Chinese Medicine
Vol. 3 No. 3 (2012) , Article ID: 22713 , 8 pages DOI:10.4236/cm.2012.33022
Hepatoprotective Diterpenoids Isolated from Andrographis paniculata
1Department of Nutrition and Health Sciences, School of Healthcare Management, Kainan University, Taipei, Taiwan
2Department of Biochemical Science and Technology, College of Life Science, National Taiwan University, Taipei, Taiwan
Email: *bifong@ntu.edu.tw, d91623701@ntu.edu.tw, wwchao@mail.knu.edu.tw
Received May 28, 2012; revised June 28, 2012; accepted July 9, 2012
Keywords: Andrographis paniculata
ABSTRACT
Andrographis paniculata (Burm.f.) Nees (Acanthaceae), a plant widely used as traditional herbal medicine in many countries, has drawn attention of the researchers in recent years. Its major constituents are diterpenoids and flavonoids. This article reviews the anti-hepatotoxic effects of A. paniculata extract and derivative compounds, such as andrographolide, the major active compound, most studied for its bioactivities. Neoandrographolide shows anti-inflammatory and anti-hepatoxic properties. 14-deoxy-11,12-didehydroandrographolide and 14-deoxyabdrographolide have immunostimulatory, anti-atherosclerotic, and anti-hepatotoxic activities. The hepatoprotective activities include 1) inhibiting carbontetrachloride (CCl4), tert-butylhydroperoxide (t-BHP)-induced hepatic toxicity; 2) acting as cytochrome P450 enzymes (CYPs) inducers; 3) modulating glutathione (GSH) content; 4) influence glutathione S-transferase (GSTP) activity and phosphatidylinositol-3-kinase/Akt (PI3k/Akt) pathway; 5) synergistic effect with anti-cancer drugs induced apoptosis contributing to the bioactivities of A. paniculata extracts and isolated bioactive compounds. The articles reviewed suggest that the above compounds could be candidates for research and development as potential hepatoprotective drugs.
1. Cytochrome P450 Enzymes and Liver Function
Cirrhosis may result from chronic metabolizing of xenobiotics including drugs, toxins, and chemical carcinogens in the liver. The cytochrome P450 enzymes (CYPs) are known as a superfamily of haemoproteins with a unique spectrophotometric absorbance peak at 450 nm when reduced by a reducing agent and bound by carbon monoxide [1]. Anti-hepatotoxic enzymes include cytochrome P450s (P450) super-family, or normalizing the levels of marker enzymes for the liver function test, such as glutamate pyruvate transaminase (GPT), glutamate oxaloacetate transaminase (GOT), alkaline phosphatase (ALP) and acid phosphatase (ACP) [2]. Phase I and phase II biotransformation enzymes are involved in the metabolic activation and detoxification of various carcinogens. Phase I enzymes convert xenobiotics to active intermediates and phase II enzymes catalyze the conjugation of these active intermediates with endogenous cofactors to increase their water solubility and facilitate their excretion through urine or bile. Glutathione S-transferase (GST) is one of phase II enzymes [3].
P450s, CYP1A1 and CYP1A2 have been shown to be the major enzymes in the metabolism of potential procarcinogens such as polycyclic aromatic hydrocarbons (PAHs) and aryl and heterocyclic arylamines. CYP1A1 is expressed constitutively in several extrahepatic tissues. But, while CYP1A1 expression has been demonstrated in liver after inducer treatment, CYP1A2 is constitutively and inducibly expressed only in the liver. CYP1B1 is a relatively new member of family 1 which is constitutively expressed in steroidogenic tissues, but is not detected in liver, kidney and lung [4]. Cytochrome P450 2C19 (CYP2C19), is a major hepatic CYP isoform involved in metabolism of many clinical drugs such as Smephenytoin [5]. Human cytochrome P450s are concentrated in the liver, the major isoforms include CYP 1A2 (13%), CYP 2C9 and 2C19 (20%), CYP 2E1 (7%), CYP 2A6 (4%), CYP 2D6 (2%) and CYP 3A4 (30%). The 5 CYP isoforms 3A4, 2D6, 2C9, 1A2 and 2C19 account for almost 70% of all drug clearance [6].
2. Bioactive Constituents from Andrographis paniculata
Andrographis paniculata (Burm.f.) Nees (Acanthaceae) has been used as a traditional medicine in Taiwan, China, India and Thailand [7-9]. In traditional Chinese medicine, it is an important cold property herb used for antipyretic properties. And it is commonly used to prevent and treat the common cold [10].
