Paper Menu >>
Journal Menu >>
 Journal of Biomaterials and Nanobiotechnology, 2011, 2, 293-300 doi:10.4236/jbnb.2011.23036 Published Online July 2011 (http://www.SciRP.org/journal/jbnb) Copyright © 2011 SciRes. JBNB 293 Macrophage Inflammatory Response to TiO2 Nanotube Surfaces Lisa M. Chamberlain1,2, Karla S. Brammer1, Gary W. Johnston1, Shu Chien2, Sungho Jin1* 1Materials Science & Engineering, University of California, San Diego, USA; 2Department of Bioengineering, Institute of Engineer- ing in Medicine, University of California, San Diego, USA. Email: *jin@ucsd.edu Received February 9th, 2011; revised March 2nd 2011; accepted April 18th, 2011. ABSTRACT It is well known that the native oxide layer on titanium (Ti) implants is responsible for its superior biocompatibility and tissue integration. Recent efforts have targeted titanium dioxide (TiO2) as a good candidate for surface modification at the nanoscale, leading to improved nanotextures for enhancing host integration properties. Here we explore the in vitro inflammatory response of macrophages to TiO2 nanotube surface structures with different diameters (30, 50, 70, and 100 nm) created by a simple electrochemical anodization process. This work was designed to study the nanosize effect for controlling and optimizing inflammatory response to a Ti implant surface utilizing nanotechnology. Using intracellu- lar staining and flow cytometry for detecting macrophage TNF cytokine expression, we have found that 70 nm diameter nanotube surfaces have the best advantage in terms of diameter size by producing the weakest inflammatory response, compared to a commercially available Ti surface without oxide modification. We also present cell-freedata on free radical scavenging using the nanotube surfaces with different diameters to test the removal of nitric oxide from solution; again, our findings indicate that 70 nm titanium dioxide nanotubes exhibit optimal removal of nitric oxide from solution, making them excellent candidates for use in medical devices that would benefit from decreased inflammatory response. Keywords: Macrophage, Inflammation, Nitric Oxide, Nanotube, TiO2 1. Introduction Medical devices, which are commonly used to improve the health of patients, serve to repair joints, reopen blood vessels, and trigger electrical stimuli among numerous applications. Implantation of medical devices is compli- cated by the creation of trauma caused by the necessary surgery which is neededfor the placement of these de- vices [1]. The natural wound healing sequel to this sur- gical trauma is in turn challenged by the presence of me- dical devices, leading to an alternate wound healing re- sponse around the device generally termed the “foreign body response.” The foreign body response is an inflam- matory response to the implanted material, and the inten- sity of this immediate inflammatory response to im- planted devices directly impacts healing around and tis- sue integration of the implant, as well as down-the- road functioning of the medical device [1]. Because the body “sees” the surface of the implant, many methods to change the surface properties of bio- materials have been undertaken in hopes of 1) improving the desired native cell/tissue adhesion and overall host integration, 2) reducing the unwanted inflammatory re- sponse of macrophages and various defense cells, and 3) eliminating the subsequent foreign body response and rejection of the medical device. One method of inquiry towards improving the integration of materials with na- tive tissue, has been to incorporate nanotopography on the surface, which presents a surface with features that are on the same nanoscale as the in vivo biological mate- rials such as biomolecules or enzymes, proteins and ex- tracellular matrices (e.g. collagen), cell surface receptors or integrins, etc. that are nanometer in dimension. When incorporated into culture surfaces, nanotopographies can vary greatly, e.g., nanoneedles, nanorods, nanoporesor- nanospheres, etc. In terms of immune cell reactions to nanotopography, previous studies on macrophages grown on zinc oxide nanorods exhibited low viability on tall, thin nanorod