Green and Sustainable Chemistry
Vol.1 No.4(2011), Article ID:8507,6 pages DOI:10.4236/gsc.2011.14023
Simple and Practical Aerobic Oxidation of AlcoholsCatalyzed by a (μ-Oxo)tetraruthenium Cluster*
1Advanced Biomedical Engineering Research Unit, Kyoto University, Kyoto, Japan
2Department of Energy and Hydrocarbon Chemistry, Graduate School of Engineering, Kyoto University, Kyoto, Japan
3Division of Multidisciplinary Chemistry, Institute for Chemical Research, Kyoto University, Kyoto, Japan
E-mail: #teruyuki@scl.kyoto-u.ac.jp
Received September 19, 2011; revised October 25, 2011; accepted November 4, 2011
Keywords: Ruthenium, Cluster, Catalyst, Aerobic Oxidation, Alcohol
Abstract
A (μ-oxo)tetraruthenium cluster showed high catalytic activity for a simple and practical aerobic oxidation of alcohols to aldehydes and ketones under 1 atm of O2 or air. After the reaction, this cluster catalyst was recovered from the reaction mixture, and we believe that this (μ-oxo)tetraruthenium cluster acts as an active catalytic species throughout the reaction.
1. Introduction
The selective oxidation of primary and secondary alcohols to the corresponding aldehydes and ketones, respecttively, is a ubiquitous and pivotal reaction in organic synthesis and the chemical industry [1,2]. Such oxidation reactions have been performed traditionally with stoichiometric inorganic oxidants, represented by CrO3, KMnO4, MnO2, and SeO2 [3]. However, these oxidation methods have serious drawbacks, such as their cost and the production of environmentally hazardous/toxic byproducts. From both economic and environmental viewpoints [4], there is an urgent demand for greener, more atom-efficient methods that use O2 and air as readily available terminal and ideal oxidants, and produce only H2O as a byproduct. Among the various transition-metal catalysts, much attention has been focused on the ability of ruthenium catalysts, since they have often been used for hydrogenation and transfer hydrogenation reactions [5] as well as dehydrogenation and oxidation reactions [6,7]. In fact, some success has been realized using ruthenium catalysts in alcohol oxidation with NaIO4 [8], tert-butyl hydroperoxide [9,10], chloramine-T [11], amine-N-oxide [11], iodosylbenzene [12], NaBrO3 [13,14], benzoquinone/Co(salen) [15-17], H2O2 [18], and TEMPO [19] as oxidants. However, there are few examples of a ruthenium-catalyzed real aerobic oxidation of alcohols without hydrogen acceptors [20,21]. Most recently, Hill and coworkers reported the ruthenium-catalyzed practical aerobic oxidation of alcohols in a continuous-flow reactor [22].
We have been interested in ruthenium chemistry, and have successfully synthesized many novel low-valent ruthenium complexes [23,24], which can be used as highly efficient catalysts for characteristic organic synthesis [25,26]. In addition, we have recently succeeded in the synthesis of a novel and isolable (μ-oxo)tetraruthenium cluster (2) [27] by an unusual reaction of a zero-valent ruthenium complex, Ru(η6-cot)(η2-dmfm)2 (1) [cot = 1,3, 5-cyclooctatriene, dmfm = dimethyl fumarate] [28], with H2O (Scheme 1).
Here, we report that this novel (μ-oxo)tetraruthenium cluster (2) efficiently catalyzes a simple, practical, and selective oxidation of primary and secondary alcohols to the corresponding aldehydes and ketones, respectively, under 1 atm of O2 or air.
2. Results and Discussion
Benzyl alcohol was smoothly oxidized in N,N-dimethylacetamide (DMA) at 80˚C for 20 h under 1 atm of O2 (balloon) to give benzaldehyde in 95% yield (Scheme 2). Neither benzoic acid nor acetal of benzaldehyde was obtained at all.
