Paper Menu >>
Journal Menu >>
 World Journal of Nano Science and Engineering, 2011, 1, 51-61 doi:10.4236/wjnse.2011.12008 Published Online June 2011 (http://www.SciRP.org/journal/wjnse) Copyright © 2011 SciRes. WJNSE Studies on the Effect of t h e Capping Materials on the Spherical Gold Nanoparticles Catalytic Activity Roshdi Seoudi1,2, Doaa A. Said3 1Spectroscopy Department, Physics Division, National Research Center, Cairo, Egypt 2Department of Physics, College of Applied Science, Umm Al-Qura University, Makkah, Saudi Arabia 3Physics Department, Faculty of Women, Ain Shams University, Cairo, Egypt E-mail: rsmawed@yahoo.com Received March 30, 2011; revised April 11, 2011; accepted April 18, 2011 Abstract Size-controlled gold nanoparticles (AuNPs) were prepared in the presence of different capping materials (sodium citrate, cetyltrimethylammonium bromide (CTAB), and chitosan). The results obtained suggest that the AuNPs were synthesized with different particle size, which is controlled by changing the molar ratio be- tween sodium citrate, (CTAB), and chitosan to Au (III). The catalytic activities of the AuNPs with different capping materials were studied for 4-nitrophenol reduction by NaBH4 as a model reaction. AuNPs with dif- ferent capping materials is comparable from the value of the apparent rate constant of 4-nitrophenol reduc- tion (0.6 × 10–3, 1.9 × 10–3, and 2.4 × 10–3 s–1) for sodium citrate, CTAB and chitosan. From the results, it is concluded that, AuNPs catalyzed the electron transfer process between 4 BH and nitro compounds with all the capping materials used AuNPs capped by chitosan were more active for the reduction than the other two. Keywords: Gold Nanoparticle, Catalytic Activity, TEM, UV-VIS Spectroscopy 1. Introduction Nanomaterials and its composites have become increa- singly popular due to their size-specific, unique proper- ties, and promising a breakthrough in the development of novel methods in medicine [1,2], and sensor [3,4]. Gold nanoparticle AuNPs, have received more attention due to their strong optical absorption in the visible region [5,6], catalysis properties [7-9], and enhanced sensitivity in surface-enhanced Raman scattering (SERS) studies [10]. All the above properties are strongly affected by the size and shape of the AuNPs. Several methods are known and have been exploited for centuries and there are still many deficiencies in the development of stable colloids con- taining AuNPs of various sizes and shapes for precise application. All synthesis procedures must overcome thermodynamic principles, which predict that AuNPs will tend to agglomerate, collapsing the colloid via pre- cipitation or flocculation of the particles. This tendency can be hindered by chemical species, which surround the particles, making particle agglomeration difficult. The synthesis of AuNPs in aqueous solution is still the gener- al route. The most popular method for preparing AuNPs in water used the citrate to reduction of HAuCl4 under boiling conditions [11]. Colloidal Au nanospheres were prepared in CTAB [12]. Therefore, diverse approaches have been developed to the reduction of Au(III) salts in water [13-17] using different ligands as colloidal particle stabilizers [18-23]. Until