Health
Vol.5 No.6A1(2013), Article ID:33133,6 pages DOI:10.4236/health.2013.56A1005
Mother’s intrahepatic cholestasis does not affect her daughter’s health*
![]()
1Department of General Practice, School of Medicine, University of Tampere, Tampere, Finland; #Corresponding Author: tiina.vimpeli@uta.fi
2Centre for General Practice, Pirkanmaa Hospital District, Tampere, Finland
Copyright © 2013 Tiina Vimpeli et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received 8 March 2013; revised 9 April 2013; accepted 1 May 2013
Keywords: Intrahepatic Cholestasis of Pregnancy; Inheritance; Epilepsy; Maternity Clinics; Primary Health Care
ABSTRACT
Objective: To establish whether a mother’s intrahepatic cholestasis of pregnancy (ICP) has connections to her daughter’s health. Design: A retrospective study of daughters to ICP mothers. Setting: The region of Tampere University Hospital in Finland. Subjects: The study population comprised 575 women diagnosed with ICP during at least one pregnancy in the obstetric department of Tampere University Hospital in the period 1969-1988, and two controls chosen for each. Questionnaires were sent to these women and to their daughters: 305 daughters to ICP mothers and 642 to controls in autumn 2010. Eventually 187 daughters to ICP mothers responded (61.3%) and 373 to controls (58.1%). Main Outcome Measures: Evaluated health, symptoms and complaints, diagnosed diseases, mental health and use of medicines. Results: Only minor differences were detected between the two groups regarding the majority of items inquired. The only prominent difference between daughters to ICP mothers and those to controls concerned epilepsy, which was significantly more frequent among ICP mothers’ daughters, the prevalence being fourfold (3.2% vs 0.8%, p = 0.033). Conclusion: A mother’s ICP does not generally affect her daughter’s health. This can be considered an encouraging new finding for mothers with ICP in primary health care.
1. INTRODUCTION
Intrahepatic cholestasis of pregnancy (ICP) is a liver disorder which usually manifests in the third trimester of gestation as skin itching especially on the palms and soles and as increased serum bile acids and transaminases [1]. Shortly after delivery the itching attenuates and abnormal laboratory values normalize, but the disease nonetheless recurs in future pregnancies in about 40% - 60% of patients [2]. The incidence of ICP varies geographically; in Finland and in Europe in general the figure is approximately 1% of pregnancies [3].
The ultimate cause of ICP remains unknown but the condition appears to be related to increased sex hormone synthesis and metabolism during pregnancy [1,4]. In addition to this hormonal component there would also appear to be heritable and environmental factors [5-7]. ICP is more common in multiple pregnancies [8] and with a positive family history [9]. Advanced maternal age has been identified as a risk factor [10]. Selenium deficiency may be implicated in the pathogenesis of ICP [11].
ICP has no significant maternal effects during pregnancy (albeit itching can be intensive), but may have severe consequences for the fetus, including fetal distress, premature deliveries and intrauterine fetal death [1,12]. Active management such as early delivery by induced labor at 37 - 38 weeks gestation has been commonly recommended to prevent especially intrauterine fetal death [13]. Although delivery often occurs at earlier weeks of gestation, the newborns’ Apgar scores are only slightly lower than normally [14].
In the Nordic countries, maternity care is organized in primary health care [15]. Serum aminotransferase levels are routinely measured for early detection of ICP if a pregnant woman has experienced itching, has a history of ICP or in cases of a multiple pregnancy [9]. In Finnish maternity care ICP is generally detected and appropriately managed, being thus only a minor risk for mothers and their children, good pregnancy outcome normally being attained [14].
A positive correlation has been discovered between the severity of ICP and fetal complication rates; when maternal serum bile acids exceed 40 µmol/L, the incidence of fetal complications begins to increase significantly [16]. Currently the best pharmacologic treatment for the condition seems to be ursodeoxycholic acid (UDCA) [17]. In severe ICP, treatment with UDCA improves all biochemical markers of cholestasis in the mother, itching included. Dexamethasone is also sometimes used. However, it remains unknown whether fetal complications could be reduced by pharmacologic treatment.
