World Journal of Condensed Matter Physics
Vol. 2 No. 4 (2012) , Article ID: 24649 , 5 pages DOI:10.4236/wjcmp.2012.24032
ZnO Spin-Coating of TiO2 Photo-Electrodes to Enhance the Efficiency of Associated Dye-Sensitized Solar Cells
![]()
1Department of Physics, Taif University, Taif, KSA; 2Department of Chemistry, Taif University, Taif, KSA.
Email: *merazga_amar@yahoo.fr
Received April 26th, 2012; revised May 26th, 2012; accepted June 9th, 2012
Keywords: TiO2 Photo-Electrodes; ZnO Sol-Gel Spin-Coating; Dye-Sensitized Solar Cells; Power Conversion Efficiency
ABSTRACT
Dye-sensitized solar cells (DSSCs) with ZnO spin-coated TiO2 photo-electrodes are compared to DSSC with a bare TiO2 photo-electrode. It is demonstrated that the deposited ZnO of controlled amount, by varying the precursor concentration in the coating sol, can indeed enhance the performance of the DSSC. The measured power conversion efficiency shows a maximum around the precursor concentration 0.1 M and falls down sharply to 0% beyond this point. The results are interpreted on the basis of two competing factors: At ZnO concentrations less than 0.1 M, the formation of an energy barrier increases the photocurrent by reducing the rate of interfacial back-recombination. At ZnO concentrations greater than 0.1 M, the screening of the TiO2 film by thicker ZnO layers decreases the photocurrent through the reduction of TiO2 dye-adsorption efficiency.
1. Introduction
The ZnO sol-gel spin-coating method is a combination of two techniques applied to produce thin nanostructured ZnO films: The sol-gel technique consists of the preparation, under defined conditions, of a ZnO solution from the mixture of a ZnO precursor [Zn(CH3COO)2·2H2O: zinc acetate dehydrate] and a solvent (2-methoxyethanol) with the addition of a stabilizing substance (ethanolamine). The clear transparent colloidal solution thus obtained, containing suspended ZnO nanoparticles, is deposited on the surface of a substrate by the spin-coating technique [1-4]. Nanostructured ZnO films prepared by this method have been used as photo-electrodes in DSSC fabrication [5,6], but low efficiencies were recorded compared to TiO2-based DSSCs. However, the deposition of a ZnO layer on the surface of a nanoporous TiO2 film to improve the DSSC photovoltaic properties is an active issue in the context related to surface treatment of TiO2 photo-electrodes by thin metal oxide films [7-11]. Kao et al. [12] employed the sol-gel spin-coating method to prepare TiO2 and ZnO spin-coated TiO2 electrodes for DSSCs. Multilayer coating was applied to TiO2 to reach the required thickness and a monolayer of ZnO was applied to spin-coat the TiO2 film. All the photovoltaic parameters were increased upon ZnO spin-coating, resulting in the promotion of the power conversion efficiency η from 2.5% to 3.25%. In this paper, we use the sol-gel technique to prepare ZnO solutions of 5 different precursor concentrations (0.1, 0.2, 0.3 and 0.4 M). An amount of 5 drops from each solution is used to deposit a ZnO layer on the TiO2 photo-electrode surface by spincoating. The measured efficiency of the associated DSSC shows a variation with the ZnO precursor concentration, exhibiting a maximum at 0.1 M, beyond which it shows a sharp decrease to 0%. It is explained that this optimal amount of the deposited ZnO forms the thinnest layer that can create the required energy barrier to reduce the rate of back-recombination of electrons to dye molecules and electrolyte species, enhancing the photocurrent without affecting the dye-adsorption efficiency of the TiO2 film.