around 1980, gold was not fully recognized for its catalytic ability. Recent techniques have shown AuNPs to be highly active, and it has the distinction of being the most highly active catalyst for the hydrochlorination of acetylene to produce vinyl chlo- ride [24]. The complexities of catalyst fouling have given rise to a number of simulations that attempt to explain and predict catalytic fouling [25-28]. Gold has many unexpected properties that prevented earlier discovery of its catalytic potential. Unlike most other elements, gold does not have a stable oxide. It also does not follow the rule of thumb that other catalysts. The atomic radius of gold is smaller and its most stable state is gold III [29,30]. However, application prospects of conventional chemi- cally synthesized nanomaterials are often complicated because of their toxicity, caused by contamination with chemical precursors or additives during their synthesis procedures [31]. Recently, much attention has been paid to chitosan due to its excellent properties such as bio- compatibility, biodegradability, nontoxicity [32-36]. Chi-  R. SEOUDI ET AL. Copyright © 2011 SciRes. WJNSE 52 tosan is a natural polymer with abundant primary amino groups in its molecular structure; the structure is shown in Figure 1. Chitosan was chosen as a protecting agent in synthesis of metal nanoparticles. Esumi et al. [37] reported the formation of gold–chitosan nanocomposites by adsorp- tion of chitosan molecules on particle surfaces. Ballauff et al. [38] have shown that it is possible for chitosan to not only stabilize, but also reduce gold chloride into gold nanoparticles. AuNPs are highly active and it can be used to decrease the temperature catalysts in the number of important reactions such as CO oxidation and propyl- ene epoxidation [39]. In this work, gold nanoparticle was prepared with different capping materials (sodium citrate, CTAB and chitosan) and used for catalysis of the reduc- tion of 4-nitrophenol (4-NP) to study the effect of cap- ping on the catalytic reduction which has not been dem- onstrated earlier. 2. Experimental Cetlytrimethylammonium, sodium borohydried (NaBH4), tri-sodium citrate, and hydrogen tetrachloroaurate (III) were purchased from Sigma-Aldrich. Medium-molecu- lar-weight chitosan (2-amino-2-deoxy-(1-4)-β-D-glu- copyranose) with a degree of deacetylation of 100% and, 4-Nitrophenol (reagent grade) was purchased from Fluka. All aqueous solutions were made with ultra-high-purity water purified. 2.1. Samples Preparation 2.1.1. Preparation of AuNPs Capped by Sodium Citrate A 20 mL aqueous solution containing 2.5 × 10–4 M HAuCl4 and 2.5 × 10–4 tri-sodium citrate was mixed in a conical flask. Next, 0.6 mL of ice cold 0.1 M NaBH4 solution was added to the solution all at once while stir- ring. The solution turned pink immediately after adding NaBH4, indication particle formation. The nanoparticle solutions were used in a catalytic reduction within 12 h after preparation. Citrate serves only as the capping agent since it cannot reduce gold salt at room temperature (25˚C). 