 American Journal of Anal yt ical Chemistry, 2011, 2, 194-199 doi:10.4236/ajac.2011.22022 Published Online May 2011 (http://www.SciRP.org/journal/ajac) Copyright © 2011 SciRes. AJAC Voltammetric Determination of Vitamin B6 at Glassy Carbon Electrode Modified with Gold Nanoparticles and Multi-Walled Carbon Nanotubes Yuzhong Zhang, Yuehong Wang College of Chemistry and Materials Science, Anhui Key Laboratory of Chemo-Biosensing, Anhui Normal University, Wuhu, China E-mail: zhyz65@mail.ahnu.edu.cn Received January 9, 2011; revised February 25, 2011; accep ted M arch 1, 2011 Abstract In this work, the gold nanoparticles (Au NPs)/multi-walled carbon nanotubes (MWCNTs) composite film modified glassy carbon electrode (GCE) was fabricated, and scanning electron microscopy (SEM) was used to investigate the assemble process of the composite film. In pH 7.0 PBS, an oxidation peak of the vitamin B6 (VB6) was only observed at composite film modified electrode. Under the optimized conditions, the cur- rent intensity was linear with the concentrations of VB6 in the range of 1.59 to 102.74 μg·mL–1 with a detec- tion limit of 0.53 μg·mL–1 (S/N = 3). The modified electrode had been applied in pharmaceutical analysis, and obtained good results. Keywords: Vitamin B6, Gold Nanoparticles, Multi-Walled Carbon Nanotubes, Differential Pulse Voltammetry 1. Introduction Vitamin B6 (VB6), also known as pyridoxine, is part of the B group vitamins and plays an important role in the synthesis and metabolism of amino acid. The deficiency of VB6 has been suggested as the cause of many types of illness and disease [1]. Several analytical methods have been described in literature for determination of VB6, including spectrophotometry [2,3], liquid chromatogram- phy [4,5] and electrochemistry method [6,7]. R. Jimé- nez-Prieto et al determined VB6 by spectrophotometric techniques in the presence of other vitamins [8]. Fang et al [9] detected VB6 in pharmaceutical preparations by micellar electrokinetic chromatography with amperomet- ric electrochemical method. Söderhjelm and Lindquist [10] firstly detected VB6 in pharmaceutical preparations with electrochemical method; however, the interferences from ascorbic acid were existed and had not been re- pelled. Until now, few papers had been described on the determination of VB6 using electrochemical method. Carbon nanotubes (CNTs) have been an important group of nanomaterials with attractive geometrical, elec- tronic and chemical properties. Recently, composite ma- terials based on integration of CNTs and other materials had gained growing interest, including conducting poly- mers, redox mediators and metal nanoparticles [11-14]. Gold nanoparticles (Au NPs) were another important nanomaterial, and it had also been used in biosensor of glucose [15,16], DNA [17], pesticides [18], and arsenic (III) [19]. We have used Au NPs/CNTs composite film modified