Among the solvents examined, toluene also gave the good results, and benzaldehyde was obtained in 94% yield, while in dioxane, propionitrile, and pyridine the conversion of benzyl alcohol decreased drastically to
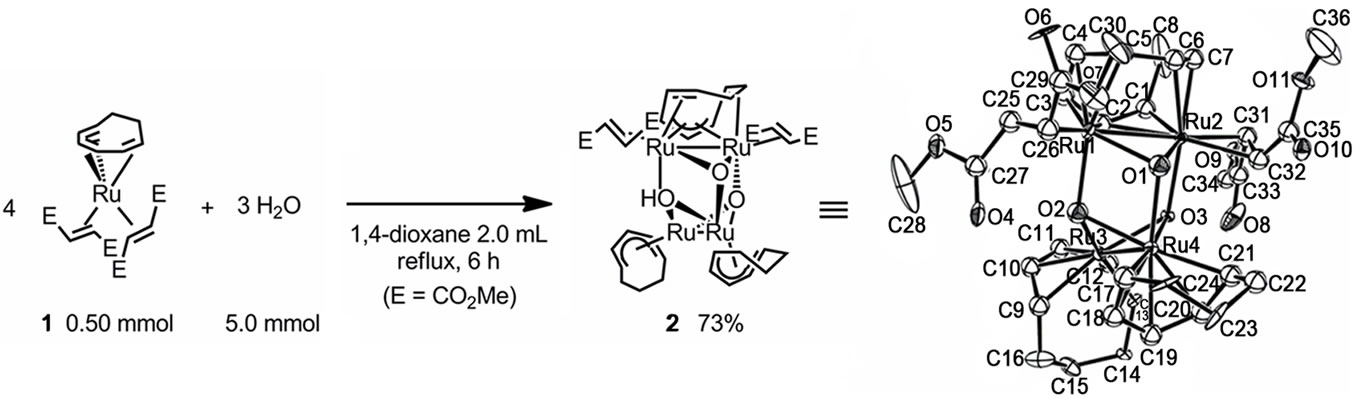
Scheme 1. Synthesis of 2 by the reaction of 1 with H2O.
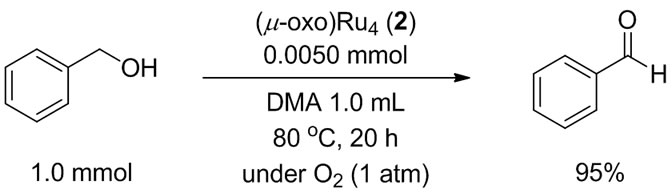
Scheme 2. Aerobic oxidation of benzyl alcohol to benzaldehyde catalyzed by 2.
give benzaldehyde in only 33%, 7%, and 2% yield, respectively.
Thus, the oxidation of primary alcohols bearing heteroaromatic rings, such as 2-thiophenemethanol and 2-pyridinemethanol, proceeded at an elevated temperature of 110˚C (Table 1, Entries 3 and 4) to give the corresponding aldehydes in good to high yields, while with aliphatic alcohols, such as 1-octanol and cyclohexanemethanol, the yield of the desired aldehydes decreased somewhat due to the formation of carboxylic acids (overoxidation) and esters (Tischenko-type-reaction) (Entries 5 and 6).
Secondary alcohols were also converted into the corresponding ketones when the reaction was carried out at 130˚C (Table 2). For example, benzophenone was obtained quantitatively by the aerobic oxidation of diphenylmethanol (Entry 1). Methyl 2-hydroxy-2-phenylacetate was also converted into methyl 2-oxo-2-phenylacetate in 81% yield (Entry 3). In all reactions, no ketals were formed at all.
The present catalyst system is highly active, and the oxidation of benzyl alcohol proceeded smoothly at 110 ˚C for 48 h under 1 atm of air (balloon) to give benzaldehyde in 87% yield with 95% selectivity (Scheme 3), while benzaldehyde was obtained in only 26% yield under an argon atmospherea. Diphenylmethanol also gave benzophenone in 93% yield under 1 atm of air.