A. paniculata contains diterpenes, lactones and flavonoids. Flavonoids mainly exist in the root, but also can be isolated from the leaves. The leaves contain two bitter lactone andrographolides, and kalmeghin. Active compounds extracted with ethanol or methanol from the whole plant, leaf and stem of A. paniculata include over 20 diterpenoids and over 10 flavonoids [11-13]. The active constituents of A. paniculata are diterpene lactonesincluding andrographolide, 14-deoxy-11,12-didehydroandrographolide, neoandrographolide and 14-deoxyandrographolide (Figure 1). Andrographolide is the most active and important compound of the plant [14-19]. Neoandrographolide shows anti-inflammatory and anti-hepatoxic properties. 14-deoxy-11,12-didehydroandrographolide and 14-deoxyabdrographolide have immunostimulatory, anti-atherosclerotic, and anti-hepatotoxic activities. Two flavonoids identified as 5,7,2’,3’-tetramethoxyflavanone and 5-hydroxy-7,2’,3’-trimethoxyflavone were also isolated from the whole plant. Extracts of the plant have been reported to exhibit a wide range of biological activities of therapeutic effects such as anti-inflammation, anti-cancer, immunomodulation, anti-infection, anti-hepatotoxicity, anti-atherosclerosis, anti-hyperglycemic effect and anti-oxidation [20].

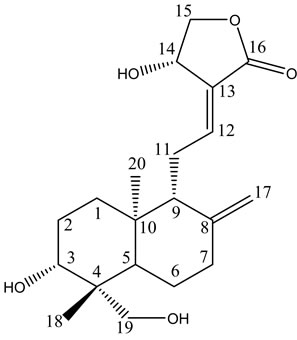
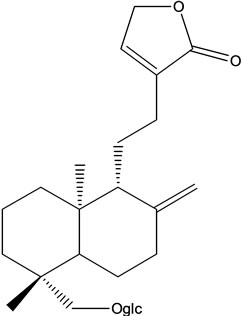
Andrographis paniculata Andrographolide Neoandrographolide
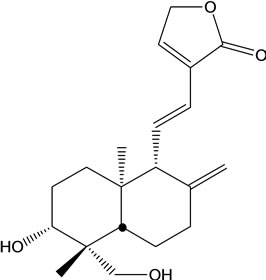
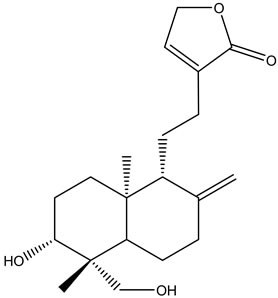
14-deoxy-11,12-didehydroandrographolide 14-deoxyandrographolide (14-DAG)
Figure 1. Chemical structures of the active compound for Andrographis paniculata.


Table 1. Anti-hepatotoxic mechanisms of A. paniculata and its bioactive compounds.
3. A. Paniculata and Pure Compounds Anti-Hepatotoxic Mechanisms (Table 1)
3.1. Against Carbontetrachloride (CCl4) and Tert-Butylhydroperoxide (t-BHP)
It is well known that CCl4 is converted by cytochrome P450 mixed function oxygenases in smooth endoplasmic reticulum of liver into toxic metabolite, mainly trichloromethyl radical (CCl3·). In the presence of oxygen this free radical can induce peroxidation of lipids on target cells resulting in extensive damage [21]. Choudhury and Poddar reported that oral administration of kalmegh leaf extract (500 mg/kg) or its bitter compound, andrographolide (5 mg/kg), to adult male albino rats produced no significant change in nicotinamide adenine dinucleotide phosphate (NDAPH)-induced hepatic microsomal lipid peroxidation. The data suggests that kalmegh leaf has greater protective effect on carbontetrachloride (5 ml/kg)- induced hepatic toxicity than andrographolide [22].
t-BHP has often been used as a model to investigate the mechanism of cell injury initiated by acute oxidative stress [23]. t-BHP can be metabolized to free radical intermediates by cytochrome P450 (in hepatocytes) or hemoglobin (in erythrocytes), which can subsequently initiate lipid peroxidation [24] and affect cell integrity. Pretreatment of mice with andrographolide, andrographiside and neoandrographolide (100 mg/kg, i.p.) reduced CCl4 or t-BHP-induced malondialdehyde (MDA) levels and release of GPT and ALP in the serum, as effective as the known hepatoprotective agent silymarin [25]. Oral treatment of rats with the A. paniculata methanol extract followed by CCl4 administration restored plasma lipid peroxidation, alanine transaminase (ALT) and aspartate transaminase (AST) levels [26]. Oral or i.p. pretreatment with andrographolide was also protective against galactosamine-induced liver damage in rats. On the other hand, protection was also observed when rats were treated with andrographolide post acetaminophen challenge and on an ex vivo preparation of isolated rat hepatocytes by increasing the viability of the hepatocytes after parcacetamol-induced toxicity [27,28]. Ye et al. demonstrated that andrographolide prevents acute liver injury induced by CCl4 via induction of heme oxygenase-1 (HO-1) and inhibition of an inflammatory response such as tumor necrosis factor-a (TNF-a) production in mice [29]. Oral administration of A. paniculata (100 - 200 mg/kg) exerted a significant dose dependent protection against paracetamol-induced hepatotoxicity in mice [30].
