Journal of Cancer Therapy
Vol.5 No.6(2014), Article ID:45947,6 pages DOI:10.4236/jct.2014.56059
Detection of Epstein-Barr Virus in Nasopharyngeal Carcinoma in Sudanese by in Situ Hybridization
Ameera A. M. Adam1, Nazik E. Abdullah2, Lamyaa A. M. El Hassan3, Elwaleed M. Elamin4, Muntaser E. Ibrahim1, Ahmed M. El Hassan1
1Department of Molecular Biology, Institute of Endemic Diseases, University of Khartoum, Khartoum, Sudan
2Department of Surgery, Faculty of Medicine, University of Khartoum, Khartoum, Sudan
3School of Medicine, Ahfad University for Women, Omdurman, Higher Specialization in Histopathology, Khartoum, Sudan
4Faculty of Medical Laboratory Sciences, Alzaeim Alazhari University, Khartoum, Sudan
Email: amiera_adam2001@yahoo.co.uk
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/


Received 3 March 2014; revised 2 April 2014; accepted 10 April 2014
ABSTRACT
Objectives: The aim of this study is to detect Epstein-Barr virus (EBV) in nasopharyngeal carcinoma (NPC) biopsies of Sudanese patients using EBV-encoded RNA (EBER) in Situ hybridization (EBER-ISH). Study Design: This is a descriptive cross-sectional study conducted at the National Center for ENT diseases and Head and Neck Surgery and the Institute of Endemic Diseases, University of Khartoum, Khartoum City, Sudan. Subjects and Methods: Biopsies from 43 patients with nasopharyngeal carcinoma were examined for the presence of Epstein-Barr virus using EBER-ISH. Ten normal samples were used to assess the presence of the virus in non cancer tissues. Results: Fifty three samples were examined for the presence of the virus by EBER-ISH, 43 biopsies were NPC and ten were normal. Histologically the cases were, 20 (46.5%), 20 (46.5%) and 3 (7%) of the biopsies were classified as WHO types II, III and mixed type II and III, respectively; there were no cases of type I NPC. All nasopharyngeal carcinoma biopsies (100%) were positive for EBER1 in almost all carcinoma cells with focal and intense dark-blue staining limited to the nucleus; no hybridization was observed in the cytoplasm. No hybridization was observed in all ten non cancer tissues. Conclusion: All NPC cells are clearly EBV-infected. The virus is located in the nucleus of the tumour cells. The presence of Epstein-Barr virus in normal nasopharyngeal epithelia is not a common event.
Keywords:EBV, NPC, Sudan, ISH, EBER

1. Introduction
Nasopharyngeal carcinoma (NPC) is an epithelial tumor that has a distinct geographical distribution. It affects Chinese in southeast Asia, Arabs in north Africa and Eskimos in Antarctica [1] . Sudan is considered an intermediate area of endemicity for this type of cancer [2] [3] . According to the World Health Organization (WHO), NPC is classified histologically into three types: Type I (keratinizing squamous cell carcinoma), type II (non keratinizing carcinoma), and type III (undifferentiated carcinoma) [4] . An additional mixed type consisting of types II and III was reported in 2011 [5] . It is well-documented that Epstein-Barr virus (EBV) is associated with this neoplasm [6] . EBV is detected in both II and III histological types but not in type I in low endemic areas [7] . In high risk areas the virus is detected in all histological types [8] .
Evidence for a role of the virus in the pathogenesis of NPC includes: 1) elevated antibody titers to the virus preceding development of the neoplasm; 2) presence of the viral genome within the neoplastic cells but not in associated, adjacent non-neoplastic cells; 3) clonality of the viral genome in the tumor, and 4) expression of viral genes in the neoplastic cells [9] . Different methods for investigating the association between EBV and nasopharyngeal carcinoma have been used. These include serology [10] , polymerase chain reaction (PCR) [11] , immunohistochemistry [12] , and in situ hybridization (ISH) [13] . Of these methods, the in situ hybridization technique that detects EBER is the only way to directly verify the existence of the EBV genome in the nuclei of tumor cells. EBER-1 does not code any protein and its function is still unknown, but it is known to be abundantly expressed in infected cells (106 copies per cell), making itself an appropriate target for detection of EBV [14] . EBER-ISH has been established as the most sensitive and practical method for detecting EBV. It is reported to have a sensitivity of 98% and a specificity of 100% in detecting primary NPC [15] . It also has a strong impact on the prognosis of NPC according to Nakado et al. and Kengjian et al. who concluded that it is an independent and excellent predictor of NPC [16] [17] .