After the reactions, (μ-oxo)tetraruthenium cluster (2) was recovered in 20% yield by simple column chromatography, which was confirmed by 1H NMR [27]. In
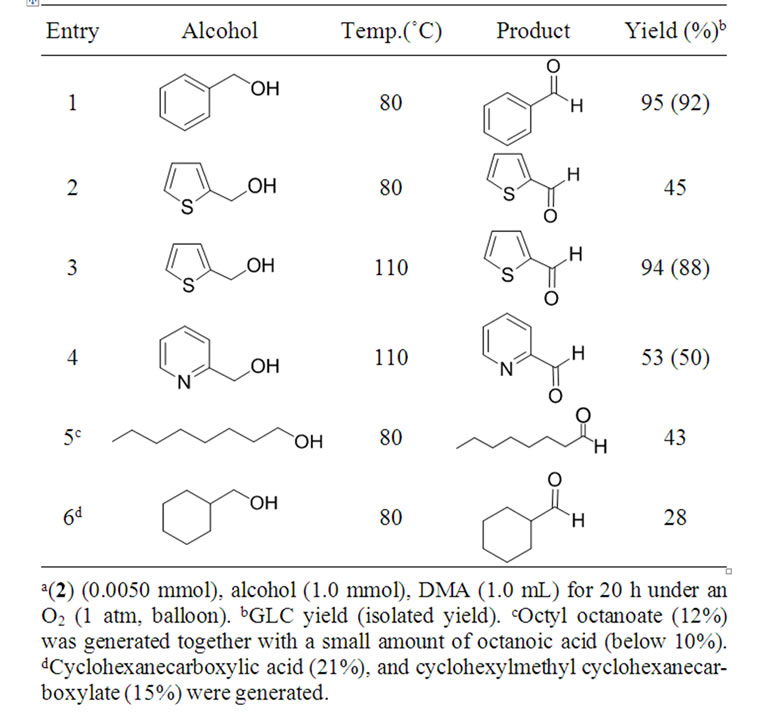
Table 1. Aerobic oxidation of primary alcohols to aldehydes catalyzed by 2a.
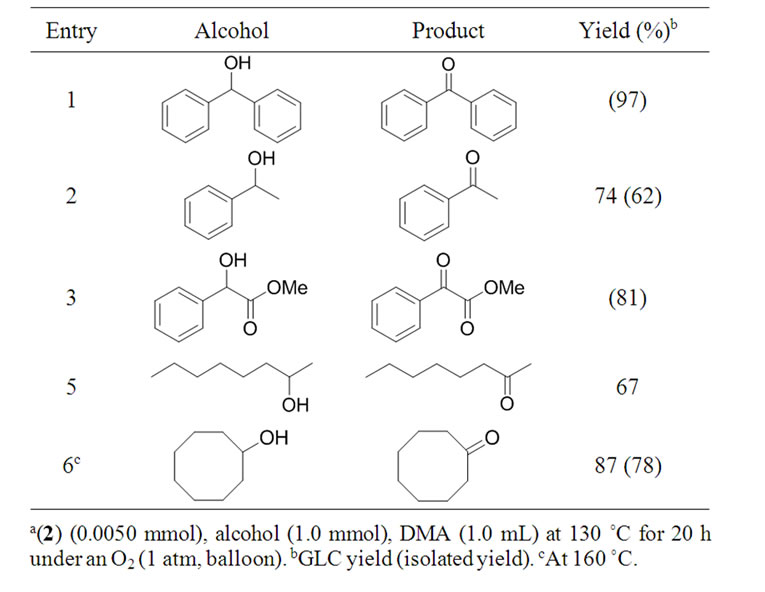
Table 2. Aerobic oxidation of secondary alcohols to ketones catalyzed by 2a.
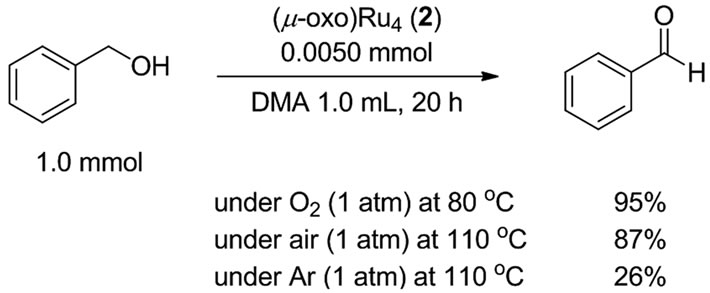
Scheme 3. Oxidation of benzyl alcohol to benzaldehyde under O2, air and Ar atmosphere.
addition, mononuclear Ru(η6-cot)(η2-dmfm)2 (1), which is the starting material for the synthesis of (μ-oxo) tetraruthenium cluster (2), showed no catalytic activity in either the absence or presence of a small amount of H2O. No induction period was observed, and benzaldehyde was generated from the initial stage of the reaction (Figure 1).