surfaces of approximately 50 nm diameter [2]. On the other hand, titanium dioxide surfaces with nanofeatures slightly larger in diameter (~70 nm) and shorter in height have demonstrated much better macrophage adhesion and survival, but exhibited less production of inflamma-  Macrophage Inflammatory Response to TiO2 Nanotube Surfaces Copyright © 2011 SciRes. JBNB 294 tory cytokines by the macrophages cultured on the nanotopographic surfaces compared to a flat surface of titanium without a nanostructure [3]. Glass nanowires have also demonstrated a trend of greater in vitro inflam- matory response with taller than shorter nanofeatures, although the same study did not detecta significant trend for inflammatory response related to the height for poly- mer nanofeatures [3]. In addition, the culture of macro- phages on patterned nanofeatures using several different polymers exhibited no significant differences in the re- lease of inflammatory cytokines [4]. Overall, it appears that there is no clear trend regarding macrophage re- sponse and inflammation due to nanotopography (for review see reference [5]). Therefore further studies to elucidate the effect of nanotopography on macrophage response would be extremely valuable for understanding and controlling the inflammatory cell behavior for im- plantable devices. Several labs have been investigating the use of TiO2 nanotubes as a titanium surface modification to the na- tive metal-oxide for use as an improved biomaterial sur- face, and many interesting cellular responses have been observed with these materials [6-10]. This type of TiO2 surface nano-configuration is advantageous in regulating many positive cell and tissue responses for various ap- plications for tissue engineering and regenerative medi- cine; therefore it was chosen as the primary substrate of investigation in this research on the macrophage inflam- matory cell response. Previously, when comparing the effect of different diameters or inner pore sizes of TiO2 nanotubes, it was found that there were distinct size re- gimes for controlling the behaviors of osteoblast [7], chondrocyte [11], and mesenchymal stem cells [8,10,12]. As well, a study comparing 20 nm vs. 200 nm pores of aluminum oxide revealed unique differences in the in- flammatory response of macrophages [13]. Because pore size seems to play an important role in controlled cell behavior, here we investigate the potential influence of differently sized TiO2 nanotubes on the in vitro inflam- matory responses of macrophages, and we hypothesize that the geometry of the nanotube may have significant impacts on the inflammatorycell behavior. 2. Materials and Methods 2.1. Substrate Preparation Annealed titanium foil (0.25 mm thick) was purchased from Alpha Aesar (Ward Hill, MA) and cut into 2 cm × 5 cm samples. Samples were cleaned with an ultrasonic cleaner for an hour in acetone, liberally rinsed with DI water, and dried overnight in a 60˚C oven. Nanotube surfaces were prepared in a 1:7 volumetric ratio of acetic acid (99.99% purity, Sigma–Aldrich) to 0.5% w/v hydro- fluoric acid in water (48% w/v, EM Science, USA) by anodizing 2 cm × 5 cm samples for 30 minutes at differ- ent voltages (5 V, 10 V, 15 V, and 20 V). Surfaces were then rinsed liberally with DI water for at least 30 seconds and dried in a 60˚C oven overnight. Following the crea- tion of the TiO2 nanotubes, samples of these and un- treated titanium were placed in a tube furnace and baked at 500˚C for 2 hours to anneal the material and crystallize the fabricated amorphousTiO2 to anatase phase for opti- mal cell culture conditions, as the effect of crystallinity has already been determined and previously reported [6, 14]. Samples were then cut into 1 cm × 1 cm pieces for cell culture, and baked again in a tube furnace at 260˚C for 2 hours to ensure the degradation of any endotoxin that may have been introduced to the samples through handling. Samples were further sterilized in an autoclave prior to use in cell culture and oxygen radical elimination experiments. The presence of nanotubes was visualized by scanning electron microscopy (SEM, XL30, FEI Co., USA). 