3.2. The Role of A. paniculata as CYPs Inducers
A crude extract of A. paniculata induce mouse hepatic cytochrome P450 isoforms CYP1A1 and CYP2B via increases in ethoxyresorufin O-dealkylase (EROD) and pentoxyresorufin O-dealkylase activities [31]. Andrographolide, a single substance extracted from A. paniculata, was further demonstrated to significantly up-regulate the CYP1- A1 and CYP1A2 mRNA expression, as did the CYP1A inducers such as benzanthracene and b-naphthoflavone [32]. Andrographolide significantly induced the expression of CYP1A1 and CYP1B1 mRNAs in a concentration-dependent manner, and also synergistically induced CYP1- A1 expression with the typical CYP1A inducers [33].
In addition, the A. paniculata 60% ethanol extract or andrographolide may cause herb-drug interactions through CYP3A and CYP2C9 inhibition in vitro or CYP2C11 inhibition in vivo [34,35]. A synergistically enhanced the expression of CYP1 family gene after andrographolide plus 3-methylcholanthrene (3-MC, a polycyclic aromatic hydrocarbon) treatment in male B6 mice. They demonstrated that a male hormone associated system to have a positive role in the synergistic effect [36]. 14-deoxy-11, 12-didehydroandrographolide and andrographolide have been shown to inhibit CYP1A2, CYP2D6 and CYP3A4 expressions in HepG2 cells [37]. A. paniculata ethanol and methanol extracts inhibited CYP3A4 and CYP2C9 activities more than aqueous and hexane extracts. On the other hand, andrographolide was found to weakly inhibit CYP3A4 activity [38]. Molecular docking analysis data supported that andrographolide and 14-deoxy-11,12- didehy-droandrographolide induced CYP1A1 expression or co-treatment with CYP1A1 inducer (b-naphthoflavone, BNF) showed a synergistic increase expression of CYP1A1. In contrast, neoandrographolide suppressed BNF induced CYP1A1 expression [39]. Qiu et al. study the herb-drug interactions in combination therapy. They demonstrated that andrographolide (1, 10, 100 mM) significantly down regulates the mRNA level and protein level of CYP3A4 in Caco-2 cells [40].
3.3. The Modulatory Effect on Glutathione (GSH) Content
GSH is abundant in liver cells and a major protective factor against oxidative stress. Andrographolide interaction with GSH significantly enhanced the BNF inducible CYP1A1 mRNA expression in C57BL/6 mouse hepatocytes [41]. Pan et al. reported that A. paniculata ethanol extract weakly inhibited CYP 2C19 activity [42].
3.4. Influence Glutathione S-Transferase (GSTP) Activity and Phosphatidylinostol-3-Kinase/Akt (PI3k/Akt) Pathway
The p class of GST (GSTP) belongs to the cytosolic class. GSTP is not expressed in healthy liver but it is increased in both chemically induced and spontaneously arising precancerous lesions and hepatomas in experimental carcinogenesis studies [3]. Induction of drug-metabolizing enzymes is considered to be an adaptive response to a cytotoxic environment. The A. paniculata ethanol extract, EtOAc extract and andrographolide induce the expression of GSTP, a phase II biotransformation enzymes involved in detoxification of various classes of environmental carcinogens, in rat primary hepatocytes [43]. Furthermore, they also demonstrated that andrographolide induced GSTP expression is mediated by the PI3K/Akt pathway in SD rat primary hepatocytes [44].
