Journal of Diabetes Mellitus
Vol.1 No.4(2011), Article ID:8420,9 pages DOI:10.4236/jdm.2011.14015
Bone marrow increases human islets insulin positive cells in co-culture: quantification with flow cytometry
![]()
1Center for Stem Cell Biology, Roger Williams Hospital, Providence, USA; *Corresponding Author: Lluo@rwmc.org
2Alpert Medical School, Brown University, Providence, USA.
Received 30 August 2011; revised 4 October 2011; accepted 15 October 2011.
Keywords: Bone marrow; Human Islets; Flow Cytometry
ABSTRACT
We have previously demonstrated that allogeneic human bone marrow (BM) supports human islet function and longevity in vitro. We hypothesize that BM supporting human islets may include to increase β-cell in cultured islets. In this study, we developed a method to quantify insulin-producing β cells from cultured islets by using immunofluorescent staining and flow cytometry analysis to explore this possibility. The results show that human islets cocultured with BM for 39 days contained a significantly higher number of insulin-positive β cells (42.3% ± 4.5%) compared to the islet-only cultures (1.15% ± 0.78%), and increased insulin release levels evaluated by ELISA is consistent with increased β cells in same culture condition. Human islet culture with BM significantly increase β-cells while islet only culture lost β-cells in same culture period supports the possibility of BM increasing β-cells in cultured islets.
1. INTRODUCTION
Diabetes is a condition whereby the body is not able to regulate levels of glucose (a sugar) in the blood, resulting in too much glucose being present in the blood. Pancreatic islet transplantation is a promising treatment for patients with Type 1 diabetes mellitus, which results in hyperglycemia due to islet β cells damage [1-4] . However, maintaining the structural integrity and insulin release of post-isolated human islets is a pre-requisite for achieving successful islet transplantation. Existing protocols for maintaining human islets in vitro are limited, allowing only short-term (less than 72 h) storage of cells under conventional static culture conditions [5]. Extending the duration of culture beyond this period usually results in a significant loss of structural integrity within the islets and a decline in glucose-stimulated insulin release [6-8].
Our previous studies have indicated that bone marrow can maintain β cell function by evaluating insulin secretion even after 200 days of in vitro culture [9]. Exploration of mechanisms how BM support human islets in long term suggests that BM cultured human islet may increase β-cell population in islet during culture. However, quantification of β-cell from cultured human islet is a challenge. There are several methods to identify β-cells, including methods to determine the function and purity of islets. At the mRNA level, polymerase chain reaction (PCR) and in situ hybridization are performed. Enzymelinked immunosorbent assay (ELISA) is used to measure insulin secretion [10-12] or c-peptide release [13] in supernatants. However, ELISA measures only β cell insulin secretion over time, but not the number of insulinproducing cells within this heterogeneous cell population. For insulin producing cell numbers, insulin-specific immunohistochemistry combined with light microscopy [14, 15] can be used. This method has its limitations in terms of reliability and comprehensiveness. Another method is the use of flow cytometry on single cell suspensions with fluorescent zinc probes [16,17] . This method uses FluoZin-3 staining to reveal higher amounts of chelatable zinc ions in β-cells [18]. However, we did not successfully detect positive FluoZin-3 in dissociated monolayer islet cells in long term culture in vitro because this method also stained some kind bone marrow cell like monocyte [19] and T cell [20]. So this method will result false positive β-cells in islet and bone marrow co-culture. Other methods include the use of cell surface molecule Poly-Sialated Neural Cell Adhesion Molecule (PSA-NCAM) as a marker of β cells, which is also suitable for flow cytometry analysis and cell sorting [21] . However, PSA-NCAM lacks specificity since it expresses in different tissues such as nerve cells [22,23] . In this study, a two-step protocol using intracellular insulin staining and flow cytometry was established in our group and applied to quantify insulin positive cells from a culture containing a heterogeneous mixture of islet cells. Using this technique, we were able to quantitate insulin-producing β cells from culture islets with or without BM. Our studies support the hypothesis that BM supporting human islet β cell function includes increase β cell population while not in islet only culture.