2.2. Primary Cell Sourcing Pathogen-free female C57BL/6 mice, 6 to 8 weeks old, were purchased from Charles River Laboratories. Mice were maintained in the University of California San Diego animal facilities, and were given sterile water, and mouse chow for the duration of the experiments. Animal guidelines for the care and use of laboratory animals have been observed; all experimental protocols used in this study were approved by the Institutional Animal Care and Use Committee of the University of California San Diego. Bone marrow cells were harvested from murine tibias and femurs and differentiated into macrophage cells us- ing previously described methods [15-17]. Bone marrow cells were flushed from long bones, and then differenti- ated into bone-marrow derived macrophages (BMMΦ) by incubating in complete DMEM (cDMEM: DMEM supplemented with 10% heat inactivated FBS, 10% of supernatant from L-929 fibroblast cells (ATCC, Manas- sas, VA), 1% penicillin-streptomycin (Gibco®, Carlsbad, CA), 0.01M Hepes buffer (Gibco®, Carlsbad, CA), 1mM sodium pyruvate (Gibco®, Carlsbad, CA), and 1% of a 100X MEM non-essential amino acids solution (Gibco®, Carlsbad, CA)). The cells were cultured for 7 days on ti- ssue culture treated dishes, with media changes every 2 days. Adherent day 7 cultured cells were selected as ma- ture macrophages for further studies. This protocol has been shown to produce a mature macrophage phenotype [16]. Replicates are defined as cells from different mice. A minimum of three replicates were completed for all experiments.  Macrophage Inflammatory Response to TiO2 Nanotube Surfaces Copyright © 2011 SciRes. JBNB 295 2.3. Macrophage Adhesion and Growth on Substrates Mature macrophages were removed from culture sur- faces by rinsing with non-cationic PBS (Gibco®, Carls- bad, CA), soaking for 5 minutes in non-cationic PBS, and finally scraping with a cell scraper. Cell suspensions were counted, and 500,000 cells were seeded into each well of a 24-well plate containing one 1cm X 1cm model material in each well in the complete DMEM. Cells were allowed to adhere and proliferate for 24 hours. Samples were then rinsed 3 times with PBS, and fixed by soaking in a solution of 4% paraformaldahyde in PBS for 20 minutes. Nuclei were stained using DAPI (1:1000, Che- micon) in PBS overnight and rinsed 3 times with PBS. Substrates were mounted onto glass slides using Fluor- mount-G (Southern Biotech), visualized and photo- graphed using a LEICA DM IRB microscope. Five ran- dom fields were imaged from each sample, and nuclei were counted using Image J software (NIH). 2.4. Scanning Electron Microscopy (SEM) for Cell Morphological Examination After 24 hours of incubation, the cells on the substrates were washed with PBS and fixed with 2.5 w/v% glu- taraldehyde (Sigma, USA) in PBS for 1 hour. After fixa- tion, they were washed three times with PBS for 15 min- utes perwash. Then the cells were dehydrated in a graded series of ethanol (50%, 70%, 90%, and 100% v/v) for 30 minutes each and left in 100% ethanol until they were dried with acritical point dryer (EMS 850, Electron Mi- croscopy Science Co., USA). Next, the dried samples were sputter-coated with metal for SEM (scanning elec- tron microscopy) examination. The morphology of the adhered cells were observed using SEM (XL30, FEI Co., USA). 2.5. Macrophage Cytokine Expression Following Exposure to Experimental Substrates Measurement of intracellular TNF in all cell types from all substrate cultures were conducted by plating cells at sub-confluent levels in tissue culture polystyrene (TCPS) dishes (used as a control), and in 24-well plates with 1 cm × 1 cm substrates with 1 μL/mL of monensin (ebio- sciences, San Diego, CA). In a separate TCPS dish, 5 μg/mL LPS (E. coli LPS, Sigma–Aldrich, St. Louis, MO) was added for a positive control. The cells were incu- bated for 8 hours under normal culture conditions. The treatment in this protocol stops the export of cellular pro- ducts, thus allowing for the buildup of cytokines within the cell. The cells were removed from the culture surface, fixed and permeabilized in suspension using a fixation and permeabilization kit (ebiosciences, San Diego, CA) and finally stained for intracellular tumor necrosis factor, TNF- (clone MP6-XT22, rat IgG1). Antibodies were purchased from eBioscience (San Diego, CA) as direct conjugates of FITC, and PE, respectively. Data acquisi- tion and analysis for this study were done using a FAC- Scan (BD Biosciences, Mountain View, CA), CellQuest TM software (BD Biosciences, San Jose, CA), and WinMDI software (Joseph Trotter, The Scripps Research Institute, San Diego, CA). Data presented represent at least three replicates, with each replicate of cells coming from a separate mouse. 