2.1.2. Preparation of AuNPs Capped by CTAB AuNPs capped by CTAB was prepared by mixing CTAB solution (5.0 mL, 0.20 M) with 5.0 mL of 0.00050 M HAuCl4. To the stirred solution, 0.60 mL of ice-cold 0.010 M NaBH4 was added, which resulted in the forma- tion of a brownish yellow solution. Vigorous stirring of the solution was continued for 2 min, then the solution was stirred, it was kept at 25˚C without further stirring. 2.1.3. Preparati on of Au NPs cap ped by Ch i tos an A completely dissolved of chitosan solution, 0.04 gm (20 mg/ml) in 1% acetic acid solution was prepared first; due to the poor solubility of chitosan, the mixture was vor- texed to completely dissolve it and kept overnight. The solution was filtered through 30 μm Millipore syringe filters to remove any impurities before use. 20 ml of aqueous solution of HAuCl4 (10 mM) was added to a 40 ml chitosan solution under magnetic stirring for two hours and 8 ml of NaBH4 (0.1 M) freshly prepared was added drop by drop. The solution turned brown imme- diately after addition of NaBH4; stirring continued until a transparent wine-red solution was obtained. The sample was kept at room temperature. The same procedure was repeated by adding 10 mM of HAuCl4 to 40 ml of chito- san nanoparticle. 2.2. Catalytic Reduction of 4-Nitrophenol The photocatalysis reactions were carried out in standard quartz cuvette with a 1-cm path length containing the reaction mixture, 1.4 cm3 of water, and 300 μL of 2 mmol dm–3 4-nitrophenol were taken. Addition and proper mixing of 1 cm3 of aqueous 0.03 mol dm–3 sodium bo- rohyride and 30 μL of gold nanoparticle solution to the reaction mixtures caused the decrease in the intensity of the peak of 4-nitrophenol. The progress of the reduction of 4-NP was monitored in situ using a UV-visible spec- trophotometer (Oceanoptics HR4000 Cg). The reaction temperature was held constant at room temperature Figure 1. Chemical structure of chitosan. 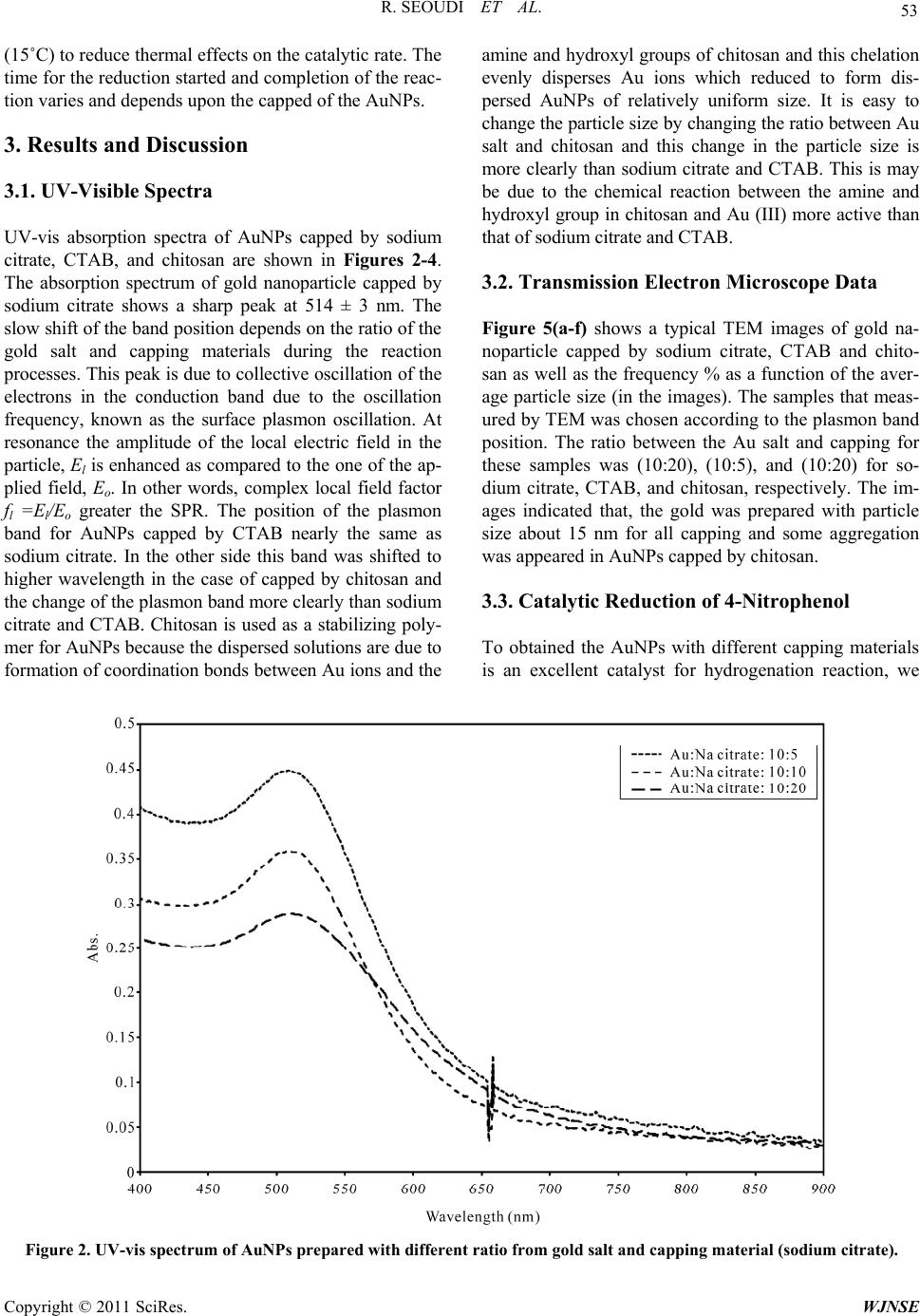 R. SEOUDI ET AL. Copyright © 2011 SciRes. WJNSE 53 (15˚C) to reduce thermal effects on the catalytic rate. The time for the reduction started and completion of the reac- tion varies and depends upon the capped of the AuNPs. 3. Results and Discussion 3.1. UV-Visible Spectra UV-vis absorption spectra of AuNPs capped by sodium citrate, CTAB, and chitosan are shown in Figures 2-4. The absorption spectrum of gold nanoparticle capped by sodium citrate shows a sharp peak at 514 ± 3 nm. The slow shift of the band position depends on the ratio of the gold salt and capping materials during the reaction processes. This peak is due to collective oscillation of the electrons in the conduction band due to the oscillation frequency, known as the surface plasmon oscillation. At resonance the amplitude of the local electric field in the particle, El is enhanced as compared to the one of the ap- plied field, Eo. In other words, complex local field factor fl =El/Eo greater the SPR. The position of the plasmon band for AuNPs capped by CTAB nearly the same as sodium citrate. In the other side this band was shifted to higher wavelength in the case of capped by chitosan and the change of the plasmon band more clearly than sodium citrate and CTAB. Chitosan is used as a stabilizing poly- mer for AuNPs because the dispersed solutions are due to formation of coordination bonds between Au ions and the amine and hydroxyl groups