Approximately one out of six ICP patients has a positive family history, and in such cases the levels of serum transaminases are higher than in sporadic cases and the recurrence rate in subsequent pregnancies is 92% [9]. In addition, the widely varying prevalence of ICP between different countries and ethnic groups [3,13] indicates a genetic basis for the disease.
ICP would appear to have some long-term effects on women’s health. There is an increased occurrence of a number of liver, biliary and pancreatic diseases among women with a history of ICP [18]. In addition, a higher frequency of breast cancer and hypothyreosis has recently been discovered [19]. Conversely, the frequency of hypertension, high cholesterol and cardiac arrhythmia is lower.
It has been proposed that the dominant mode of inheritance of ICP is either autosomal or X-chromosomelinked [20-22]. Several different cholestatic genes are evidently associated with the pathogenesis [23-25]. In view of the genetic predisposition to the disease, the aim of this study was to investigate whether a mother’s ICP has connections to her daughter’s health.
2. MATERIAL AND METHODS
To assess the health history of women who had suffered from ICP, a postal survey was conducted among ICP patients and control women in autumn 2010. The cohort comprised 575 women who had been diagnosed with ICP during at least one pregnancy in the obstetric department of Tampere University Hospital in the period 1969-1988, and two control women chosen for each [19].
At the same time, a slightly different questionnaire was sent to the daughters of these women. Postal addresses were obtained from the Population Register Centre in Finland. After excluding 11 stillborn and seven deceased daughters as well as 60 whose addresses could not be obtained, the questionnaire was sent to 947 daughters (305 to ICP and 642 to control mothers’ daughters); 187 daughters to ICP mothers (61.3%) and 373 to controls (58.1%) responded (Figure 1).
While categorizing the respondents’ age, 30 years was set as a cut-off point. Education was classified as “high” for those who had taken the matricular examination (gymnasium/grammar school) and “low” for those who had not. Concerning respondents’ BMI, 25 kg/m² was set as a cut-off point. With respect to age, educational level and body mass index (BMI) the two groups were comparable (Table 1).
The two groups received identical questionnaires. The questionnaire comprised 65 items and the main aspects pertinent to this study were present health, symptoms and complaints, diseases diagnosed by a doctor, use of medicines and mental health.
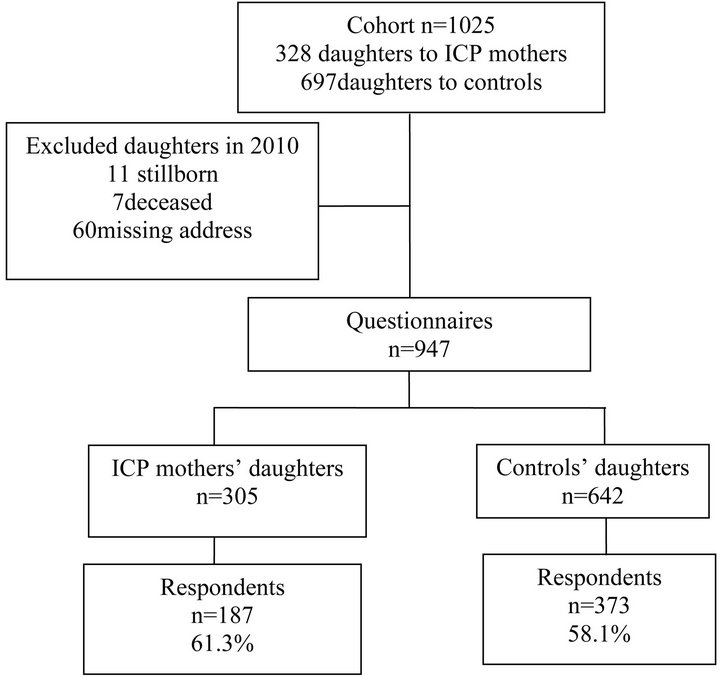
Figure 1. Flow chart of the survey population.