2. MATERIALS AND METHODS
2.1. Antibodies and Reagents
4% paraformaldehyde (Sigma) solution was prepared by dissolving paraformaldehyde powder in phosphate buffered saline (PBS, Invitrogen, Carlsbad, CA, USA). Insulin antibody (ab7842) and insulin + proinsulin antibody (ab20756) were purchased from Abcam (Cambridge, MA, USA). Proinsulin antibody (10-P14B) was purchased from Fitzgerald Industries (Acton, MA, USA). Normal IgG of goat (sc-2028), rabbit (sc-2027), mouse (sc-2025) and guinea pig (SC-2711) were purchased from Santa Cruz biotechnology Inc (Santa Cruz, CA, USA). Fluorescein isothiocyanate (FITC) conjugated goat polyclonal to Guinea pig secondary antibody (ab6904) was purchased from Abcam. FITC conjugated horse antimouse secondary antibody (FI-2000) was purchased from Vector Laboratories (Burlingame, CA, USA).
2.2. Tissue and Cell Culture
Human pancreatic islets. Human islet tissue from normal donors was obtained through the Islet Resource Centers (ICRs) Basic Science Islet Distribution Program from the Human Islet Laboratories at the University of Pennsylvania (Philadelphia, PA, USA), Massachusetts General Hospital (Boston, MA, USA), City of Hope National Medical Center (Duarte, CA, USA), University of Miami, School of Medicine, Diabetes Research Institute (Miami, FL, USA). The use of these cells is approved by the Institutional Review Board (IRB) at Roger Williams Hospital and the ICR Committee [9].
Human BM. Human BM from normal donors was obtained under a separate Roger Williams Hospital IRBapproved protocol. Bone marrow erythrocytes were eliminated by Ficoll-Paque™ Plus (Amersham Biosciences; Amersham, UK) per manufacturer directions. Cells were then washed twice with 5% Fetal Bovine Serum (FBS) in phosphate buffered saline (PBS) and resuspended in culture medium (see below). Trypan blue (Gibco, 15250-061) staining was used to assess cell viability. The use of these cells is approved by the Institutional Review Board (IRB) at Roger Williams Hospital and the ICR Committee [9].
Allogeneic BM co-culture with human islets. Human islets were received from ICR within 48 h of harvest from cadaveric donors. The viability of whole islets upon arrival was >95% as determined by trypan blue dye exclusion. Islets were placed in culture at 50 islet equivalents (IEQs) per ml with 1 × 106 allogeneic BM cells/ ml. Cultures were maintained in RPMI 1640 (Gibco) supplemented with 10% heat inactived FBS (Hi-FBS, Hyclone), 5.5 mM glucose, 10 mM HEPES, and 1% P/S, pH 7.3 [9].
2.3. Enzyme-Linked Immunosorbent Assay for Insulin Measurement
Insulin concentrations in each specimen (culture medium and cell extracts) were measured using Ultra Sensitive Human Insulin ELISA Kit (Millipore, EZHI-14K) according to the vendor’s instructions. Insulin concentrations were measured and calculated using a μQuant microplate reader equipped with KC Junior® microplate reader software (Bio-Tek Instruments, Inc., Winooski, VT) [24] . Briefly, insulin standards and appropriately diluted samples were added to 96-well microplates precoated by the manufacturer with an anti-insulin antibody and incubated for 1 hour at room temperature. Samples were removed, plate was washed five times, and the enzyme-linked anti-human insulin conjugate was added to the well for 30 minutes incubation at room temperature. After washing five times, the enzyme substrate solution was added and incubated 10 to 15 minutes at room temeperature in the dark. The reaction was halted by adding 1 N sulfuric acid. Absorbance at 450 nm was read with a μQuant microplate reader and insulin concentrations were calculated based on the standard curve by KC Junior® microplate reader software (Bio-Tek Instruments, Inc.) [9].
