Journal of Water Resource and Protection
Vol.09 No.02(2017), Article ID:73943,20 pages
10.4236/jwarp.2017.92012
Energetic Consequences of Stranding of Juvenile Atlantic Salmon (Salmo salar L.)
Michael Puffer1, Ole Kristian Berg1*, Sigurd Einum1, Svein Jakob Saltveit2, Torbjørn Forseth3
1Department of Biology, Norwegian University of Science and Technology (NTNU), Trondheim, Norway
2Freshwater Ecology & Inland Fisheries Laboratory, Natural History Museum, University of Oslo, Oslo, Norway
3Norwegian Institute for Nature Research, Trondheim, Norway

Copyright © 2017 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: December 13, 2016; Accepted: February 3, 2017; Published: February 6, 2017
ABSTRACT
An effect of hydropower and hydropeaking regulation in rivers is stranding of fish. Those fishes that survive stranding may experience stranding as a stressful situation. In four experimental stranding experiments (each with 6 individuals in 10 control and 10 treatment replicates), the energetic consequences of two forms of stranding (i.e. trapping and beaching) of juvenile Atlantic salmon (Salmo salar) were investigated in summer and winter. Restricted food access in the experimental channels ensured that effects of hydropeaking could be revealed. Mean fish length ranged between 60 mm and 110 mm among experiments. Both during the winter and summer experiments fish did not grow in length, neither in the control nor in the treatment channels and fish lost body mass as well as body fat in all experiments (body fat in summer trapping experiment not determined). The four experiments revealed similar results: stranding did not affect growth or energy content. Despite the severity of the stranding and the resulting mortality, which was especially high during summer, no stranding related effects on fish performance could be detected.
Keywords:
Hydropeaking, Trapping, Beaching, Growth, Lipid Stores, Salmonids

1. Introduction
Climate change is a driver for increased demand of renewable energy, i.e. hydropower, wind and solar energy [1] [2] [3] [4] . Contrary to most renewable energy sources, hydropower has high production flexibility (i.e. load balancing) [5] [6] , and is the only renewable energy source with a feasible storage potential. Thus, reservoir based hydropower can be used to store energy when demands are low and produce energy when demands are high [7] [8] [9] [10] . The flexibility of hydropower has led to increased use of hydropeaking, i.e. short-term changes in hydropower production causing frequent and rapid flow changes downstream of power plants.
Riverine aquatic organisms are adapted to natural flow fluctuations, but the amplitude and high rate of flow changes during hydropeaking have a suite of effects on river ecosystems [11] [12] [13] . It may alter the riverine habitat dramatically, leading to dewatered riverbeds strongly affecting riverine organisms through stranding. A number of field experiments (usually without control, though) have been performed to assess the stranding rate and to assess the conditions when stranding prevail [7] [14] [15] [16] [17] . Important factors affecting stranding rates in young wild Atlantic salmon (Salmo salar) and brown trout (Salmo trutta) were water temperature, time of year, time of day, minimum discharge, wetted history, shape of the river bed, and dewatering rate [17] [18] [19] [20] . However, death is not necessarily a consequence of stranding. In a series of stranding experiments, fishes from previous trials were found alive in subsequent experiments [17] . When water levels drop rapidly during hydropeaking episodes, juvenile salmonids may be trapped in bankside gravel, in ponds or within the substrate, where inflow of groundwater or interstitial water is important for survival (reviewed by [21] ). If they do not suffer from oxygen deficiency, drying-out or capture by predators, they may survive. On the other hand, such experiences may be stressful [22] or cause damage that may affect their long- term performance and survival.
Hunter [14] defines two types of fish stranding, i.e. trapping and beaching. Trapping is the isolation of fish in pockets of water with no access to the free- flowing surface water, while beaching is when fishes flounder out-of-water on the substrate. Here we compare the effects of experimentally stranded fish with unaffected controls (channels with stable water level) to explore the performance (i.e. growth in length and mass, body lipids) of juvenile Atlantic salmon exposed either to trapping or beaching. Experiments in stream channels were performed both during winter and summer conditions to ensure seasonal validity of the results. The channels have been used in previously published experiments and are known to have an invertebrate fauna sufficient to feed small parr (e.g. [23] [24] [25] ), but the experiments described below were designed to avoid abundance of food which may let the fish compensate negative growth effects of hydropeaking [26] .
2. Materials and Methods
2.1. Experimental Facility and Design
The experiments were conducted at the Norwegian Institute for Nature Research (NINA) research station at Ims, southwest Norway (59°N 6°E). These stranding experiments were performed in 20 artificial outdoor channels (each 485 × 25 cm) with about 10 cm of water above the substrate between stranding episodes. The fish were deliberately left unfed in the channels in order to investigate the effects of hydropeaking under conditions with restricted food access. The channels had an approximately 5 cm thick substrate layer consisting mainly of cobbles and pebbles (size: 1 - 7 cm), offering appropriate shelter for juvenile Atlantic salmon (Figure 1). Water originated from a nearby lake. The channels were pairwise coupled to the water supply and water outlet. At the lower end of each channel pair, a tube (height: 20 cm; diameter: 5 cm) was inserted vertically into the outlet opening at the bottom of the channel to ensure a stable water level. Channel pairs with odd numbers were assigned to the treatment group (i.e. varying water flow and water level), whereas channel pairs with even numbers were assigned to the control group (i.e. stable water flow and water level). The channels were covered with black netting material to avoid avian predation in winter and with white tarpaulin to offer shade from the sun in summer.
At the lower and upper end of the channels a wooden, netting covered frame (height of wooden beam = 2.5 cm) covered the cross-section of the channel and thereby ensured a minimum water level. The netting ensured that fish were retained in the gravel-covered area. In the trapping experiments, the netting material had a small mesh size (1.2 × 1.2 mm) that likely restricted drift of larger aquatic invertebrates into the channels. Based on the poor growth in these experiments, the netting was replaced with coarser netting (3.6 × 3.6 mm) in the beaching experiments.
Figure 1. Photo of one pair of the artificial stream channels at normal water level during the summer trapping experiment. Top: water intake; bottom: Water outlet.
In the trapping experiments (experimental details are given in Table 1), water levels in the treatment channels were episodically lowered for three h at a time by removing the tube from the outlet, thereby effectively lowering the water level to the upper edge of the wooden frame at the bottom of the channel. Water was not turned off and the wooden frame ensured a water level of approx. 2.5 cm at the lower end, thus creating pockets of water between exposed stones. This set-up prevented mortality from desiccation during the low water periods. In contrast, in the beaching experiments (Table 1), the tube was removed from the outlet, the water supply was turned off, and the wooden frames at the lower end of the channels were lifted approximately 5 mm so that the channels were nearly drained. Two people observed the fish in the treatment channels during this stage, and water was turned on and the tube was set in place as soon as fish were observed to struggle which usually occurred after about 30 min of exposure (depending on the season and the weather). Since there was no remaining water in the channels, the beaching experiment was considered to be much harder on the fish and thus draining of the channels was only repeated twice in the course of the whole experimental period (see Table 1). However, in the trapping experiment lowering of the water level was repeated 30 - 60 times during the experimental period (see Table 1).