In a previous study we detected EBV in nasopharyngeal carcinoma among Sudanese patients using PCR. Two markers were used: Epstein-Barr virus nuclear Antigen-1 (EBNA-1) and Latent Membrane protein-1 (LMP-1). Both markers gave a high frequency of the presence of the virus in the tumor. The frequencies were 77.6% and 84.5% for EBNA-1 and LMP-1 primers, respectively [5] . PCR technique gives the key step in the contribution of EBV in the pathology of NPC, but it could not provide an evidence for the cellular localization of the virus, since tumor extracts contained DNA from both malignant epithelial cells and infiltrating lymphocytes, so there is a need to demonstrate the virus in its local cellular environment using morphology-based technique and so confirm its contribution in this cancer.
The present study is the first in Sudan that investigated the presence of EBV genome in NPC by using ISH.
The aim of this study is to detect EBV genome, to determine its cellular localization in NPC biopsies and to assess its presence in non cancer tissue.
2. Subjects and Methods
2.1. Patients
Forty-three NPC biopsies from patients, who attended the nasopharyngeal carcinoma clinic at the ENT Khartoum teaching Hospital and confirmed to be NPC histologically, were included in this study. Informed consent was obtained from all patients. In the case of the children, consent was signed by the patients [5] . Ten histologically normal samples from normal subject were used as non cancer tissue. The study was approved by the Ethical Review Committee Board of the Institute of Endemic Diseases, University of Khartoum in March 2005.
2.2. Samples
Samples were fixed in 10% neutral formalin and two sections of 5 µm thickness were made. One was stained with Haematoxylin and Eosin for histopathological examination. The tumors were classified as described previously [5] . The other section was used for ISH. ISH was made with EBV peptide nucleic acid (PNA) Probe/ Fluorescein (Dako, Denmark) and detected by PNA ISH detection Kit. Hybridization lasted 1.5 hours at 55˚C and was visualized by alkaline phosphatase (AP)-conjugated anti-fluorescein antibodies. Nitro blue tetrazolium (NBT)/Bromo chloroIndoyl phosphate (BCIP) (Dako, Denmark) was used as a substrate for AP. To test the sensitivity and specificity of ISH, both negative and positive controls supplied by the manufacturer were used. A Burkitt’s lymphoma biopsy served as an additional positive control. A case was considered to be positive if the nucleus of a tumor cell stained dark blue or black.
3. Results
The age of the patients ranged from 10 to 80 years and the median age was 45 years. Thirty four (79.1%) were males and 9 (21.1%) were females. Histological, twenty (46.5%), 20(46.5%) and 3 (7%) of the biopsies were classified as types II, III and mixed type II and III, respectively; there were no cases of type I NPC (Table 1).
All NPC biopsies (100%) expressed EBER1 in all carcinoma cells with an intense dark-blue staining limited to the nucleus; no hybridization was observed in the cytoplasm (Figure 1). No hybridization was observed in all ten non cancer tissues.
Abbreviations and Acronyms
EBV: Epstein-Barr Virus; NPC: Nasopharyngeal Carcinoma; WHO: World Health Organization; EBNA-1: Epstein-Barr Virus Nuclear Antigen-1; LMP-1: Latent Membrane Protein-1; ISH: In Situ Hybridization;
Table 1. Charactristics of the sudied pateints.
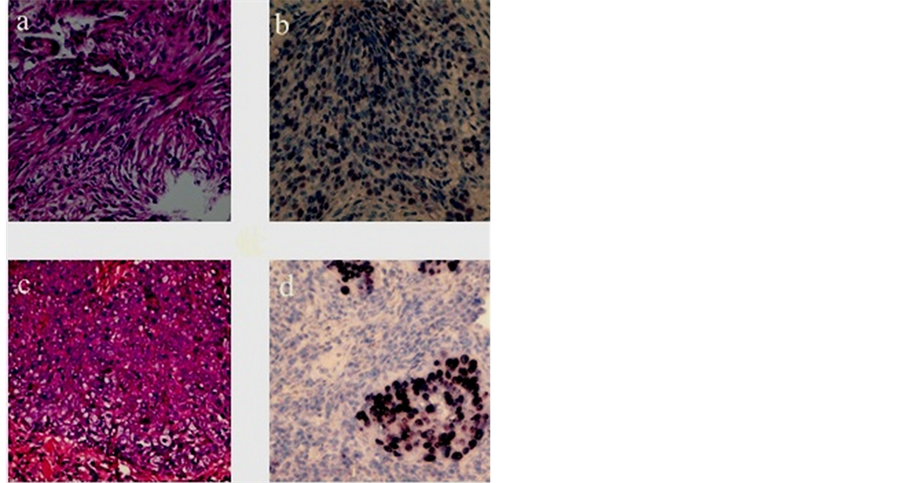
Figure 1. (a) non-kernatinizing carcinoma (WHO type II) (H & E ×40). (b) Nuclear staining with ISH for EBER in nonkeratinizing carcinoma (WHO type II). (c) Undifferentiated carcinoma (WHO type III) (H & E ×40). (d) Nuclear staining with ISH for EBER in undifferentiated carcinoma (WHO type III).
