Paper Menu >>
Journal Menu >>
 Optics and Photonics Journal, 2011, 1, 24-35 doi:10.4236/opj.2011.12005 Published Online June 2011 (http://www.SciRP.org/journal/opj/) Copyright © 2011 SciRes. OPJ Trace Gas Detection from Plant Leaves, Flowers and Seeds Using Conventional and Photothermal Light Deflection Spectroscopy Mohammad Ibrahim Abu-Taha, Yasmeen Abduljaleel Abu-Rayan Physics Department, College of Science and Technology, Al-Quds University, Jerusalem, Palestine E-mail: mabutaha@science.alquds.edu Received March 14 , 20 11; revised April 14, 2011; accepted April 25, 2011 Abstract Photothermal deflection spectroscopy is a method used indirectly to measure optical absorption of a sample. Different techniques can be employed to measure the amount of deflection, hence evaluate optical absorption of sample. This work investigates an alternative method both in principle and technique to measure sample’s optical absorption. The new method employed for the first time, relies on the simple idea of light beam de- flection from the medium under investigation as a result of change in the index of refraction in its vicinity. The amount of deviation executed by the deviated beam is estimated using new technique that is used for the first time in deflection spectroscopy. As the deviated beam is allowed to pass through a single slit, the value of beam deflection is estimated from the resulting diffraction pattern, i.e. indicating the value of changes taking place in the sample and or measure sample’s optical absorption. The new detection technique used in the estimation of probe beam deflection was also applied in photothermal spectroscopy. Results from both methods were compared and revealed the ease of use of the new method, in addition it cuts cost and experi- mental efforts although its sensitivity is less than the conventional photothermal method. Keywords: Photothermal Deflection Spectroscopy, Deflection Spectroscopy, Trace Detection 1. Introduction In photothermal deflection spectroscopy, sample under investigation undergoes a photo-excitation, i.e. optical absorption, which subsequently causes a change in state of the sample and a thermal response. The method hence, constitutes an indirect method to measure optical ab- sorption. In regular optical absorption spectroscopy, the intensity of the exciting light beam and passing through the sample is monitored and compared with the intensity of light beam before it enters the sample. However, in photothermal spectroscopy, the transmission of light is not used in measuring, and instead, sample heating, which is a direct consequence of optical absorption, is what is studied by measuring the value of deflection suffered by a probe beam. Many researchers carried out different investigations for the theoretical aspects of photothermal method [1-3]. Photothermal deflection spectroscopy (PTDS) is widely used in many applications for example in the investiga- tions of thermal diffusivity of bulk solids and thin films [4], measurements of low