Measurements (16 in each channel) of water depth and water velocity were performed every 25 cm in each channel to ensure that the control and treatment channels had a similar physical environment. Water depth in the control channels was 105 ± 14 mm (mean ± SD) and in the treatment channels 108 ± 14 mm. Surface water velocity in the control channels was 0.9 ± 0.6 cm sec−1 and in the treatment channels 0.8 ± 0.6 cm sec−1 (Schiltknecht Messtechnik; Gossau, Switzerland; MiniAir 20 Multiprobe Anemometer with water probe; precision ± 1 cm sec−1; 3 s measurement time). Similar measurements at the low water stage were only taken for the trapping experiment (water depth: 34 ± 14 mm; water velocity: 5.7 ± 3.6 cm∙sec−1) since there was no measureable depth or current in the beaching experiment.
Water temperature was recorded during the experimental period, using Hobo H8 (www.onsetcomp.com) and Tinytags TG4100 (www.geminidataloggers.com) temperature loggers (for details of the experiments, see: Table 1), but it should
Table 1. Details of the experimental set-up for the trapping and beaching experiments during both winter and summer.
be noted that the fish in both summer experiments experienced periods with water temperatures above 20˚C.
2.2. Fish and Treatment
The experimental protocol was deliberately adjusted between experiments in order to induce a strong standing effect to the experimental fish, but still try to avoid mortality which could affect density between experimental and control treatments. Two seasons with trapping experiments were performed before the beaching experiments (see Table 1).
The juvenile Atlantic salmon used in these experiments were hatched from eggs originating from the local River Imsa and reared at the research station. One week prior to stocking into experimental channels, all fish were anaesthetized with benzocaine and length (fork length; in mm) and wet mass (in g) measured (scale type UWE-NJW-150, precision: ±0.005 g). The first experimental bout was the trapping experiments, where the fish were individually tagged with PIT-tags (length 13 mm; diameter 2 mm; Oregon RFID, U.S.A.). During the summer trapping experiments fish got fungi infection (Saprolegnia sp.) and consequently no PIT-tagging was done in the following beaching experiments in order to avoid these problems.
For each experiment, the fish were divided into 21 groups of similar body size. One group (Nfish = 20) was sacrificed for establishment of pre-experimental body fat content. Both the 10 control groups as well as the 10 treatment groups consisted of N = 6 individuals each. The groups were randomly distributed among the channels and left under stable conditions (i.e. water level and flow) for recovery and habituation for five to seven days prior to the experiment. There were no differences in initial body length between the channels (Tukey HSD: p ≥ 0.21) or between the control and treatment groups (t-test: p ≥ 0.07) for each one of the four experiments.
Trapping experiments were conducted with three h of low water during weekdays, but water levels were not reduced when air temperatures were below 0˚C. The experimental period for the trapping experiments lasted 61 days in winter and 85 days in summer (for further details see: Table 1). The consecutive beaching experiments were set-up with another time schedule, since the beaching treatment is much harder on the fish than the trapping treatment. At the first day of the beaching experiment, fish in the treatment channels were beached for about 30 min, then given two h to recover at a normal water level and thereafter beached again for another 30 min. After these two beaching episodes, the water flow and levels in the treatment channels were set to the same levels as the control channels and fish were left undisturbed until the end of the experiment. The experimental period for the beaching experiment lasted 35 days during winter and 23 days during summer (for further details see: Table 1). This beaching schedule is not similar to what fish might experience in regulated rivers, but the sole goal of this experiment was to stress fish, not to suffocate them to death. A more realistic beaching schedule with more beaching episodes would probably have resulted in no surviving fish left to measure.
In the winter trapping experiment, channels were visually checked for dead fish on a daily basis, whereas in the summer trapping experiment, channels were only checked several times a week throughout the entire experimental period. In the beaching experiment, channels were visually checked for dead fish only during the first week of the experimental period because that was the period when fish experienced stranding and thus the immediate lethal effects should have happened then and there.
At the termination of the experiments, fish were caught with an aquarium dip net, killed with a blow to their heads, PIT-tag codes were recorded (when applicable), fork length and wet mass measured, and body cavity opened to remove the PIT-tag and stomach contents (only small remains were detected). The fish were thereafter frozen for body fat analyses, following a standard procedure for analysing body lipid stores [27] [28] .
In the winter trapping experiment, one fish died in a treatment channel, but was not replaced. In the summer trapping experiment, a total of 27 fish in the control and 20 fish in the treatment channels died. The fish in this experiment were infected by Saprolegnia sp., and infection appeared to increase due to handling before the experiment started. Since channels were not visually checked each day, it was not possible to determine when individual fish died. Additionally, because of the warm water temperatures dead fish decayed rapidly and thus made it often impossible to find their remains. A total of 12 fish were replaced (5 experimental and 7 control fish) in the summer trapping experiment. These were not tagged and therefore not included in the later processing. Because most of the dead fish were completely decayed, it was not possible to measure neither their length or weight nor to use them for the body fat analyses.
2.3. Data Analysis
The statistical analyses were performed with R, v. 2.15.3 [29] . The function lme, a linear mixed effects model from the package nlme [30] , was used with channel as a random effect to analyse the effects of the treatment on final body length, body mass, and percentage of body fat for the trapping experiments. For body length and body mass, the corresponding initial value was used as a covariate. For percentage of body fat, final body mass was used as a covariate. In addition, final fish density, i.e. when mortality caused densities to vary among channels (only summer experiments), was used as a covariate. The same linear mixed-ef- fects model, with channel as a random effect, was used to test whether final fork length, body mass, and percentage of body fat were affected by the treatment in the beaching experiments. For the summer experiment, final fish density was included as a covariate. Other covariates like initial body mass could not be used in the analyses because of the lack of PIT-tags and thus no individual identification. Model simplification followed the protocol described in [31] and final model residuals were checked for normal distribution, homogeneity and independence. Because three different response variables were tested in each experiment, the p-values were adjusted for multiple comparisons by the Holm-Bonferroni method [32] using the function p. adjust in R.
3. Results
3.1. Mortality
In the winter beaching experiment, no fish died during the beaching episodes, and by the end of the experimental period, only one fish had died in a treatment channel. In the summer beaching experiment, two fish died during the first and eleven during the second beaching episode. These first two dead fish were found in the two-hour recovery period between the two beaching episodes, and the other eleven dead fish were found after the second beaching episode had taken place at the end of the day. In the following weeks, seven more fish died in the treatment channels, whereas five fish died in the control channels. As already explained above, since we did not visually check these channels after the first week of the experiment, it was not possible to determine when individual fish died following the first week. Dead fish were not replaced with new fish. Summing up, for both summer experiments (i.e. trapping and beaching) mortality caused some variation in final density among channels.
3.2. Winter Trapping
Fish experiencing the trapping treatment during winter lost significantly more body mass than fish in the control treatment. Both body length and body fat were not affected by the trapping treatment (Figure 2(a) and Figure 2(b)). The detailed results for body length, body mass and body fat are as follows:
For final body length (Figure 2(a)), treatment could be excluded from the full model during model simplification (p = 0.42) and final length was best explained by initial length (slope value ± SE: 1.04 ± 0.02; t = 48.7; adjusted p < 0.0004). Final length was 111 ± 18 mm (mean ± SD) in the control and 110 ± 15 mm in the treatment.