3.5. The Role in Apoptosis Pathway
The combination of andrographolide and anti-cancer drug 5-fluorouracil (5-FU) could enhance the apoptosis in hapatocellular carcinoma (SMMC-7721) cells through a caspase-8 dependent mitochondrial pathway involving p53, Bax, cytochrome c, caspase-9 and caspase-3 [45]. Copper accumulation within the hepatocyte results in oxidative stress and promotes apoptosis. One study demonstrated that andrographolide combined with D-penicillamine could decrease caspase-8 activation and upstream events of caspase-3 activation and inflammatory cytokines production for treatment of copper toxicosis [46]. Bile duct ligation (BDL) could induce hepatic fibrosis. The BDL SD rats treated with andrographolide (50, 100, 200 mg/kg, i.p.) significantly reduced serum ALT, AST, TNF-a and interleukin-1b (IL-1b) levels and Bax, cytochrome c [47]. Zhou et al. reported that andrographolide promoted anti-cancer drug doxorubicin-induced apoptosis in HepG2 cell, indicating that andrographolide enhances cancer cells to anticancer drug doxorubicin via signal transducers and activators of transcription-3 (STAT3) pathway suppression [48]. 14-deoxyandrographolide (14-DAG), a bioactive compound of A. paniculata, was shown to protective on TNF-a-mediated apoptosis. Pre-treatment of primary rat hepatocytes with 10 nM 14-DAG accentuated microsomal Ca-ATPase activity through induction of NO/cGMP pathway and desensitizes hepatocytes to TNF-a-mediated apoptosis through the release of tumor necrosis factor receptor 1 (TNFR1) [49]. In nude mice bearing xenografted Hep3B tumors, buthionine sulfoximine (BSO, an inhibitor of cellular GSH biosynthesis) improved the inhibition of tumor growth by andrographolide [50]. Concanavalin A (Con A)-induced hepatitis model was widely used for the investigation of immune mediated acute liver injury [51-53]. Shi and coworkers demonstrated that andrographolide attenuated Con A-induced liver injury through reducing oxidative stress and inhibited hepatocyte apoptosis [54].
4. Conclusion
The major compounds andrographolide, neoandrographolide, 14-deoxy-11,12-didehydroandro-grapholide and 14-deoxyandrographolide of A. paniculata are most well studied for both bioactivities and functional mechanisms. They exert anti-hepatotoxic actions, such as 1) inhibiting CCl4, t-BHP induced hepatic toxicity; 2) acting as CYPs inducers; 3) modulating GSH content; 4) influencing GSTP activity and PI3k/Akt pathway; 5) synergistic effects with anti-cancer drugs inducing apoptosis are suggested to be the bioactivities of A. paniculata extracts and isolated active compounds. With more in vitro and in vivo studies, the A. paniculata extracts and purified active compounds might be developed into potential anti-hepatotoxic drug that warrant further research and development in the future.
REFERENCES
- F. J. Gonzalez, “The Molecular Biology of Cytochrome P450s,” Pharmacological Reviews, Vol. 40, No. 4, 1988, pp. 243-288.
- P. K. Singha, S. Roy and S. Dey, “Protective Activity of Andrographolide and Arabinogalactan Proteins from Andrographis paniculata Nees. against Ethanol Induced Toxicity in Mice,” Journal of Ethnopharmacology, Vol. 111, No. 1, 2007, pp. 13-21. doi:10.1016/j.jep.2006.10.026
- J. D. Hayes, J. U. Flanagan and I. R. Jowsey, “Glutathione Transferases,” Annual Review of Pharmacology and Toxicology, Vol. 45, 2005, pp. 51-88. doi:10.1146/annurev.pharmtox.45.120403.095857
- M. Iwanari, M. Nakajima, R. Kizu, K. Hayakawa and T. Yokoi, “Induction of CYP1A1, CYP1A2 and CYP1B1 mRNAs by Nitropolycyclic Aromatic Hydrocarbons in Various Human Tissue Derived Cells: Chemical-, Cytochrome p450 Isoforms-, and Cell Specific Differences,” Archives of Toxicology, Vol. 76, No. 5-6, 2002, pp. 287- 298. doi:10.1007/s00204-002-0340-z
- P. J. Wedlund, “The CYP2C19 Enzyme Polymorphism,” Pharmacology, Vol. 61, No. 3, 2000, pp. 174-183. doi:10.1159/000028398
- T. Shimada, H. Yamazaki, M. Mimura, Y. Inui and F. P. Guengerich, “Interindividual Variations in Human Liver Cytochrome P450 Enzymes Involved in the Oxidation of Drugs, Carcinogens and Toxic Chemicals: Studies with Liver Microsomes of 30 Japanese and 30 Caucasians,” Journal of Pharmacology and Experimental Therapeutics, Vol. 270, No. 1, 1994, pp. 414-423.
- B. Kligler, C. Ulbricht, E. Basch, C. D. Kirkwood, T. R. Abrams, M. Miranda, K. P. Singh Khalsa, M. Giles, H. Boon and J. Woods, “Andrographis paniculata for the Treatment of Upper Respiratory Infection: A Systematic Review by the Natural Standard Research Collaboration,” Explore, Vol. 2, No. 1, 2006, pp. 25-29. doi:10.1016/j.explore.2005.08.008
- M. Roxas and J. Jurenka, “Colds and Influenza, a Review of Diagnosis and Conventional, Botanical and Nutritional Considerations,” Alternative Medicine Review, Vol. 12, No. 1, 2007, pp. 25-48.