2.4. Two-Step Intracellular Anti-Insulin Staining and Flow Cytometry
After human islets were dissociated into single cells with 0.05% trypsin-EDTA and 0.2% collagenase, cell suspensions were washed and then re-suspended in PBS for cell counting. The cell viability was measured with trypan blue. Cells were fixed in 2% paraformaldehyde solution for 20 min in the dark at 4˚C with slight shaking every 5 minutes. Cells were then washed in ice-cold 5% Hi-FBS PBS and permeabilized in 0.05% triton x-100 solution in 5% Hi-FBC PBS for 15 minutes at room temperature. Cells were washed and resuspended with icecold PBS buffer (including 0.05% triton x-100 and 5% Hi-FBS). Cell suspensions were added to each roundbottom polystyrene tube for the antibody staining. Cells were stained with isotype control or antibody to insulin or proinsulin and an antibody that recognizes both proinsulin+insulin at 1:50, 1:100 and 1:200 dilution diluted in 0.05% triton x-100 and 5% Hi-FBS PBS for 1 hour in ice or overnight at 4˚C. After incubation, cells were washed and resuspended in ice-cold permeablization buffer. Cells were then stained with the secondary antibody conjugated with fluorescein isothiocyanate (FITC) at 1:300 (FITC conjugated goat polyclonal to Guinea pig secondary antibody) or 1:100 (FITC conjugated horse antimouse secondary antibody) dilution diluted in ice-cold permeablization buffer for 30 minutes on ice and in the dark. For the non-specific staining, cells were only stained with specific secondary antibody conjugated with FITC for 30 minutes on ice and in the dark. Finally, cells were washed in permeablization buffer by centrifugation at 300 g for 10 minutes and resuspended in 400 μL 5% Hi-FBS PBS buffer. Flow cytometry analysis was performed on Becton Dickinson LSRII system (BD Biosciences, CA, USA). In all cases, more than 10,000 cellular events were analyzed per tube, and in several cases, 100,000 cellular events were analyzed per tube. This method is relative stable than other reported methods [25] .
2.5. Data Analysis
Results are displayed as mean ± standard deviation (SD). Statistical significance was determined using the Student’s t test; p values less than 0.05 are considered statistically significant.
3. RESULTS
3.1. Optimizing Antibodies for Labeling β-Cell
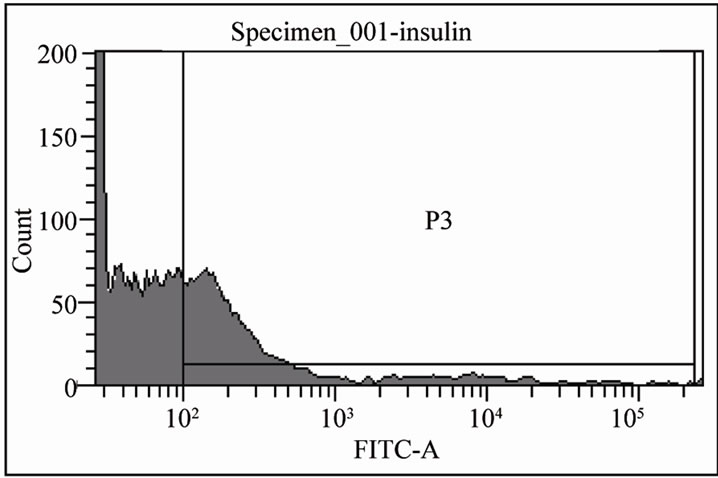
Cultured human islets are cluster of cell group, which have to be dissociated into single cells. We established a protocol to dissociate these cultured human islet into single cells. Human islets were dissociated under the optimized digestive conditions (0.05% trypsin and 0.2% collagenase solution) into single cells (Figure 1). To find an optimized antibody and concentration for labeling β cells, cells were stained with various concentrations of antibodies against insulin, proinsulin, or insulin + proinsulin. Optimum staining for insulin occurs at a 1:100 dilution of anti-insulin antibody, at 1:200 of anti-proinsulin antibody, and 1:100 dilution of anti-insulin + proinsulin antibody. Under same condition of islet dissociation samples, results showed that anti-insulin antibodies identified the least amount of cells (17.2% ± 0.44%) whereas anti-proinsulin detected 45.4% ± 3.48% cells and the antibody for insulin + proinsulin detected 43.2% ± 7.92% cells. The antibodies that recognized either proinsulin or proinsulin + insulin were two folds greater versus insulin only antibody within similar groups (Figure 2), then we used anti-proinsulin antibody in following studies.