electrode to detect Sal and target DNA [20,21] and obtained good results. From our practical experiment, we find the Au NPs/CNTs composite are good materials, and hope to open more applied window. We want to study medicament analysis based on this ideas. To our knowledge, there was no report of VB6 detection based on Au NPs and MWCNTs composite film modified elec- trode. In this work, Au NPs was electrodeposited on the sur- face of the MWCNTs modified electrode, and the elec- trochemical behavior of VB6 at this modified electrode was investigated, the experiment results showed an oxi- dation peak of VB6 was only observed in CVs and the electrochemical response of VB6 was higher in contract to MWCNTs or Au NPs modified electrode alone. Under optimal experiment condition, the current intensity was linear with the concentrations of VB6 in the range of 1.59 to 102.74 μg·mL–1 with a detection limit of 0.53 μg·mL–1  Y. Z. ZHANG ET AL.195 (S/N = 3). When the modified electrode was used to de- tect VB6 in pharmaceutical preparations, a satisfied result was obtained. 2. Experimental 2.1. Reagents Multi-walled carbon nanotubes with carboxylic acid groups (MWCNTs, with a diameter of about 30 nm and length of around 30 μm, purity > 95%) were obtained from Chengdu Institute of Organic chemistry, Chinese Academy of Sciences. Vitamin B6, Vitamin B1 (VB1), Vi- tamin B2 (VB2), Ascorbic acid (AA), and HAuCl4·4H2O were obtained from Shanghai Chemical Reagent Co., Ltd (China). The phosphate buffer solution (PBS, 0.10 mol·L –1) was prepared by NaH2PO4 and Na2HPO4, and adjusted the pH with H3PO4 and NaOH solutions. All chemicals were of analytical grade and used without fur- ther purification. All solutions were prepared with twice- quartz-distilled water. 2.2. Apparatus Cyclic voltammetry (CV) and differential pulse voltam- metry (DPV) were performed on a CHI 650C electro- chemical workstation (Shanghai Chenhua Instruments Co., China). The three-electrode system consisted of a bare GCE or modified electrode as a working electrode, a platinum wire as a counter electrode, and a saturated calomel electrode (SCE) as a reference electrode. All potentials given in this work were referenced to the SCE reference electrode. The morphologies of different modified electrodes were obtained using a scanning electron microscopy (SEM) with JEOL JSM-4800F microscopy (Hitachi, Japan). 2.3. Preparation of the Au NPs/MWCNTs/GCE The bare GCE was carefully polished sequentially with 0.3 and 0.05 m alumina slurries on microcloth pads followed by rinsing successively in an ultrasonic bath with acetone, absolute alcohol and twice-quartz-distilled water for 3 min, respectively. Afterwards, the electrode was electrochemically treated by cycling the potential between − 0.3 and + 1.5 V in 0.50 mol·L–1 H2 SO4 with a scan rate of 100 mV·s–1 until a reproducible cyclic volt- ammogram was observed, followed it is rinsed with twice-quartz-distilled water thoroughly and dried in a nitrogen stream. 