For final body mass (Figure 2(b)), no variable could be excluded during model simplification. Trapping had a significant negative effect on the body mass of the treatment fish (−0.49 ± 0.11; t = −4.6; adjusted p < 0.0004) and the covariate initial body mass having a significant positive effect on final body mass (0.89 ± 0.01; t = 113.7; adjusted p < 0.0004; Figure 1). All fish lost body mass during the experimental period, on average 11% in the control and 15% in the treatment. Final body mass was 13.9 ± 5.9 g in the control and 12.6 ± 5.3 g in the treatment.
For final body fat, treatment could be excluded from the full model during model simplification (p = 0.12), leaving only final body mass in the final model (0.11 ± 0.02; t = 7.3; adjusted p < 0.0004). The fat content changed from a pre-experimental 10.1% ± 2.2% to 7.4% ± 1.2% in the control and 7.5% ± 1.2% in the treatment.
3.3. Winter Beaching
Fish experiencing the beaching treatment during winter did not perform differently than fish in the control treatment. The detailed results for body length, body mass and body fat are as follow:
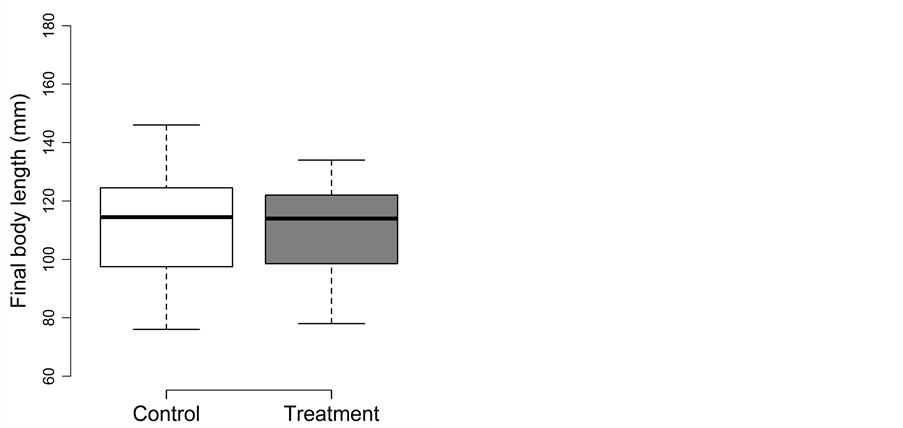
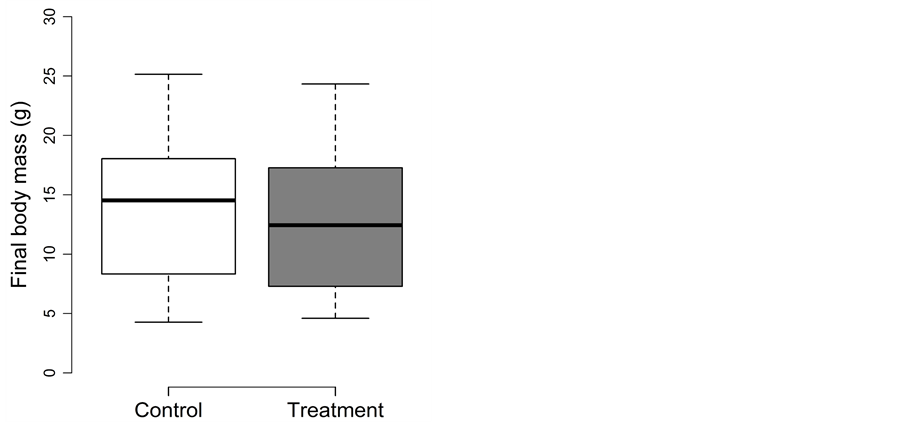
Figure 2.Trapping experiment in winter: Box-plots for (a) the final body length (in mm) and (b) the final body mass (in g) for the control (□) and treatment (■). Whiskers extent to data points no more than 1.5 times the interquartile range from the box, and open circles represent outliers.
For final body length, treatment could be removed from the full model (p = 0.83). Thus, final fork length did not differ between the control (82 ± 9 mm) and treatment (81 ± 8 mm) (Figure 3(a)).
For final body mass, treatment could be removed from the full model (p = 0.91). Thus, final body mass did not differ between control (6.2 ± 2 g) and treatment (6.2 ± 2 g) channels (Figure 3(b)). Fish lost body mass both in the control (9%) and in the treatment (8%).
For final body fat, treatment was excluded from the full model during model simplification (p = 0.7). Thus, final body fat did not differ between the two treatments. Initial body fat of the pre-experimental base group was 7.6% ± 4.0%,
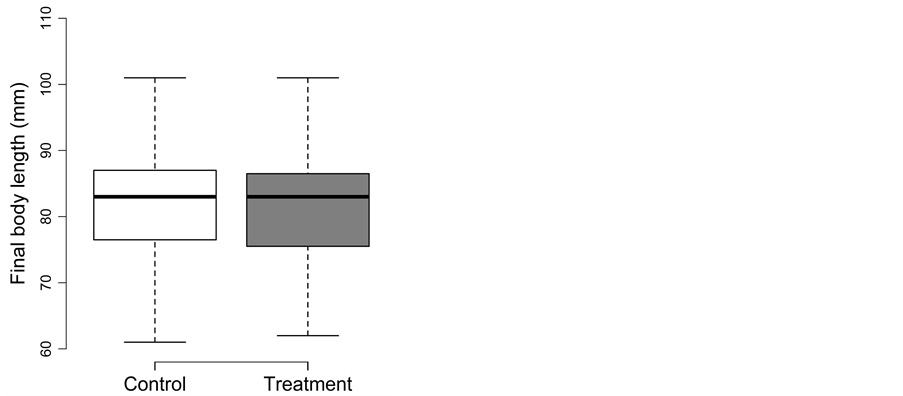
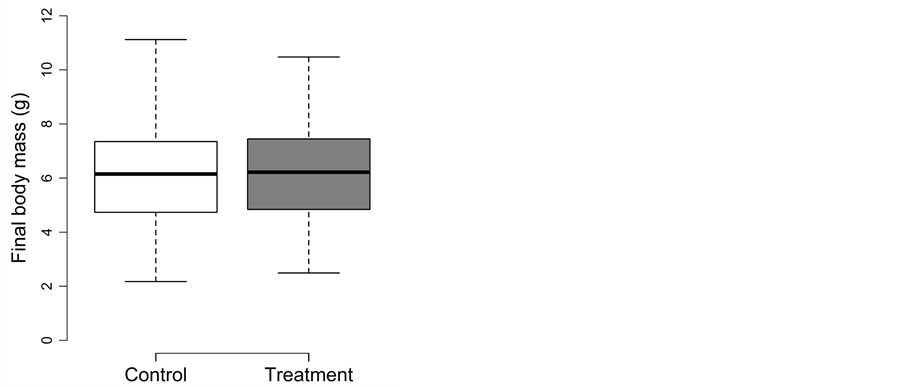
Figure 3. Beaching experiment in winter: Box-plots for (a) the final body length (in mm) and (b) the final body mass (in g) for the control (□) and treatment (■). Whiskers extent to data points no more than 1.5 times the interquartile range from the box, and open circles represent outliers.
and final body fat was 7.6% ± 5.2% and 7.2% ± 4.9% in the control and treatment, respectively.