absorption coefficients [5], temperature measurem ent s i n flam e by photothermal spec- troscopy was also used [6-7], study of solids [8] and electrochromism of synthetic metals [9]. In most of the experimental set ups employing PTDS a photosensitive position sensor [10] is used and a com- plicated equipment and electronic circuitry involved [11 ]. Some studies even used two sensors, and concluded that PTDS is very sensitive for measurements of low absorp- tion coefficients [12]. In this work the conventional optical effect, i.e. when light passes from one medium to another, it changes speed, and bends depending on the refractive index change of the mediums is used as a spectroscopic indic ator. This means, that idea of light bending toward or away from the normal drawn perpendicular to the line dividing the two mediums can act as a spectroscopic indicator, for example if light is passing in the same medium and in center part of it an index change does occur, then the beam change position accordingly confirming the index change, hence leading to a spectroscopic result related to the concerned medium.  M. I. ABU-TAHA ET AL. Copyright © 2011 SciRes. OPJ 25 The amount of deflection is proportional to the mount of change in the medium properties. 2. Experimental 2.1.Experimental Set Up A schematic diagram showing the complete experimental setup for conventional deflection and phtothermal spec- troscopy used in the present work are shown in the Fig- ure 1 and Figure 2 respectively. Experiments are carried out using the two methods, i.e. the conventional light deflection (CLD) and the photothermal light deflection (PTLD) spectroscopic techniques. In both methods we applied one technique for measuring the deflection of the probe beam resulting from the effect taking place in the cell. Firstly, results are taken for known samples used to authenticate the systems, followed by the use of un- known samples being taken using bo th method s and their results compared. It is worth noting that both arrangements comprise: Power function generator, simple rectangular glass box, He-Ne laser to act as a probe beam, single slit, light photometer and a plane mirror fixed at one side of the chamber. The only difference between the two set ups is that the PTLD method employs an Infra red light source to act as the power beam. The infrared light source used in this study [14], is a wideband pulsed mid-IR thermal source based on elec- trical heating of a thin metal alloy foil up to red heat (~900˚C) and cooled by its own radiation. The device, emitting a wideband in the rang e 2 - 15 µm with emitting area diameters of 25 mm, is based on a suspended bispi- ral metal foil geometry which offers substantial thermal isolation from the supporting structure. Although its power is in the range of microwatts it proved enough in the above arrangement, making the use of the source advantageous in replacing hefty laser sources. 