of chitosan and this chelation evenly disperses Au ions which reduced to form dis- persed AuNPs of relatively uniform size. It is easy to change the particle size by changing the ratio between Au salt and chitosan and this change in the particle size is more clearly than sodium citrate and CTAB. This is may be due to the chemical reaction between the amine and hydroxyl group in chitosan and Au (III) more active than that of sodium citrate and CTAB. 3.2. Transmission Electron Microscope Data Figure 5(a-f) shows a typical TEM images of gold na- noparticle capped by sodium citrate, CTAB and chito- san as well as the frequency % as a function of the aver- age particle size (in the images). The samples that meas- ured by TEM was chosen according to the plasmon band position. The ratio between the Au salt and capping for these samples was (10:20), (10:5), and (10:20) for so- dium citrate, CTAB, and chitosan, respectively. The im- ages indicated that, the gold was prepared with particle size about 15 nm for all capping and some aggregation was appeared in AuNPs capped by chitosan. 3.3. Catalytic Reduction of 4-Nitrophenol To obtained the AuNPs with different capping materials is an excellent catalyst for hydrogenation reaction, we Figure 2. UV-vis spectrum of AuNPs prepared with different ratio from gold salt and capping material (sodium citrate). 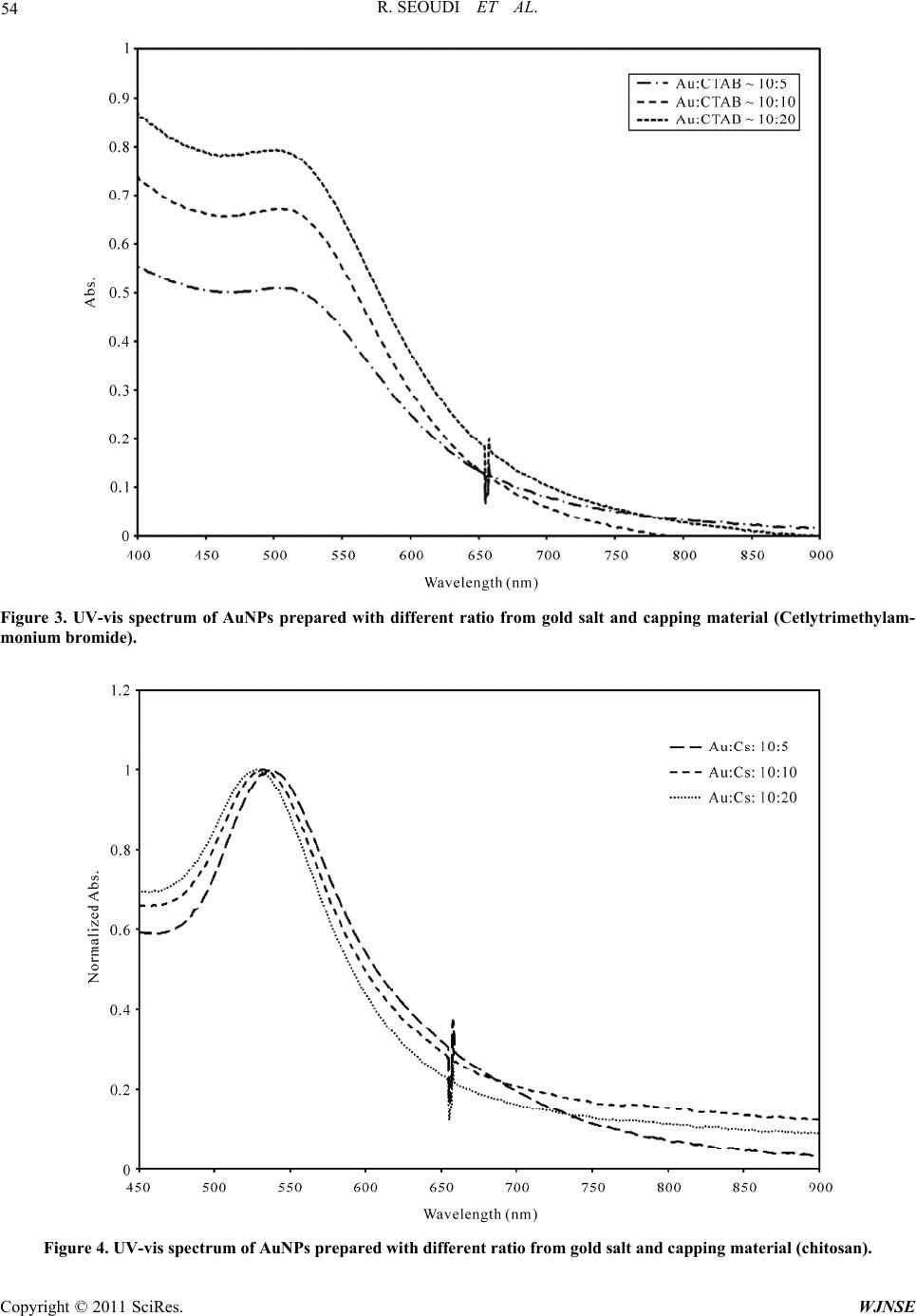 R. SEOUDI ET AL. Copyright © 2011 SciRes. WJNSE 54 Figure 3. UV-vis spectrum of AuNPs prepared with different ratio from gold salt and capping material (Cetlytrimethylam- monium bromide). Figure 4. UV-vis spectrum of AuNPs prepared with different ratio from gold salt and capping material (chitosan). 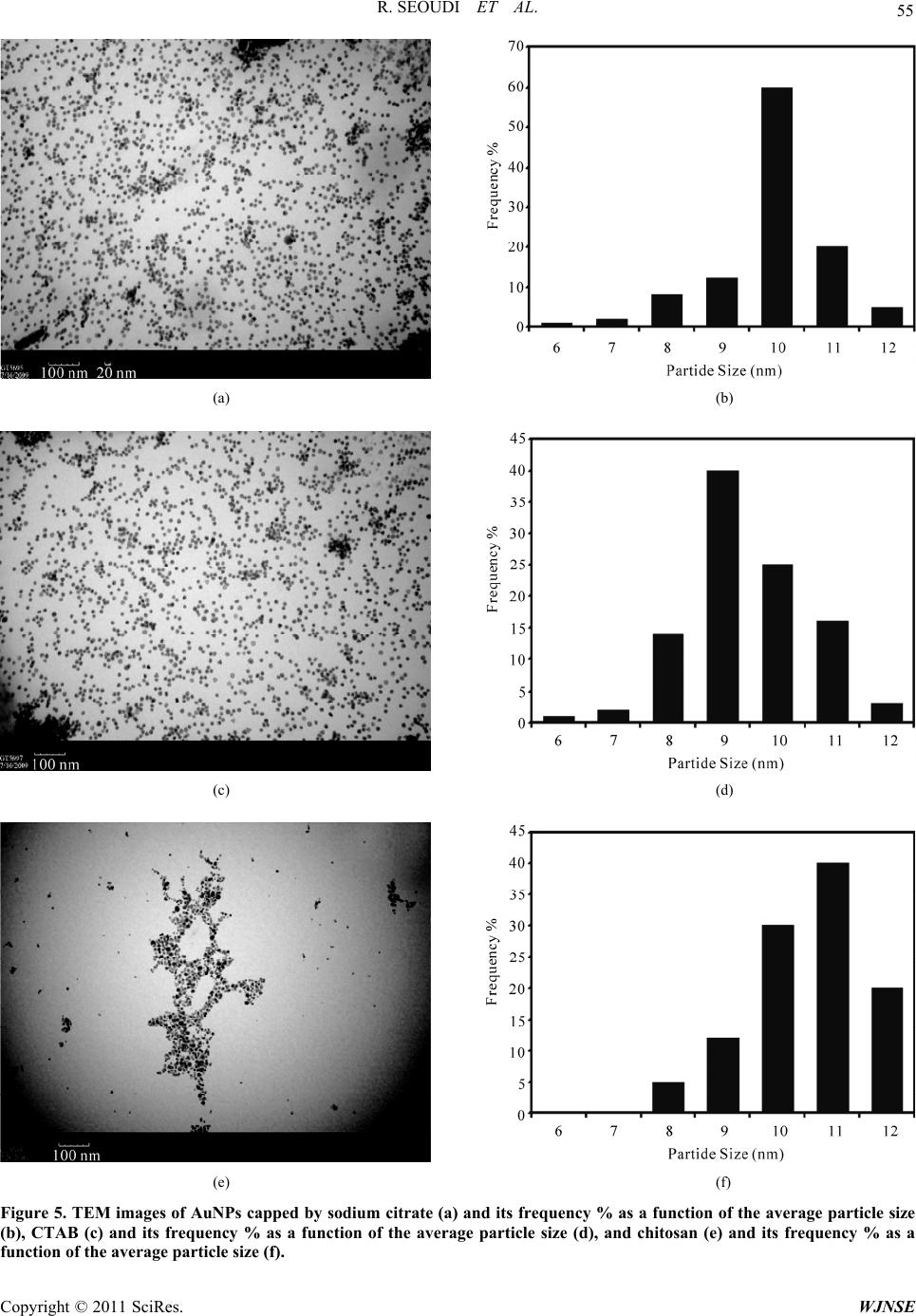 R. SEOUDI ET AL. Copyright © 2011 SciRes. WJNSE 55 (a) (b) (c) (d) (e) (f) Figure 5. TEM images of AuNPs c apped by sodium citrate (a) and its frequency % as a function of the average particle size (b), CTAB (c) and its frequency % as a function of the average particle size (d), and chitosan (e) and its frequency % as a function of the average particle size (f). 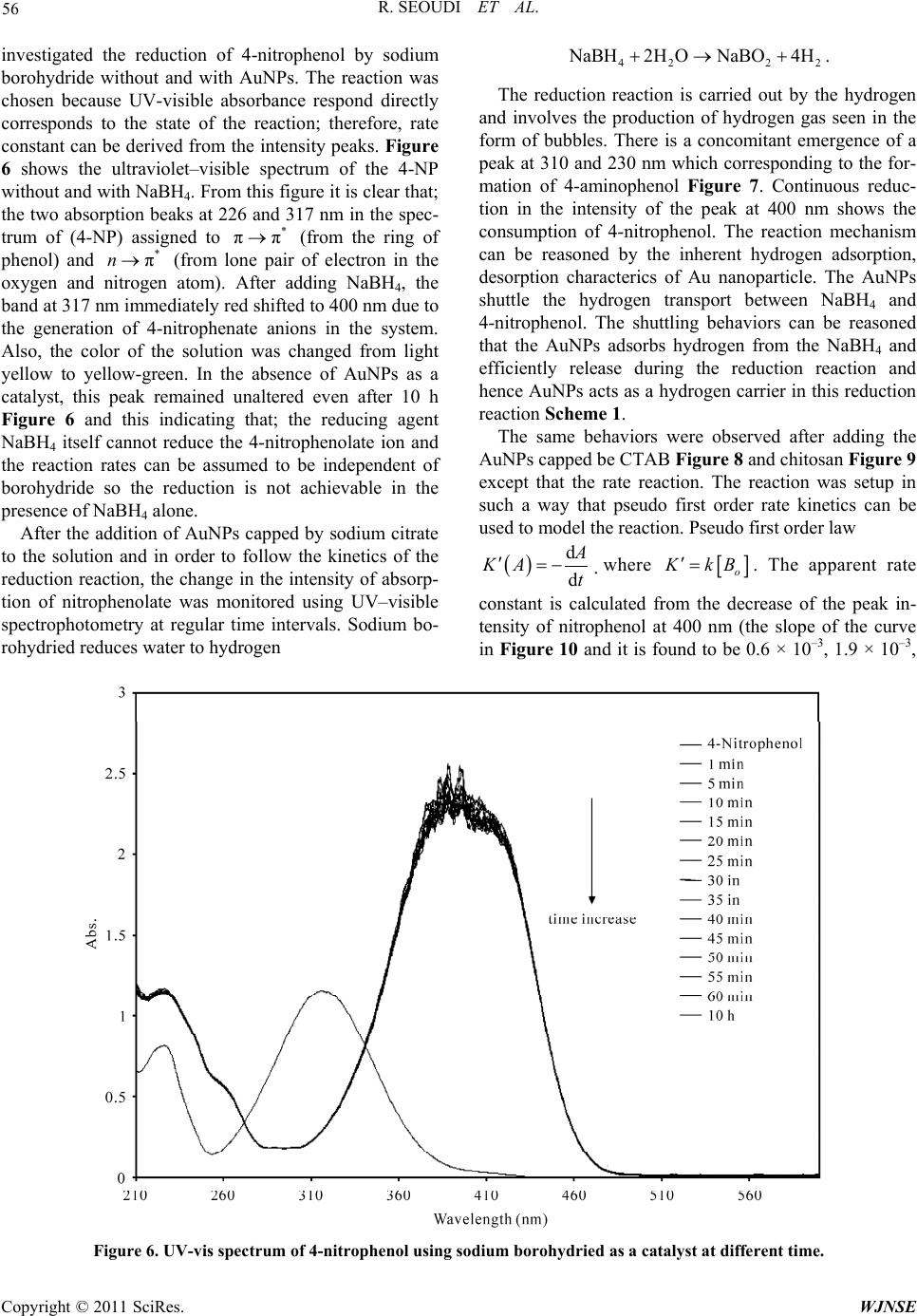 R. SEOUDI ET AL. Copyright © 2011 SciRes. WJNSE 56 investigated the reduction of 4-nitrophenol by sodium borohydride without and with AuNPs. The reaction was chosen because UV-visible absorbance respond directly corresponds to the state of the reaction; therefore, rate constant can be derived from the intensity peaks. Figure 6 shows the ultraviolet–visible spectrum of the 4-NP without and with NaBH4. From this figure it is clear that; the two absorption beaks at 226 and 317 nm in the spec- trum of (4-NP) assigned to * ππ (from the ring of phenol) and * πn (from lone pair of electron in the oxygen and nitrogen atom). After adding NaBH4, the band at 317 nm immediately red shifted to 400 nm due to the generation of 4-nitrophenate anions in the system. Also, the color of the solution was changed from light yellow to yellow-green. In the absence of AuNPs as a catalyst, this peak remained unaltered even after 10 h Figure 6 and this indicating that; the reducing agent NaBH4 itself cannot reduce the 4-nitrophenolate ion and the reaction rates can be assumed to be independent of borohydride