3.4. Summer Trapping
Fish experiencing the trapping treatment during summer did not perform differently than fish in the control treatment (Figure 4(a) and Figure 4(b)). The detailed results for body length, body mass and body fat are as follow:
Both density and treatment could be removed during model simplification from the full model of body length (p ≥ 0.41). Final body length was best described by initial body length (1.05 ± 0.06; t = 17.6; adjusted p < 0.001) and did not differ between the control (98 ± 7 mm) and treatment (97 ± 8 mm) (Figure 4(a)).
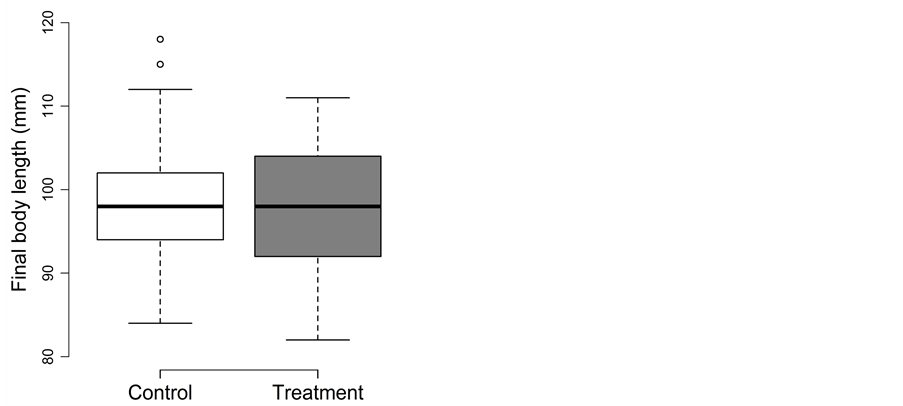
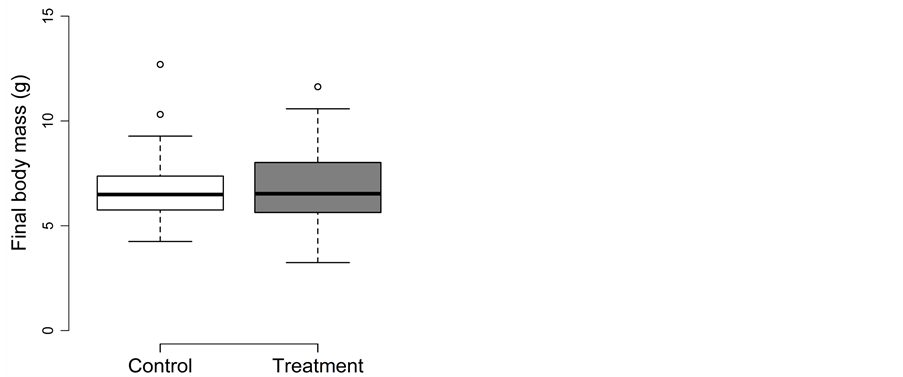
Figure 4. Trapping experiment in summer: Box-plots for (a) the final body length (in mm) and (b) the final body mass (in g) for the control (□) and treatment (■). Whiskers extent to data points no more than 1.5 times the interquartile range from the box, and open circles represent outliers.
Treatment and density could be removed from the full model of body mass during model simplification (p ≥ 0.14). Final body mass did not differ between the control (98.5 ± 7.4 g) and treatment (97.1 ± 7.5 g; Figure 4(b)) and were best explained by initial body mass (0.77 ± 0.05; t = 15.5; adjusted p < 0.0002). Growth in body mass was negative in both the control (−4.8 g ≙ −41%) and the treatment (−4.4 g ≙ −39%).
3.5. Summer Beaching
Fish experiencing the beaching treatment during summer were significantly longer and heavier than fish in the control treatment (Figure 5(a) and Figure 5(b)). Body fat was not affected by the beaching treatment. The detailed results for body length, body mass and body fat are as follows:
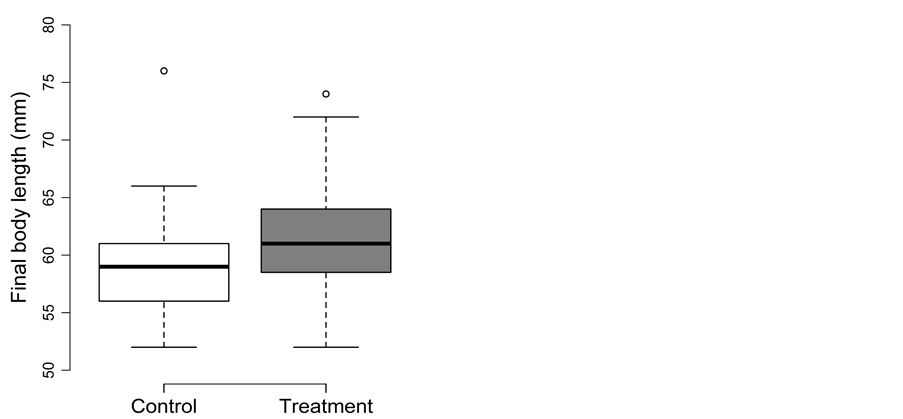
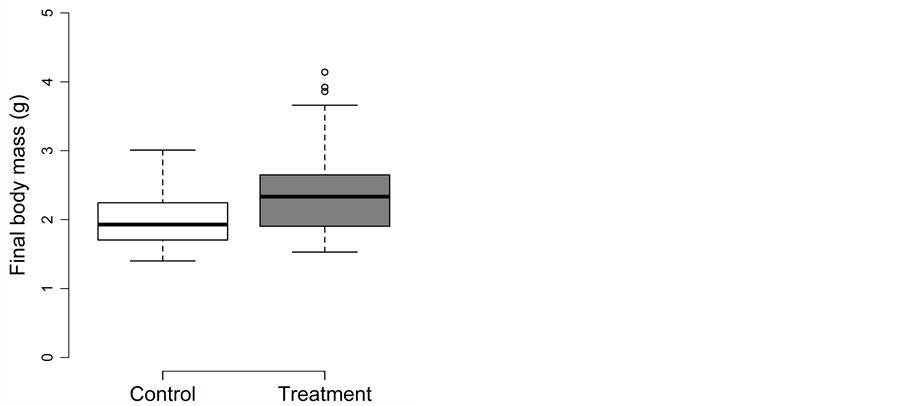
Figure 5. Beaching experiment in summer: Box-plots for (a) the final body length (in mm) and (b) the final body mass (in g) for the control (□) and treatment (■). Whiskers extent to data points no more than 1.5 times the interquartile range from the box, and open circles represent outliers.
For final body length (Figure 5(a)), the covariate density could be removed from the full model during model simplification (p = 0.65), but all other factors remained in the model. Final body length differed significantly between the control and treatment (2.95 ± 0.98; t = 3.0; adjusted p = 0.008; Figure 5(a)), with treatment fish being about 5% longer than control fish (control: 59 ± 4 mm; treatment: 62 ± 5 mm). Only fish in the treatment gained length (2.5 mm ≙ 4%), whereas no change in length was observed in the control.