2.2. Light Deflection Detection Scheme It is customary in PTLDS to use sensitive position sensors. Various types of posit i on sens ors can be used to detect the change in the position of the light beam that is reflected from a specific t arget absor bi n g zo ne, employing different measuring principles and involving difficulties and limit- ing efficiencies accordingly. In the present work a new detection scheme is suggested that can avoid experimen- tal complications and cost. The new detection scheme serves two goals: Firstly, and most importantly, it allows the simple conventional light deflection to become a practical spectroscopic method that uses only probe beam laser; And secondly, it replaces position sensors in the known PTLDS method, making it more manageable and less complicated. The suggested method can be summa- rized as follows: When the light beam from He-Ne laser is allowed to pass through the medium under investiga- tion and reflected from a mirror to a single slits, then bright and a dark spots are formed on a screen, i.e. dif- fraction pattern. A central peak containing most of the light intensity accompanied by secondary higher-order maxima and intensity minima governed according to: dsin(θ) = mλ, and the first dark fringe occurs at the angle given by: sin(θ) = λ/d, where θ is the angle between the central incident propagation direction and the first mini- mum of the diffraction pattern, and m indicates the se- quential number of the higher-order maxima, λ is the wavelength of the probe beam and d is the width of the slit. Usually, interest is in the location of the first mini- mum, when m = 1, because most of the light energy is located in the central diffraction maximum. Light inten- sity is maximum at θ = zero degrees, and decreases as we move to away from the center point at angles dictated by the equation above, see Figure 3 belo w. In the present work the diffraction pattern is initiated with a heliumneon laser beam allowed to fall on a single aperture after being reflected from the region of interest where a sample under investigation is placed. Abu-Taha, 2008, suggested the technique using a light photometer head to be placed right at the center of the principal maxima, then any slight changes in the index of refrac- tion of the sample due to an activity or physical effect, will result in a shift of the reflected beam, hence, a cor- responding shift in the center of the principal maxima. This resulted in certain angle change corresponding to a change in the relative intensity amplitude falling on the photometer sensor. In this case a change due to any ac- tivity/effect taking place in the sample contained in the cell will be translated into change in intensity amplitude being recorded b y the photometer. This technique, allows determining any minute sh ifts of the light beam position, hence, the complicated position sensor in photothermal deflection spectroscopy can be replaced by a simple slit. 2.3. Detection Scheme and Sensitivity The reflected beam passes through the single slit forming diffraction pattern. Photometer’s head is