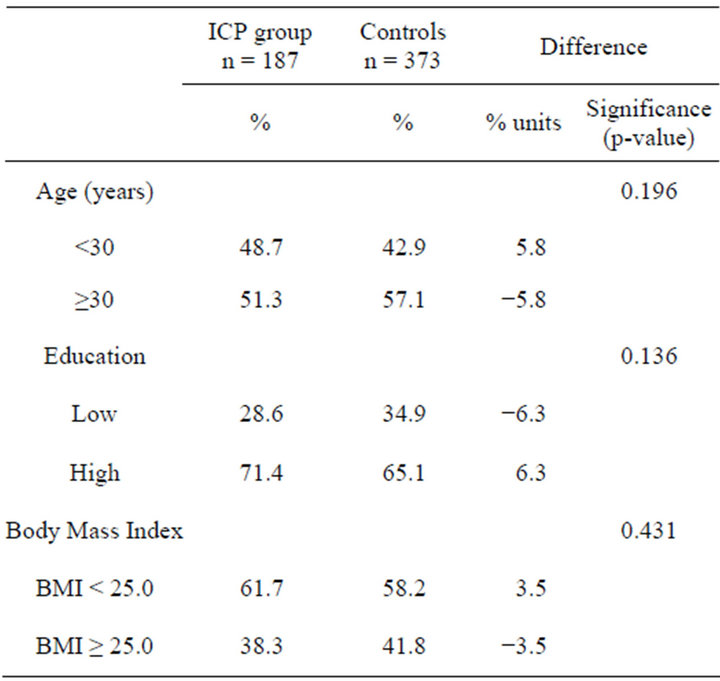
Table 1. Characteristics of daughters to ICP mothers and to controls.
Respondents were asked to evaluate their current health by choosing one of the following alternatives: good, fairly good, moderate, fairly poor and poor [19]. They were also asked whether any of 24 common symptoms or complaints had bothered them during the past year. Further, they were asked whether a doctor had ever diagnosed any of the diseases listed in the questionnaire, including gastrointestinal, endocrinological, urological, gynecological, oncological, boneand articulation-related, respiratory and cardiovascular diseases. A few separate disorders were also listed, for example migraine, epilepsy and anemia. Respondents were further asked whether they had ever undergone any major medical surgery, and a history of hip, wrist and vertebral fractures was inquired after.
Respondents’ mental health was estimated by two mental health-related questions and the Depression Scale. The two questions were devised to establish whether a respondent had ever suffered from a mental disorder and been treated for a mental disorder. The Depression Scale is a validated Finnish test screening for the risk of present clinical depression [26].
Use of medicines, natural health drugs as well as vitamins and trace elements during the last 12 months were inquired after. Three options were given for the mode of use: non-use, occasional use and regular use.
Statistical analyses were made using the SPSS System for Windows, release 20.0. Results are presented as frequencies and percentages. Statistical significance was tested by the chi-squared test.
3. RESULTS
The two groups did not differ in respect of respondents’ own evaluation of their current health status. Of ICP mothers’ daughters 87.1% and of controls’ daughters 86.0% assessed their health as good or fairly good. Symptoms and complaints are shown in Table 2 and diagnosed diseases in Table 3.
There were only minor differences between the two
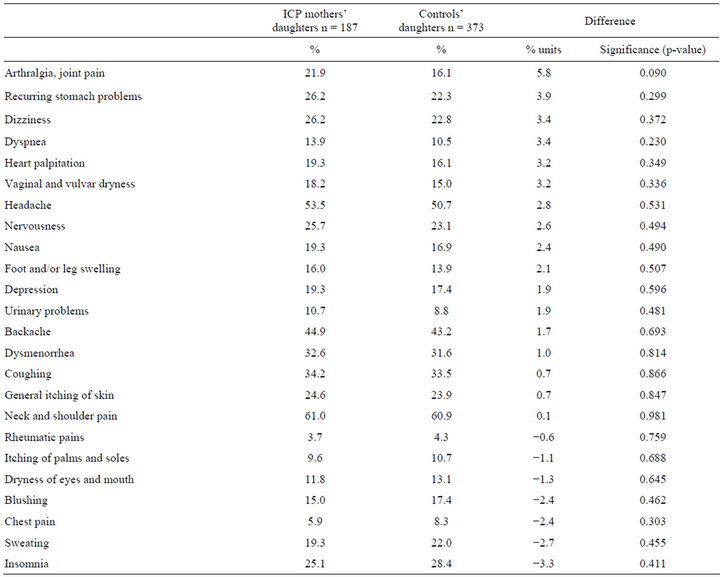
Table 2. Symptoms and complaints during the past 12 months among daughters to ICP mothers and to controls.