 (a)
(a)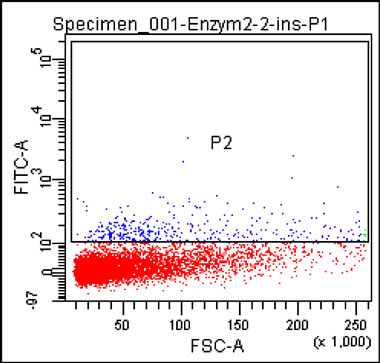 (b)
(b)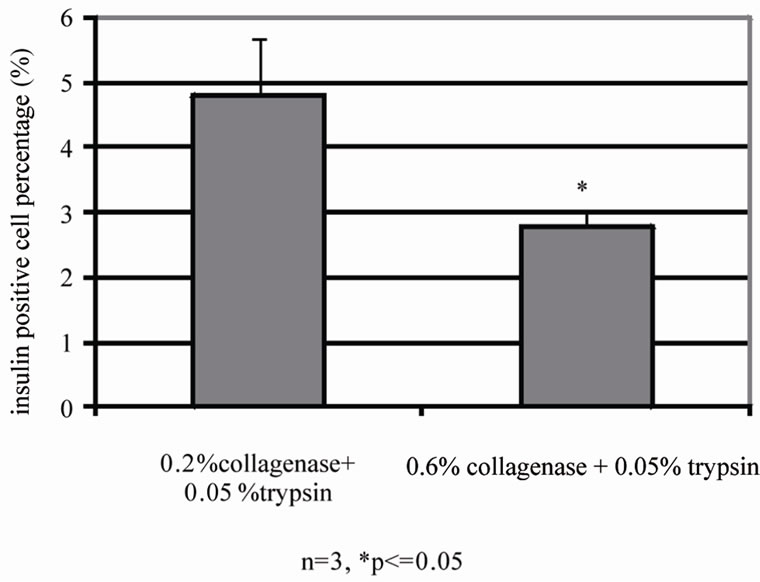 (c)
(c)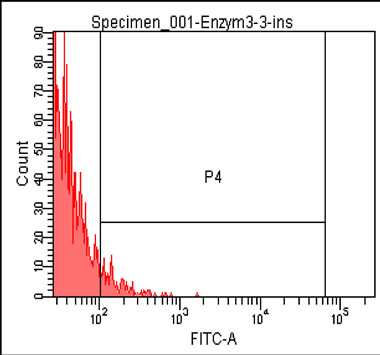 (d)
(d)
Figure 1. Optimized tissue dissociation enzyme condition and quantified insulin positive cells by flow cytometry. After human islets were incubated with digestion enzyme, partly dissociated human islets (a) were digested into single cells (b). Single cells were stained with insulin antibody (c). Insulin positive cells were detected with anti-insulin for flow cytometry for human islets dissociated with 0.2% collagenase + 0.05% trypsin (left) or 0.6% collagenase + 0.05% trypsin (top) (d) and then stained (n = 3, *p ≤ 0.05), bottom panels indicated Flow Cytometry data (Magnification × 20 ((a) and (b)), × 40 (c)).
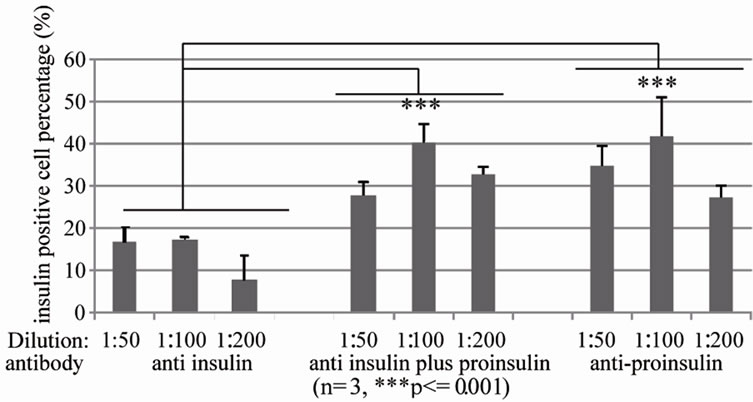
Figure 2. Optimized anti-insulin antibodies for identifying human islet β cells dissociated and recovered human islet cells were fixed and stained with anti insulin antibody (left), anti insulin plus proinsulin antibody (middle) and anti-proinsulin antibody (right) at 1:50, 1:100, 1:200 dilution respectively, and then subjected to flow cytometry analysis (n = 3, ***p ≤ 0.001).