- A. S. Negi, J. K. Kumar, S. Luqman, K. Sbanker, M. M. Gupta and S. P. S. Kbanuja, “Recent Advances in Plant Hepatoprotectives: A Chemical and Biological Profile of Some Important Leads,” Medicinal Research Reviews, Vol. 28, No. 5, 2008, pp. 746-772. doi:10.1002/med.20115
- D. D. Caceres, J. L. Hancke, R. A. Burgos and G. K. Wikman, “Prevention of Common Colds with Andrographis paniculata Dried Extract: A Pilot Doudle-Blind Trial,” Phytomedicine, Vol. 4, 1997, pp. 101-104.
- H. Y. Cheung , C. S. Cheung and C. K. Kong, “Determination of Bioactive Diterpenoids from Andrographis paniculata by Micellar Electrokinetic Chromatography,” Journal of Chromatography A, Vol. 930, No. 1-2, 2001, pp. 171-176. doi:10.1016/S0021-9673(01)01160-8
- T. Matsuda, M. Kuroyanagi, S. Sugiyama, K. Umehara, A. Ueno and K. Nishi, “Cell Differentiation Inducing Diterpenes from Andrographis paniculata Nees.,” Chemical & Pharmaceutical Bulletin, Vol. 42, No. 6, 1994, pp. 1216- 1225. doi:10.1248/cpb.42.1216
- N. Pholphana, N. Rangkadilok, S. Thongnest, S. Ruchirawat, M. Ruchirawat and J. Satayavivad, “Determination and Variation of Three Active Diterpenoids in Andrographis paniculata (Burm.f.) Nees,” Phytochemical Analysis, Vol. 15, No. 6, 2004, pp. 365-371. doi:10.1002/pca.789
- P. H. Kishore, M. V. Reddy, M. K. Reddy, D. Gunasekar, C. Caux and B. Bodo, “Flavonoids from Andrographis lineate,” Phytochemistry, Vol. 63, No. 4, 2003, pp. 457- 461. doi:10.1016/S0031-9422(02)00702-1
- J. Li, W. Huang, H. Zhang, X. Wang and H. Zhou, “Synthesis of Andrographolide Derivatives and Their TNFAlpha and IL-6 Expression Inhibitory Activities,” Bioorganic & Medicinal Chemistry Letters, Vol. 17, No. 24, 2007, pp. 6891-6894. doi:10.1016/j.bmcl.2007.10.009
- W. Parichatikanond, C. Suthisisang, P. Dhepakson and A. Herunsalee, “Study of Anti-Inflammatory Activities of the Pure Compounds from Andrographis paniculata (burm.f.) Nees and Their Effects on Gene Expression,” International Immunopharmacology, Vol. 10, No. 11, 2010, pp. 1361-1373. doi:10.1016/j.intimp.2010.08.002
- W. W. Chao, Y. H. Kuo, W. C. Li and B. F. Lin, “The Production of Nitric Oxide and Prostaglandin E2 in Peritoneal Macrophages Is Inhibited by Morus alba, Angelica sinensis and Andrographis paniculata Ethyl Acetate Fraction Extracts,” Journal of Ethnopharmacology, Vol. 122, No. 1, 2009, pp. 68-75. doi:10.1016/j.jep.2008.11.029
- W. W. Chao, Y. H. Kuo, S. L. Hsieh and B. F. Lin, “Inhibitory Effects of Ethyl Acetate Extract of Andrographis paniculata on NF-κB Trans-Activation Activity and LPSInduced Acute Inflammation in Mice,” eCAM, Vol. 2011, 2011, p. 254531.
- W. W. Chao, Y. H. Kuo and B. F. Lin, “Anti-Inflammatory Activity of New Compounds from Andrographis paniculata by NF-kB Trans-Activation Inhibition,” Journal of Agricultural and Food Chemistry, Vol. 58, No. 4, 2010, pp. 2505-2512. doi:10.1021/jf903629j
- W. W. Chao and B. F. Lin, “Isolation and Identification of Bioactive Compounds in Andrographis paniculata (Chuanxinlian),” Chinese Medicine, Vol. 5, 2010, p. 17. doi:10.1186/1749-8546-5-17
- R. O. Recknagel, “Carbon Tetrachloride Hepatotoxicity: Status Quo and Future Prospects,” Trends in Pharmacological Sciences Vol. 4, 1983, pp. 129-131. doi:10.1016/0165-6147(83)90328-0
- B. R. Choudhury and M. K. Poddar, “Andrographolide and Kalmegh (Andrographis paniculata) Extract: In Vivo and in Vitro Effect on Hepatic Lipid Peroxidation,” Methods & Findings in Experimental & Clinical Pharmacology, Vol. 6, No. 9, 1984, pp. 481-485.