adjusted such that it is sensing the principle maximum central spot, then the head was manually moved across the maxima spot in both sides. This is a trial carried out to confirm the sensitivity of the detection scheme and to make sure that data will result in a diagram similar to that of the diffraction pattern as shown in Figure 4 below. The sen- sitivity of the system is limited by the sensitivity of the photometer being used. In the system used a displacement  M. I. ABU-TAHA ET AL. Copyright © 2011 SciRes. OPJ 26 Glass box Reflected beam after the introduction of sam p le Photometer’s head Reflected beam prior to introduction of sample He Ne Laser beam Sample Mirror Slit Figure 1. chematic sh owing the conventional Light Deflection cell u s ed to detect gas trace emissions from d ifferent samples. IR Source Glass box Aft er a bs orp tion Photometer’s head Before absorption Mirror Sample He Ne Laser b eam Slit Figure 2. Schematic showing the PTLD experimen tal s etup used to detect trace gas IR abs orptions. 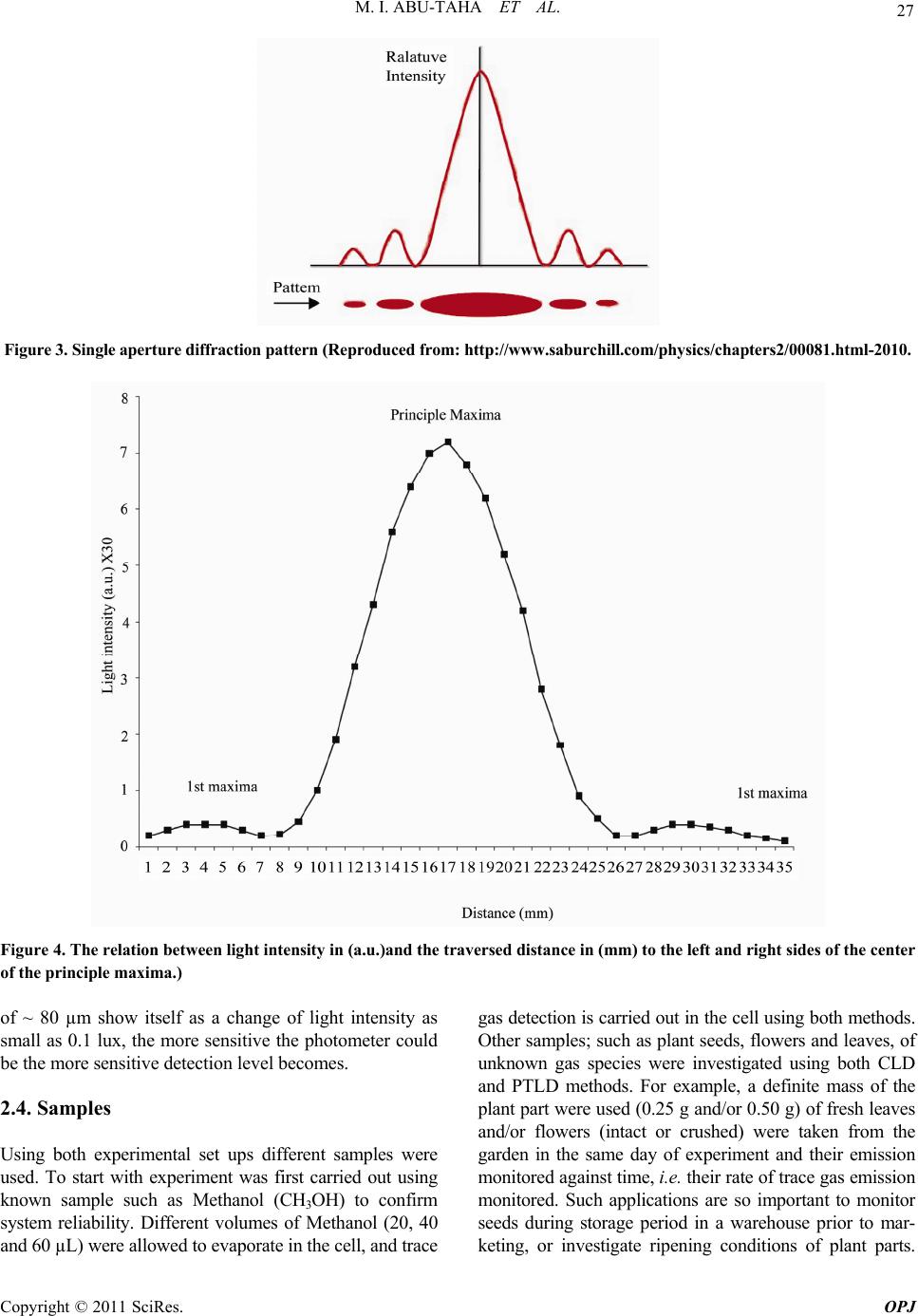 M. I. ABU-TAHA ET AL. Copyright © 2011 SciRes. OPJ 27 Figure 3. Single aperture diffraction pattern (Reproduced from: http://www.saburchill.com/physics/cha pter s2/00081.html-2010. Figure 4. The relation between light intensity in (a.u.)and the traversed distance in (mm) to the left and right sides of t he c enter of the principle maxima.) of ~ 80 µm show itself as a change of light intensity as small as 0.1 lux, the more sensitive the photometer could be the more sensitive detection level becomes. 2.4. Samples Using both experimental set ups different samples were used. To start with experiment was first carried out using known sample such as Methanol (CH3OH) to confirm system reliability. Different volumes of Methanol (20, 40 and 60 µL) were allowed to evaporate in the cell, and trace gas detection is carried out in the cell using both methods. Other samples; such as plant seeds, flowers and leaves, of unknown gas species were investigated using both CLD and PTLD methods. For example, a definite mass of the plant part were used (0.25 g and/or 0.50 g) of fresh leaves and/or flowers (intact or crushed) were taken from the garden in the same day of experiment and their emission monitored against time, i.e. their rate of trace gas emission monitored. Such applications are so important to monitor seeds during storage period in a warehouse prior to mar- keting, or investigate ripening conditions of plant parts.  M. I. ABU-TAHA ET AL. Copyright © 2011 SciRes. OPJ 28 Healthy and caries infected samples were used so as to monitor the live activity of insects on seeds. 