5 µL of 1.0 mg·mL–1 MWCNTs suspensions (10.0 mg of MWCNTs was dispersed in 10.0 mL of ethanol with the help of ultrasonication) were dropped on the surface of pretreated GCE, and then it was dried at room tem- perature. The Au NPs/MWCNTs/GCE was obtained by immersing the MWCNTs/GCE into 0.10 mol·L–1 KNO3 solution containing 1 mmol·L–1 HAuCl4 and electrode- posited 20 s at – 0.2 V (vs. SCE). 2.4. Procedure The preparation of sample: 1.0 mL of injection (Henan Topfond Pharmaceutical Co., Ltd.) was transferred to a 10.0 mL volumetric flask, diluted to the mark with twice- quartz-distilled water. Next, 2.0 mL of solution above was taken to 10.0 mL volumetric flask, diluted to the mark with twice-quartz-distilled water. The preparation of tablets: 20 tablets VB6 (Nanjing Baijingyu Pharmaceutical Co., Ltd.) were taken to grind a fine powder. Next, a suitable amount powder (about one tablet mount) was taken to be dissolved with twice- quartz-distilled water, and it was then transferred to 50 mL volumetric flask, diluted to the mark with twice- quartz-distilled water. Final, it was filtered to remove infusible substance. Electrochemical measurement: 5.0 mL of 0.10 mol·L –1 PBS containing a suitable amount of VB6 was added to the 10.0 mL cell. Before experiment, the solu- tion was purged with nitrogen for 10 minutes, and the flow of nitrogen was maintained during the experiment. In DPV measurements, the experiment parameters were following: initial potential: 0.3 V; final potential: 0.9 V; amplitude: 0.05 V; pulse width: 0.05 s; sample width: 0.0167. 3. Results and Discussion 3.1. SEM of the Different Modified Electrodes Figure 1 displayed SEM morphologies of MWCNTs/ GCE (a) and Au NPs/MWCNTs/GCE (b). It was ob- served that the MWCNTs distributed uniformly on the surface of GCE and Au NPs had been uniformly deco- rated onto the surface of MWCNTs (Figure 1(b)). The diameter of Au NPs was about 50 nm. 3.2. Electrochemical Behaviors of VB6 at Bare or Modified Electrode Electrochemical behaviors of VB6 at various modified electrode in 0.10 mol·L–1 PBS were investigated by CV technique. Figure 2 showed the CVs of VB6 at the bare GCE, the Au NPs/GCE, the MWCNTs/GCE, and the Au NPs/MWCNTs/GCE. For the bare GCE (Figure 2(a)) and Au NPs/GCE (Figure 2(b)), the responses of VB6 ere poor. When MWCNTs or Au NPs/MWCNTs was w \ Copyright © 2011 SciRes. AJAC 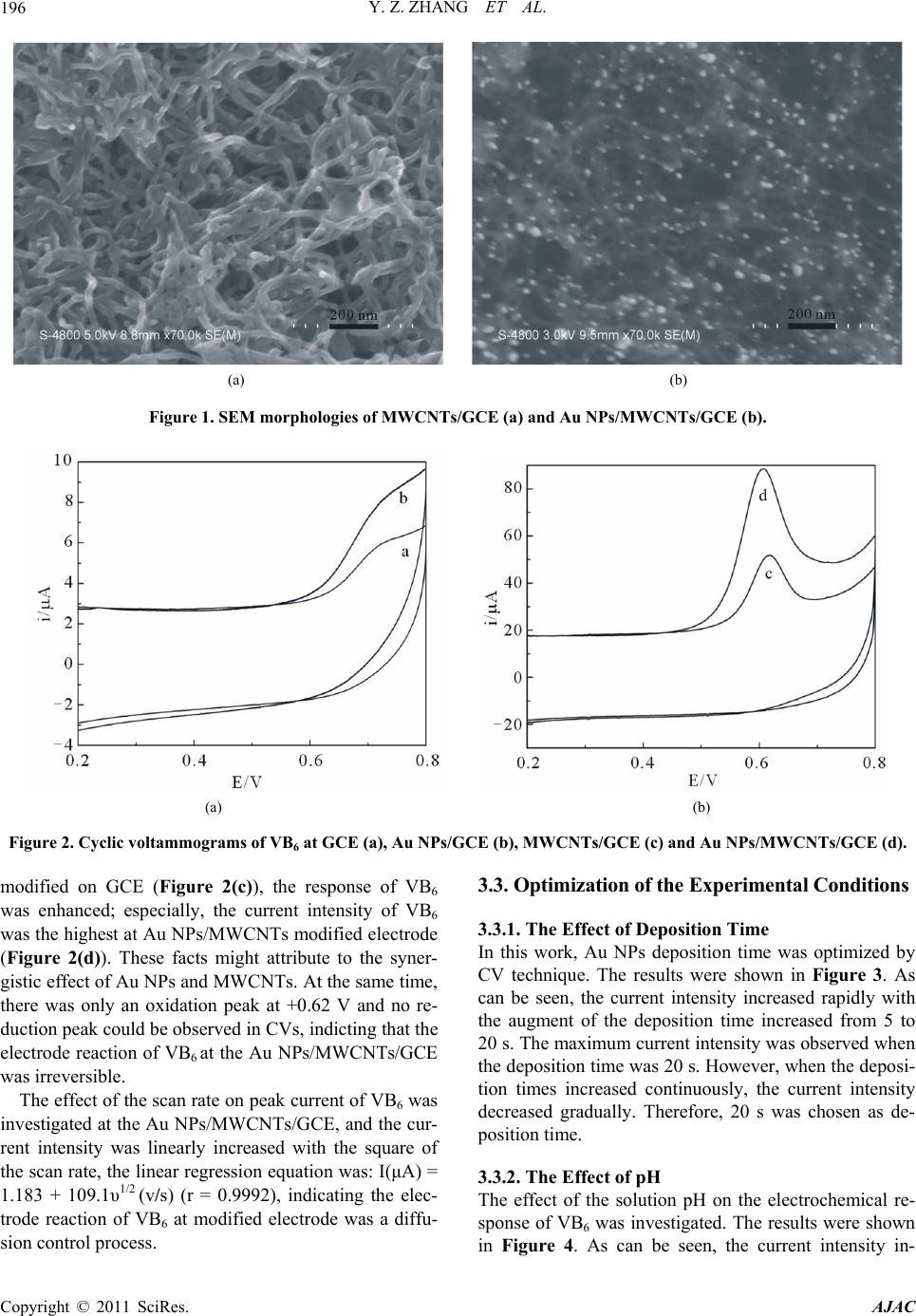 Y. Z. ZHANG ET AL. Copyright © 2011 SciRes. AJAC 196 (a) (b) Figure 1. SEM morphologies of MWCNTs/GCE (a) and Au NPs/MWCNTs/GCE (b). (a) (b) Figure 2. Cyclic voltammograms of VB6 at GCE (a), Au NPs/GCE (b), MWCNTs/GCE (c) and Au NPs/MWCNTs/GCE (d). 