- G. F. Rush, J. R. Gorski, M. G. Ripple, J. Sowinski, P. Bugelski and W. R. Hewitt, “Organic Hydroperoxide-Induced Lipid Peroxidation and Cell Death in Isolated Hepatocytes,” Toxicology and Applied Pharmacology, Vol. 78, No. 3, 1985, pp. 473-483. doi:10.1016/0041-008X(85)90255-8
- J. Hogberg, S. Orrenius and P. J. O’Brien, “Further Studies on Lipid-Peroxide Formation in Isolated Hepatocytes,” European Journal of Biochemistry, Vol. 59, No. 2, 1975, pp. 449-455. doi:10.1111/j.1432-1033.1975.tb02473.x
- A. Kapil, I. B. Koul, S. K. Banerjee and B. D. Gupta, “Antihepatotoxic Effects of Major Diterpenoid Constituents of Andrographis paniculata,” Biochemical Pharmacology Vol. 46, No. 1, 1993, pp. 182-185. doi:10.1016/0006-2952(93)90364-3
- G. A. Akowuah, I. Zhari, A. Mariam and M. F. Yam, “Absorption of Andrographolides from Andrographis paniculata and Its Effect on CCl4-Induced Oxidative Stress in Rats,” Food and Chemical Toxicology, Vol. 47, No. 9, 2009, pp. 2321-2326. doi:10.1016/j.fct.2009.06.022
- S. S. Handa and A. Sharma, “Hepatoprotective Activity of Andrographolide against Galactosamine and Paracetamol Intoxication in Rats,” Indian Council of Medical Research, Vol. 92, 1990, pp. 284-292.
- P. K. Visen, B. Shukla, G. K. Patnaik and B. N. Dhawan, “Andrographolide Protects Rat Hepatocytes against Paracetamol Induced Gamage,” Journal of Ethnopharmacology, Vol. 40, No. 2, 1993, pp. 131-136. doi:10.1016/0378-8741(93)90058-D
- J. F. Ye, H. Zhu, Z. F. Zhou, R. B. Xiong, X. W. Wang, L. X. Su and B. D. Luo, “Protective Mechanism of Andrographolide against Carbon Tetrachloride Induced Acute Liver Injury in Mice,” Biological and Pharmaceutical Bulletin, Vol. 34, No. 11, 2011, pp. 1666-1670. doi:10.1248/bpb.34.1666
- R. Nagalekshmi, A. Menon, D. K. Chandrasekharan and C. K. K. Nair, “Hepatoprotective Activity of Andrographis paniculata and Swertia Chirayita,” Food and Chemical Toxicology, Vol. 49, No. 12, 2011, pp. 3367-3373. doi:10.1016/j.fct.2011.09.026
- K. Jarukamjorn, K. Don-in, C. Makejaruskul, T. Laha, S. Daodee, P. Pearaksa and B. Sripanidkulchai, “Impact of Andrographis paniculata Crude Extract on Mouse Hepatic Cytochrome P450 Enzymes,” Journal of Ethnopharmacology, Vol. 105, No. 3, 2006, pp. 464-467. doi:10.1016/j.jep.2005.11.024
- A. Jaruchotikamol, J. Kanokwan, S. T. S. Wanna, K. Yuki and N. Nobuo, “Strong Synergistic Induction of CYP1A1 Expression by Andrographolide plus Typical CYP1A Inducers in Mouse Hepatocytes,” Toxicology and Applied Pharmacology, Vol. 224, No. 2, 2007, pp. 156-162. doi:10.1016/j.taap.2007.07.008
- W. Chatuphonprasert, K. Jarukamjorn, S. Kondo and N. Nemoto, “Synergistic Increases of Metabolism and Oxidation Reduction Genes on Their Expression after Combined Treatment with a CYP1A Inducer and Andrographolide,” Chemico-Biological Interactions, Vol. 182, No. 2-3, 2009, pp. 233-238. doi:10.1016/j.cbi.2009.09.001
- D. Pekthong, H. Martin, C. Abadie, A. Bonet, B. Heyd, G. Mantion and L. Richert, “Differential Inhibition of Rat and Human Hepatic Cytochrome P450 by Andrographis paniculata Extract and Andrographolide,” Journal of Ethnopharmacology, Vol. 115, No. 3, 2008, pp. 432-440. doi:10.1016/j.jep.2007.10.013
- D. Pekthong, N. Blanchard, C. Abadie, A. Bonet, B. Heyd, G. Mantion, A. Berthelot, L. Richert and H. Martin, “Effects of Andrographis paniculata Extract and Andrographolide on Hepatic Cytochrome P450 mRNA Expression and Monooxygenase Activities after in Vivo Administration to Rats and in Vitro in Rat and Human Hepatocyte Cultures,” Chemico-Biological Interactions, Vol. 179, No. 2-3, 2009, pp. 247-255.