For final body mass, density could be removed from the full model (p = 0.97). Body mass differed significantly between the control and treatment (0.44 ± 0.11; t = 3.8; adjusted p = 0.0026; Figure 5(b)), with treatment fish being on average 22% heavier than control fish. Fish lost body mass in both the control (17%) and treatment (4%).
Both density and treatment could be removed from the full model of body fat during model simplification (p ≥ 0.3). Body fat did not differ between the control (3.99% ± 1.94%) and treatment (3.53% ± 1.90%). All fish lost body fat in the course of the experiment when compared to the pre-experimental base group (6.6% ± 0.7%): 40% in the control and 47% in the treatment.
4. Discussion
In spite of stranding procedures, which were severe enough to create mortality, relatively small differences were seen between control and treatment groups in the four experiments. Where differences were revealed, the effect of stranding was insignificant in most: Growth in length did not differ between the control and treatment in the four experiments, except for the summer beaching experiment. Body mass decreased in all four experiments, both in control and treatment channels, but body mass decrease was highest for fish exposed to winter trapping and for fish in the control channels during the summer beaching. Stranding had no significant effect on body fat in three out of four experiments (fourth experiment not analysed). The four different experiments involved a total 40 experimental channels and 40 control channels, but none of the experiments displayed any major stranding effects.
Fish did grow poorly in length in all experiments and growth was negative in some, a property that has been reported as a sign of unfavourable environmental conditions [33] . In three out of four experiments, we did not find a difference in final length between the control and the treatment fish. Only the fish from the treatment channels in the summer beaching experiment had a larger final body length (5% larger). This could have been caused by higher mortality in the treatment channels leading to improved feeding conditions, i.e. more food items and space per capita. Fish density was added as a covariate in the analyses to test for this effect and did not explain the differences in final body length, however. Regardless of the final fish density, final body length was always higher in the treatment groups, than in the controls (see Table 2). Since the channels had a natural gravel substrate, dead fish could lie hidden in substrate and the
Table 2. Details of the summer beaching experiment, listing the number of channels with different final densities for the two treatments, as well as details (final body length, final body mass, final body fat) of the fish for each final density.
channels were not checked for dead fish on a daily basis. It was therefore not possible to know when individual fish died and how fish density developed in the different channels over time, but the experimental protocol tried to ensure a similar density between channels after the stranding. Thus, it is impossible to accurately tell which actual densities each individual fish experienced and for how long, which could have helped to explain the growth differences between the channels and treatments. Although the difference in initial body length between the control and treatment fish was not significant, fish that were assigned to the treatment channels were by chance 1.7% longer than fish in the control channels. The beaching could have caused some selective mortality, i.e. eliminated the smallest fish in these channels and thus increased the mean final length. Because of the lack of PIT-tags in these experiments, it was not possible to follow individual fish and thus the reasons for the increased body length will therefore only be speculations.
Fish in all four experiments lost body mass, regardless of the treatment, thus indicating poor growth conditions in all channels. The change from the fine to the coarse netting at the water inflow from the trapping to the beaching experiments improved the feeding conditions in the channels since fish lost less weight in the latter experiments. Likewise as for final body length, in three out of four experiments there were no differences in body mass between the control and treatment channels, thus stranding had no effect on growth in body mass. The only exception were the fish experiencing beaching during summer, which were on average 22% heavier than the control fish (see Table 2). Final fish density did not account for the differences, but selective mortality could have increased mean final body mass (as was discussed above for final body length). The fluctuations in water level may also induce catastrophic drift in invertebrates [34] and thus affected the fish mass change in the experimental channels less negatively. Both too little or too abundant food may mask difference between control and experimentally stranded fish, as the fish may lose or gain their energy as fast as possible. It is therefore important that conditions are not too adverse or benign for the fish in order to test effects of stranding and the channels used have previously been used for growth experiments [24] [35] . We are fully aware of the lack of growth in both control and experimental groups in several of the experiments, but this underpins the need to do these experiments in an environment where the fish cannot compensate the extra energy used in the experimental situation.
Lipid stores in juvenile salmonids seem to be important for winter survival, and relatively small increases in metabolism may have severe consequences (e.g. [35] ). A long-term consequence of depletion or not being able to build up energy stores for winter might be reduced over-winter survival [36] [37] . In our experiments, fish lost body fat during the winter trapping and the summer beaching experiment, but body fat content did not differ between the control and treatment fish. There were no changes in body fat during the winter beaching experiment (when compared to a pre-study base group), probably due to low metabolism (Wootton, 1998) and thus low energy expenditure of the fish. Since body fat was the same for the control and treatment fish in all four experiments, stranding did not increase metabolism and thus did not influence winter survival in our experiments. In contrast to the present study, other studies [26] [38] showed that non-lethal effects of hydropeaking (i.e. changes in water discharge and water level without actual stranding) result in decreased growth and body fat in juvenile salmonids.
Our experiments did not measure stress, but it has been shown that juvenile brown trout exposed to a fluctuating flow regime quickly recover from this artificial stressor and that their blood cortisol levels normalise already after 6 h after the first low flow stage [22] . When measuring blood cortisol after consecutive days of fluctuating flow and water levels, [22] did not find differences in cortisol levels between the control and treatment fish. Although fish showed an initial stress-response to the changes in water flow, the effect was only short lived and brown trout became accustomed to the stressor. The mortality may be slightly underestimated, as it has been shown that delayed mortality may occur due to experiences with harsh hydraulic conditions [39] . Adaptation to variable flow conditions could explain the absence of effects of stranding on fish growth or body fat content in the present experiments. In the trapping experiments, where fish experienced stranding on a daily basis, fish might have shown a stress response at the beginning of the experiments, which evened out in the course of the experimental period. In the beaching experiment, where fish experienced stranding only at the first day of the experiment, the stress response to the treatment was probably only short-lived. Based on another experiment, [40] concluded that juvenile brown trout exposed to fluctuating or stable low water levels (8 cm) had a reduced growth rate compared to fish exposed to stable high water levels (49 cm). Our experimental channels were rather small, being only little more than 1 m2 in size and having a mean water depth of 10 cm, thus our control fish experienced stable but low water levels which could have affected their growth, as is suggested by [40] . Although stranding is most likely more stressful than a stable low water level and negatively affected the fish in the treatment channels (as can be seen by the mortalities), the differences in impact on growth might have been too small to detect growth differences between stranded and control salmon.
One could argue that the small size of our experimental channels had a major effect on our results and that the poor growth conditions inhibited us to find differences between the control and treatment channels. In our view, it is not necessary to have positive growth in either length or mass to be able to detect negative effects of stranding on growth. If non-lethal stranding has negative effects on growth in fish our experimental setup, which included control channels exactly for that purpose, should have shown us this negative impact. However, we could not find such a negative impact, neither during summer and winter, nor during trapping and beaching. Hatchery reared fish may have lower ability to regulate their metabolism down in periods with low food availability than wild fish [41] , but we cannot see that this should have affected our interpretation of results from the experiments. As mentioned in methods, the channels have been used for growth experiments based on available food in the channels (e.g. [25] ), but conditions has been insufficient to provide growth under the present experimental situation. The advantage of downscaling from a natural to an experimental system is that it allows us to study our focal factor (in our case: stranding) in a controlled environment, which is nearly impossible in a natural system. However, results from experimental studies have to be verified in nature to ensure their generality.
