Journal of Materials Science and Chemical Engineering
Vol.03 No.06(2015), Article ID:57051,6 pages
10.4236/msce.2015.36002
Synthesis of (2S,4S)-2-Substituted-3- (3-Sulfanylpropanoyl)-6- Oxohexahydropyrimidine-4-Carboxylic Acids as Potential Antihypertensive Drugs
Andrei Ershov*, Dmitry Nasledov, Igor Lagoda, Valery Shamanin
Institute of Macromolecular Compouns of Russian Academy of Sciences, Saint Petesburg, Russia
Email: *ershov305@mail.ru


Received January 2015

ABSTRACT
Proceeding from natural amino acid L-asparagine and commercially available aldehydes a stereoselective synthesis was developed of (2S,4S)-2-alkyl(aryl)-3-(3-sulfanylpropanoyl)-6-oxohexahy- dropyrimidine-4-carboxylic acids, potential antihypertensive drugs, inhibitors of the angiotensin converting enzyme.
Keywords:
6-Oxohexahydropyrimidine-4-Carboxylic Acids, Inhibitors of the Angiotensin Converting Enzyme

1. Introduction
Efficient antihypertensive drugs function as inhibitors of the angiotensin converting enzyme (ACE). This enzyme is involved in the regulator system rennin-angiotensin-aldosterone, and the distortion of its operation results in the majority of hypertensive human diseases [1]-[3]. The first of the drugs from this series was captopril, (S)-N-(3-sulfanyl-2-methyl-1-oxopropyl)-L-proline [4]. The later research on development of antihypertensive substances, ACE inhibitors, was directed to the replacement of the natural amino acid L-proline in the captopril molecule by synthetic cyclic amino acids of the heterocyclic series, among them derivatives of 1,3-oxazolidine [5], 1,3-thiazolidine [6], pipecoline [7], quinazoline [8], indole [9], and azepine [10]. In the series of the saturated pyrimidine derivatives the only example of the inhibitor activity with respect to ACE is known to be shown by 3-(3-sulfanylpropanoyl)-6-oxohexahydropyrimidine-4-carboxylic acid [11].
It was found by the analysis of published data that sodium (potassium) salts of 2-substituted 6-oxohexahydropyrimidine-4-carboxylic acids formed in the reaction of the natural amino acid L-asparagine with carbonyl compounds in alkaline medium. This reaction was investigated mainly by an example of derivatives of isobutyric, trimethylacetic, and benzoic aldehydes [12]-[16]. The process was found to occur with a high stereoselectivity giving prevailingly the spatial isomer with (2R,4S)-configuration of the substituents in the pyrimidine ring.
The derivatives of 6-oxohexahydropyrimidine-4-carboxylic acids are used as key intermediates in the synthesis of β-amino acids [12] [13], in the asymmetric cycloaddition reactions [14], in liposomal microencapsulation of biopreparations [15].
2. Results and Discussion
The goal of this study is the search for new and synthetically accessible potential antihypertensive substances, ACE inhibitors, compounds where the cyclic amino acid fragment is a derivative of pyrimidine-4-carboxylic acid.
Compounds 2а-i were obtained in 70% - 90% yields after maintaining L-asparagine, an appropriate aliphatic or aromatic aldehyde, and equivalent quantity of sodium hydroxide in methanol solution for 10 - 12 h at 25˚С (see Scheme 1 and Table 1).
The first objects of our investigation were the condensation products of L-asparagine with a series of aliphatic aldehydes, compounds 2а-f. The cyclic pyrimidine structure of these compounds is unquestionable, as shows the appearance in the 1Н NMR spectra of the typical АВХ system due to the diastereotopic character of Н-5 and Н-4 protons, of the signals of Н-2 atom in the region 4.2 - 4.5 ppm, and in the 13С NMR spectra, of the signal of sp3- hybridized С-2 atom in the region 65 - 70 ppm. In the spectra of all obtained compounds 2а-f two sets of resonance signals from (2R,4S)-and (2S,4S)-stereoisomeric forms of the pyrimidine ring are observed. The assign- ment of the stereoisomeric forms of compounds 2а-f is based on the formerly established criteria and rules ob-
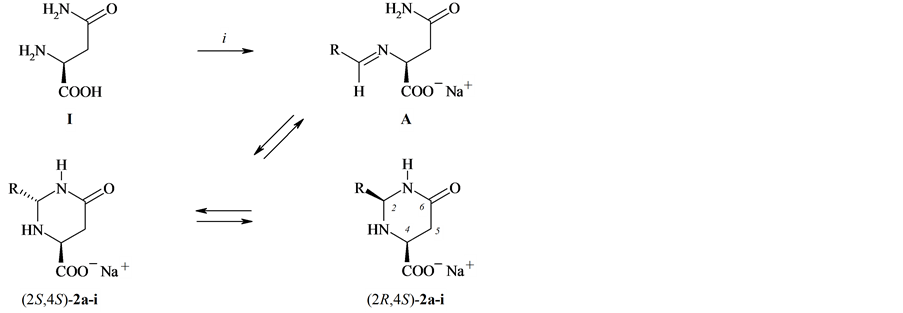
Scheme 1. i: RCH=O, NaOH/MeOH, 25˚C, 10 - 12 h. R = Me (a), Et (b), Bu (c), i-Pr (d), i-Bu (e), CH2CH2Ph (f), R =XC6H4, X = H (g), 4-Cl (h), 4-MeO (i).