so the reduction is not achievable in the presence of NaBH4 alone. After the addition of AuNPs capped by sodium citrate to the solution and in order to follow the kinetics of the reduction reaction, the change in the intensity of absorp- tion of nitrophenolate was monitored using UV–visible spectrophotometry at regular time intervals. Sodium bo- rohydried reduces water to hydrogen 42 22 N aBH2H ONaBO4H . The reduction reaction is carried out by the hydrogen and involves the production of hydrogen gas seen in the form of bubbles. There is a concomitant emergence of a peak at 310 and 230 nm which corresponding to the for- mation of 4-aminophenol Figure 7. Continuous reduc- tion in the intensity of the peak at 400 nm shows the consumption of 4-nitrophenol. The reaction mechanism can be reasoned by the inherent hydrogen adsorption, desorption characterics of Au nanoparticle. The AuNPs shuttle the hydrogen transport between NaBH4 and 4-nitrophenol. The shuttling behaviors can be reasoned that the AuNPs adsorbs hydrogen from the NaBH4 and efficiently release during the reduction reaction and hence AuNPs acts as a hydrogen carrier in this reduction reaction Scheme 1. The same behaviors were observed after adding the AuNPs capped be CTAB Figure 8 and chitosan Figure 9 except that the rate reaction. The reaction was setup in such a way that pseudo first order rate kinetics can be used to model the reaction. Pseudo first order law d d A KA t where o K kB . The apparent rate constant is calculated from the decrease of the peak in- tensity of nitrophenol at 400 nm (the slope of the curve in Figure 10 and it is found to be 0.6 × 10–3, 1.9 × 10–3, Figure 6. UV-vis spectrum of 4-nitrophenol using sodium borohydried as a catalyst at different time.  R. SEOUDI ET AL. Copyright © 2011 SciRes. WJNSE 57 Figure 7. UV-vis spectrum of 4-nitrophenol using sodium borohydried and AuNP s capped by sodium citrate as a catalyst at different times. Figure 8. UV-vis spectrum of 4-nitrophenol using sodium borohydried and AuNPs capped by CTAB as a catalyst.  R. SEOUDI ET AL. Copyright © 2011 SciRes. WJNSE 58 Figure 9. UV-vis spectrum of 4-nitrophenol using sodium borohydried and AuNPs capped by chitosan as a catalyst. Figure 10. UV-Vis absorption changes vs. time in seconds for the disappearance of 4-nitrophenol absorption at 400 nm.  R. SEOUDI ET AL. Copyright © 2011 SciRes. WJNSE 59 O2NOH OHH2N NaBH4 & AuNPs hydrogen adsorption AuNPs H2 Scheme 1. O H HO H H O O OO H O H NH H n AuNPsN O O AuNPs AuNPs AuNPs AuNPs AuNPs AuNPs Structure 1 of AuNPs capped by chitosan. and 2.4 × 10–3 S–1 for AuNPs capped by sodium citrate, CTAB and chitosan, respectively. It was found that was higher in case of chitosan and it is demonstrate that the AuNPs capped by chitosan is a highly effective carrier for catalysis. This seems obvious when we consider that the mobility of NH and OH groups on the AuNPs-chitosan increase the surface activ- ity of gold nanoparticles Structure 1. 5. Conclusions The rate of the gold nanoparticle reduction of 4-nitrophenol with sodium borohydried is faster in case of AuNPs capped by chitiosan because the surface activ- ity is more active in the catalytic activity application. So that chitosan brush particles will work more effectively as a template for gold in the water solution compared to other capping. 