EBER: Epstein-Barr Virus Encoded RNA; PCR: Polymarase Chain Reaction; PNA: Peptide Nucleic Acid; NBT: Nitro blue tetrazolium; BCIP: Bromo chloroIndoyl phosphate; AP: Alkaline Phosphatase.
4. Discussion
The association between EBV and NPC is well documented [6] [18] . The most sensitive method for investigating this association is the EBER-ISH [19] . It is well known that NPC incidence in Western countries is lower than in the Far East while the Mediterranean basin countries have an intermediate incidence of this type of cancer [20] . EBV frequencies using EBER-ISH were reported from different areas in the world with variable degrees of endemicity for NPC. In this study the virus was detected in all biopsies (100%).
Malaysia, Japan and Taiwan, are areas of high endemicity [21] . In a study of 107 cases of NPC from Taiwan, EBV was detected by ISH in 105 cases [15] . In another study among Japanese patients, EBV genome was detected by the same method in all 39 cases of NPC, including the type I, keratinizing subtype [22] . In Malaysian patients, the virus genome was demonstrated in all 120 patients [23] .
Using ISH for EBER, similar studies were conducted in regions with intermediate endemicity for NPC such as Italy, Spain, the United States of America, Morocco, Iran and Turkey. Interestingly, these studies revealed high frequencies of EBV infection [24] -[29] .
In normal epithelia of the non cancer tissues, no hybridization was detected. This result is in agreement with Sam et al. who reported no signal detection of EBV using ISH in normal nasopharyngeal biopsies from Malaysian subjects [30] . Also Dickens et al. detected no signal for EBV in eight normal samples among the Chinese, a known high area for NPC expressing EBV [31] . It is worth mentioning that in our cases, no virus was detected in the normal lymphocyte and epithelium surrounding the tumor.
These findings also raise several questions that remain largely unanswered: when does EBV enter the epithelial cell, how it does that and what type of receptors expressed by cells allow its entry. It might also be that EBV infection is not an early event in the carcinogenesis of NPC.
5. Conclusion
In conclusion, EBV is found in a high frequency among Sudanese patients which could indicate its role in the pathogenesis of the tumor. This high incidence of the virus among NPC patients can be used as a tumor marker for the diagnosis and prognostication of the disease and also as a tool for screening for this tumor. However, the number of biopsies examined was relatively small and further work needs to be carried out to test the validity of this provisional conclusion.
Acknowledgements
This research was supported by The Academy of Sciences for the Developing World (TWAS) Research Grant 07-044/BIO/AF/AC. We also acknowledge the Center for ENT diseases and Head and Neck Surgery medical staff for their help with sampling the patients.
References
- Yu, M.C. and Yuan, J. (2006) Nasopharyngeal Cancer. In: Schottenfeld, D. and Fraumeni, J.F., Eds., Cancer Epidemiology and Prevention, Oxford University Press, New York, 620-626. http://dx.doi.org/10.1093/acprof:oso/9780195149616.003.0031
- Abdullah, N.E., Adam, A.A.M., Khalifa, E.H., El Hassan, L.A.M., Elamin, E.M., Hamad, K.M. et al. (2011) Nasopharyngeal Cancer in Sudan: Epidemiology, Clinical and Histological Characteristics. Clinical Medicine Insights: Ear, Nose and Throat, 4, 5-11. http://dx.doi.org/10.4137/CMENT.S5825
- Hidayatalla, A.M.O., Malik, A.E., El Hadi, A.A., Osman, A.A and Hutt, M.S. (1983) Studies on Nasopharyngeal Carcinoma in the Sudan—I Epidemiology and Aetiology. European Journal of Cancer and Clinical Oncology, 19, 705-710. http://dx.doi.org/10.1016/0277-5379(83)90001-9
- Wei, W.I. and Sham, J.S. (2005) Nasopharyngeal Carcinoma. Lancet, 365, 2041-2054. http://dx.doi.org/10.1016/S0140-6736(05)66698-6
- Adam, A.A.M., Abdullah, N.E., Khalifa, E.H., El Hassan, L.A.M., Elamin, E.M., Hamad, K.M., et al. (2011) Pathology of Nasopharyngeal Carcinoma in Sudanese Patients and Its Association with Epstein-Barr Virus: A Report from a Single Center in Khartoum. Advances in Tumor Virology, 2, 1-6. http://dx.doi.org/10.4137/ATV.S5817
- Kieff, E. (1995) Epstein-Barr Virus—Increasing Evidence of a Link to Carcinoma. The New England Journal of Medicine, 333, 724-726. http://dx.doi.org/10.1056/NEJM199509143331110
- Neel, H.B. 3rd, Pearson, G.R. and Taylor, W. (1984) Antibodies to Epstein-Barr Virus in Patients with Nasopharyngeal Carcinoma and in Comparison Groups. Annals of Otology, Rhinology, and Laryngology, 93, 477-482.