Table 1. Characteristics of compounds 2a-i.
tained at the use of 1Н and 13С NMR spectroscopy [13]-[15], and also on the data of X-ray diffraction analysis [13] [16]. In keeping with the comparison of the 1Н and 13С NMR spectra of compounds 2а-f to the analogous spectral characteristics published in [13]-[16] the (2R,4S)-configuration was assigned to the main isomer. In the 1Н NMR spectrum of this stereoisomer the protons Н-5 and Н-4 are characterized by larger spin-spin coupling constant (JAВ = 17 Hz, JAX + JBX = 16 Hz), and the signals of Н-5 and Н-4 protons are considerably shifted upfield than the analogous signals of the stereoisomer with (2S,4S)-configuration. The corresponding coupling constant in the latter isomer is smaller (JAВ = 15 Hz, JAX + JBX = 13 Hz).
The position of the configurational equilibrium of compounds 2а-f is shifted to a large extent to (2R,4S)-ste- reoisomer, therefore it is impossible to obtain a clear correlation between the logarithms of the constant of the configurational equilibrium and the Taft steric constants of alkyl substituents. The largest fraction of the minor (2S,4S)-isomer (13%) was observed in the D2O solution of compound 2e, isovaleric aldehyde derivative.
In the 1Н and 13С NMR spectra in D2O of the L-asparagine condensation products with aromatic aldehydes (compounds 2g-i) alongside the signals of two configuration isomers of the pyrimidine form signals appear of a linear form А. Not entering into details of the previously found by us [17] spectral distinctions between the cyclic and linear forms of compounds 2g-i we only mention that the typical signs of the latter in the 1Н NMR spectrum is a significant downfield shift of the protons of СН2СН group as compared with analogous signals Н-5 and Н-4 of cyclic forms, the appearance of the proton signal of the azomethine group at 8.25 - 8.35 ppm, and in the 13С NMR spectrum, of the downfield signal at 165 ppm (C=N).
By an example of compound 2g, the condensation product of L-asparagine with benzaldehyde, we studied the dependence of the position of the tautomeric equilibrium on the nature of the applied solvent (see Table 2). In the crystalline state compound 2g exists in the cyclic pyrimidine (2R,4S)-form as confirmed by the solid state 13С NMR spectrum [17]. In all solvents the coexistence of tautomeric forms was observed, and in going to aprotic polar solvents (DMSO-D6 and DMF-D7) the fraction of the linear form A significantly increased.
Hence the products of L-asparagine condensation with acetic, propionic, valeric, isobutyric, isovaleric, and hydrocinnamic aldehydes in alkaline medium have the cyclic pyrimidine structure, and in D2O solutions they are present as two cyclic spatial stereoisomers with a significant prevalence of the (2R,4S)-form. In neither case the appearance in the solutions of the linear imine form А was observed. In this respect compounds 2а-f fundamentally differ from the condensation products of L-asparagine with a series of aromatic aldehydes 2g-i where the occurrence of the ring-chain tautomerism has been found and alongside two pyrimidine stereoisomers a linear form exists in solutions.
The acylation of 2-substituted 6-oxohexahydropyrimidine-4-carboxylic acids results in the formation of optically pure N-acyl derivatives with (2S,4S)-configuration of the substituents at the pyrimidine ring [13]-[16]. In some studies the ability was demonstrated of (2S,4S)-3-acyl-2-substituted-6-oxohexahydropyrimidine-4-carboxylic acids to the conformational cis,trans-isomerization in solutions due to effect of the hindered rotation relative to C?N bond of the amide fragment [13] [15] [18].
It was found that acylation of compounds 2а-i with 3-(acetylthio)propionyl chloride proceeded in acetone and completed in 10-12 h with the formation of 3-(3-acetylsulfanylpropanoyl)-2-alkyl(aryl)-6-oxohexahydropyrimidine-4-carboxylic acids 3а-i (see Scheme 2 and Table 3).