2. Experiment
Titanium dioxide (TiO2) was coated on F-doped SnO2 (FTO) glass using the classical screen printing technique. The TiO2 film was then dried and sintered at 450˚C for 1 hour to obtain the bare TiO2 photo-electrode to be used in this work for the preparation of the DSSCs. The sol-gel spin-coating method is employed to deposit thin layers of ZnO on the surface of TiO2 photo-electrodes. The precursor (Zn(CH3COO)2·2H2O) is mixed with the solvent (2-methoxyethanol) and the stabilizer (monoethanolamine) is added drop wise until satisfying a molar ratio of 1 with the precursor. The solution is heated at 60˚C under continuous magnetic stirring for one hour. The prepared solution containing suspended ZnO nanoparticles is then kept firmly enclosed in a clean flask before spin-coating. The spin-coater (SPIN-150, SPS) is adjusted to run at a speed of 3000 rpm for 30 s to produce reasonable thin films. The ZnO amount is monitored by the number of ZnO sol drops injected through the needle of a “smart” dispenser by carefully advancing the plunger into the barrel. The ZnO-coated TiO2 photoelectrode is annealed at 400 K for one hour. Rutheniumbased dye (C26H20O10N6S2Ru) known as N3 (Solaronix) is used to sensitize the bare and ZnO-coated TiO2 photoelectrodes by immersing in 3 × 10–4 M ethanolic solution for 24 hours. Pt-coated FTO-glass (Solaronix) is assembled as the counter-electrode against the dye-loaded photoelectrode using two paper clips. The electrolyte solution (Solaronix) containing Iodide/Triodide Redox couple is injected through a capillary channel originally drilled across the counter-electrode. The I-V characteristics of the DSSCs are measured using a solar simulator (Solar-Light) and an electrometer (Keithley 2400). The latter is computer-controlled to acquire and plot the I-V data, while AM1.5-filtered light from the 300 W-Xenon lamp of the solar simulator shines the DSSC at a power density Pi = 100 mW/cm2. The transmittance and absorbance spectra of the different films are measured in the visible range by means of a UV/VIS/NIR spectrophotometer (Jasco V-570).
3. Results and Discussions
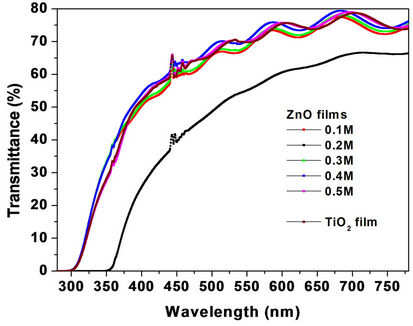
Before studying the ZnO as a spin-coating layer to improve the performance of TiO2 DSCs, it is worth presenting first some optical characteristics of this ZnO layer, with the aim to identify the coating material prior to its application. The transmittance spectra of five ZnO films with different ZnO precursor concentrations (0.1, 0.2, 0.3, 0.4 and 0.5 M) are shown in Figure 1(a) together with the transmittance spectrum of the TiO2 film for comparison. The ZnO layers were deposited by the sol-gel spin-coating technique on FTO-glass substrates using 5 sol drops for each layer, while the Solaronix TiO2 film was screen-printed on FTO-glass substrate. The transmission wavelength threshold of the ZnO (~300 nm) is lower than that of the TiO2 (~350 nm), suggesting a wider energy gap for the ZnO, which can be the physical reason for the observed higher ZnO transmittance.
In Figure 1(b), we present the plots (αhν)2 versus hν for the curves of Figure 1(a), with hν = hc/λ the photon energy (c = 3 × 108 m/s is the light velocity, h = 6.66 × 10–34 Js is the Plank constant and λ is the light wavelength) and α the optical absorption coefficient determined by the approximate relation T = exp(−α·d) which
 (a)
(a)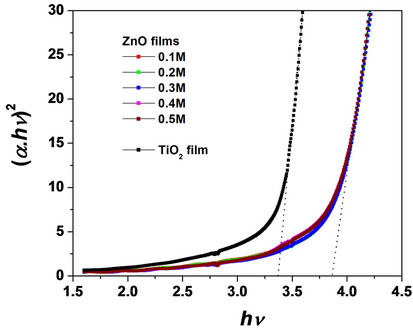 (b)
(b)
Figure 1. (a) Transmittance spectra for five ZnO films spincoated on FTO-glass at different ZnO precursor concentrations (0.1, 0.2, 0.3, 0.4 and 0.5 M). (b) (α·hν)2 versus hν curves plotted from the transmittance spectra of Figure 1 (a).
ignores the film reflectance (d is the film thickness). The plots of Figure 1(b) are based on the assumption of direct electron transitions in ZnO and TiO2 semiconductors where the relation αhν ~ (hν − Eg)1/2 holds. The parameter Eg called “optical gap” can be experimentally determined by extrapolating the line portion of the plot (αhν)2 versus hν to zero absorption coefficient. Thus, average optical gap values of 3.8 eV and 3.4 eV for the ZnO and TiO2 films are respectively determined from the curves of Figure 1(b).