Table 3. Diseases diagnosed by a doctor among daughters to ICP mothers and to controls.
groups. Chronic choledochitis, cholelithiasis, rise in liver function test results (other than during pregnancy), acute hepatitis and fatty liver were all more common among daughters to ICP mothers. Hypothyreosis and goiter were likewise more frequent among ICP mothers’ daughters. Polycystic ovary syndrome was more frequent among ICP mothers’ daughters, whereas endometriosis was less frequent. Cancers other than breast or gynecologic were more common among daughters to ICP mothers than among the controls’ daughters. None of these differences were statistically significant. Concerning bone fractures, mental health, medication allergies and use of medicines there were no differences between the two groups, either.
A prominent difference between daughters of ICP mothers and those of controls concerned epilepsy, which was significantly more frequent among ICP mothers’ daughters, the prevalence being fourfold (3.2% vs 0.8%, p = 0.033).
4. DISCUSSION
There were only minor differences between the two groups regarding most of the survey questions. The main finding was a higher frequency of epilepsy among daughters to ICP mothers, the difference being fourfold.
Postal addresses were found for the majority of the study population. Response rates did not differ markedly between the groups (61.3% and 58.1%) and they were relatively high for a postal survey. Data were collected by a questionnaire to be filled in at home. As most of the respondents were relatively young, a better response rate might have been achieved by an online survey.
Since we only studied daughters we cannot generalize the findings to apply also to the sons of ICP mothers. However, the majority of the diseases observed in this study were not gender-dependent. Antenatal corticosteroids are sometimes administered in ICP [17], and also to women expected to deliver preterm because they reduce the newborn’s respiratory and cardiac problems [27]. However, corticosteroids have increased the risk of epilepsy especially among male infants. Neuro-sensory development seems to be more sensitive to external factors in the fetal life of boys than of girls.
Although higher frequencies of several liver, biliary and pancreatic diseases, breast cancer and hypothyreosis have been discovered among women with a history of ICP, this was not the case regarding their daughters. However, by reason of the considerable age difference between mothers and daughters at the time of the postal survey, the two groups cannot be compared with each other as to the incidence of the above-mentioned conditions. On the other hand, at least hypothyreosis often emerges at a relatively young age.
It may be speculated that epilepsy can be the result of fetal distress or premature delivery, though there are contradictory study results concerning such a hypothesis. Recently it has been noted that very premature birth and perinatal distress have associations with epilepsy [28] and that preterm infants have higher rates of seizures than full-term infants [29]. Conversely, it has been also claimed that low birth weight and fetal distress do not cause epilepsy [30].
To our knowledge this is the first study to explore the association between mothers’ ICP and their daughters’ health in later life. Based on our findings a mother’s ICP will not affect her daughter’s health. It has recently been reported that women with a history of ICP have, in most cases unnecessarily, limited their number of children for health reasons more often than women without ICP, and they also more often have a single child compared to controls [31].
Pregnancy follow-up and screening ICP takes place at maternity clinics in primary health care. Based on the findings here, our conclusion is that healthcare professionals can reassure mothers with ICP that the condition does not generally have any long-term adverse effects on their daughters’ health in later life.
