ac unit consists of a 300 mL vacuum insulated chamber, fitted with a withdrawal tube and nozzle liquid nitrogen dispensing system. The unit dispenses a spray of liquid nitrogen by utilizing pressure inside of the unit that builds as vaporization of the liquid nitrogen occurs. A nozzle with an aperture diameter of 0.04in was used for the experiments. The nature of the device dictates a variable flow of liquid nitrogen based on the pressure in the tank. A spring operated automatic release valve controls the pressure in the tank.
Several setups were used in the experiments to replicate the different substrate geometries. To replicate two dimensional bonds, a disk-slab geometry was created using a DAP disk formed in the Simplimet 1000 Automatic Mounting Press. A thin layer of epoxy was applied to the DAP disk which was then bonded to a PVC slab
Table 1. Coefficient of thermal expansion.
by heavy finger pressure (approximately 45 Newtons) for a period of 20 seconds. The slab and disk were then cured in an oven for 3 hours at 80˚C. To replicate three-dimensional bonds, a slot was cut in the DAP disks and a rectangular prism was epoxied inside the slot. These substrate geometries are shown in Figure 3. To account for various glue patterns, epoxy was applied to the entire bonded surface on some samples and only in two drop patterns on other samples. The glue patterns for three dimensional bonding patterns are shown in Figure 4.
3. Experimental Procedures
The Cry-Ac was used to spray liquid nitrogen either along one long seam between the PVC and DAP (denoted as single in Table 1) or along both long seams (denoted as double). After applying the liquid nitrogen, the sample warmed back up to room temperature and the bond was tested with light finger strength (tested to be less than five pounds force with a spring scale). If the bond held, the cycle was repeated until failure. The results of this test are denoted in Table 2.
4. Results and Discussion
Table 2 and Table 3 document the results from the thermal cycling experiments. In two dimensional bonding pattern tests, the bond in all samples was broken after relatively few thermal cycles. In the three dimensional bonding pattern tests, One-drop glue patterns required only a few cycles of liquid nitrogen spray to induce epoxy failures. On the excessive epoxy samples, 14 spray cycles of 10 seconds did not cause epoxy failure in the worst-case scenario. These results led to the submerging of the two fully covered samples. This full submersion produced breakage after 2 and 4 cycles. This suggests the ability of thermal cycling to break even the worst-case bond scenarios as long as the entire geometry reaches extremely low temperatures. Based on the experimental results, cryogenic cycling provides an acceptable method for breaking BARCO BOND epoxy bonds between DAP and PBX 9501. With the exception of one test, each DAP-PVC substrate tested successfully separated through cryogenic thermal cycles of liquid nitrogen. The data show the number of cycles required to break the bond does show correlation with the amount of epoxy applied to each sample. Fully coated samples required full submersion in liquid nitrogen to break the epoxy bonds. Application of liquid nitrogen by way of a Cry-Ac® was demonstrated to be an effective means for delivering the drastic temperature gradient required to break the epoxy bonds. Samples F2 and F3 in Table 3 refer to the samples with a full coverage of epoxy.
5. Conclusion
Initially, ultraviolet radiation was identified as a potential method for weakening epoxy bonds. Testing based on the UV experiment indicates the ineffectiveness of exposure to ultraviolet radiation at weakening the BARCO BOND even though it is not a UV stabilized epoxy. Several solvents were demonstrated to be effective methods for weakening the epoxy bonds and in some cases completely dissolving the BARCO BOND. Unfortunately, due to their corrosiveness, flammability, and tendency to degrade the DAP and PBX 9501, as well as the extended time required to dissolve the BARCO BOND, the solvents are unsuitable for application in processes at Pantex Plant. Cryogenic thermal cycling proved successful as an effective method for breaking the BARCO BOND resulting in the separation of the PBX 9501 from the DAP. Cryogenic thermal cycling through the application of liquid nitrogen appears to be a suitable method to incorporate into processes at Pantex Plant.
6. Recommendations
The successful degredation of epoxy bonds by thermal cycling provides a positive indication of potential success
Figure 3.Two dimensional (left) and three dimensional (right) bonding pattern substrates.
Figure 4.Two drop (left) and complete (right) epoxy patterns.
Table 2. Two dimensional bonding pattern thermal cycling results.
Table 3. Three dimensional bonding pattern thermal cycling results.
on weapon assemblies. Future work entails physics-based calculations to verify the safety of the process on PBX 9501, and the setup of a remote system to use the CRY-Ac® on the weapon disassembly processes at B & W Pantex. Further work has been done regarding the heat transfer rates of the Brymill Cry-Ac® and will be published at a later date.
Acknowledgements
This project would not have been accomplished in such a timely manner without support from Gregg Chambliss and Kathy Mitchell providing assistance to obtain funding for a mid-year start Plant Directed Research and Development (PDRD) project. The students that worked on this project did a fantastic job collaborating with Pantex and developing ideas to make the project succeed.
Cite this paper
Matt Jackson,Benton Allen,Trent Kelly,Courtney Waddell,Emily M. Hunt,Stephanie Steelman,Neil Koone, (2015) Breaking the Adhesive Bond between Dialyll Phthlate, Barco Bond 185 and PBX 9501. Journal of Materials Science and Chemical Engineering,03,196-201. doi: 10.4236/msce.2015.37026
References
- 1. Yeager, J.D., Ramos, K.J., Pesce-Rodriquez, R.A. and Piraino, S.M. (2013) Microstructural Effects of Processing in the Plastic-Bonded Explosive Composition A-3. Materials Chemistry and Physics, 139, 305-313. http://dx.doi.org/10.1016/j.matchemphys.2013.01.041
- 2. Yan, Q.-L., Svatopluk, Z. and Elbeih, A. (2012) Recent Advances in Thermal Analysis and Stability Evaluation of Insensitive Plastic Bonded Explosives (PBXs). Thermochi-mica Acta, 537, 1-12. http://dx.doi.org/10.1016/j.tca.2012.03.009
- 3. Shackelford, J.F. and Alexander, W. (2012) CRC Materials Science and Engineering Handbook. 3rd Edition, CRD Press, Boca Raton, 2000.
- 4. Sumitomo Bakelite North America Inc. Sumitomo Bakelite North America 52-01 Short-Glass Reinforced Diallyl Phthalate.
- 5. Dobratz, B.M. and Crawford, P.C. (1985) LLNL Explosives Handbook. Lawrence Livermore National Laboratory, Livermore.
- 6. Royal Building Products (2014) Royal Decorative Moulding.
- 7. Suelflow, W. (2013) PBX 9501 Mechanical Properties Cycle Report Data. B & W Pantex, Amarillo.
- 8. Thompson, D.G. (2009) Thermal Expansion of PBX 9501 and PBX 9502 Plastic Bonded Explosives.
NOTES

*Corresponding author.