3.3. Optimization of the Experimental Conditions modified on GCE (Figure 2(c)), the response of VB6 was enhanced; especially, the current intensity of VB6 was the highest at Au NPs/MWCNTs modified electrode (Figure 2(d)). These facts might attribute to the syner- gistic effect of Au NPs and MWCNTs. At the same time, there was only an oxidation peak at +0.62 V and no re- duction peak could be observed in CVs, indicting that the electrode reaction of VB6 at the Au NPs/MWCNTs/GCE was irreversible. 3.3.1. The Effect of Deposition Time In this work, Au NPs deposition time was optimized by CV technique. The results were shown in Figure 3. As can be seen, the current intensity increased rapidly with the augment of the deposition time increased from 5 to 20 s. The maximum current intensity was observed when the deposition time was 20 s. However, when the deposi- tion times increased continuously, the current intensity decreased gradually. Therefore, 20 s was chosen as de- position time. The effect of the scan rate on peak current of VB6 was investigated at the Au NPs/MWCNTs/GCE, and the cur- rent intensity was linearly increased with the square of the scan rate, the linear regression equation was: I(μA) = 1.183 + 109.1υ1/2 (v/s) (r = 0.9992), indicating the elec- trode reaction of VB6 at modified electrode was a diffu- sion control process. 3.3.2. The Effect of pH The effect of the solution pH on the electrochemical re- sponse of VB6 was investigated. The results were shown in Figure 4. As can be seen, the current intensity in-  Y. Z. ZHANG ET AL.197 Figure 3. The plots of the peak currents of VB6 vs electro- deposition time of Au NPs, c = 30 μg·mL–1. Figure 4. The effect of pH on peak currents (a) and poten- tial of VB6 (b). The pH value are 5.0, 6.0, 7.0, 8.0, 9.0 (from a to e). creased with solution pH from 5.0 - 7.0, then it decreased when the pH was over 7.0. The maximum current inten- sity was observed when pH was 7.0. The peak potential was linearly shifted negatively with the augment of solu- tion pH, indicating that protons had taken part in the electrode reaction process of VB6. Therefore, pH 7.0 was selected in all following experiments. 3.3.3. Calibration Curve The calibration curve of VB6 detection was performed with DPV technique. Under the optimized experimental conditions, a series of different concentrations of VB6 were measured. The current intensity at + 0.62 V was used to obtain the calibration curve. The results showed that the current intensity was linear with the concentra- tions of VB6 in the range of 1.59 to 102.74 μg·mL–1 as shown in Figure 5. The linear regression equation was Ipa (μA) = 0.2759 c + 0.7007 (C- μg·mL–1) (r = 0.9994) with the detection limit of 0.53 μg·mL–1 (S/N = 3). 3.3.4. Stability and Reproducibility The reproducibility of the modified electrode was inves- tigated by successive measurements of 30 μg·mL–1 VB6 by DPV (n = 6), the obtained peak current value was about the range of 8.79 to 9.24 μA with a relative stan- dard deviation (RSD) of 2.05%. The stability of the modified electrode was investigated after it was storage in air for 2 weeks. The obtained peak currents were al- most constant. Therefore, the modified electrode had a good stability and reproducibility. 