The acylation products 3а-i possess the (2S,4S)-configuration of the substituents of the pyrimidine ring, in their solutions in DMSO-D6, in keeping with the data of 1Н and 13С NMR spectra, the presence of two cis, trans- conformers has been found in the ratio ~3:2. It was confirmed by the coalescence of doubled signals at the re-
Table 2. Tautomeric composition (%) of compound 2g in various solvents 48 h after dissolution.
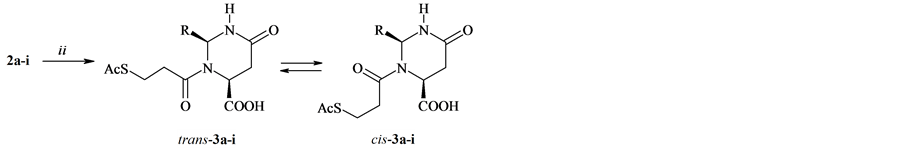
Scheme 2. ii: AcSCH2CH2COCl/Me2CO, C5H5N, 5˚C - 10˚C, 10 - 12 h. R = Me (a), Et (b), Bu (c), i-Pr (d), i-Bu (e), CH2CH2Ph (f), R =XC6H4, X = H (g), 4-Cl (h), 4-MeO (i).
Table 3. Characteristics of compounds 3a-i.
gistering 1Н NMR spectra in DMSO-D6 at higher temperature (~80˚С - 85˚С). Proceeding from the comparison of the 1Н and 13С NMR spectra of compounds 3а-i with the published [13]-[16] spectral characteristics of 3-alkanoyl-2-substituted 6-oxohexahydropyrimidine-4-carboxylic acids the trans-configuration was ascribed to the main isomer. This conformer is characterized by the downfield shift of Н-4 and Н-2 signals in the 1Н NMR spectrum compared the analogous signals of the minor cis-isomer. The position of the conformational equilibrium is governed by the nature of the applied solvent, and in going from СDCl3 to polar solvents, as show the data for compounds 3а-i, the stability of the more polar cis-conformer increases.
The removal of S-acetyl protection in compounds 3а-i occurs in the ammonia solution within several hours at room temperature and leads to the formation of (2S,4S)-2-alkyl(aryl)-3-sulfanylpropanoyl-6-oxohexahydropyrimidine-4-carboxylic acids 4а-i in 50% - 65% yields (see Scheme 3 and Table 4). In the 1Н NMR spectra in DMSO-D6 of compounds 4а-i a triplet signal is observed from the proton of the SH group in the region 1.45 - 2.10 ppm. Like in compounds 3а-i, in the solutions of products 4а-i cis- and trans-conformers are present due to the effect of the hindered amide rotation.
3. Experimental
1Н and 13С NMR spectra were registered on a spectrometer Bruker AV-400 at operating frequencies 400 and 100 MHz respectively (internal reference HMDS). The stereoisomeric composition of obtained compounds was estimated by the integration of the appropriate signals in the 1Н NMR spectra. The specific optical rotation was measured on a polarimeter P-161M at the wavelength of the plane-polarized light 589 nm. Elemental analysis of newly obtained compounds was carried out on a CHN Analyzer Hewlett Packard 185B. The purity of prepared compounds was checked by TLC on Silufol UV-254 plates, eluent benzene-acetone, 1:1.
Compounds 2а-i are white or light yellow amorphous hygroscopic powders, mp >250˚С. Spectral characteristics of compounds 2a-i, 3a-i, and 4a-i were described previously [19]. 3-(Acetylsulfanyl)propanoyl chloride was obtained by the method [20].
4. Conclusions
Hence we developed a three-stage stereoselective synthesis from L-asparagine of previously unknown (2S,4S)-

Scheme 3. iii: NH3(aq.), 25˚C, 5 - 10 h. R = Me (a), Et (b), Bu (c), i-Pr (d), i-Bu (e), CH2CH2Ph (f), R = XC6H4, X = H (g), 4-Cl (h), 4-MeO (i).
Table 4. Characteristics of compounds 4a-i.
3-(3-sulfanylpropanoyl)-2-alkyl(aryl)-6-oxohexahydropyrimidine-4-carboxylic acids 4а-i, potential antihypertensive drugs, ACE inhibitors [21]; the compounds otained are structural analogs of the known antihypertensive drug methiapril, (2S,6S)-1-[(3-acetylsulfanyl)propanoyl]-6-methylpipecolinic acid [7]. However unlike methiapril whose production requires difficultly available synthetic (2S,6S)-6-methylpipecolinic acid [22], the stereoselective synthesis of compounds 4а-i is underlain by natural or commercially available reagents. Besides, by varying the structure of the initial aldehyde component the developed procedure makes it possible to introduce in the position 2 of pyrimidine ring substituents of diverse nature, length, and branching.