2.6. Surface Interaction with Nitric Oxide To investigate the nitric oxide quenching properties of difference surfaces, an NO donor DPTA-NO was used to make a solution of known NO concentration, and a 500 µL sample of this solution was then subjected to each of the surfaces for 15minutes. Samples in duplicate were taking from each well and a Griess reagent was used to stabilize the NO concentration by converting it into ni- trite (NO2). The measured concentration of NO2 in each of the extracted samples correlates to the remaining con- centration of NO after the surfaces have scavenged some of free radicals.The concentration of the total nitrate was determined from the absorbance at wavelength λ = 550 nm by using a UV-Vis spectrophotometer (BiomateTM 3, Thermo Electron Co., USA) and calculated with the aid ofa dilution standard curve. 2.7. Statistical Significance All bar graphs are displayed as the mean ± standard error. Sigma Plot software (2001) which specializes in scien- tific data analysis and presentation, was utilized for demonstrating statistical significance for the assays. One- way ANOVAs were performed using the pairwisemulti- ple comparison procedure. 3. Results 3.1. Substrate Properties Are Consistent with Previously Published Results The experimental TiO2 nanotube surfaces appear similar to previously fabricated surfaces [6-8,11,12,18], and have similar size characteristics and discrete uniform geome- tries (Figure 1). Figure 1 shows highly ordered, verti- cally aligned nanotube structures with different diameters fabricated by varying the anodization potential. Applied voltage for creating nanotube surfaces with different di- ameters were from the same as in previous studies: 5 V = 30 nm, 10 V = 50 nm, 15 V = 70 nm, 20 V = 100 nm. 3.2. Macrophage Adhesion to Substrates Following 24 Hours of Culture As seen in Figure 2, the number of adhered macrophages  Macrophage Inflammatory Response to TiO2 Nanotube Surfaces Copyright © 2011 SciRes. JBNB 296 Figure 1. Physical characteristics of titanium dioxide nano- tube surfaces. SEM images of nanotube surfaces and meas- urements of nanotube phy sical dimensions are repr oducible and consistent with previous reports. to unmodified commercial Ti (with thin native oxide layer) and anodized TiO2 nanotube substrates is inde- pendent of oxide structure. The average number of cells per 10Xmicroscopic field ranged from 150 on titanium to 230 on 30 nm TiO2 nanotube surfaces, with the anodized TiO2 surfaces generally supporting more cell adhesion. Although there was a general trend of a reduction in the number of adherent macrophages as the nanotube size increased, this was not statistically significant. No sig- nificant differences in cell adhesion among the substrates were observed. 3.3. Macrophage Morphology on the Experimental Substrates Figure 3 illustrates SEM micrographs of macrophages on the Ti and TiO2 nanotube surfaces after 24 hours of incubation, which is an adequate time to reveal the acute inflammatory response onthe different surfaces. The re- action of the cells to the surfaces can be seen from cell- spreading, ruffled membranes, and extended filipodia. On the smaller 30 nm diameter and flat Ti, there seemed to be more filipodia extensions, with a spiky appearance of the cell, as shown by the arrows in Figure 3. Larger spreading areas with less filipodia are seen on the 50 - 100 nm TiO2 surfaces. Both the cell spreading and filipo- dia extension are typical signs of macrophage activation. 