3.3.5. Interference Study In these experiments, we investigated the interference of several species on the determination of VB6, such as VB1, VB2, AA, glucose, starch. The experiment results were shown in Table 1, from Table 1, there were no interfer- ences existed for the determination of 30 μg·mL–1 VB6 in the presence of 20-fold VB1, a 50-fold VB2, 100-fold AA, glucose and starch. Figure 5. Differential pulse voltammograms of VB6 at the modified electrode. The concentrations of VB6 (μg·mL–1) are: 1.59, 6.299, 9.375, 18.32, 22.64, 33.03, 40.31, 47.27, 60.93, 74.20, 90.28, 102.74. Copyright © 2011 SciRes. AJAC  Y. Z. ZHANG ET AL. 198 3.3.6. Analytical Applications In this work, the injection and tablet of VB6 were ana- lyzed by the standard addition method. For comparison purposes, the obtained results were compared with that of China Pharmacopoeia Method [22] (shown in Tables 2, 3). From Table 2 and Table 3 we could observe that the obtained values were agreement with the results found by the China Pharmacopoeia Method. 4. Conclusions The Au NPs/MWCNTs composite film modified elec- trode showed a strong electrochemical response towards VB6 in pH 7.0 and AA showed no interference during VB6 detection. The modified electrode had been applied in medication analysis, and obtained good results. Table 1. The inference of coexistent substances. Coexisting Currenta Multiple amount Currentb Relative substance μA(n = 3) added μA(n = 3) deviation % Glucose 200.0 8.738 –2.39 Starch 200.0 8.821 –1.46 Ascorbic acid 8.952 100.0 8.708 –2.73 Vitamin B1 20.0 9.233 3.14 Vitamin B2 50.0 9.134 2.03 a. the average peak current of VB6. b. the average peak current of VB6 after adding different coexisting substances. Table 2. Results for the determination of VB6 in tablets. This method Pharmacopeia method Sample Labeled mg per tablet Found mg per tablet RSD, n = 5 (%) Found mg per tablet RSD, n = 5 (%) 1 10.0 9.95 3.3 9.96 1.1 2 10.0 9.89 3.4 9.85 0.9 3 10.0 9.82 2.8 9.85 0.8 The tables were obtained from Nanjing Baijinhyu Pharmaceutical Co., Ltd. Table 3. Results for the determination of VB6 in injections. This method Pharmacopeia method Sample Labeled mg· mL –1 Found mg· mL –1 RSD, n = 5 (%) Found mg· mL –1 RSD, n = 5 (%) 1 50.0 50.4 1.7 50.1 0.9 2 50.0 49.5 2.4 49.3 0.6 3 50.0 49.3 3.2 49.2 0.5 4 50.0 50.2 1.9 50.1 0.9 5 50.0 49.7 2.7 49.9 0.8 The injections were obtained from Henan Topfond Pharmaceutical Co., Ltd. 5. Acknowledgements This project was financially supported by the Nature Science Foundation of China (NSFC) (No. 20675002). 