Based on the results described above, we believe that a (μ-oxo)tetraruthenium cluster (2) acts as an active catalytic species throughout the reaction [29,30]. There are two possible catalytic cycles to be considered (A and B), which are illustrated in Scheme 4. In catalytic cycle A, the reaction starts with the alcoholysis of a hydroxoRu(II) bond in 2 with an alcohol to give an (alkoxo)Ru- (II) cluster together with H2O. Oxidation of the alkoxoattached ruthenium atom with O2 then occurs to give a cluster containing an (alkoxo)(peroxo)Ru(III) part, followed by β-hydrogen elimination from an alkoxo functionality and hydrogen transfer to a peroxyo group to give a (hydroperoxo)Ru(II) cluster and the corresponding aldehyde. Final alcoholysis of a (hydroperoxo)Ru(II) cluster would give H2O2 with the regeneration of an (alkoxo)Ru(II) cluster. In the other possible catalytic cycle B, a (μ-oxo)- tetraruthenium cluster (2) is first converted into an (alkoxo)(hydroxo)Ru(II) cluster by addition of an alcohol to a bridging oxo-Ru bond. Subsequent β-hydrogen elimination releases an aldehyde, and a (hydrido)(hydroxo)Ru(II) cluster is formed. Insertion of O2 into a hydrido-Ru bond gives a (hydroperoxo)(hydroxo)Ru(II) cluster, and subsequent hydrogen abstraction from a hydroxo functionality gives H2O2 with the regeneration of 2. After the reaction, no peroxide was detected in the reaction mixture by a peroxide test (see Experimental), and we consider that H2O2 is decomposed immediately to H2O and O2 under the present reaction conditions.
Although there is not yet much evidence for full consideration and discussion of the mechanism, the recent study on the mechanism of ruthenium-catalyzed alcohol oxidation, in which the active [Ru]-OH species could be generated, supports the mechanism according to the catalytic cycle B [19,31,32], and the real oxidant in the present reaction is O2, not H2O2. A further mechanistic study including observation of KIE (Kinetic Isotope Effect) for alcohols is now in progress.

Figure 1. Time-course of aerobic oxidation of benzyl alcohol to benzaldehyde catalyzed by 2.
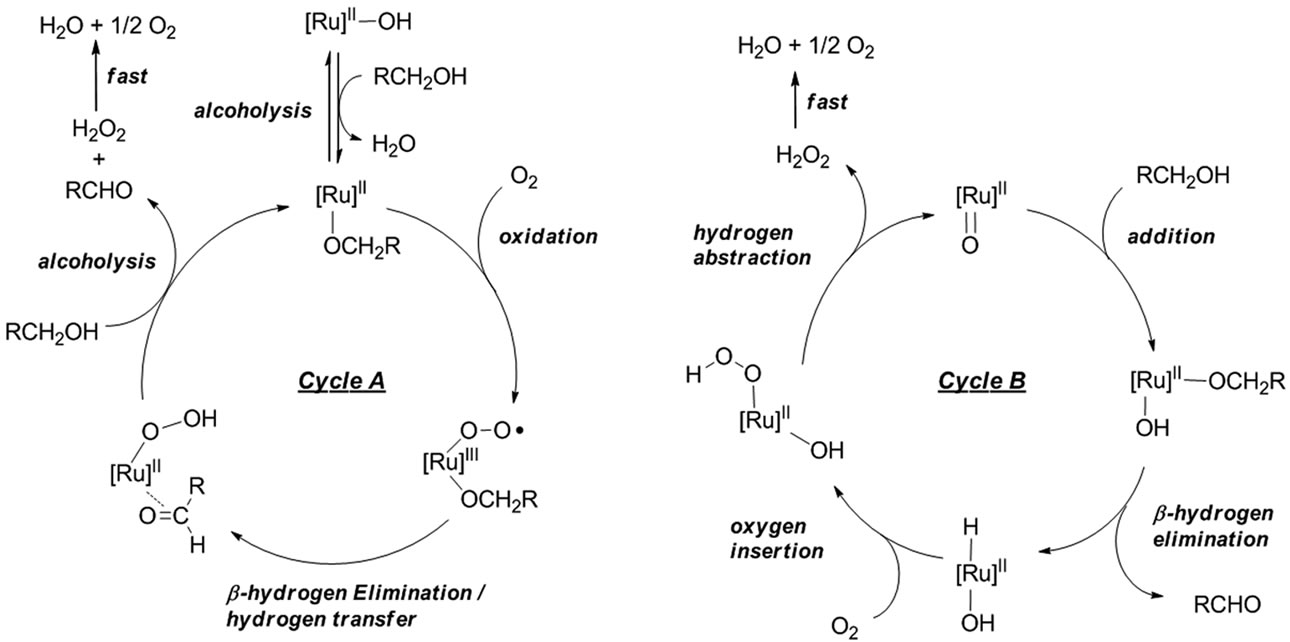
Scheme 4. Possible catalytic cycles for (μ-oxo)Ru4 (2)-catalyzed aerobic oxidation of primary alcohols to aldehydes.
3. Experimental
3.1. Materials