6. Acknowledgements The author expresses their thanks to Professor M. A. El-Sayed and his group, LDL, Gatech for helping me during preparation this work. 7. References [1] T. Xu, N. Zhang, H. L. Nichols, D. L. Shi and X. J. Wen, “Modification of Nanostructured Materials for Biomedi- cal Applications,” Materials Science and Engineering: C, Vol. 27, No. 3, April 2007, pp. 579-594. doi:10.1016/j.msec.2006.05.029 [2] K. Xu, J. Huang, Z. Ye, Y. Ying and Y. Li, “Recent De- velopment of Nano-Materials Used in DNA Biosensors,” Sensors, Vol. 9, No. 7, 2009, pp. 5534-5557. doi:10.3390/s90705534 [3] A. Józefczak and A. Skumiel, “Ultrasonic Investigation of Magnetic Nanoparticles Suspension with PEG Bio- compatible Coating,” Journal of Magnetism and Mag- netic Materials, Vol. 323, No. 11, June 2011, pp. 1509- 1516. doi:10.1016/j.jmmm.2011.01.009 [4] S. Thangavel and R. Ramaraj, “Electroless Synthesis of Multibranched Gold Nanostructures Encapsulated by Poly(o-phenylenediamine) in Nafion,” Journal of Colloid and Interface Science, Vol. 355, No. 2, March 2011, pp. 293-299. doi:10.1016/j.jcis.2010.12.035 [5] R. Güzel, Z. Üstündağ, H. Ekşi, S. Keskin, B. Taner, Z. G. Durgun, A. A. Turan and A. O. Solak, “Effect of Au and Au@Ag Core–Shell Nanoparticles on the SERS of Bridging Organic Molecules,” Journal of Colloid and In- terface Science, Vol. 351, No. 1, Novemner 2010, pp. 35- 42. [6] O. Estévez-Hernández, E.M. Molina-Trinidad, P. Santia- go-Jacinto, L. Rendón and E. Reguera, “Gold Nanopar- ticles Conjugated to Benzoylmercaptoacetyltriglycine and l-Cysteine Methylester,” Journal of Colloid and Interface Science, Vol. 350, No. , 2010, pp. 161-167. [7] J. Wang and W. De Zhang, “Sputtering Deposition of Gold Nanoparticles onto Vertically Aligned Carbon Na- notubes for Electroanalysis of Uric Acid,” Journal of Electroanalytical Chemistry, Vol. 654, No. 1-2, May 2011, pp. 79-84. doi:10.1016/j.jelechem.2011.01.023 [8] L. Wang, F. Montagne, P. Hoffmann, H. Heinzelmann and R. Pugin, “Hierarchical Positioning of Gold Nano- particles into Periodic Arrays Using Block Copolymer Nanoring Templates,” Journal of Colloid and Interface Science, Vol. 356, No. 2, April 2011, pp. 496-504. doi:10.1016/j.jcis.2010.12.081 [9] I. H. Hsu, W. H. Chen, T. K. Wu and Y. C. Sun, “Gold nanoparticle-Based Inductively Coupled Plasma Mass Spectrometry Amplification and Magnetic Separation for the Sensitive Detection of a Virus-Specific RNA Se- quence,” Journal of Chromatography A, Vol. 1218, No. 14, April 2011, pp. 1795-1801. doi:10.1016/j.chroma.2011.02.005 [10] Z. Guo, X. Fan, L. Liu, Z. Bian, C. Gu, Y. Zhang, N. Gu, D. Yang and J. Zhang, “Achieving High-Purity Colloidal Gold Nanoprisms and Their Application as Biosensing Platforms,” Journal of Colloid and Interface Science, Vol. 348, No. 1,August 2010, pp. 29-36. doi:10.1016/j.jcis.2010.04.013 [11] S. Nath, S. Jana, M. Pradhan and T. Pal, “Ligand-Stabi- lized Metal Nanoparticles in Organic Solvent,” Journal of Colloid and Interface Science, Vol. 341, No. 2, 2010, pp. 333-352. [12] J. R. Peralta-Videa, L. Zhao, M. L. Lopez-Moreno, G. Rosa, J. Hong and J. L. Torresdey, “Nanomaterials and the