3.2. Quantification of Human Islet β Cells with or without BM from Different Culture Days
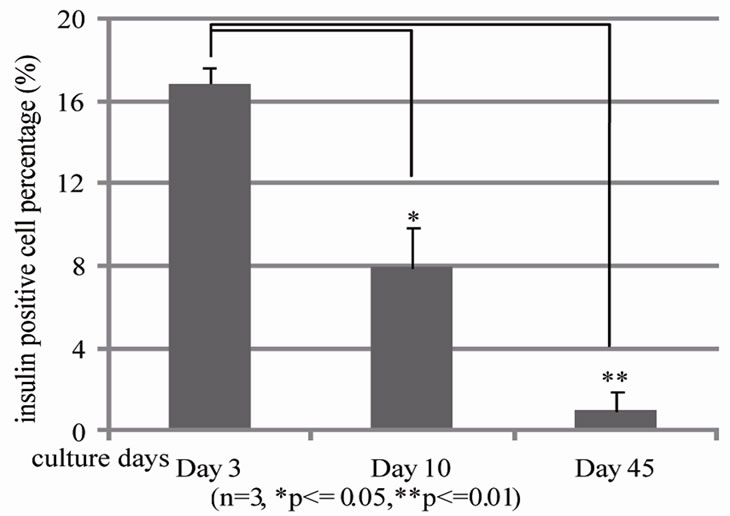
We evaluated β cell populations in human islets with or without BM during culture from day 3 to 45 as indicated by insulin positive cells with the established β-cell quantification method. We demonstrated that the β-cell population decreases in islet only cultures from 16.8% ± 0.8% on Day 3 to 7.9% ± 1.9% on Day 10 and then to 0.93% ± 0.06% on Day 45 (Figure 3(a)).Consequently, insulin release decreased from 19412.4 ± 104.3 µU/ml on Day 3 to 11386.7 ± 5920 µU/ml on Day 10 and to 418.0 ± 134.5 µU/ml on Day 45 (Figure 3(b)).
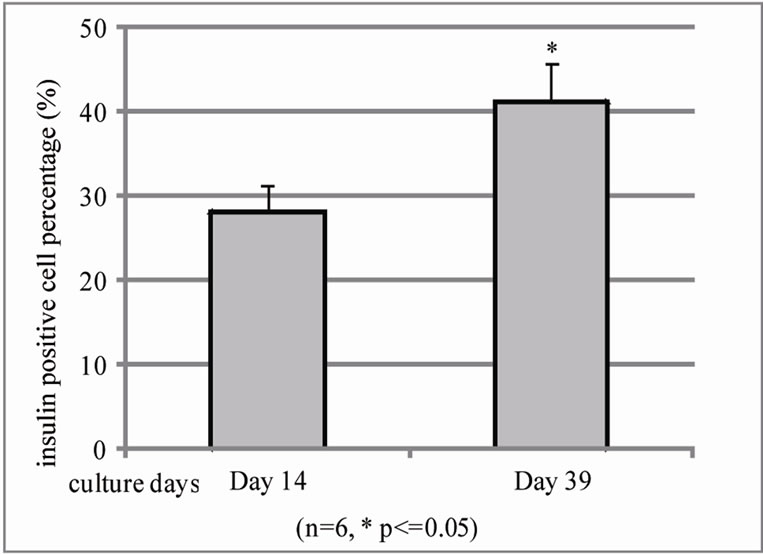
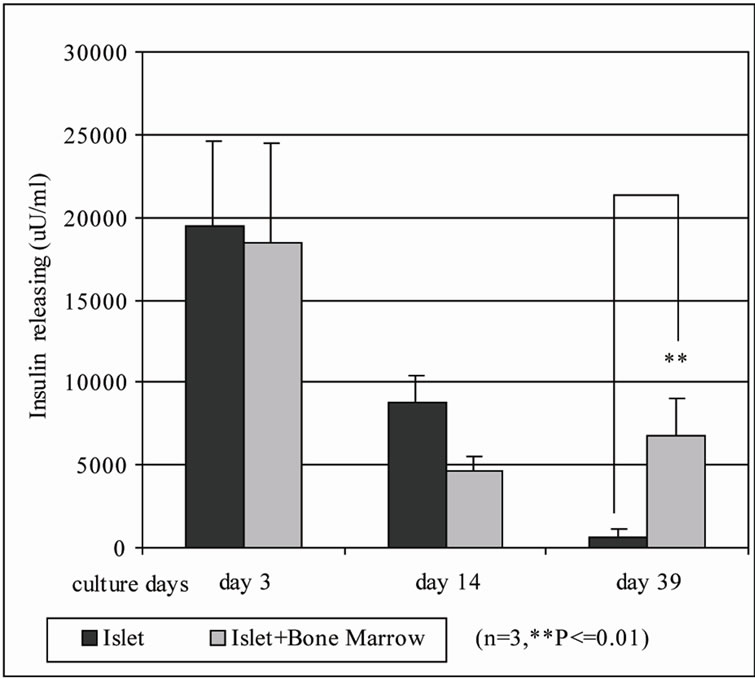
In evaluation of BM co-culture human islet during different culture days using same method, we found that β cell population from islet with BM co-culture significantly increases from 14 to 39 day in cultures (28.15% ± 