Figure 2 shows the I-V characteristics of DSSCs with ZnO spin-coated TiO2 photo-electrodes at five different precursor concentrations (0.1, 0.2, 0.3, 0.4 and 0.5 M). The I-V characteristic of a DSSC with bare TiO2 photoelectrode is included for comparison. In Table 1, we present the PV parameters extracted from the I-V curves of Figure 2. At the smallest precursor concentration 0.1 M, there is a clear enhancement of the short circuit photocurrent JSC from 5.73 to 7.48 mA/cm2, while small shifts
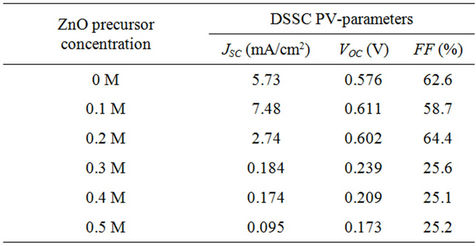
Table 1. DSSC PV-parameters at different ZnO precursor concentrations.
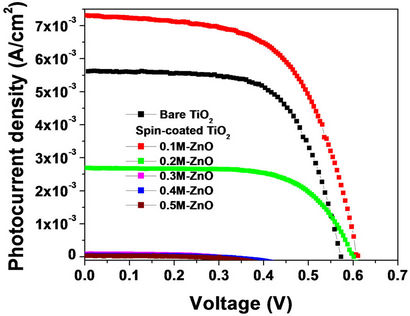
Figure 2. I-V characteristics of DSSCs with ZnO spincoated TiO2 photo-electrodes at 5 different precursor concentrations. The I-V characteristic of the DSSC with uncoated TiO2 photo-electrode is included for comparison.
upward from 0.58 to 0.61 V and downward from 62% to 59% for the open circuit voltage VOC and the fill factor FF are respectively observed. Further increase of the ZnO precursor concentration, to 0.2 M, causes JSC to decrease remarkably to 2.74 mA/cm2, whereas small shifts to 0.6 V and to 64.4% for VOC and FF are again respectively observed. Beyond 0.2 M, the PV-parameters decrease drastically and almost no photovoltaic effect is detected (JSC ~ 0.2 - 0.1 mA/cm2, VOC ~ 0.2 V and FF ~ 25%).
The resulting power conversion efficiency
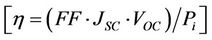 is plotted in Figure 3 as a function of the ZnO precursor concentration. An optimum η ~ 2.7% is observed at the smallest concentration 0.1 M followed by a sharp decrease to 0%. While the small upward shift of VOC can be explained by a small shift of the Fermi-level upon ZnO coating, it is clear from Table 1 and Figure 3 that the variation of η is mainly due to the variation of JSC. It is known in the literature that the deposition of a metal oxide layer, with a higher conduction band energy minimum than that of TiO2, on the surface of a TiO2 photo-electrode creates an energy barrier that can inhibit the injected electrons from the dye molecules to recombine back with the electrolyte species
is plotted in Figure 3 as a function of the ZnO precursor concentration. An optimum η ~ 2.7% is observed at the smallest concentration 0.1 M followed by a sharp decrease to 0%. While the small upward shift of VOC can be explained by a small shift of the Fermi-level upon ZnO coating, it is clear from Table 1 and Figure 3 that the variation of η is mainly due to the variation of JSC. It is known in the literature that the deposition of a metal oxide layer, with a higher conduction band energy minimum than that of TiO2, on the surface of a TiO2 photo-electrode creates an energy barrier that can inhibit the injected electrons from the dye molecules to recombine back with the electrolyte species
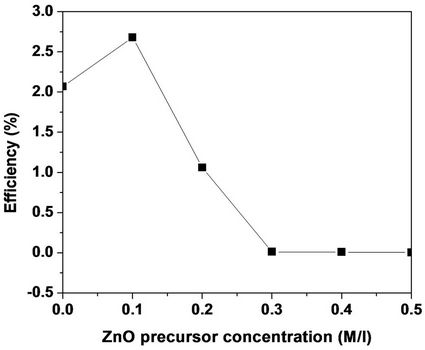
Figure 3. Variation of η with the ZnO precursor concentration.