3. Results It is worth noting that since, the center of the principle maxima spot was made to coincide with the fixed pho- tometer’s fiber versus optic head at the start of measure- ments, then the photometer measurement of intensity would be maximum at first. As time goes intensity read- ing would be decreased as the beam deflected away from the principle max-spot, hence, for measurements to re- flect the physics behind the effect taking place in the cell the intensity difference is plotted versu s time as ind icated in all figures of the experimental results. Figure 5(a) and Figure 5(b) show the rate of evapora- tion from different volumes of methanol placed in the cell as they monitored using CLD and PTLD respectively. This experiment with known chemical sample is used to authenticate the viability of t he CLD method in particul ar. Plant leaves is an important source of essential oil of some plants. The level of trace emission also reflects the ripening situation of the plant. Mint is chosen as an im- portant example as Figur es 7(a)-(d) show. Two different kinds of flowers namely; Jasmine and Rose were investigated using CLD and PTLD techniques. For each flower type two flower colors were used. Two different plant seeds namely wheat and chickpeas were investigated using CLD and PTLD techniques. It’s known that seeds can be attacked by caries insects or start to germinate during the storage period, so the avai- lability of a monitoring technique in the warehouse is advantageous. The present study was employed to con- firm the ability of the system to check for insect actions in seeds, this is done by detecting their gas emissions when insect contaminated seeds were introduced into the testing box. In the above trace gas results using CLD and PTLD are presented. A comparison between both techniques could help identify the technique that is more sensitive. Com- parison results between the two techniques are shown in Figures 9(a)-(e). To further prove the technique which is most sensitive the PTLD technique was used to detect trace gas from only one Jasmine flower, see Figure 9(d). It is believed that each plant leaves emit a trace gas that contain part of the gases emitted by its flower. To inves- tigate this fact the relation between gas emissions from jasmine leaves and flowers are shown in Figure 9(e). 4. Discussion The data obtained from the experiments using both me- thods were discussed along three paths; one involves re- sults of gas emissions using CLD technique and the sec- ond involves results of gas emission from samples using the well known PTLD technique. And, th e third involves comparison between results of CLD and PTLD. Both CLD and PTLD signals were monitored as a function of time. In PTLD experimental part samples were allowed to absorb wideband IR radiation from a pulsed IR source, i.e. for the experiment a probe and a pump beams are used; contrary to CLD technique which involves only one beam namely the probe beam. As mentioned earlier, no position sensor is used to determine the deflection of beams in both techniques, and instead a diffraction slit proved very useful and satisfactory. The new less sophisticated technique CLD method was used to detect trace gas emissions from different chemical and plant part samples. The first impression confirms our hypothesis that CLD method are giving comparable re- sults to that of