Alcohols were obtained commercially and purified before use by standard procedures. Anhydrous DMA and dioxane were obtained commercially and used as received. Anhydrous toluene, THF, and CH2Cl2 were obtained by filtration through a drying column on a Glass Contour system [33], and used after bubbling with argon. Ru(η6-cot)- (η2-dmfm)2 (1) and (μ-oxo)tetraruthenium cluster (2) were prepared as described in the literature [27,28]. Quantofix® peroxide test paper was used to detect H2O2.
3.2. General
GLC analyses were carried out on a Shimadzu GC14BPF (FID) gas chromatograph equipped with a glass column (2.8 mm i.d. × 3 m) packed with Silicone OV-17 (2% on Chromosorb W(AW-DMCS), 60 mesh - 80 mesh). The formation of hydrogen was analyzed carefully by a Shimadzu 8AIT (TCD) gas chromatograph equipped with a stainless column (3.0 mm i.d. × 2 m) packed with MS-5A. GC/MS analyses were performed using a Shimadzu Parvum2 mass spectrometer connected to a Shimadzu GC-2010 gas chromatograph [column: J & W Scientific capillary column DB-1, 0.25 mm i.d. × 25 m (film thickness 0.25 μm)]. 1H-NMR spectra were recorded at 400 MHz, and 13C-NMR spectra were recorded at 100 MHz. Samples were analyzed in CDCl3, and the chemical shift values are expressed relative to Me4Si as an internal standard. IR spectra were obtained on a JASCO FT/IR-4200 spectrometer.
3.3. General Procedure for the (μ-Oxo)Tetraruthenium Cluster-Catalyzed Oxidation of Alcohols to Aldehydes and Ketones
A mixture of alcohol (1.0 mmol), (μ-oxo)tetraruthenium cluster (2) (0.0050 mmol), and N,N-dimethylacetamide (1.0 mL) was placed in a 20 mL Pyrex flask equipped with a magnetic stirring bar and a reflux condenser under a flow of argon. The reaction vessel was then connected to a balloon containing O2 or air (1 atm). The reaction was carried out at 80˚C - 110 ˚C (for primary alcohols) or 130 ˚C (for secondary alcohols) for 20 h with stirring. After being allowed to cool, the reaction mixture was analyzed by GLC. The products were isolated by Kugelrohr distillation, if necessary, followed by silica gel column chromatography (KANTO CHEMICAL, Silica Gel 60N (spherical, neutral), 40 μm - 50 μm, eluent: hexane/EtOAc = 8/1), and identified by comparing the spectral (1H and 13C NMR spectra [34]) and analytical data with those of authentic samples.