3.4. Production of the Inflammatory Cytokine TNF Tumor necrosis factor (TNF) is a cytokine involved in inflammation and is a member of a group of cytokines Figure 2. Macrophage adhesion to substrates is independent of nanotexture. No significant differences were observed in the number of adherent macrophages on substrates follow- ing 24 hours of culture. Figure 3. Macrophage morphology. The SEM images show macrophages on the experimental surfaces after 24 hours of incubation. Arrow indicate abundant filipodia extensions on the Ti and 30 nm TiO2 nanotubes. Dotted lines show cell outlines and large spreading area on the 50 - 100 nm TiO2 nanotubes surfaces. that stimulate the acute phase reaction. This cytokine was used to test the inflammatory response of the macro- phages to the different surfaces. As presented in Figure 4, the production of TNF per cell varied on the different substrates. Mean channel fluorescence (MCF) values exhibited several significant differences for cells grown on the various substrates. MCF for cells grown on tissue culture polystyrene was 8.85, a value significantly lower than that for cells cultured on flat titanium surfaces (9.81). Cells grown on 30, 50 and 100 nm surfaces had no significant differences in MCF values compared to other surfaces (9.43, 9.45 and 9.40, respectively). How- ever, cells cultured on 70 nm surfaces had significantly lower MCF values than flat Ti (9.13). Percent positive data show no significant differences among the culture substrates. 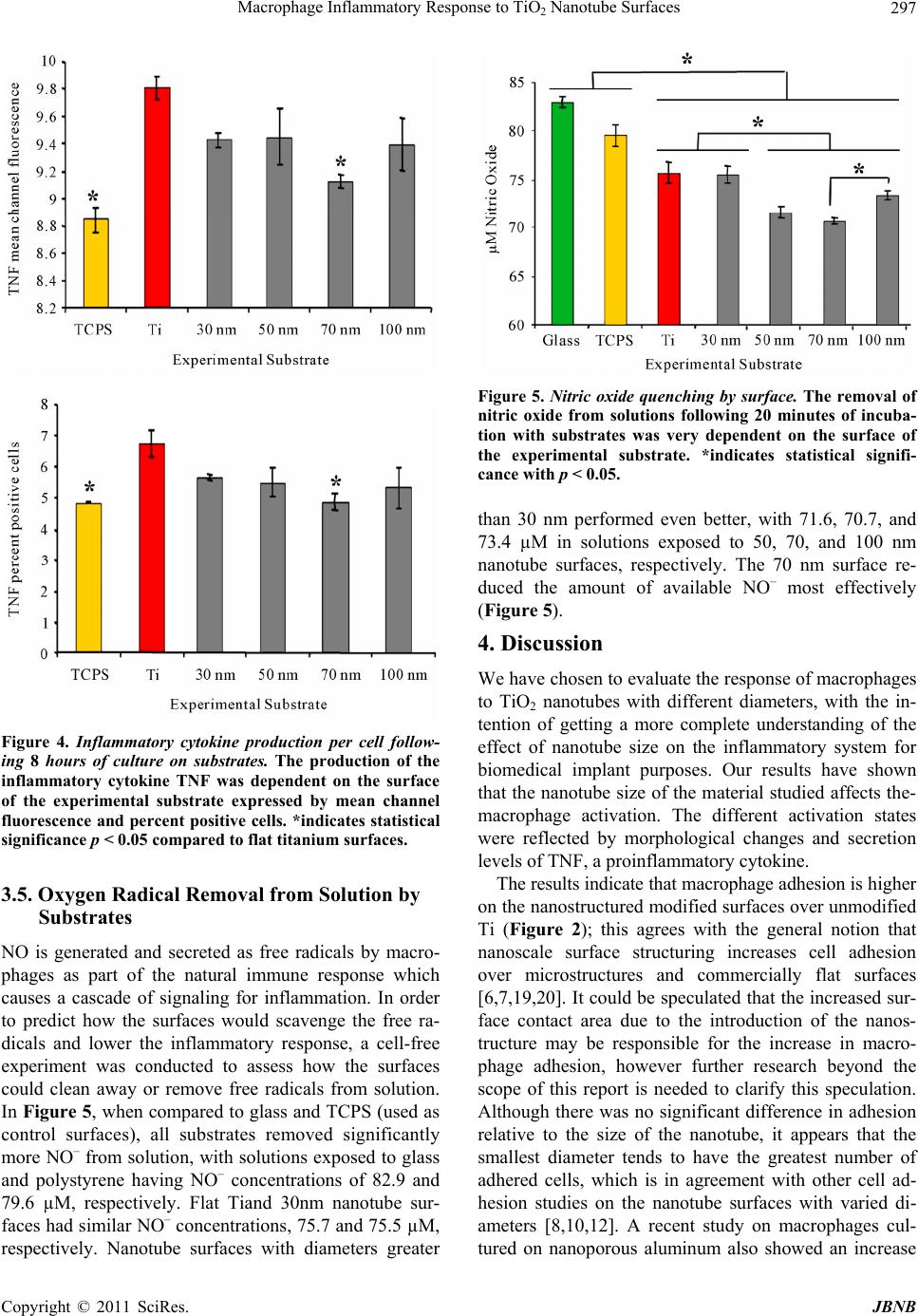 Macrophage Inflammatory Response to TiO2 Nanotube Surfaces Copyright © 2011 SciRes. JBNB 297 Figure 4. Inflammatory cytokine production per cell follow- ing 8 hours of culture on substrates. The production of the inflammatory cytokine TNF was dependent on the surface of the experimental substrate expressed by mean channel fluorescence and percent positive cells. *indicates statistical significance p < 0.0 5 compared to flat titanium surfaces. 3.5. Oxygen Radical Removal from Solution by Substrates NO is generated and secreted as free radicals by macro- phages as part of the natural immune response which causes a cascade of signaling for inflammation. In order to predict how the surfaces would scavenge the free ra- dicals and lower the inflammatory response, a cell-free experiment was conducted to assess how the surfaces could clean away or remove free radicals from solution. In Figure 5, when compared to glass and TCPS (used as control surfaces), all substrates removed significantly more NO– from solution, with solutions exposed to glass and polystyrene having NO– concentrations of 82.9 and 79.6 µM, respectively. Flat Tiand 30nm nanotube sur- faces had similar NO– concentrations, 75.7 and 75.5 µM, respectively. Nanotube surfaces with diameters greater Figure 5. Nitric oxide quenching by surface. The removal of nitric oxide from solutions following 20 minutes of incuba- tion with substrates was very dependent on the surface of the experimental substrate. *indicates statistical signifi- cance with p < 0.05. than 30 nm performed even better, with 71.6, 70.7, and 73.4 µM in solutions exposed to 50, 70, and 100 nm nanotube surfaces, respectively. The 70 nm surface re- duced the amount of available NO– most effectively (Figure 5). 4. Discussion We have chosen to evaluate the response of macrophages to TiO2 nanotubes with different diameters, with the in- tention of getting a more complete understanding of the effect of nanotube size on the inflammatory system for biomedical implant purposes. Our results have shown that the nanotube size of the material studied affects the- macrophage activation. The different activation states were reflected by morphological changes and secretion levels of TNF, a proinflammatory cytokine. The results indicate that macrophage adhesion is higher on the nanostructured modified surfaces over unmodified Ti (Figure 2); this agrees with the general notion that nanoscale surface structuring increases cell adhesion over microstructures and commercially flat surfaces [6,7,19,20]. It could be speculated that the increased sur- face contact area due to the introduction of the nanos- tructure may be responsible for the increase in macro- phage adhesion, however