- K. Jarukamjorn, S. Kondo, W. Chatuphonprasert, T. Sakuma, Y. Kawasaki and N. Nemoto, “Gender-Associated Modulation of Ineucible CYP1A1 Expression by Andrographolide in Mouse Liver,” European Journal of Pharmaceutical Sciences, Vol. 39, No. 5, 2010, pp. 394-401. doi:10.1016/j.ejps.2010.01.009
- J. P. Ooi, M. Kuroyanagi, S. F. Sulaiman, T. S. T. Muhammad and M. L. Tan, “Andrographolide and 14-Deoxy-11, 12-Didehydroandrographolide Inhibit Cytochtome P450s in HepG2 Hepatoma Cells,” Life Sciences, Vol. 88, No. 9-10, 2011, pp. 447-454. doi:10.1016/j.lfs.2010.12.019
- Y. Pan, B. A. Abd-Rashid, Z. Ismail, R. Ismail, J. W. Mak, P. C. K. Pook, H. M. Er and C. E. Ong, “In Vitro Determination of the Effect of Andrographis Paniculata Extracts and Andrographolide on Human Hepatic Cytochrome P450 Activities,” Journal of Natural Medicines, Vol. 65, No. 3-4, 2011, pp. 440-447. doi:10.1007/s11418-011-0516-z
- W. Chatuphonprasert, T. Remsungnen, N. Nemoto and K. Jarukamjorn, “Different AhR Binding Sites of Diterpenoid Ligands from Andrographis Paniculata Caused Differential CYP1A1 Induction in Primary Culture in Mouse Hepatocytes,” Toxicology in Vitro, Vol. 25, No. 8, 2011, pp. 1757-1763. doi:10.1016/j.tiv.2011.09.004
- F. Qiu, X. L. Hou, K. Takahashi, L. X. Chen, J. Azuma and N. Kang, “Andrographolide Inhibits the Expression and Metabolic Activity of Cytochrome P450 3A4 in the Modified Caco-2 Cells,” Journal of Ethnopharmacology, Vol. 141, No. 2, 2012, pp. 709-713. doi:10.1016/j.jep.2011.09.002
- S. Kondo, W. Chatuphonprasert, A. Jaruchotikamol, T. Sakuma and N. Nemoto, “Cellular Glutathione Content Modulates the Effect of Andrographolide on b-Naphtho-Flavone Induced CYP1A1 mRNA Expression in Mouse Hepatocytes,” Toxicology, Vol. 280, No. 1-2, 2011, pp. 18- 23. doi:10.1016/j.tox.2010.11.002
- Y. Pan, B. A. Abd-Rashid, Z. Ismail, R. Ismail, J. W. Mak, P. C. K. Pook, H. M. Er and C. E. Ong, “In Vitro Modulatory Effects of Andrographis paniculata, Centella asiatica and Orthosiphon stamineus on Cytochrome P450 2C19 (CYP2C19),” Journal of Ethnopharmacology, Vol. 133, No. 2, 2011, pp. 881-887. doi:10.1016/j.jep.2010.11.026
- K. T. Chang, C. K. Lii, C. W. Tsai, A. J. Yang and H. W. Chen, “Modulation of the Expression of the p Class of Glutathione S-Transferase by Andrographis paniculata Extracts and Andrographolide,” Food and Chemical Toxicology, Vol. 46, No. 3, 2008, pp. 1079-1088. doi:10.1016/j.fct.2007.11.002
- C. Y. Lu, C. C. Li, C. K. Lii, H. T. Yao, K. L. Liu, C. W. Tsai and H. W. Chen, “Andrographolide Induced Pi Class of Glutathione S-Transferase Gene Expression via PI3K/ AKt Pathway in Rat Primary Hepatocytes,” Food and Chemical Toxicology, Vol. 49, No. 1, 2011, pp. 281-289. doi:10.1016/j.fct.2010.10.030
- L. Yang, D. Wu, K. Luo, S. Wu and P. Wu, “Andrographolide Enhances 5-Fluorouracil-Induced Apoptosis via Caspase-8 Dependent Mitochondrial Pathway Involving p53 Participation in Hepatocellular Carcinoma (SMMC- 7721) Cells,” Cancer Letters, Vol. 276, No. 2, 2009, pp. 180-188. doi:10.1016/j.canlet.2008.11.015
- D. N. Roy, G. Sen, K. D. Chowdhury and T. Biswas, “Combination Therapy with Andrographolide and D-Penicillamine Enhanced Therapeutic Advantage over Monotherapy with D-Penicillamine in Attenuating Fibrogenic Response and Cell Death in the Periportal Zone of Liver in Rats during Copper Toxicosis,” Toxicology and Applied Pharmacology, Vol. 250, No. 1, 2011, pp. 54-68. doi:10.1016/j.taap.2010.09.027