4. Conclusions
One of the major goals of catalysis today is the design of environmentally benign and highly atom-efficient processes to replace traditional waste-forming reactions, including some of the most fundamental synthetic methods. Our method for the catalytic oxidation of alcohols to aldehydes and ketones can be used with a wide range of alcohols, is highly atom-efficient, and is particularly convenient with respect to a simple operation under 1 atm of O2 or air as the real oxidants.
5. Acknowledgements
This work was supported in part by a Grant-in-Aid for Challenging Exploratory Research (No. 22655025) from the Japan Society for the Promotion of Science (JSPS). T.K. acknowledges financial support from the Asahi Glass Foundation and the Yazaki Memorial Foundation for Science and Technology. This research was conducted in part at the Advanced Research Institute (Research Project of Engineering for Sustainable Environment), Katsura-Int’tech Center, Graduate School of Engineering, Kyoto University.
6. References
[1] I. W. C. E. Arends and R. A. Sheldon, “Modern Oxidation of Alcohols Using Environmentally Benign Oxidants,” In: J.-E. Bäckvall, Ed., Modern Oxidation Methods, Wiley-VCH, Weinheim, 2004, pp. 83-118.
[2] I. W. C. E. Arends and R. A. Sheldon, “Modern Oxidation of Alcohols Using Environmentally Benign Oxidants,” In: J.-E. Bäckvall, Ed., Modern Oxidation Methods, 2nd Edition, Wiley-VCH, Weinheim, 2010, pp. 147- 185. doi:10.1002/9783527632039.ch5
[3] M. B. Smith and J. March, “March’s Advanced Organic Chemistry: Reactions, Mechanisms, and Structure,” 6th Edition, John Wiley & Sons, Hoboken, 2007, pp. 1703- 1786.
[4] P. T. Anastas, “Green Catalysis,” Wiley-VCH, Weinheim, 2009.
[5] M. Kitamura and R. Noyori, “Hydrogenation and Transfer Hydrogenation,” In: S.-I. Murahashi, Ed., Ruthenium in Organic Synthesis, Wiley-VCH, Weinheim, 2004, pp. 3-52.
[6] S.-I. Murahashi and N. Komiya, “Oxidation Reactions,” In: S. -I. Murahashi, Ed., Ruthenium in Organic Synthesis, Wiley-VCH, Weinheim, 2004, pp. 53-93. doi:10.1002/3527603832
[7] I. W. C. E. Arends, T. Kodama and R. A. Sheldon, “Oxidation Using Ruthenium Catalysts,” In: C. Bruneau and P. H. Dixneuf, Eds., Ruthenium Catalysts and Fine Chemistry, Springer, Berlin, 2004, pp. 277-320.
[8] W. S. Trahanovsky, “Oxidation in Organic Chemistry,” Academic Press, Cleveland, 1973.
[9] Y. Tsuji, T. Ohta, T. Ido, H. Minbu and Y. Watanabe, “Ruthenium-Catalyzed Oxidation of Alcohols and Catechols Using t-Butyl Hydroperoxide,” Journal of Organometallic Chemistry, Vol. 270, No. 3, 1984, pp. 333-341. doi:10.1016/0022-328X(84)80381-2
[10] S.-I. Murahashi and T. Naota, “Ruthenium-Catalyzed Oxidations for Selective Syntheses of Ketones and Acyl Cyanides. Selective Acylation of Amino Compounds with Acyl Cyanides,” Synthesis, Vol. 50, No. 4, 1993, pp. 433-440. doi:10.1055/s-1993-25877
[11] K. B. Sharpless, K. Akashi and K. Oshima, “RutheniumCatalyzed Oxidation of Alcohols to Aldehydes and Ketones by Amine-N-Oxides,” Tetrahedron Letters, Vol. 17, No. 29, 1976, pp. 2503-2506. doi:10.1016/S0040-4039(00)78130-5