PTLD. Different trials to detect trace gas emission from plant parts were carried out. Group of Figures 6(a)-(d), show detection possibility for trace detection using plant leaves at different conditions using both methods. Plant flowers are important in the perfume industry, both experimental techniques were checked for their ability to detect trace emissions from different types of flowers and even different flower colors in the same flower type (see Figures 7(a)-(d). Trace emissions from seeds provides a way to follow up the storage conditions in the warehouse. Trials to mimic storage conditions of humidity and insect attack on the stored seeds were stud- ied (see Figures 8(a)-(c)). The activity of only 5 caries attacking chickpeas could be monitored using both meth- ods. Finally, Comparisons Figures 9(a)-(e) between se- lected situations using both methods speak for them- selves. It obvious that the PTLD is superior to the CLD method, since it employs the advantage of trace gas ab- sorption of IR radiation of the wide band light source emitting in the range 2 - 15 µm. As far as the sensitivity of the methods are concerned, it can be estimated from a known volume of methanol, detection levels were estimated to be as low as 18 and 24 ppm for PTLD and CLD respectively. It is obvious that the PTLD methods is more sensitive than CLD, but the reward from the later is simplicity and easy to implement. Open area detection (no cell used to hold sample) was carried out using the simple conventional light deflection method, which was used to detect smoke emission from one cigarette placed such that its smoke is flown across the path of the laser beam. More, the effect of air speed going through the path of the laser beam were invest- tigated too. In this experiment the laser beam allowed to fall on a mirror fixed on the wall inside the lab room and be reflected through a slit into the photometer’s head. The CLD method is found applicable for both smoke 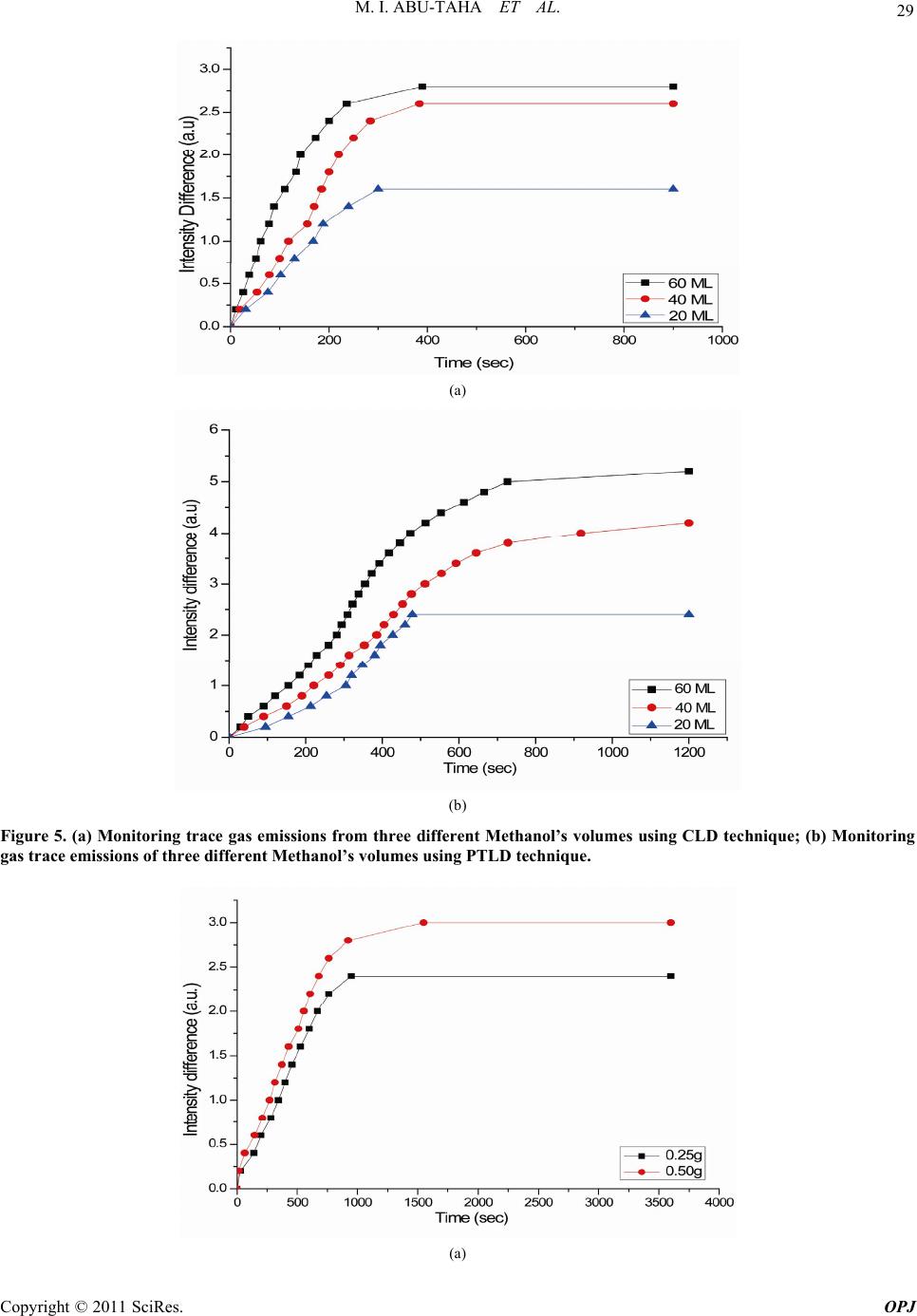 M. I. ABU-TAHA ET AL. Copyright © 2011 SciRes. OPJ 29 (a) (b) Figure 5. (a) Monitoring trace gas emissions from three different Methanol’s volumes using CLD technique; (b) Monitoring gas trace emissions of three different Methanol’s volumes using PTLD technique. (a) 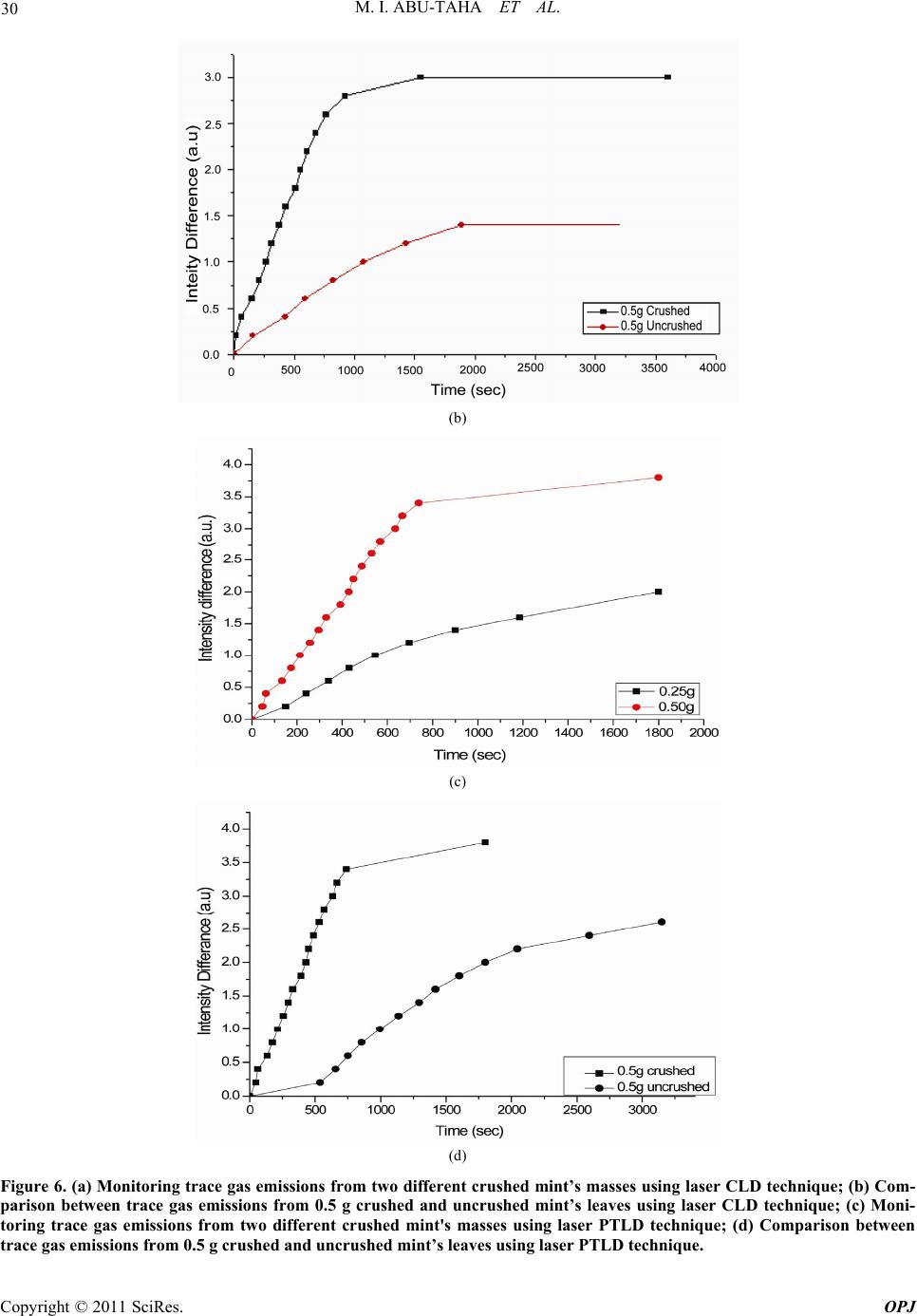 M. I. ABU-TAHA ET AL. Copyright © 2011 SciRes. OPJ 30 (b) (c) (d) Figure 6. (a) Monitoring trace gas emissions from two different crushed mint’s masses using laser CLD technique; (b) Com- parison between trace gas emissions from 0.5 g crushed and uncrushed mint’s leaves using laser CLD technique; (c) Moni- toring trace gas emissions from two different crushed mint's masses using laser PTLD technique; (d) Comparison between trace gas emissions from 0.5 g crushed and unc r ushe d mint’s le ave s using laser PTLD technique.  