Acknowledgements
This work received financial support from the Ministry of Education and Science of the Russian Federation (contract 14.547.21.0002, no. RFMEFI57414X0002).
Cite this paper
Andrei Ershov,Dmitry Nasledov,Igor Lagoda,Valery Shamanin, (2015) Synthesis of (2S,4S)-2-Substituted-3- (3-Sulfanylpropanoyl)-6- Oxohexahydropyrimidine-4-Carboxylic Acids as Potential Antihypertensive Drugs. Journal of Materials Science and Chemical Engineering,03,7-12. doi: 10.4236/msce.2015.36002
References
- 1. Wyvratt, M.J. and Patchett, A.A. (1985) Medicinal Research Reviews, 5, 483-531. http://dx.doi.org/10.1002/med.2610050405
- 2. Brown, N.J. and Vaughan, D.E. (1998) Circulation, 97, 1411-1420. http://dx.doi.org/10.1161/01.CIR.97.14.1411
- 3. Ondetti, M.A., Rubin, B. and Cushman, D.W. (1977) Science, 196, 441-444. http://dx.doi.org/10.1126/science.191908
- 4. Ondetti, M.A. and Cushman, D.W. (1978) US Patent No. 4105776.
- 5. Ryono, D.E. and Ondetti, M.A. (1982) US Patent No. 4321392.
- 6. Ondetti, M.A. (1981) US Patent No. 4282235.
- 7. Yakhontov, L.N., Mashkovskij, M.D., Kugaevskaya, E.V., Orekhovich, V.N., Pavlikhina, L.V., Eliseeva, Y.E., Mastafanova, L.I., Evstratova, M.I. and Shvarts, G.Y. (1993) SU Patent No. 1838298.
- 8. Nozawa, K., Yoshizawa, T. and Kuroda, N. (1985) US Patent No. 4537967.
- 9. Stanton, J.L. and Gruenfeld, N. (1986) UA Patent No. 4599357.
- 10. Robl, J.A. (1999) US Patent No. 5856476.
- 11. Nakamura, S., Tsuda, Y. and Doi, T. (1983) JP Patent No. 58110573.
- 12. Juaristi, E., Lopez-Ruiz, H., Madrigal, D., Ramirez-Quiros, Y. and Escalante, J. (1998) Journal of Organic Chemistry, 63, 4706-4710. http://dx.doi.org/10.1021/jo980367a
- 13. Juaristi, E., Quintana, D., Balderas, M. and Gareia-Perez, E. (1996) Tetrahedron: Asymmetry, 7, 2233-2246. http://dx.doi.org/10.1016/0957-4166(96)00278-9
- 14. Lakner, F.J. and Negrete, G.R. (2002) Synlett, 643-645. http://dx.doi.org/10.1055/s-2002-22712
- 15. Mfuh, A.M., Mahindaratne, M.P.D., Quintero, M.V., Lakner, F.J., Bao, A., Goins, B.A., Phillips, W.T. and Negrete, G.R. (2011) Langmuir, 27, 4447-4455. http://dx.doi.org/10.1021/la105085k
- 16. Kundu, S.K., Mahindaratne, M.P.D., Quinones, B., Negrete, G.R. and Tiekink, E.R.T. (2010) Acta Crystallographica, E66, o3.
- 17. Ershov, A.Yu., Nasledov, D.G., Nasonova, K.V., Sezyavina, K.V., Susarova, T.V., Lagoda, I.V. and Shamanin, V.V. (2013) Chemistry of Heterocyclic Compounds, 49, 598-603. http://dx.doi.org/10.1007/s10593-013-1287-0
- 18. Negrete, G.R., Mahindaratne, M.P.D., Mfuh, A.M. and Quintero, M.V. (2011) US Patent No. 20110268653.
- 19. Ershov, A.Yu., Nasledov, D.G., Parilova, E.V., Lagoda, I.V. and Shamanin, V.V. (2013) Russian Journal of Organic Chemistry, 50, 1164-1174. http://dx.doi.org/10.1134/S1070428014080168
- 20. Daeniker, H.U. and Druey, J. (1957) Helvetica Chimica Acta, 40, 2148-2156. http://dx.doi.org/10.1002/hlca.19570400714
- 21. Ershov, A.Yu., Susarova, T.V., Chernitsa, B.V. and Shamanin, V.V. (2011) RF Patent No. 245528.
- 22. Kadouri-Puchot, C. and Comesse, S. (2005) Amino Acids, 29, 101-130. http://dx.doi.org/10.1007/s00726-005-0193-x
NOTES

*Corresponding author.