[12,13]. This recombination inhibition is the direct cause of the photocurrent enhancement occurring at ZnO precursor concentrations around 0.1 M. However, there must be an optimal amount of ZnO in the deposited layer above which the illuminated TiO2 surface is partially obscured by the ZnO layer. The original dye-adsorption efficiency of the TiO2 film decreases when the coating ZnO of much lower dye-adsorption efficiency [14] screens the TiO2 surface. The light harvest rate and the electron injection rate will consequently decrease and this will be reflected by the decrease of JSC and η (Figure 3).
Figure 4 shows the absorbance spectra of dye-loaded ZnO-coated TiO2 films at five different ZnO precursor concentrations (0.1, 0.2, 0.3, 0.4 and 0.5 M), plotted together with the spectrum of the dye-loaded uncoated TiO2 film. The spectrum of the bare TiO2 film is also plotted in Figure 4 for comparison. We can first compare the spectrum of the dye-loaded TiO2 with that of the bare TiO2: It is noticeable that the spectrum of the bare TiO2 film (black line) is much lower than the spectrum of the film with loaded dye (black curve), which means that the dye molecules form the main light absorber in the visible range. Since ZnO is more transparent in the visible range than TiO2, one would expect that the dye molecules will still form the main absorber in the presence of the ZnO layer and that the spectra of the dye-loaded ZnO-coated TiO2 films will be similar to that of the dye-loaded uncoated TiO2 film. In contrast, ZnO coating of the TiO2 film using only 0.1 M-ZnO sol results in a much higher spectrum (red curve) than that of the uncoated TiO2 film. In fact, the chemical instability of ZnO against acidic dye molecules results in partial dissociation of Zn2+ ions from ZnO particles during the dye-loading process [5,15]. The Zn2+-dye aggregates thus formed on the TiO2 film must be the cause of the observed high excess absorption. This light loss by the Zn2+-dye absorption does not affect the
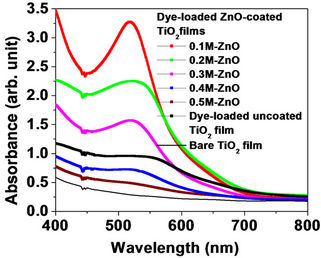
Figure 4. Absorbance spectra for dye-loaded ZnO-coated TiO2 films at five different precursor concentrations. The absorbance spectra for the dye-loaded uncoated TiO2 film and the bare TiO2 film are also presented for comparison.
photocurrent because the 0.1 M-ZnO layer is very thin and the low surface density of the deposited ZnO particles on the TiO2 film will not affect the dye-adsorption efficiency of the TiO2. Yet, the necessary energy barrier to minimize the rate of electron back-recombination is established. The photocurrent density JSC and the cell efficiency η of the associated DSSC will then be enhanced, as detailed above (Figures 2 and 3). With increasing the concentration of the ZnO precursor to 0.2 M, the deposited ZnO particles will partially screen the dye molecules from being efficiently adsorbed by the TiO2 material. The effective dye-covered TiO2 area reduces and the electron light harvest efficiency reduces as a consequence. The ZnO sol amount of 5 drops must then be sufficiently high, so that an increase of the ZnO concentration to 0.2 M will cause considerable reduction of the TiO2 dye adsorption efficiency. Thereby, JSC shifts well below that of the bare TiO2 case (Figure 2) and η reduces to about 1%. Further increase of the ZnO concentration to 0.3, 0.4 and 0.5 M will vanishingly reduce the dye adsorption efficiency of the TiO2, resulting in absorbance spectra lower than that of the uncoated TiO2 film (bleue curve for 0.4 M ZnO and brown curve for 0.5 M ZnO).
4. Conclusion
The role of ZnO, as spin-coated on TiO2 phto-electrodes, to enhance the power conversion efficiency of the corresponding DSSC is studied. The I-V characteristics of ZnO-coated TiO2 DSSCs with 5 different ZnO precursor concentrations (0.1, 0.2, 0.3, 0.4 and 0.5 M) are compared to that of a bare TiO2 DSSC. The results show maximum power conversion efficiency at 0.1 M with enhancement of about 35%. A sharp decrease of η to 0% with increasing ZnO concentration follows this maximum. The efficiency enhancement upon ZnO coating is due to the formation of an energy barrier which limits the rate of electron back-recombination without affecting the dye adsorption efficiency of the TiO2 film. The decrease of the efficiency with increasing ZnO concentration is attributed to the reduction of the dye-adsorption efficiency of the TiO2 film due to screening by thicker ZnO layers.