6. References [1] G. Melentyeva and L. Antonova, “Pharmaceutical Chem- istry,” Mir Publishers, Moscow, 1988, pp. 375-393. [2] García, S. Blázquez, M. P. S. Andrés and S. Vera, “De- termination of Thiamine, Riboflavin and Pyridoxine in Pharmaceuticals by Synchronous Fluorescence Spec- trometry in Organized Media,” Analytica Chimica Acta, Vol. 434, No. 2, May 2001, pp. 93-99. [3] P. O. Barrales, M. L. F. Córdova and A. M. Díaz, “Mi- crodetermination of Vitamin B1 in the Presence of Vita- mins B2, B6, and B12 by Solid-Phase UV Spectropho- tometry,” Analytical Chemistry, Vol. 70, No. 2, January 1998, pp. 271-275. [4] W. Y. Hou, H. M. Ji and E. K. Wang, “Liquid Chroma- tography of Vitamin B6 with Electrochemical Detection Using a Carbon Fibre Electrode,” Analytica Chimica Acta, Vol. 230, March 1990, pp. 207-211. doi:10.1016/S0003-2670(00)82784-1 [5] G. Chen, X. G. Ding, Z. G. Cao and J. N. Ye, “Determi- nation of Melatonin and Pyridoxine in Pharmaceutical Preparations for Health-Caring Purposes by Capillary Electrophoresis with Electrochemical Detection,” Ana- lytica Chimica Acta, Vol. 408, No. 1-2, March 2000, pp. 249-256. doi:10.1016/S0003-2670(99)00809-0 [6] F. S. T. Marcos, S. Aline, C. M. Fernando, J. L. H. Mar- colino, F. O. Fatibello and É. T. G. Cavalheiro, “Deter- mination of Vitamin B6 (Pyridoxine) in Pharmaceutical Preparations by Cyclic Voltammetry at a Copper (II) Hexacyanoferrate (III) Modified Carbon Paste Elec- trode,” Journal of the Brazilian Chemical Society, Vol. 14, No. 2, April 2003, pp. 316-321. doi:10.1590/S0103-50532003000200021 [7] F. S. T. Marcos, M. Glimaldo, E. R. Dockal and É. T. G. Cavalheiro, “Voltammetric Determination of Pyridoxine (vitamin B6) at a Carbon Paste Electrode Modified with Vanadyl (IV)-Salen Complex,” Analytica Chimica Acta, Vol. 508, No. 1, April 2004, pp. 79-85. doi:10.1016/j.aca.2004.03.021 [8] R. Jiménez-Prieto, M. Silva and D. Pérez-Bendito D, “Analytical Assessment of the Oscillating Chemical Re- actions by Use Chemiluminescence Detection,” Talanta, Vol. 44, No. 8, August 1997, pp. 1463-1472. doi:10.1016/S0039-9140(97)00047-7 [9] Q. Hu, T. S. Zhou, L. Zhang, H. Li and Y. Z. Fang, “Se- paration and Determination of Three Water-Soluble Vi- tamins in Pharmaceutical Preparations and Food by Mi- cellar Electrokinetic Chromatography with Ampero- met- ric Electrochemical Detection,” Analytica Chimica Acta, Vol. 437, No. 1, June 2001, pp. 123-129. [10] P. Söderhjelm and J. Lindquist, “Voltammetric Determi- nation of Pyridoxine by Use of a Carbon Paste Elec- Copyright © 2011 SciRes. AJAC  Y. Z. ZHANG ET AL. Copyright © 2011 SciRes. AJAC 199 trode,” Analyst, Vol. 100, No. 1190, May 1975, pp. 349- 354. doi:10.1039/an9750000349 [11] M. Y. Li, G. Q. Zhao, Z. L. Yue and S. S. Huang, “Sen- sor for Traces of Hydrogen Peroxide Using an Electrode Modified by Multiwalled Carbon Nanotubes, a Gold- Chitosan Colloid, and Prussian Blue,” Microchimica Acta, Vol. 167, No. 2, November 2009, pp. 167-172. doi:10.1007/s00604-009-0238-z [12] J. Z. Xu, J. J. Zhu, Q. Wu, Z. Hu