further research beyond the scope of this report is needed to clarify this speculation. Although there was no significant difference in adhesion relative to the size of the nanotube, it appears that the smallest diameter tends to have the greatest number of adhered cells, which is in agreement with other cell ad- hesion studies on the nanotube surfaces with varied di- ameters [8,10,12]. A recent study on macrophages cul- tured on nanoporous aluminum also showed an increase  Macrophage Inflammatory Response to TiO2 Nanotube Surfaces Copyright © 2011 SciRes. JBNB 298 in adhesion on the small 20 nm pore size incomparison to the larger 200 nm pore size [13]. Activated macrophagespresent a large number of mor- phologic, functional, andmetabolic differences from nor- mal resting cells. They are largerin size, anddisplay pro- nounced ruffling of the plasma membrane, increased ca- pacity for adherence and spreading on surfaces, increased formation of pseudopods, as well as functional differ- ences [21]. Macrophage spreading is the preliminary stepin of macrophage activation andconsidered to be an important marker of this event [22]. The active process of spreading represents alterations including filipodia and rough plasma membranes. It appears that cells present on the experimental surfaces had activated morphologies withruffled membranes and filipodia extensions. The Ti and 30 nm TiO2 nanotubes had extensive filipodia, whereas less filipodia but greater spreading area with some membrane ruffling was observed on the 50 - 100 nm TiO2 surfaces (Figure 3). Because cells on the Ti and 30 nm TiO2 show more abundant and well-established filipodia extensions, it can be assumed that there was a higher degree of activation on these surfaces.Although less filipodia were observed on the 50 - 100 nm nano- tubes, cells exhibited a larger spreading area. While the mechanism for filipodia activation (Ti and 30 nm TiO2) vs. cell spreading (50 - 100 nm TiO2) on the different nanotube diameters was not determined in the scope of this report it would be valuable in the future to shed light on this morphological phenomenon in determining the immune response. A previous study onmacrophage production of in- flammatory cytokines showed higher production of many cytokines on flat Ti compared to nanostructured TiO2 [3]. In the current study on TNF expression on similar nanos- tructured surfaces with the same chemistry but varying geometries, we observed the same general trend of lower TNF production by cells on the nanotubes, suggesting that the nanotopography itself contributes to lowering the TNF production. No statistically significant correlation- was observed as a function of the diameter size, but the largest decrease in TNF values was significant for macrophages cultured on 70 nm nanotube surfaces (Fig- ure 4). A possible reason for the lower inflammatory response in general of the TiO2 nanotubesurfaces could be due to radical quenching by the surface, aknown property of TiO2 [23,24], which was observed in our re- sults with higher levels of NO– quenching on TiO2 nano- tube surfaces compared to the other experimental materi- als (Figure 5). Oxygen radical production is a common occurrence in inflammation [25], and the reduction of oxygen radicals at the surface of a biomaterial could re- duce the local inflammatory response. Although the mechanism for oxygen radical reduction is not explained by the extent of this report, it appears that the 70 nm di- ameter nanotubes are most advantageous in terms of radical reduction for decreasing the inflammatory re- sponse to the surface. Interestingly, the surfaces that had the most radical reduction or oxygen quenching, i.e. 50 - 100 nm TiO2 nanotubes (Figure 5), showed the least amount of filipodia activation (Figure 3) and possibly a lower degree of inflammatory response of the macro- phages. However, to shed further light on the complete understanding the effect of surface nanotopography on macrophage behaviors, additional studies are needed to determine the fate of the cells as well as the secretion of other growth factors. In summary, this study demonstrated that the TiO2 