5. Acknowledgements
The authors would like to acknowledge financial support from Taif University for the project No. 2-432-762.
REFERENCES
- S. Ilican, Y. Caglar and M. Caglar, “Preparation and ChaRacterization of ZnO Thin Films Deposited by the SolGel Spin-Coating Method,” Journal of Optoelectronics and Advanced Materials, Vol. 10, No. 10, 2008, pp. 2578- 2583.
- D. Raoufi and T. Raoufi, “The Effect of Heat Treatment on the Physical Properties of Sol-Gel Derived ZnO Thin Films,” Applied Surface Science, Vol. 255, No. 11, 2009, pp. 5812-5817. doi:10.1016/j.apsusc.2009.01.010
- M. Smirnov, C. Baban and G. I. Rusu, “Structural and Optical Characteristics of Spin-Coated ZnO Thin Films,” Applied Surface Science, Vol. 256, No. 8, 2010, pp. 2405- 2408. doi:10.1016/j.apsusc.2009.10.075
- K. Balachandra and P. Raji, “Synthesis and Characterization of Nano ZnO by Sol-Gel Spin-Coating,” Recent Research in Science and Technology, Vol. 3, No. 3, 2011, p. 48.
- Y.-J. Shin, et al., “Enhancement of Photovoltaic Properties of Ti-Modified Nanocrystalline ZnO Electrode for Dye-Sensitized Solar Cells,” Bulletin of the Korean Chemical Society, Vol. 26, No. 12, 2005, p. 1929.
- M. C. Kao, H. Z. Chen and S. L. Young, “Effects of PreAnnealing Temperature of ZnO Thin Films on the Performance of Dye-Sensitized Solar Cells,” Applied Physics A, Vol. 98, No. 3, 2010, pp. 595-599. doi:10.1007/s00339-009-5467-9
- D. B. Menzies, et al., “Modification of Mesoporous TiO2 Electrodes by Surface Treatment with Titanium (IV), Indium (III) and Zirconium (IV) Oxide Precursors,” Nanotechnology, Vol. 18, No. 12, 2007, Article ID: 125608. doi:10.1088/0957-4484/18/12/125608
- S. Wu, et al., “Improvement in Dye-Sensitized Solar Cells Employing TiO2 Electrodes Coated with Al2O3 by Reactive Direct Current Magneton Sputtering,” Journal of Power Sources, Vol. 182, No. 1, 2008, pp. 119-123.
- S. Wu, et al., “Enhancement in Dye-Sensitized Solar Cells Based on MgO-Coated TiO2 Electrodes by Reactive DC Magnetron Sputtering,” Nanotechnology, Vol. 19, No. 21, 2008, Article ID: 215704.
- L. Li, et al., “Improved Performance of TiO2 Electrodes Coated with NiO by magnetron sputtering for Dye-Sensitized Solar Cells,” Applied Surface Science, Vol. 256, No. 14, 2010, pp. 4533-4537. doi:10.1016/j.apsusc.2010.02.042
- Y. Liu, et al., “Efficiency Enhancement in Dye-Sensitized Solar Cells by Interfacial Modification of Conducting Glass/Mesoporous TiO2 Using a Novel ZnO Compact Blocking Film,” Journal of Power Sources, Vol. 196, No. 1, 2011, pp. 475-481. doi:10.1016/j.jpowsour.2010.07.031
- M. C. Kao, H. Z. Chen and S. L. Young, “Effects of ZnOCoating on the Performance of TiO2 Nanostructured Thin Films for Dye-Sensitized Solar Cells,” Applied Physics A, Vol. 97, No. 2, 2009, pp. 469-474. doi:10.1007/s00339-009-5244-9
- S. S. Kim, J. H. Yum and Y. E. Sung, “Improved Performance of a Dye-Sensitized Solar Cell Using a TiO2/ ZnO/Eosin Y Electrode,” Solar Energy Materials and Solar Cells, Vol. 79, No. 4, 2003, pp. 495-505. doi:10.1016/S0927-0248(03)00065-5
- T. Soga, “Nanostructured Materials for Solar Energy Conversion,” Elsevier, Amsterdam, 2006.
- K. S. Kim, et al., “Photovoltaic Properties of Nanoparticulate and Nanorod Array ZnO Electrodes for Dye-Sensitized Solar Cells,” Bulletin of the Korean Chemical Society, Vol. 27, No. 2, 2005, p. 295.
NOTES
*Corresponding author.