M. I. ABU-TAHA ET AL. Copyright © 2011 SciRes. OPJ 31 (a) (b) (c)  M. I. ABU-TAHA ET AL. Copyright © 2011 SciRes. OPJ 32 (d) Figure 7. (a) Comparison between trace gas emissions from white and yellow Jasmine flowers using CLD technique; (b) Comparison between trace gas emissions from crushed and uncrushed yellow Jasmine flowers using CLD technique; (c) Comparison between trace gas emissions from white and yellow Jasmine flowers using PTLD technique; (d) Comparison between tr ac e gas emissions from white and red ro se flowers using CLD technique. (a) (b)  M. I. ABU-TAHA ET AL. Copyright © 2011 SciRes. OPJ 33 (c) Figure 8. (a) Comparison between trace gas emissions from non contaminated and insect contaminated chick peas using CLD technique; (b) Comparison between trace gas emissions from non contaminated and insect contaminated chick peas using PTLD technique; (c) Comparison be tween trace gas emissions from germinated and un germinated wheat grains using CLD technique. (a) (b) 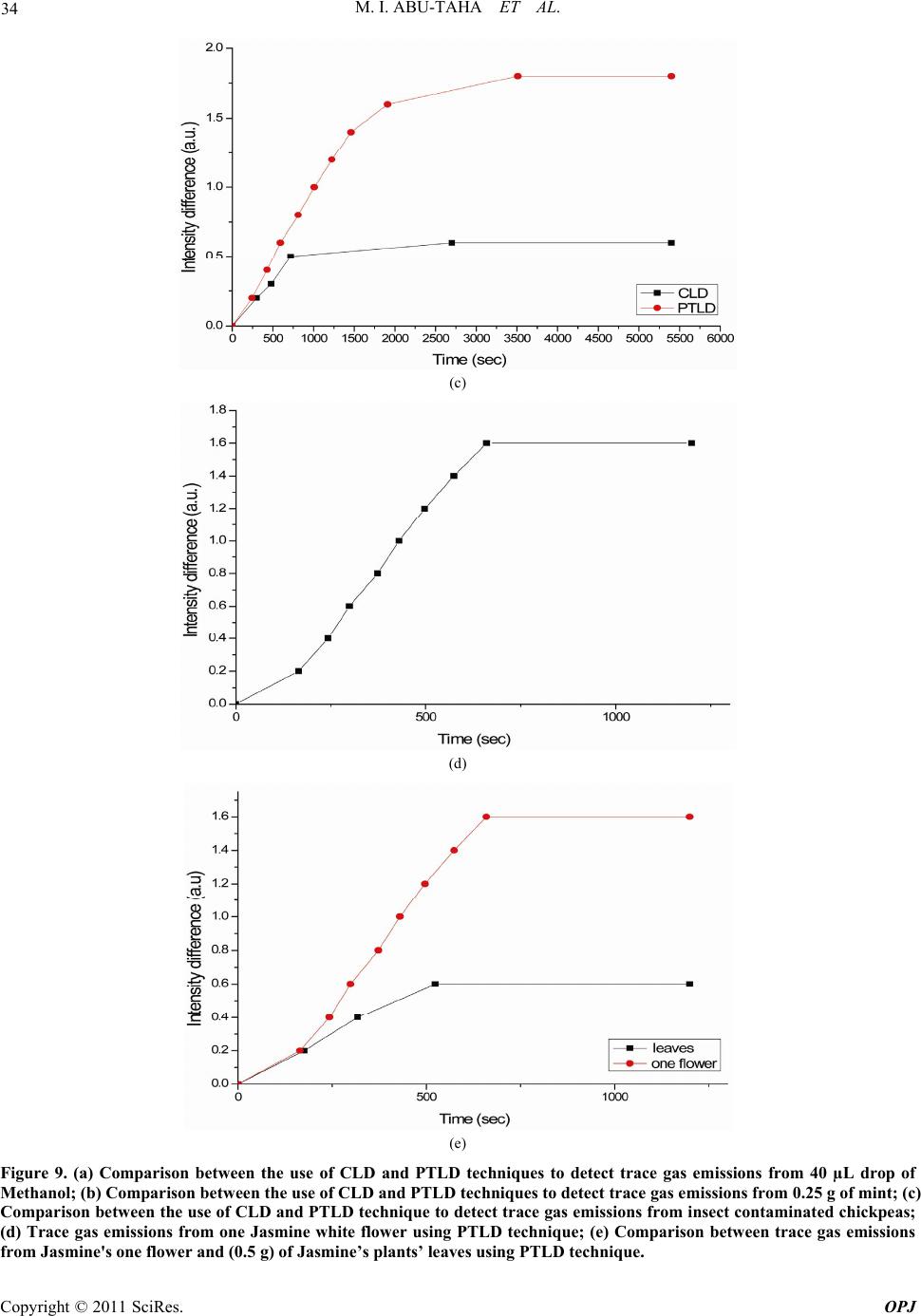 M. I. ABU-TAHA ET AL. Copyright © 2011 SciRes. OPJ 34 (c) (d) (e) Figure 9. (a) Comparison between the use of CLD and PTLD techniques to detect trace gas emissions from 40 µL drop of Methanol; (b) Comparison between the use of CLD and PTLD techniques to detect trace gas e missions from 0.25 g of mint; (c) Comparison betwee n the use of CLD and PTLD technique to detect trace gas emissions from insect contaminated chickpeas; (d) Trace gas emissions from one Jasmine white flower using PTLD technique; (e) Comparison between trace gas emissions from Jasmine's one flower and (0.5 g) of Jasmine’s plants’ leaves using PTLD technique.  M. I. ABU-TAHA ET AL. Copyright © 2011 SciRes. OPJ 35 detection and changes of air speed in the form of draft. It was possible to determine the side into which air is blown. This experiment indicates the ability of CLD method for open area applications, for example detecting insect sprays vapors carried away from fields toward inhabited areas. 5. Conclusions A set of experiments were performed successfully using two simple, sensitive, easy to handle, safely, inexpensive and accurate systems. The basic conclusions of this work, is drawn from the use of CLD and PTLD methods to de- tect gaseous species at the trace level for different sam- ples. This experiment prov ed the ability of the two me th- ods to distinguish different kinds of samples emitting at low ppm level. Both methods were used without the need for position sensor, instead a slit initiated diffraction pat- tern was used, i.e. an expensive difficult to handle posi- tion sensor could be replaced successfully with a single diffraction slit. This is considered an achievement for such experiments, resulting in the enhancement and ease of their use. Although the CLD gave encouraging results comparable to the PTLD method, its sensitivity co uld be improved using more sensitive light photometer. CLD can have a wide variety of agricultural applications, for example, to monitor storage conditions in a warehouse, conditions of animal barns, industrial plants, hospitals and many different environmental studies. 