Environment: A Review for the Biennium 2008- 2010,” Journal of Hazardous Materials, Vol. 186, No. 1, 2011, pp. 1-15. doi:10.1016/j.jhazmat.2010.11.020 [13] E. Falletta, F. Ridi, E. Fratini, C. Vannucci, P. Canton, S. Bianchi, V. Castelvetro and P. Baglioni, “A Tri-Block  R. SEOUDI ET AL. Copyright © 2011 SciRes. WJNSE 60 Copolymer Templated Synthesis of Gold Nanostruc- tures,” Journal of Colloid and Interface Science, Vol. 357, No. 1, May 2011, pp. 88-94. doi:10.1016/j.jcis.2010.12.074 [14] Y. C. Lin, B. Y. Yu, W. C. Lin, S. H. Lee, C. H. Kuo and J. J. Shyue, “Tailoring the Surface Potential of Gold Na- noparticles with Self-Assembled Monolayers with Mixed Functional Groups,” Journal of Colloid and Interface Science, Vol. 340, No. 1, December 2009, pp. 126-130. doi:10.1016/j.jcis.2009.08.014 [15] W. K. Lee, S. H. Cha, K. H. Kim, B. W. Kim and J. C. Lee, “Shape-Controlled Synthesis of Gold Icosahedra and Nanoplates Using Pluronic P123 Block Copolymer and Sodium Chloride,” Journal of Solid State Chemistry, Vol. 182, No. 12, December 2009, pp. 3243-3248. doi:10.1016/j.jssc.2009.09.020 [16] C. Contado and R. Argazzi, “Size Sorting of Citrate Re- duced Gold Nanoparticles by Sedimentation Field-Flow Fractionation,” Journal of Chromatography A, Vol. 1216, No. 52, December 2009, pp. 9088-9098. doi:10.1016/j.chroma.2009.08.026 [17] J. P. Douliez, B. Novales and C. Gaillard, “Synthesising Gold Nanoparticles within Bola Fatty Acid Nanosomes,” Journal of Colloid and Interface Science, Vol. 337, No. 2, September 2009, pp. 610-613. doi:10.1016/j.jcis.2009.05.042 [18] S. I. Stoeva A. B. Smetana, C. M. Sorensen and K. J. Klabunde, “Gram Scale Synthesis of Aqueous Gold Col- loids Stabilized by Various Ligands,” Journal of Colloid and Interface Science, Vol. 309, No. 1, May 2007, pp. 94-98. doi:10.1016/j.jcis.2006.12.064 [19] Y. Sun and C. Lei, “Synthesis of Out-of-Substrate Au-Ag Nanoplates with Enhanced Stability for Catalysis,” An- gewandte Chemie, Vol. 121, No. 37, September 2009, pp. 6956-6959. doi:10.1002/ange.200902305 [20] N. Yan, J. Zhang, Y. Yuan, G. T. Chen, P. J. Dyson, Z. C. Li and Y. Kou, “Thermoresponsive Polymers Based on Poly-Vinylpyrrolidone: Applications in Nanoparticle Ca- talysis,” Chem Commun, Vol. 46, No. 10, 2010, pp. 1631- 1633. [21] S. S. Kinge and M. C. Calama, “Gold Nanoparticle As- semblies through Hydrogen-Bonded Supramolecular Me- diators,” Langmuir, Vol. 23, No. 17, 2007, pp. 8772- 8777. doi:10.1021/la700514u [22] S. S. Kumar, C. S. Kumar, J. Mathiyarasu and K. L. Phani, “Stabilized Gold Nanoparticles by Reduction Us- ing 3,4-Ethylenedioxythiophene-Polystyrenesulfonate in Aqueous Solutions: Nanocomposite Formation, Stability, and Application in Catalysis,” Langmuir. Vol. 23, No. 6, 2007, pp. 3401-3408. doi:10.1021/la063150h [23] M. Montalti, L. Prodi, N. Zaccheroni, M. Beltrame, T. Morotti and S. Quici, “Stabilization of Gold Nanopar- ticles by Metal Ions Complexation,” New Journal of Chemistry, Vol. 31, No. 1, 2007, pp.102-108. [24] N. Nilius, M. V. Ganduglia-Pirovano, V. Brázdová, M. Kulawik, J. Sauer and H.