- Cohen, J. (2002) Epstein-Barr Virus Infection. The New England Journal of Medicine, 343, 481-492. http://dx.doi.org/10.1056/NEJM200008173430707
- Pagano, J.S., Blaser, M., Buendia, M.A., Damania, B., Khalili, K., Raab-Traub, N. and Roizman, B. (2004) Infectious Agents and Cancer: Criteria for a Causal Relation. Seminars in Cancer Biology, 14, 453-471. http://dx.doi.org/10.1016/j.semcancer.2004.06.009
- Lanier, A.P., Bornkamm, G.W., Henle, W., Henle, G., Bender, T.R., Talbot, M.L., et al. (1981) Association of EpsteinBarr virus with Nasopharyngeal Carcinoma in Alaskan Native Patients: Serum Antibodies and Tissue EBNA and DNA. International Journal of Cancer, 28, 301-305. http://dx.doi.org/10.1002/ijc.2910280308
- Akao, I., Sato, Y., Mukai, K., Uhara, H., Furuya, S., Hoshikawa, T., et al. (1991) Detection of Epstein-Barr Virus DNA in Formalin-Fixed Paraffin-Embedded Tissue of Nasopharyngeal Carcinoma Using Polymerase Chain Reaction and in Situ Hybridization. Laryngoscope, 101, 279-283. http://dx.doi.org/10.1288/00005537-199103000-00010
- Niedobitek, G., Fahraeus, R,. Herbst, H., Latza, U., Ferszt, A., Klein, G., et al. (1992) The Epstein-Barr Virus Encoded Membrane Protein (LMP) Induces Phenotypic Changes in Epithelial Cells. Virchows Archiv B, 62, 55-59. http://dx.doi.org/10.1007/BF02899665
- Weiss, L.M., Movahed, L.A., Butler, A.E., Swanson, S.A., Frierson, H.F.J., Cooper, P.H., et al. (1989) Analysis of Lymphoepithelioma and Lymphoepithelioma-Like Carcinomas for Epstein-Barr Viral Genomes by in Situ Hybridization. American Journal of Surgical Pathology, 13, 625-631. http://dx.doi.org/10.1097/00000478-198908000-00001
- Howe, J.G. and Shu, M.D. (1988) Isolation and Characterization of the Genes for Two Small RNAs of Herpesvirus papio and Their Comparison with Epstein-Barr Virus-Encoded EBER RNAs. Journal of Virology, 62, 2790-2798.
- Tsai, S.T., Jin, Y.T., Mann, R.B. and Ambinder, R.F. (1998) Epstein-Barr Virus Detection in Nasopharyngeal Tissues of Patients with Suspected Nasopharyngeal Carcinoma. Cancer, 82, 1449-1453.
- Nakado, K., Mochiki, M., Nibu, K., Sugasaywa, M. and Uozaki, H. (2006) Analysis of Prognostic Factors of Nasopharyngeal Carcinoma: Impact of in Situ Hybridization for Epstein-Barr Virus Encoded Small RNA 1. Otolaryngology—Head and Neck Surgery, 134, 639-645. http://dx.doi.org/10.1016/j.otohns.2005.11.022
- Kengjian, K., Haiyun, W., Zichen, S., Liping, D., Dabo, L. and Jin, Y. (2014) Epstein-Barr Virus-Encoded RNAs as a Survival Predictor in Nasopharyngeal Carcinoma. Chinese Medical Journal, 2, 294-299.