REFERENCES
- Reyes, H. (1997) Intrahepatic cholestasis: A puzzling disorder of pregnancy. Journal of Gastroenterology and Hepatology, 12, 211-216. doi:10.1111/j.1440-1746.1997.tb00410.x
- Reyes, H. and Sjövall, J. (2000) Bile acids and progesterone metabolites in intrahepatic cholestasis of pregnancy. Annals of Medicine, 32, 94-106. doi:10.3109/07853890009011758
- Geenes, V. and Williamson, C. (2009) Intrahepatic cholestasis of pregnancy. World Journal of Gastroenterology, 15, 2049-2066. doi:10.3748/wjg.15.2049
- Reyes, H. (2008) Sex hormones and bile acids in intrahepatic cholestasis of pregnancy. Hepatology, 47, 376- 379. doi:10.1002/hep.22139
- Arrese, M. and Reyes, H. (2006) Intrahepatic cholestasis of pregnancy: A past and present riddle. Annals of Hepatology, 5, 202-205. http://www.medigraphic.com/pdfs/hepato/ah-2006/ah063r.pdf
- Hay, J. and Eileen, E. (2008) Liver disease in pregnancy. Hepatology, 47, 1067-1076. doi:10.1002/hep.22130
- Lammert, F., Marschall, H.U., Glantz, A. and Matern, S. (2000) Intrahepatic cholestasis of pregnancy: Molecular pathogenesis, diagnosis and management. Journal of Hepatology, 33, 1012-1021. doi:10.1016/S0168-8278(00)80139-7
- Gonzalez, M.C., Reyes, H., Arrese, M., Figueroa, D., Lorca, B., Andresen, M., et al. (1989) Intrahepatic cholestasis of pregnancy in twin pregnancies. Journal of Hepatology, 9, 84-90. doi:10.1016/0168-8278(89)90079-2
- Savander, M., Ropponen, A., Avela, K., Weerasekera, N., Cormand, B., Hirvioja, M.-L., et al. (2003) Genetic evidence of heterogeneity in intrahepatic cholestasis of pregnancy. Gut, 52, 1025-1029. doi:10.1136/gut.52.7.1025
- Heinonen, S. and Kirkinen, P. (1999) Pregnancy outcome with intrahepatic cholestasis. Obstetrics & Gynecology, 94, 189-193. doi:10.1016/S0029-7844(99)00254-9
- Kauppila, A., Korpela, H., Mäkilä, U.M. and Yrjänheikki, E. (1987) Low serum selenium concentration and glutathione peroxidase activity in intrahepatic cholestasis of pregnancy. British Medical Journal, 294, 150-152. doi:10.1136/bmj.294.6565.150
- Rioseco, A.J., Ivankovic, M.B., Manzur, A., Hamed, F., Kato, S.R., Parer, J.T., et al. (1994) Intrahepatic cholestasis of pregnancy: A retrospective case-control study of perinatal outcome. American Journal of Obstetrics & Gynecology, 170, 890-895. doi:10.1016/S0002-9378(94)70304-3
- Bacq, Y. (2011) Liver diseases unique to pregnancy: A 2010 update. Clinics and Research in Hepatology and Gastroenterology, 35, 182-193. doi:10.1016/j.clinre.2010.11.011
- Turunen, K., Sumanen, M., Haukilahti, R.L., Kirkinen, P. and Mattila, K. (2010) Good pregnancy outcome despite intrahepatic cholestasis. Scandinavian Journal of Primary Health Care, 28, 102-107. doi:10.3109/02813431003784001
- Sigurdsson, J. (2003) The GP’s role in maternity care. Scandinavian Journal of Primary Health Care, 21, 65. doi:10.1080/02813430310000302302
- Glantz, A., Marschall, H.U. and Mattsson, L.Å. (2004) Intrahepatic cholestasis of pregnancy: Relationships between bile acid levels and fetal complication rates. Hepatology, 40, 467-474. doi:10.1002/hep.20336
- Glantz, A., Marschall, H.U., Lammert, F. and Mattsson, L.Å. (2005) Intrahepatic cholestasis of pregnancy: A randomized controlled trial comparing dexamethasone and ursodeoxycholic acid. Hepatology, 42, 1399-1405. doi:10.1002/hep.20952