-J. Freund, “Electronic Proper- ties and Charge State of Gold Monomers and Chains Adsorbed on Alumina Thin Films on NiAl(110),” Physi- cal Review B, Vol. 81, No. 4, 2010, pp, 45422-45429. [25] M. T. Javed, N. Irfan and B. M. Gibbs, “Control of Combustion-Generated Nitrogen Oxides by Selective Non-Catalytic Reduction,” Journal of Environmental Management, Vol. 83, No. 3, May 2007, pp. 251-289. doi:10.1016/j.jenvman.2006.03.006 [26] E. J. Beckman, “Supercritical and Near-Critical CO2 in Green Chemical Synthesis and Processing,” The Journal of Supercritical Fluids, Vol. 28, No. 2-3, March 2004, pp. 121-191. doi:10.1016/S0896-8446(03)00029-9 [27] M. Sahimi and T. T. Tsotsis, “A Percolation Model of Catalyst Deactivation by Sit Coverage and Pore Block- age,” Journal of Catalysis, Vol. 96, No. 2, December 1985, pp. 552-562. doi:10.1016/0021-9517(85)90323-9 [28] R. R. Melkote and K. F. Jensen, “Models for Catalytic Pore Plugging, Application to Hydrodemetallation,” Che- mical Engineering Science, Vol. 44, No. 3, 1989, pp. 649-663. doi:10.1016/0009-2509(89)85041-9 [29] G. C. Bond, “Gold: A Relatively New Catalyst,” Cataly- sis Today, Vol. 72, No. 1-2, February 2002, pp. 5-9. doi:10.1016/S0920-5861(01)00522-3 [30] G. J. Hutchings, “Nanocrystalline Gold Catalysts: A Ref- lection on Catalyst Discovery and the Nature of Active Sites,” Gold Bulletin, Vol. 42, No. 4, 2009, pp. 260-266. doi:10.1007/BF03214947 [31] P. T. Anastas and J. C. Warner, “Green Chemistry: Theory and Practice,” Oxford University Press, New York, 1998. [32] X. G. Zhang, D. Y. Teng, Z. M. Wu, X. Wang, Z. Wang, D. M. Yu and C. X. Li, “PEG-Grafted Chitosan Nanopar- ticles as an Injectable Carrier for Sustained Protein Re- lease,” Journal of Materials Science: Materials in Medi- cine, Vol. 19, No. 12, 2008, pp. 3525-3533. doi:10.1007/s10856-008-3500-8 [33] A. S. Karikari, W. F. Edwards, J. B. Mecham and T. E. Long, “Influence of Peripheral Hydrogen Bonding on the Mechanical Properties of Photo-Cross-Linked Star- Shaped Poly(D,L-Lactide) Networks,” Biomacromole- cules, Vol. 6, No. 5, 2005, pp. 2866-2874. doi:10.1021/bm050375i [34] R. Song, R. Xuea, L. He, Y. Liu and Q. Xiao, “The Structure and Properties of Chitosan/Polyethylene Gly- col/Silica Ternary Hybrid Organic-Inorganic Films,” Chinese Journal of Polymer Science Vol. 26, No. 5, 2008, pp. 621-630. doi:10.1142/S0256767908003357 [35] J. G. Rouse and M. E. Van Dyke, “A Review of Kera- tin-Based Biomaterials for Biomedical Applications,” Materials, Vol. 3, No. 2, 2010, pp. 999-1014. doi:10.3390/ma3020999 [36] M. Bodnar, J. F. Hartmann and J. Borbely, “Synthesis and Study of Cross-Linked Chitosan-N-Poly(ethylene glycol) Nanoparticles,” Biomacromolecules, Vol. 7, No. 11, 2006, pp. 3030-3036. doi:10.1021/bm0605053 [37] K. Esumi, N. Takei and T. Yoshimura, “Antioxidant- Potentiality of Gold–Chitosan Nanocomposites,” Colloids and Surfaces B: Biointerfaces, Vol. 32, No. 2, October 2003, pp. 117-123. doi:10.1016/S0927-7765(03)00151-6  R. SEOUDI ET AL. Copyright © 2011 SciRes. WJNSE 61 [38] S. Wunder, F. Polzer, Y. Lu, Y. Mei and M. Ballauff, “Kinetic Analysis of Catalytic Reduction of 4-Nitrophe- nol by Metallic Nanoparticles Immobilized in Spherical Polyelectrolyte Brushes,” Journal of Physical Chemistry C, Vol. 114, No. 19, 2010, pp. 8814-8820. [39] M. Haruta, “Catalysis of Gold Nanoparticles Deposited on Metal Oxides,” CATTECH, Vol. 6, No. 3, 2002, pp. 102-115. |