6. References [1] J. Ravi, S. Lekshmi, K. Nair and T. Rasheed, “A Simple Theoretical Extension to the Analysis of Photothermal Deflection Signal for Low Thermal Diffusivity Evalua- tion,” Journal of Quantitative Spectroscopy & Radiative Transfer, Vol. 83, No. 2, 2004, pp. 193-202. doi:10.1016/S0022-4073(02)00351-5 [2] A. Mandelis, A. Salnic, J. Opsal and A. Roencwaig, “Nonlinear Fundamental Photothermal Response in Three Dimensional Geometry: Theoretical Model,” Journal of Applied Physics, Vol. 85, No. 3, 1999, pp. 1811-1820. doi:10.1063/1.369328 [3] S. Bialkowski, “Progress Toward a Better Understanding of Signal Generation in Laser-Excited Photothermal Spec - trometry of Homogeneous Samples,” Trends in Analyti- cal Chemistry, Vol. 17, No. 8-9, 1998, pp. 520-532. doi:10.1016/S0165-9936(98)00045-4 [4] B. X. Shi, C. W. Ong and K. L. Tam, “Measurements of Thermal Diffusivity of Boron-Silicon Film-on-Glass Structure Using Phase Detection Method of Photothermal Deflection Spectroscopy,” Journal of Materials Science, Vol. 34, No. 21, 1999, pp. 5169-5173. doi:10.1023/A:1004751627438 [5] M. Moser, S. Tasch and G. Leising, “Photothermal De- flection Spectroscopy of a Poly—(Para Phenelyne)— Type Ladder Polymer,” Synthetic Metals, Vol. 84, No. 1-3, 1997, pp. 651-652. doi:10.1016/S0379-6779(96)04092-1 [6] Y. Li and R. Gupta, “Simultaneous Measurements Of Absolute OH Concentration, Temperature, and Flow Ve- locity in Flame by Photothermal Spectroscopy,” Applied Physics B: Lasers and Optics, Vol. 75, No. 8, 2002, pp. 903-906. doi:10.1007/s00340-002-1048-8 [7] Y. Li and R. Gupta, “An Investigation of the Photother- mal Deflection Spectroscopy Technique for Temperature Measurement in Flame,” Applied Physics B: Lasers and Optics, Vol. 75, No. 1, 2002, pp. 103-112. doi:10.1007/s00340-002-0937-1 [8] S. Yun and H. Seo, “Photothermal Beam Deflection Technique For The Study of Solids, Chinese Journal of Physics, Vol. 30, No. 5, 1992, pp. 753-767. [9] C. Barbero, R. KÖtz and O. Haas, “Defferential photo- thermal deflection spectroscopy. A Technique to Study Electrochromism of Synthetic Metals,” Synthetic Metals, Vol. 101, No. 3, 1999. pp. 170. doi:10.1016/S0379-6779(98)00766-8 [10] J. Spear, R. Russo and R. Silva, “Collinear Photothermal Deflection Spectroscopy with Light Scattering Samples,” Applied Optics, Vol. 29, No. 28, 1990, pp. 4225-4234. doi:10.1364/AO.29.004225 [11] F. Sogandares and E. Fry, “Absorption Spectrum (340 - 640 nm) of Pure Water I. Photothermal Measurement,” Applied Optics, Vol. 36, No. 33, 1997, pp. 8699-8709. doi:10.1364/AO.36.008699 [12] M. Mosear, S. Tasch and G. Leising “Photothermal De- flection Spectroscopy of a Poly (Para-Phynelene)-Type Polymer,” Synthetic Metals, Vol. 84, No. 1-3, 1997, pp. 651-652. doi:10.1016/S0379-6779(96)04092-1 [13] M. I. Abu-Taha, “Use of single slit in photothermal spec- troscopy experiments,” Private Communications, 2008. [14] D. C. Laine, M. Al-Jourani, S. Carpenter and M. Sedg- beer, “Pulsed Wideband Infrared Thermal Source,” IEE Proceedings-OptoElectronics, Vol. 144, No. 5, 1997, pp. 315-323. |