nanotubes with 70 nm diameter is the surface with the lowest level of macrophage morphological activation, the lowest macrophage production of inflammatory cyto- kines, and the greatest oxygen radical quenching capabil- ity. Hence, TiO2 nanotubes in the ~70 nm diameter re- gime ismost promising for implant surfaces for decreased inflammatory response. It should be noted that larger nanotubes in the ~70 - 100 nm range elicited a favorable response in terms of chondrocytes and osteoblast cells [7,11], mature cells also derived from mesenchymal cells in general. While TiO2 nanotubes surfaces are well knownfor or- thopedic implant technologies, it is advantageous and beneficial to consider this type of surface nanostructure for other biomedical implant technologies including vas- cular stent applications. In our previous work explor- ingthe possibility of utilizing TiO2 nanotubes as a possi- ble Ti or NiTi surface modifications in arterial stents, we have found that the TiO2 nanotube surface structuring is excellent for the growth, mobility, and endothelialization of vascular luminal surface [18]. Collectively, the reduc- tion of inflammation and the quenching of oxygen radi- cals shown in the present study bolster the potential use of TiO2 nanotubes for stents, especially be- cause nitric oxide synthase has been shown to co-localize with athe- rosclerotic plaques [26]. 5. Conclusions The present work shows that by changing the diameter of TiO2 nanotubes, in the range of 30 - 100 nm, it is possi- ble to modulate the macrophage and inflammatory re- sponse. In addition, these findings open the possibility of exploiting some of the beneficial material properties of TiO2 as oxygen radical scavengers. In general, it was found that TiO2 nanotubes surfaces had lower macro- phage activation, decreased levels of TNF cytokine ex- pression, as well as increased ability to quench free radi- cals, resulting in lower inflammatory effects compared to conventional Ti. The nanotube surface with ~70 nm di-  Macrophage Inflammatory Response to TiO2 Nanotube Surfaces Copyright © 2011 SciRes. JBNB 299 ameter has the greatest effect in reducing the inflamma- tory response. This study emphasizes the role of nanotopography in dictating inflammatory cell responses and demonstrates that nanotopography can be utilized to control the inflammatory likelihood of medical implants. 6. Acknowledgements The authors acknowledge financial support of this re- search by UC Discovery Grant No. ele08-128656/Jin. This work was also partially supported by Postdoctoral Fellowship (for Lisa Chamberlain) through the NHLBI institutional training grant 5T32HL007089. REFERENCES [1] J. M. Anderson, A. Rodriguez and D. T. Chang, “Foreign Body Reaction to Biomaterials,” Seminars in Immunology, Vol. 20, No. 2, 2008, pp. 86-100. doi:10.1016/j.smim.2007.11.004 [2] T. D. Zaveri, et al., “Contributions of Surface Topogra- phy and Cytotoxicity to the Macrophage Response to Zinc Oxide Nanorods,” Biomaterials, Vol. 31, No. 11, pp. 2999-3007. doi:10.1016/j.biomaterials.2009.12.055 [3] K. M. Ainslie, et al., “In Vitro Inflammatory Response of Nanostructured Titania, Silicon Oxide, and Polycaprolac- tone,” Journal of Biomedical Materials Research A, Vol. 91, No. 3, 2009, pp. 647-655. doi:10.1002/jbm.a.32262 [4] S. Chen, et al., “Characterization of Topographical Ef- fects on Macrophage Behavior in a Foreign Body Re- sponse Model,” Biomaterials, Vol. 31, No. 13, 2010, pp. 3479-3491. doi:10.1016/j.biomaterials.2010.01.074 [5] K. M. Ainslie, et al., “Inflammatory Response to Im- planted Nanostructured Materials,” In: D. A. Puleo and R. Bizios, Eds., Biological Interactions on Materials Sur- faces, Springer, New York, 2009, pp. 355-371. doi:10.1007/978-0-387-98161-1_18 [6] S. Oh, et al., “Significantly Accelerated Osteoblast Cell Growth on Aligned TiO2 Nanotubes,” Journal of Bio- medical Materials Research A, Vol. 78, No. 1, 2006, pp. 97-103.doi:10.1002/jbm.a.30722 [7] K. S. Brammer, et al., “Improved Bone-Forming Func- tionality on Diameter-Controlled TiO2 Nanotube Sur- face,” Acta Biomaterialia, Vol. 5, No. 8, 2009, pp. 3215- 3223. doi:10.1016/j.actbio.2009.05.008 [8] S. Bauer, et