[12] P. Müller and J. Gody, “Catalyzed Oxidation of Alcohols and Aldehydes with Iodosylbenzene,” Tetrahedron Letters, Vol. 22, No. 25, 1981, pp. 2361-2364. doi:10.1016/S0040-4039(01)82906-3
[13] Y. Yamamoto, H. Suzuki and Y. Moro-oka, “RutheniumCatalyzed Oxidation of Alcohols with Sodium Bromate,” Tetrahedron Letters, Vol. 26, No. 17, 1985, pp. 2107- 2108. doi:10.1016/S0040-4039(00)94791-9
[14] S. Kanemoto, H. Tomioka, K. Oshima and H. Nozaki, “Cerium or Ruthenium-Catalysed Oxidation of Alcohols to Carbonyl Compounds by means of Sodium Bromate,” Bulletin of the Chemical Society of Japan, Vol. 59, No. 1, 1986, pp. 105-108. doi:10.1246/bcsj.59.105
[15] J.-E. Bäckvall, R. L. Chowdhury and U. Karisson, “Ruthenium-Catalyzed Aerobic Oxidation of Alcohols via Multistep Electron Transfer,” Journal of the Chemical Society, Chemical Communications, No. 7, 1991, pp. 473-475. doi:10.1039/C39910000473
[16] C. Csjernyik, A. H. Éll, L. Fadini, B. Pugin and J.-E. Bäckvall, “Efficient Ruthenium-Catalyzed Aerobic Oxidation of Alcohols Using a Biomimetic Coupled Catalytic System,” Journal of Organic Chemistry, Vol. 67, No. 5, 2002, pp. 1657-1662. doi:10.1021/jo0163750
[17] E. V. Johnston, E. A. Karlsson, L.-H. Tran, B. Åkermark and J.-E. Bäckvall, “Efficient Aerobic Ruthenium-Catalyzed Oxidation of Secondary Alcohols by the use of a Hybrid Electron Transfer Catalyst,” European Journal of Organic Chemistry, No. 10, 2010, pp. 1971-1976. doi:10.1002/ejoc.201000033
[18] F. Shi, M. K. Tse and M. Beller, “A Novel Environmentally Benign Method for the Selective Oxidation of Alcohols to Aldehydes and Ketones,” Chemistry, an Asian Journal, Vol. 2, No. 3, 2007, pp. 411-415. doi:10.1002/asia.200600383
[19] A. Dijksman, A. Marino-González, A. M. I. Payeras, I. W. C. E. Arends and R. A. Sheldon, “Efficient and Selective Aerobic Oxidation of Alcohols into Aldehydes and Ketones Using Ruthenium/TEMPO as the Catalytic System,” Journal of the American Chemical Society, Vol. 123, No. 28, 2001, pp. 6826-6833. doi:10.1021/ja0103804
[20] C. Bilgrien, S. Davis and R. S. Drago, “The Selective Oxidation of Primary Alcohols to Aldehydes by O2 Employing a Trinuclear Ruthenium Carboxylate Catalyst,” Journal of the American Chemical Society, Vol. 109, No. 12, 1987, pp. 3786-3787. doi:10.1021/ja00246a049
[21] I. E. Markó, P. R. Giles, M. Tsukazaki, I. Chellé-Regnaut, C. J. Urch and S. M. Brown, “Efficient, Aerobic, Ruthenium-Catalyzed Oxidation of Alcohols into Aldehydes and Ketones,” Journal of the American Chemical Society, Vol. 119, No. 51, 1997, 12661-12662. doi:10.1021/ja973227b
[22] N. Zotova, K. Hellgardt, G. H. Kelsall, A. S. Jessiman and K. K. Hill, “Catalysis in Flow: The Practical and Selective Aerobic Oxidation of Alcohols to Aldehydes and Ketones,” Green Chemistry, Vol. 12, No. 12, 2010, 2157- 2163. doi:10.1039/c0gc00493f
[23] T. Mitsudo, Y. Ura and T. Kondo, “Chemistry of Ru (η6- 1,3,5-cyclooctatriene)(η2-dimethyl fumarate)2,” Chemical Record, Vol. 6, No. 3, 2006, pp. 107-116. doi:10.1002/tcr.20076