and H. Y. Chen, “An Amperometric Biosensor Based on the Coimmobilization of Horseradish Peroxidase and Methylene Blue on a Carbon Nanotubes Modified Electrode,” Electroanalysis, Vol. 15, No. 3, March 2003, pp. 219-224. doi:10.1002/elan.200390027 [13] L. Q. Jiang and L. Gao, “Modified Carbon Nanotubes: An Effective Way to Selective Attachment of Gold Nanoparticles,” Carbon, Vol. 41, No. 15, November 2003, pp. 2923-2929. doi:10.1016/S0008-6223(03)00339-7 [14] L. Wang, S. J. Guo, L. J. Huang and S. J. Dong, “Alter- nate Assemblies of Polyelectrolyte Functionalized Car- bon Nanotubes and Platinum Nanoparticles as Tunable Electrocatalysts for Dioxygen Reduction,” Electrochem- istry Communications, Vol. 9, No. 4, April 2007, pp. 827- 832. doi:10.1016/j.elecom.2006.11.009 [15] Y. Liu, S. Wu, H. X. Ju and L. Xu, “Amperometric Glu- cose Biosensing of Gold Nanoparticles and Carbon Nanotube Multilayer Membranes,” Electroanalysis, Vol. 19, No. 9, May 2007, pp. 986-992. doi:10.1002/elan.200603814 [16] R. F. Gao and J. B. Zheng, “Amine-Terminated Ionic Liquid Functionalized Carbon Nanotube-Gold Nanopar- ticles for Investigating the Direct Electron Transfer of Glucose Oxidase,” Electrochemistry Communications, Vol. 11, No. 3, March 2009, pp. 608-611. doi:10.1016/j.elecom.2008.12.060 [17] S. H. Lim, J. Wei and J. Y. Lin, “Electrochemical Genosensing Properties of Gold Nanoparticle-Carbon Nanotube Hybrid,” Chemical Physics Letters, Vol. 400, No. 4-6, December 2004, pp. 578-582. doi:10.1016/j.cplett.2004.11.023 [18] Y. Zhang, T. F. Kang, Y. W. Wan and S. Y. Chen, “Gold Nanoparticles-Carbon Nanotubes Modified Sensor for Electrochemical Determination of Organophosphate Pes- ticides,” Microchimica Acta, Vol. 165, No. 3-4, January 2009, pp. 307-311. doi:10.1007/s00604-008-0134-y [19] L. Xiao, G. G. Wildgoose and R. G. Compton, “Sensitive Electrochemical Detection of Arsenic (III) Using Gold Nanoparticle Modified Carbon Nanotubes via Anodic Stripping Voltammetry,” Analytica Chimica Acta, Vol. 620, No. 1-2, July 2008, pp. 44-49. doi:10.1016/j.aca.2008.05.015 [20] H. Y. Ma, L. P. Zhang, Y. Pan, K. Y. Zhang and Y. Z. Zhang, “A Novel Electrochemical DNA Biosensor Fab- ricated with Layer-by-Layer Covalent Attachment of Multiwalled Carbon Nanotubes and Gold Nanoparticles,” Electroanalysis, Vol. 20, No. 11, June 2008, pp. 1220- 1226. doi:10.1002/elan.200704169 [21] S. J. Zhang, M. L. Xu and Y. Z. Zhang, “Simultaneous Voltammetric Detection of Salsolinol and Uric Acid in the Presence of High Concentration of Ascorbic Acid with Gold Nanoparticles/Functionalized Multiwalled Carbon Nanotubes Composite Film Modified Electrode,” Electroanalysis, Vol. 21, No. 23, December 2009, pp. 2607-2610. doi:10.1002/elan.200900228 [22] Editorial Committee of Pharmacopoeia of People’s Re- publicof China, “People’s Republic of China Pharmaco- poeia (Part II),” Chemical Industry Press, Beijing, 2000.
|