- Ropponen, A., Sund, R., Riikonen, S., Ylikorkala, O. and Aittomäki, K. (2006) Intrahepatic cholestasis of pregnancy as an indicator of liver and biliary diseases: A population-based study. Hepatology, 43, 723-728. doi:10.1002/hep.21111
- Turunen, K., Mölsä, A., Helander, K., Sumanen, M. and Mattila, K.J. (2012) Health history after intrahepatic cholestasis of pregnancy. Acta Obstetricia et Gynecologica Scandinavica, 91, 679-685. doi:10.1111/j.1600-0412.2012.01403.x
- Holzbach, R.T., Sivac, D.A. and Braun, W.E. (1983) Familial recurrent intrahepatic cholestasis of pregnancy: A genetic study providing evidence for transmission of a sex-limited, dominant trait. Gastroenterology, 85, 175- 179. http://www.ncbi.nlm.nih.gov/pubmed/6852450
- Reyes, H., Ribalta, J. and Gonzales-Ceron, M. (1976) Idiopathic cholestasis of pregnancy in a large kindred. Gut, 17, 709-713. doi:10.1136/gut.17.9.709
- Hirvioja, M.-L. and Kivinen, S. (1993) Inheritance of intrahepatic cholestasis of pregnancy in one kindred. Clinical Genetics, 43, 315-317. doi:10.1111/j.1399-0004.1993.tb03826.x
- Pauli-Magnus, C., Lang, T. and Meier, Y. (2004) Sequence analysis of bile salt export pump (ABCB11) and multidrug resistance p-glycoprotein 3 (ABCB4, MDR3) in patients with intrahepatic cholestasis of pregnancy. Pharmacogenetics, 14, 91-102. doi:10.1097/00008571-200402000-00003
- Eloranta, M.-L., Häkli, T., Hiltunen, M., Helisalmi, S., Punnonen, K. and Heinonen, S. (2003) Association of single nucleotide polymorphisms of the bile salt export pump gene with intrahepatic cholestasis of pregnancy. Scandinavian Journal of Gastroenterology, 38, 648-652. doi:10.1080/00365520310000807
- Müllenbach, R., Bennett, A., Tetlow, N. and Patel, N. (2005) ATP8B1 mutations in British cases with intrahepatic cholestasis of pregnancy. Gut, 54, 829-834. doi:10.1136/gut.2004.058115
- Salokangas, R.K., Poutanen, O. and Stengard, E. (1995) Screening for depression in primary care. Development and validation of the depression scale, a screening instrument for depression. Acta Psychiatrica Scandinavica, 92, 10-16. doi:10.1111/j.1600-0447.1995.tb09536.x
- Eriksson, L., Haglund, B., Ewald, U., Odlind, V. and Kieler, H. (2009) Short and long-term effects of antenatal corticosteroids assessed in a cohort of 7827 children born preterm. Acta Obstetricia et Gynecologica Scandinavica, 88, 933-938. doi:10.1080/00016340903111542
- Sellier, E., Uldall, P., Calado, E., Sigurdardottir, S., Torrioli, M.G., Platt, M.J., et al. (2012) Epilepsy and cerebral palsy: Characteristics and trends in children born in 1976- 1998. European Journal of Paediatric Neurology, 16, 48- 55. doi:10.1016/j.ejpn.2011.10.003
- Saigal, S. and Doyle, L.W. (2008) An overview of mortality and sequelae of preterm birth from infancy to adulthood. Lancet, 371, 261-269. doi:10.1016/S0140-6736(08)60136-1
- Ross, E.M., Peckham, C.S., West, P.B. and Butler, N.R. (1980) Epilepsy in childhood: Findings from the National Child Development Study. British Medical Journal, 280, 207-210. doi:10.1136/bmj.280.6209.207
- Mölsä, A., Turunen, K., Mattila, K.J. and Sumanen, M. (2012) Unnecessary confusion about family planning after intrahepatic cholestasis of pregnancy. Contraception, 86, 639-644. doi:10.1016/j.contraception.2012.05.149
NOTES
*The Centre for General Practice in the Tampere University Hospital District funded the mail survey.