Juvenile Atlantic salmon are restricted to rearing and feeding habitats in the proximity of stream and river margins [42] [43] (e.g. Mitchell et al. 1998; Mäki-Petäys et al. 2004). These shallow shoreline habitats are strongly affected by hydropeaking [12] [13] [44] [45] and are obviously among the first to be drained. Atlantic salmon parr have been shown to hold position even when flow changes, often accompanied by an increased use of shelter [46] [47] [48] . In a small-scale experiment, [46] found responses of salmon parr to acute reductions in water flow to be variable and the majority of the experimental parr remained in shallow areas prone to dewatering. Actually, there appeared to be no strong selection for mechanisms facilitating emigration from shallow areas. Additionally, salmonids are known to intensify their sheltering behaviour below a certain water temperature [49] [50] [51] [52] to avoid predation and to conserve energy [53] [54] [55] . This leads to a typical winter shelter-and-move behaviour [36] [49] [54] [56] [57] [58] , which might increase the stranding risk during daytime [48] . If water levels decline, sheltering fish might get trapped and stranded, often, but not always, with lethal consequences [14] [15] [17] [48] [59] [60] [61] (Bradford et al. 1995; Cocherell et al. 2012; Hunter 1992; Hvidsten 1985; Saltveit et al. 2001; Scruton et al. 2008; Kelly et al. 2016). Our winter experiments indicate that both trapping and beaching can give low mortality in the absence of freezing or predation. If juvenile fish find pockets of water, they may survive stranding, especially at low water temperatures, as their metabolism [62] and thus oxygen demands are low. The high mortality in the summer trapping experiment was probably a result of extensive Saprolegnia sp. infections due to the injection of PIT-tags, and was not a result of stranding since it occurred in both the control and treatment channels. The high mortality in the treatment channels of the summer beaching experiment does not come as a surprise since the channels were almost completely drained of water.
5. Conclusion
Without predators or freezing, the experiments indicate that Atlantic salmon juveniles may survive trapping for at least three hours during winter in pockets of residual water between the gravel. Beaching had the most severe effects on the juveniles and short lasting periods during summer may have immediate or subsequent lethal effects on Atlantic salmon juveniles. The winter effects of beaching may also be severe in cases with longer lasting beaching periods than those used here, as fish will have no opportunity to seek shelter in pools or ground water refuges. In surviving fish, very small effects on fish performance were detected from artificial stranding.
Acknowledgements
Knut Bergesen and Kristian Pettersen (both NINA) assisted in fish tagging and the staff of the NINA research station Ims offered logistical help during the experiment. Anne Lohrmann is acknowledged for the body composition analysis in the laboratory. Wouter Koch and Øystein Kielland helped with setting up the experiment. The experiment was approved by the Norwegian Animal Research Authority FDU (ID: 5372) (licence for Knut Bergesen) and were performed according to legislation on animal protection and care. The work was part of the EnviPEAK-project and has been financed by Centre for Environmental Design of Renewable Energy (CEDREN), one of the Norwegian Research Council’s Research Centres for Environmental Friendly Energy (FME contract-number: 193818).
Cite this paper
Puffer, M., Berg, O.K., Einum, S., Saltveit, S.J. and Forseth, T. (2017) Energetic Consequences of Strand- ing of Juvenile Atlantic Salmon (Salmo salar L.). Journal of Water Resource and Protection, 9, 163-182. https://doi.org/10.4236/jwarp.2017.92012
References
- 1. Abdullah, M.A., Agalgaonkar, A.P. and Muttaqi, K.M. (2014) Climate Change Mitigation with Integration of Renewable Energy Resources in the Electricity Grid of New South Wales, Australia. Renewable Energy, 66, 305-313.
https://doi.org/10.1016/j.renene.2013.12.014 - 2. Frey, G.W. and Linke, D.M. (2002) Hydropower as a Renewable and Sustainable Energy Resource Meeting Global Energy Challenges in a Reasonable Way. Energy Policy, 30, 1261-1265.
https://doi.org/10.1016/S0301-4215(02)00086-1 - 3. IEA (2011) Deploying Renewables 2011: Best and Future Policy Practice. Organisation for Economic Co-Operation and Development, International Energy Agency, Paris.
- 4. Premalatha, M., Abbasi, T. and Abbasi, S.A. (2014) A Critical View on the Eco-Friendliness of Small Hydroelectric Installations. Science of the Total Environment, 481, 638-643.
https://doi.org/10.1016/j.scitotenv.2013.11.047 - 5. Kern, J.D., Patino-Echeverri, D. and Characklis, G.W. (2014) The Impacts of Wind Power Integration on Sub-Daily Variation in River Flows Downstream of Hydro-electric Dams. Environmental Science & Technology, 48, 9844-9851.
https://doi.org/10.1021/es405437h - 6. Latorre, J.M., Cerisola, S., Ramos, A. and Perea, A. (2014) Coordinated Hydropower Plant Simulation for Multireservoir Systems. Journal of Water Resources Planning and Management, 140, 216-227.
https://doi.org/10.1061/(ASCE)WR.1943-5452.0000306 - 7. Cushman, R.M. (1985) Review of Ecological Effects of Rapidly Varying Flows Downstream from Hydroelectric Facilities. North American Journal of Fisheries Management, 5, 330-339.
https://doi.org/10.1577/1548-8659(1985)5<330:ROEEOR>2.0.CO;2 - 8. Moog, O. (1993) Quantification of Daily Peak Hydropower Effects on Aquatic Fauna and Management to Minimize Environmental Impacts. Regulated Rivers: Research & Management, 8, 5-14.
https://doi.org/10.1002/rrr.3450080105 - 9. Oud, E. (2002) The Evolving Context for Hydropower Development. Energy Policy, 30, 1215-1223.
https://doi.org/10.1016/S0301-4215(02)00082-4 - 10. Winemiller, K., et al. (2016) Balancing Hydropower and Biodiversity in the Amazon, Congo, and Mekong. Science, 351, 128-129.
https://doi.org/10.1126/science.aac7082 - 11. Poff, N.L., et al. (1997) The Natural Flow Regime: A Paradigm for River Conservation and Restoration. Bioscience, 47, 769-784.
https://doi.org/10.2307/1313099 - 12. Schmutz, S., et al. (2015) Response of Fish Communities to Hydrological and Morphological Alterations in Hydropeaking Rivers of Austria. River Research and Applications, 31, 919-930.
https://doi.org/10.1002/rra.2795 - 13. Kennedy, T.A., et al. (2016) Flow Management for Hydropower Extirpates Aquatic Insects, Undermining River Food Webs. BioScience, 66, 561-575.
https://doi.org/10.1093/biosci/biw059 - 14. Hunter, M.A. (1992) Hydropower Flow Fluctuations and Salmonids: A Review of the Biological Effects, Mechanical Causes and Options for Mitigation. Department of Fish and Wildlife, Olympia.