- zur Hausen, H., Schulte-Holthausen, H., Klein, G., Henle, W., Henle, G., Clifford, P., et al. (1970) EBV DNA in Biopsies of Burkitt’s Tumours and Anaplastic Carcinomas of the Nasopharynx. Nature, 228, 1056-1058. http://dx.doi.org/10.1038/2281056a0
- Chang, K.L., Chen, Y.Y., Shibata, D. and Weiss, L.M. (1992) Description of an in Situ Hybridization Methodology for Detection of Epstein-Barr Virus RNA in Paraffin-Embedded Tissues, with a Survey of Normal and Neoplastic Tissues. Diagnostic Molecular Pathology, 1, 246-255. http://dx.doi.org/10.1097/00019606-199203000-00037
- Chan, A.T.C., Teo, P.M.L and Johnson, P.J. (2002) Nasopharyngeal Carcinoma. Annals of Oncology, 13, 1007-1015. http://dx.doi.org/10.1093/annonc/mdf179
- Collins, S.L. (1996) Squamous Cell Carcinoma of the Oral Cavity and Oropharynx. In: Ballenger, J. and Snow, J., Eds., Otorhinolaryngology: Head and Neck Surgery, Media, Williams & Wilkins, 249-368.
- Inoue, H., Sato, Y., Tsuchiya, B., Nagai, H., Takahashi, H. and Kameya, T. (2002) Expression of Epstein-Barr Virus Encoded Small Nuclear RNA1 in Japanese Nasopharyngeal Carcinomas. Acta Oto-Laryngologica, 547, 113-117. http://dx.doi.org/10.1080/000164802760057725
- Pathmanathan, R., Prasad, U., Chandrika, G., Sadler, R., Flynn, K. and Traub, R.N. (1995) Undifferentiated, Nonkeratinizing, and Squamous Cell Carcinoma of the Nasopharynx: Variants of Epstein Barr Virus-Infected Neoplasia. American Journal of Pathology, 146, 1355-1367.
- Gabusi, E., Lattes, C., Fiorentino, M., D’Errico, A. and Grigioni, W.F. (2001) Expression of Epstein-Barr Virus-Encoded RNA and Biological Markers in Italian Nasopharyngeal Carcinomas. Journal of Experimental & Clinical Cancer Research, 20, 371-376.
- Plaza, G., Manzanal, A.I., Fogué, L., Santón, A., Martínez-Montero, J.C. and Bellas, C. (2002) Association of Epstein-Barr Virus and Nasopharyngeal Carcinoma in Caucasian Patients. Annals of Otology, Rhinology, and Laryngology, 111, 210-216.
- Lin, Z., Khong, B., Kwok, S., Cao, H., West, R.B., Le, Q. and Kong C. (2014) Human Papiloma Virus 16 Detected in Nasopharyngeal Carcinomas in Caucasian American But Not in Endemic Southern Chinese Patients. Head & Neck, 36, 709-714. http://dx.doi.org/10.1002/hed.23362
- Laantri, N., Attaleb, M., Kandil, M., Naji, F., Mouttaki, T., Dardari, R., et al. (20011) Human Papillomavirus Detection in Moroccan Patients with Nasopharyngeal Carcinoma. Infect Agent Cancer, 6, 3.
- Mirzamani, N., Salehian, P., Farhadi, M. and Tehran, E.A. (2006) Detection of EBV and HPV in Nasopharyngeal Carcinoma by in Situ Hybridization. Experimental and Molecular Pathology, 81, 231-234 http://dx.doi.org/10.1016/j.yexmp.2006.04.006
- Ertan, Y., Hekimgil, M., Karaarslan, S. and Soydan, S. (2008) Expression of Epstein-Barr-Virus-Encoded Small Nuclear RNA in Nasopharyngeal Carcinomas of Aegean Turkish Patients. Virchows Arch, 452, 411-414. http://dx.doi.org/10.1007/s00428-008-0589-6
- Sam, C.K., Boorks, L.A., Niedoitek, G., Young, L.S., Prasad, U. and Riskinson, A.B. (1993) Analysis of Epstein-Barr Virus Infection in Nasopharyngeal Carcinoma from a Group at High Risk of Nasopharyngeal Carcinoma. International Journal of Cancer, 53, 957-962. http://dx.doi.org/10.1002/ijc.2910530616
- Dickens, P., Srivastava, G., Loke, S.L., Chan, C.W. and Liu, Y.T. (1992) Epstein-Barr Virus DNA in Nasopharyngeal Carcinomas from Chinese Patients in Hong Kong. Journal of Clinical Pathology, 45, 396-397. http://dx.doi.org/10.1136/jcp.45.5.396