- T. Y. Lee, K. C. Lee and H. H. Chang, “Modulation of the Cannabinoid Receptors by Andrographolide Attenuates Hepatic Apoptosis Following Bile Duct Ligation in Rats with Fibrosis,” Apoptosis, Vol. 15, No. 8, 2010, pp. 904-914. doi:10.1007/s10495-010-0502-z
- J. Zhou, C. N. Ong, G. M. Hur and H. M. Shen, “Inhibition of the JAK-STAT3 Pathway by Andrographolide Enhances Chemosensitivity of Cancer Cells to Doxorubicin,” Biochemical Pharmacology, Vol. 79, No. 9, 2010, pp. 1242-1250. doi:10.1016/j.bcp.2009.12.014
- D. N. Roy, S. Mandal, G. Sen, S. Mukhopadhyay and T. Biswas, “14-Deoxyandrographolide Desensitizes Hepatocytes to Tumour Necrosis Factor-Alpha Induced Apoptosis through Calcium Dependent Tumour Necrosis Factor Receptor Superfamily Member 1A Release via the NO/cGMP Pathway,” British Journal of Pharmacology, Vol. 160, No. 7, 2010, pp. 1823-1843. doi:10.1111/j.1476-5381.2010.00836.x
- L. Ji, K. Shen, P. Jiang, G. Morahan and Z. Wang, “Critical Roles of Cellular Glutathione Homeostasis and JNK Activation in Andrographolide Mediated Apoptotic Cell Death in Human Hepatoma Cells,” Molecular Carcinogenesis, Vol. 50, No. 8, 2011, pp. 580-591. doi:10.1002/mc.20741
- G. Tiegs, J. Hentschel and A. Wendel, “A T Cell Dependent Experimental Liver Injury in Mice Inducible by Concanavalin A,” The Journal of Clinical Investigation, Vol. 90, No. 1, 1992, pp. 196-203. doi:10.1172/JCI115836
- Z. L. Wang, X. H. Wu, L. F. Song, Y. S. Wang, X. H. Hu, Y. F. Luo, Z. Z. Chen, J. Ke, X. D. Peng, C. M. He, W. Zhang, L. J. Chen and Y. Q. Wei, “Phosphoinostide 3 Kinase Gamma Inhibitor Ameliorates Concanavalin A Induced Hepatic Injury in Mice,” Biochemical and Biophysical Research Communications, Vol. 386, No. 4, 2009, pp. 569-574. doi:10.1016/j.bbrc.2009.06.060
- D. H. Adams, C. Ju, S. K. Ramaiah, J. Uetrecht and H. Jaeschke, “Mechanisms of Immune Mediated Liver Injury,” Toxicological Sciences, Vol. 115, No. 2, 2010, pp. 307-321. doi:10.1093/toxsci/kfq009
- G. Shi, Z. Zhang, R. Zhang, X. Zhang, Y. Lu, J. Yang, D. Zhang, Z. Zhang, X. Li and G. Ning, “Protective Effect of Andrographolide against concanavalin A Induced Liver Injury,” Naunyn-Schmiedeberg’s Archives of Pharmacology, Vol. 385, No. 1, 2012, pp. 69-79. doi:10.1007/s00210-011-0685-z
Abbreviations
CYPs: cytochrome P450 enzymes;
GPT: glutamate pyruvate transaminase;
GOT: glutamate oxaloacetate transaminase;
ALP: alkaline phosphatase;
ACP: acid phosphatase;
GST: Glutathione S-transferase;
PAHs: polycyclic aromatic hydrocarbons;
CCl4: carbontetrachloride;
tBHP: tert-butylhydroperoxide;
NDAPH: nitotinamide adenine dinucleotide phosphate;
MDA: malondialdehyde;
ALT: alanine transaminase;
AST: aspartate transaminase;
HO-1: heme oxygenase-1;
TNF-a: tumor necrosis factor-a;
3-MC: 3-methylcholanthrene;
BNF: b-naphthoflavone;
GSH: glutathione;
GSTP: glutathione S-transferase;
PI3k/Akt: phosphatidylinositol-3-kinase/Akt;
5-FU: 5-fluorouracil;
BDL: Bile duct ligation;
IL-1b: interleukin-1b;
STAT3: signal transducers and activators of transcription-3;
14-DAG: 14-deoxyan-drographolide;
TNFR1: tumor necrosis factor receptor 1;
BSO: buthionine sulfoximine;
Con A: concanavalin A
NOTES
*WWC and BFL searched the literature and drafted the manuscript. All authors read and approved the final version of the manuscript.
#Corresponding author.