- 15. Hvidsten, N.A. (1985) Mortality of Pre-Smolt Atlantic Salmon, Salmo salar L., and Brown Trout, Salmo trutta L., Caused by Fluctuating Water Levels in the Regulated River Nidelva, Central Norway. Journal of Fish Biology, 27, 711-718.
https://doi.org/10.1111/j.1095-8649.1985.tb03215.x - 16. Nagrodski, A., Raby, G.D., Hasler, C.T., Taylor, M.K. and Cooke, S.J. (2012) Fish Stranding in Freshwater Systems: Sources, Consequences, and Mitigation. Journal of Environmental Management, 103, 133-141.
https://doi.org/10.1016/j.jenvman.2012.03.007 - 17. Saltveit, S.J., Halleraker, J.H., Arnekleiv, J.V. and Harby, A. (2001) Field Experiments on Stranding in Juvenile Atlantic Salmon (Salmo salar) and Brown Trout (Salmo trutta) during Rapid Flow Decreases Caused by Hydropeaking. Regulated Rivers: Research & Management, 17, 609-622.
https://doi.org/10.1002/rrr.652 - 18. Halleraker, J.H., et al. (2003) Factors Influencing Stranding of Wild Juvenile Brown Trout (Salmo trutta) during Rapid and Frequent Flow Decreases in an Artificial Stream. River Research and Applications, 19, 589-603.
https://doi.org/10.1002/rra.752 - 19. Irvine, R.L., et al. (2014) Why Do Fish Strand? An Analysis of Ten Years of Flow Reduction Monitoring Data from the Columbia and Kootenay Rivers, Canada. River Research and Applications, 21, 1242-1250.
- 20. Casas-Mulet, R., Alfredsen, K., Brabrand, Å. and Saltveit, S.J. (2016) Hydropower Operations in Groundwater-Influenced Rivers: Implications for Atlantic Salmon, Salmo salar, Early Life Stage Development and Survival. Fisheries Management and Ecology, 23, 144-151.
https://doi.org/10.1111/fme.12165 - 21. Heggenes, J., Bremset, G. and Brabrand, Å. (2010) Groundwater, Critical Habitats, and Behaviour of Atlantic Salmon, Brown Trout and Arctic Char in Streams. NINA Rapport 654, Norsk Institutt for Naturforskning, Trondheim, 28 p.
- 22. Flodmark, L.E.W., et al. (2002) Cortisol and Glucose Responses in Juvenile Brown Trout Subjected to a Fluctuating Flow Regime in an Artificial Stream. Journal of Fish Biology, 60, 238-248.
https://doi.org/10.1111/j.1095-8649.2002.tb02401.x - 23. Finstad, A.G., Forseth, T., Næsje, T. and Ugedal, O.L.A. (2004) The Importance of Ice Cover for Energy Turnover in Juvenile Atlantic Salmon. Journal of Animal Ecology, 73, 959-966.
https://doi.org/10.1111/j.0021-8790.2004.00871.x - 24. Skoglund, H., Einum, S. and Robertsen, G. (2011) Competitive Interactions Shape Offspring Performance in Relation to Seasonal Timing of Emergence in Atlantic Salmon. Journal of Animal Ecology, 80, 365-374.
https://doi.org/10.1111/j.1365-2656.2010.01783.x - 25. Finstad, A.G., Einum, S., Forseth, T. and Ugedal, O. (2007) Shelter Availability Affects Behaviour, Size-Dependent and Mean Growth of Juvenile Atlantic Salmon. Freshwater Biology, 52, 1710-1718.
https://doi.org/10.1111/j.1365-2427.2007.01799.x - 26. Puffer, M., Berg, O.K., Huusko, A., Vehanen, T., Forseth, T. and Einum, S. (2015) Seasonal Effects of Hydropeaking on Growth, Energetics and Movement of Juvenile Atlantic Salmon (Salmo Salar). River Research and Applications, 31, 1101-1108.
https://doi.org/10.1002/rra.2801 - 27. Berg, O.K., Rød, G., Solem, ø. and Finstad, A.G. (2011) Pre-Winter Lipid Stores in Brown Trout Salmo trutta along Altitudinal and Latitudinal Gradients. Journal of Fish Biology, 79, 1156-1166.
https://doi.org/10.1111/j.1095-8649.2011.03097.x - 28. Dobush, G.R., Ankney, C.D. and Krementz, D.G. (1985) The Effect of Apparatus, Extraction Time, and Solvent Type on Lipid Extractions of Snow Geese. Canadian Journal of Zoology, 63, 1917-1920.
https://doi.org/10.1139/z85-285 - 29. R Core Team (2015) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna.
- 30. Pinheiro, J., et al. (2015) Nlme: Linear and Nonlinear Mixed Effects Models.
- 31. Zuur, A.F., Ieno, E.N., Walker, N., Saveliev, A.A. and Smith, G.M. (2009) Mixed Effects Models and Extensions in Ecology with R. Springer, New York, 574.
https://doi.org/10.1007/978-0-387-87458-6 - 32. Holm, S. (1979) A Simple Sequentially Rejective Multiple Test Procedure. Scandinavian Journal of Statistics, 6, 65-70.
- 33. Huusko, A., Mäki-Petäys, A., Stickler, M. and Mykrä, H. (2011) Fish Can Shrink under Harsh Living Conditions. Functional Ecology, 25, 628-633.
https://doi.org/10.1111/j.1365-2435.2010.01808.x - 34. Bruno, M.C., Cashman, M.J., Maiolini, B., Biffi, S. and Zolezzi, G. (2016) Responses of Benthic Invertebrates to Repeated Hydropeaking in Semi-Natural Flume Simulations. Ecohydrology, 9, 68-82.
https://doi.org/10.1002/eco.1611 - 35. Finstad, A.G., Berg, O., Forseth, T., Ugedal, O. and Næsje, T.F. (2010) Adaptive Winter Survival Strategies: Defended Energy Levels in Juvenile Atlantic Salmon along a Latitudinal Gradient. Proceedings of the Royal Society B: Biological Sciences, 277, 1113-1120.
https://doi.org/10.1098/rspb.2009.1874 - 36. Cunjak, R.A., Prowse, T.D. and Parrish, D.L. (1998) Atlantic Salmon (Salmo salar) in Winter: The Season of Parr Discontent? Canadian Journal of Fisheries and Aquatic Sciences, 55, 161-180.
https://doi.org/10.1139/d98-008 - 37. Scruton, D.A., et al. (2005) Seasonal Response of Juvenile Atlantic Salmon to Experimental Hydropeaking Power Generation in Newfoundland, Canada. North American Journal of Fisheries Management, 25, 964-974.
https://doi.org/10.1577/M04-133.1 - 38. Korman, J. and Campana, S.E. (2009) Effects of Hydropeaking on Nearshore Habitat Use and Growth of Age-0 Rainbow Trout in a Large Regulated River. Transactions of the American Fisheries Society, 138, 76-87.
https://doi.org/10.1577/T08-026.1 - 39. Rechisky, E.L., et al. (2009) Experimental Measurement of Hydrosystem-Induced Delayed Mortality in Juvenile Snake River Spring Chinook Salmon (Oncorhynchus tshawytscha) Using a Large-Scale Acoustic Array. Canadian Journal of Fisheries and Aquatic Sciences, 66, 1019-1024.