[24] T. Mitsudo, Y. Ura and T. Kondo, “Chemistry of a Novel Zerovalent Ruthenium π-Acidic Alkene Complex, Ru(η6- 1,3,5-cyclooctatriene)(η2-dimethyl fumarate)2,” Proceedings of the Japan Academy, Series B, Vol. 83, No. 3, 2007, pp. 65-76. doi:10.2183/pjab.83.65
[25] T. Kondo, “On Inventing Catalytic Reactions via Ruthenaor Rhodacyclic Intermediates for Atom Economy,” Synlett, No. 5, 2008, pp. 629-644. doi:10.1055/s-2008-1042807
[26] T. Kondo, “New Catalytic Performances of Ruthenium and Rhodium Complexes Directed toward Organic Synthesis with High Atom Efficiency,” Bulletin of the Chemical Society of Japan, Vol. 84, No. 5, 2011, pp. 441-458. doi:10.1246/bcsj.20110019
[27] T. Kondo, F. Tsunawaki, T. Suzuki, Y. Ura, K. Wada, S. Yamaguchi, H. Masuda, K. Yoza, M. Shiro and T. Mitsudo, “Synthesis and Characterization of a Novel (μ3- Oxo)tetraruthenium Cluster,” Journal of Organometallic Chemistry, Vol. 692, No. 1-3, 2007, pp. 530-535. doi:10.1016/j.jorganchem.2006.08.062
[28] T. Mitsudo, T. Suzuki, S.-W. Zhang, D. Imai, K.i Fujita, T. Manabe, M. Shiotsuki, Y. Watanabe, K. Wada and T. Kondo, “Novel Ruthenium Complex-Catalyzed Dimerization of 2,5-Norbornadiene to Pentacyclo[6.6.02,6.03,13. 010,14]tetradeca-4,11-diene Involving Carbon-Carbon Bond Cleavage,” Journal of the American Chemical Society, Vol. 121, No. 9, 1999, pp. 1839-1850. doi:10.1021/ja9835741
[29] M. S. Thompson and T. J. Meyer, “Mechanisms of Oxidation of 2-Propanol by Polypyridyl Complexes of Ruthenium (III) and Ruthenium (IV),” Journal of the American Chemical Society, Vol. 104, No. 15, 1982, pp. 4106- 4115. doi:10.1021/ja00379a011
[30] C. S. Yi, T. N. Zeczycki and I. A. Guzei, “Highly Cooperative Tetrametallic Ruthenium-μ-Oxo-μ-Hydroxo Catalyst for the Alcohol Oxidation Reaction,” Organometallics, Vol. 25, No. 4, 2006, pp. 1047-1051. doi:10.1021/om0510674
[31] A. M. Khenkin, L. J. W. Shimon and R. Neumann, Preparation and Characterization of New Ruthenium and Osmium Containing Polyoxo-metalates, [M(DMSO)3M7O24]4– (M = Ru(II), Os(II)), and Their Use as Catalysts for the Aerobic Oxidation of Alcohols,” Inorganic Chemistry, Vol. 42, No. 10, 2003, pp. 3331-3339. doi:10.1021/ic026003p
[32] F. Nikaidou, H. Ushiyama, K. Yamaguchi, K. Yamashita and N. Mizuno, “Theoretical and Experimental Studies on Reaction Mechanism for Aerobic Alcohol Oxidation by Supported Ruthenium Hydroxide Catalysts,” Journal of Physical Chemistry C, Vol. 114, No. 24, 2010, pp. 10873-10880. doi:10.1021/jp101692j
[33] A. B. Pangborn, M. A. Giardello, R. H. Grubbs, R. K. Rosen and F. J. Timmers, “Safe and Convenient Procedure for Solvent Purification,” Organometallics, Vol. 15, No. 5, 1996, pp. 1518-1520. doi:10.1021/om9503712
[34] C. Pouchert, “Aldrich Library of 13C and 1H FT-NMR Spectra,” John Wiley & Sons, Hoboken, 1993.
NOTES
*Dedicated to Professor Christian Bruneau on the occasion of his 60th birthday.
aUnder an argon atmosphere, catalytic dehydrogenation of benzyl alcohol occurred to give benzaldehyde in 26% yield together with the formation of H2 (0.20 mmol), which was confirmed by a careful GC analysis of the gas phase.