al., “Size Selective Behavior of Mesenchy- mal Stem Cells on ZrO2 and TiO2 Nanotube Arrays,” In- tegrative Biology, Vol. 1, No. 8-9, 2009, pp. 525-532. doi:10.1039/b908196h [9] J. Park, et al., “Narrow Window in Nanoscale Dependent Activation of Endothelial Cell Growth and Differentiation on TiO2 Nanotube Surfaces,” Nano Letters, Vol. 9, No. 9, 2009, pp. 3157-3164.doi:10.1021/nl9013502 [10] J. Park, et al., “Nanosize and Vitality: TiO2 Nanotube Diameter Directs Cell Fate,” Nano Letters, Vol. 7, No. 6, 2007, pp. 1686-1691.doi:10.1021/nl070678d [11] K. S. Brammer, et al., “Nanotube Surface Triggers In- creased Chondrocyte Extracellular Matrix Production,” Materials Science & Engineering C-Materials for Bio- logical Applications, Vol. 30, No. 4, 2010, pp. 518-525. [12] S. Oh, et al., “Stem Cell Fate Dictated Solely by Altered Nanotube Dimension,” Proceedings of the National Aca- demy of Sciences of USA, Vol. 106, No. 7, 2009, pp. 2130-2135.doi:10.1073/pnas.0813200106 [13] N. Ferraz, J. Hong, S. Matteo and M. Karlsson Ott, “Nanoporosity of Alumina Surfaces Induces Different Pa- tterns of Activation in Adhering Monocytes/Macro- phages,” International Journal of Biomaterials, Vol. 2010, 2010, p. 8. [14] S. H. Oh, et al., “Growth of Nano-Scale Hydroxyapatite Using Chemically Treated Titanium Oxide Nanotubes,” Biomaterials, Vol. 26, No. 24, 2005, pp. 4938-4943. doi:10.1016/j.biomaterials.2005.01.048 [15] L. M. Chamberlain, et al., “Phenotypic Non-Equivalence of Murine (Monocyte-) Macrophage Cells in Biomaterial and Inflammatory Models,” Journal of Biomedical Mate- rials Research A, Vol. 88, No. 4, 2009, pp. 858-871. doi:10.1002/jbm.a.31930 [16] E. R. Rhoades and I. M. Orme, “Similar Responses by Macrophages from Young and Old Mice Infected with Mycobacterium Tuberculosis,” Mechanisms of Ageing and Development, Vol. 106, No. 1-2, 1998, pp. 145-153. doi:10.1016/S0047-6374(98)00113-4 [17] M. L. Godek, et al., “Rho GTPase Protein Expression and Activation in Murine Monocytes/Macrophages Is Not Modulated by Model Biomaterial Surfaces in Serum- Containing In Vitro Cultures,” Journal of Biomaterials Science, Polymer Edition, Vol. 17, No. 10, 2006, pp. 1141-1158.doi:10.1163/156856206778530731 [18] K. S. Brammer, et al., “Enhanced Cellular Mobility Guided by TiO2 Nanotube Surfaces,” Nano Letters, Vol. 8, No. 3, 2008, pp. 786-793.doi:10.1021/nl072572o [19] J. Wei, et al., “Comparison of Physical, Chemical and Cellular Responses to Nano- and Micro-Sized Calcium Silicate/Poly(Epsilon-Caprolactone) Bioactive Compos- ites,” Journal of the Royal Society Interface, Vol. 5, No. 23, 2008, pp. 617-630.doi:10.1098/rsif.2007.1267 [20] E. Martinez, et al., “Effects of Artificial Micro- and Nano-Structured Surfaces on Cell Behaviour,” Annals of Anatomy, Vol. 191, No. 1, 2009, pp. 126-135. doi:10.1016/j.aanat.2008.05.006 [21] R. J. North, “The Concept of the Activated Macrophage,” The Journal of Immunology, Vol. 121, No. 3, 1978, pp. 806-809. [22] T. Tatefuji, et al., “Isolation and Identification of Com- pounds from Brazilian Propolis Which Enhance Macro- phage Spreading and Mobility,” Biological & Pharma- ceutical Bulletin, Vol. 19, No. 7, 1996, pp. 966-970. [23] R. J. Schutte, A. Parisi-Amon and W. M. Reichert, “Cy- tokine Profiling Using Monocytes/Macrophages Cultured on Common Biomaterials with a Range of Surface Che- mistries,” Journal of Biomedical Materials Research A, Vol. 88, No. 1, 2009, pp. 128-139. doi:10.1002/jbm.a.31863  Macrophage Inflammatory Response to TiO2 Nanotube Surfaces Copyright © 2011 SciRes. JBNB 300 [24] R. Suzuki, et al., “Reactive Oxygen Species Inhibited by Titanium Oxide Coatings,” Journal of Biomedical Mate- rials Research A, Vol. 66, No. 2, 2003, pp. 396-402. doi:10.1002/jbm.a.10001 [25] H. Maeda and T. Akaike, “Nitric Oxide and Oxygen Radicals in Infection, Inflammation, and Cancer,” Bio- chemistry (Mosc), Vol. 63, No. 7, 1998, pp. 854-865. [26] K. M. Cromheeke, et al., “Inducible Nitric Oxide Syn- thase Colocalizes with Signs of Lipid Oxidation/ Peroxi- dation in Human Atherosclerotic Plaques,” Cardiovascu- lar Research, Vol. 43, No. 3, 1999, pp. 744-754. doi:10.1016/S0008-6363(99)00148-0 |