https://doi.org/10.1139/F09-078 - 40. Flodmark, L.E.W., Vollestad, L.A. and Forseth, T. (2004) Performance of Juvenile Brown Trout Exposed to Fluctuating Water Level and Temperature. Journal of Fish Biology, 65, 460-470.
https://doi.org/10.1111/j.0022-1112.2004.00463.x - 41. Auer, S.K., Salin, K., Rudolf, A.M., Anderson, G.J. and Metcalfe, N.B. (2015) Flexibility in Metabolic Rate Confers a Growth Advantage under Changing Food Availability. Journal of Animal Ecology, 84, 1405-1411.
https://doi.org/10.1111/1365-2656.12384 - 42. Mitchell, J., McKinley, R.S., Power, G. and Scruton, D.A. (1998) Evaluation of Atlantic Salmon Parr Responses to Habitat Improvement Structures in an Experimental Channel in Newfoundland, Canada. Regulated Rivers: Research & Management, 14, 25-39.
https://doi.org/10.1002/(SICI)1099-1646(199801/02)14:1<25::AID-RRR474>3.0.CO;2-1 - 43. Mäki-Petäys, A., Erkinaro, J., Niemelä, E., Huusko, A. and Muotka, T. (2004) Spatial Distribution of Juvenile Atlantic Salmon (Salmo salar) in a Subarctic River: Size-Specific Changes in a Strongly Seasonal Environment. Canadian Journal of Fisheries and Aquatic Sciences, 61, 2329-2338.
https://doi.org/10.1139/f04-218 - 44. Bain, M.B., Finn, J.T. and Booke, H.E. (1988) Streamflow Regulation and Fish Community Structure. Ecology, 69, 382-392.
https://doi.org/10.2307/1940436 - 45. Stanford, J.A. (1994) Instream Flows to Assist the Recovery of Endangered Fishes of the Upper Colorado River Basin, in National Biological Survey. US Department of the Interior, Washington DC, 53.
- 46. Armstrong, J.D., Braithwaite, V.A. and Fox, M. (1998) The Response of Wild Atlantic Salmon Parr to Acute Reductions in Water Flow. Journal of Animal Ecology, 67, 292-297.
https://doi.org/10.1046/j.1365-2656.1998.00193.x - 47. Robertson, M.J., Pennell, C.J., Scruton, D.A., Robertson, G.J. and Brown, J.A. (2004) Effect of Increased Flow on the Behaviour of Atlantic Salmon Parr in Winter. Journal of Fish Biology, 65, 1070-1079.
https://doi.org/10.1111/j.0022-1112.2004.00516.x - 48. Scruton, D.A., et al. (2008) A Synopsis of Hydropeaking Studies on the Response of Juvenile Atlantic Salmon to Experimental Flow Alteration. Hydrobiologia, 609, 263-275.
https://doi.org/10.1007/s10750-008-9409-x - 49. Fraser, N.H.C., Metcalfe, N.B. and Thorpe, J.E. (1993) Temperature-Dependent Switch between Diurnal and Nocturnal Foraging in Salmon. Proceedings of the Royal Society B: Biological Sciences, 252, 135-139.
https://doi.org/10.1098/rspb.1993.0057 - 50. Gibson, R.J. (1978) Behavior of Juvenile Atlantic Salmon (Salmo salar) and Brook Trout (Salvelinus fontinalis) with Regard to Temperature and to Water Velocity. Transactions of the American Fisheries Society, 107, 703-712.
https://doi.org/10.1577/1548-8659(1978)107<703:TBOJAS>2.0.CO;2 - 51. Heggenes, J., Bagliniere, J.L. and Cunjak, R.A. (1999) Spatial Niche Variability for Young Atlantic Salmon (Salmo salar) and Brown Trout (Salmo trutta) in Heterogeneous Streams. Ecology of Freshwater Fish, 8, 1-21.
https://doi.org/10.1111/j.1600-0633.1999.tb00048.x - 52. Rimmer, D.M. and Paim, U. (1990) Effects of Temperature, Photoperiod, and Season on the Photobehavior of Juvenile Atlantic Salmon (Salmo salar). Canadian Journal of Zoology, 68, 1098-1103.
https://doi.org/10.1139/z90-162 - 53. Cunjak, R.A. (1996) Winter Habitat of Selected Stream Fishes and Potential Impacts from Land-Use Activity. Canadian Journal of Fisheries and Aquatic Sciences, 53, 267-282.
https://doi.org/10.1139/f95-275 - 54. Metcalfe, N.B., Fraser, N.H.C. and Burns, M.D. (1999) Food Availability and the Nocturnal vs. Diurnal Foraging Trade-Off in Juvenile Salmon. Journal of Animal Ecology, 68, 371-381.
https://doi.org/10.1046/j.1365-2656.1999.00289.x - 55. Valdimarsson, S.K. and Metcalfe, N.B. (1998) Shelter Selection in Juvenile Atlantic Salmon or Why Do Salmon Seek Shelter in Winter? Journal of Fish Biology, 52, 42-49.
https://doi.org/10.1111/j.1095-8649.1998.tb01551.x - 56. Bremset, G. (2000) Seasonal and Diel Changes in Behaviour, Microhabitat Use and Preferences by Young Pool-Dwelling Atlantic Salmon, Salmo salar, and Brown Trout, Salmo trutta. Environmental Biology of Fishes, 59, 163-179.
https://doi.org/10.1023/A:1007691316864 - 57. Heggenes, J. and Dokk, J.G. (2001) Contrasting Temperatures, Waterflows, and Light: Seasonal Habitat Selection by Young Atlantic Salmon and Brown Trout in a Boreonemoral River. Regulated Rivers: Research & Management, 17, 623-635.
https://doi.org/10.1002/rrr.620 - 58. Heggenes, J., Krog, O., Lindås, O.R. and Dokk, J.G. (1993) Homeostatic Behavioral Responses in a Changing Environment-Brown Trout (Salmo trutta) Become Nocturnal during Winter. Journal of Animal Ecology, 62, 295-308.
https://doi.org/10.2307/5361 - 59. Bradford, M.J., Taylor, G.C., Andrew Allan, J. and Higgins, P.S. (1995) An Experimental Study of the Stranding of Juvenile Coho Salmon and Rainbow Trout during Rapid Flow Decreases under Winter Conditions. North American Journal of Fisheries Management, 15, 473-479.
https://doi.org/10.1577/1548-8675(1995)015<0473:AESOTS>2.3.CO;2 - 60. Cocherell, S., et al. (2012) A Lateral-Displacement Flume for Fish Behavior and Stranding Studies during Simulated Pulsed Flows. Environmental Biology of Fishes, 93, 143-150.
https://doi.org/10.1007/s10641-011-9894-z - 61. Kelly, B., Smokorowski, K.E. and Power, M. (2016) Slimy Sculpin (Cottus cognatus) Annual Growth in Contrasting Regulated and Unregulated Riverine Environments. Hydrobiologia, 768, 239-253.
https://doi.org/10.1007/s10750-015-2553-1 - 62. Wootton, R.J. (1998) Ecology of Teleost Fishes. 2nd Edition, Kuwer Academic Publishers, Dordrecht.