3.4% on day 14 and 42.3% ± 4.5% on day 39 cultures) (Figure 4(a)) and function evaluation also found insulin release increase but too much variation to statistic sig-
 (a)
(a)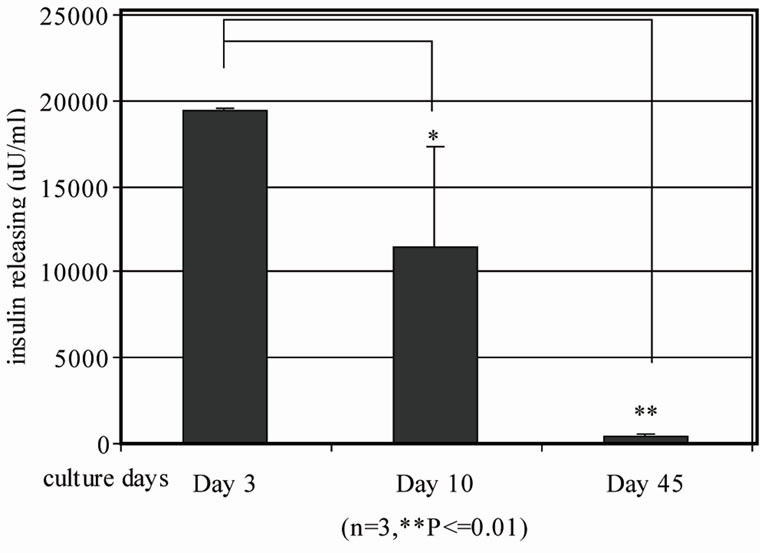 (b)
(b)
Figure 3. Quantification of insulin positive cells and insulin releasing in human islets during different culture days by flow cytometry and human insulin ELISA. Human islets were cultured for 3 days, 10 days and 45 days in vitro respectively. (a) Human islets were then dissociated with 0.2% collagenase + 0.05% trypsin into single cells. Guinea pig anti-human insulin polyclonal antibody and FITC conjugated secondary antibody were subsequently added to stain insulin positive cells and then subjected to BD LSRII flow cytometry analysis (n = 3, *p ≤ 0.05, **p < 0.01,); (b) Human islet insulin release was analyzed by human insulin ELISA kit.
nificance (4650 ± 866.51 µU/ml on day 14 and 6748.68 ± 2236.59 µU/ml, Figure 4(b)).
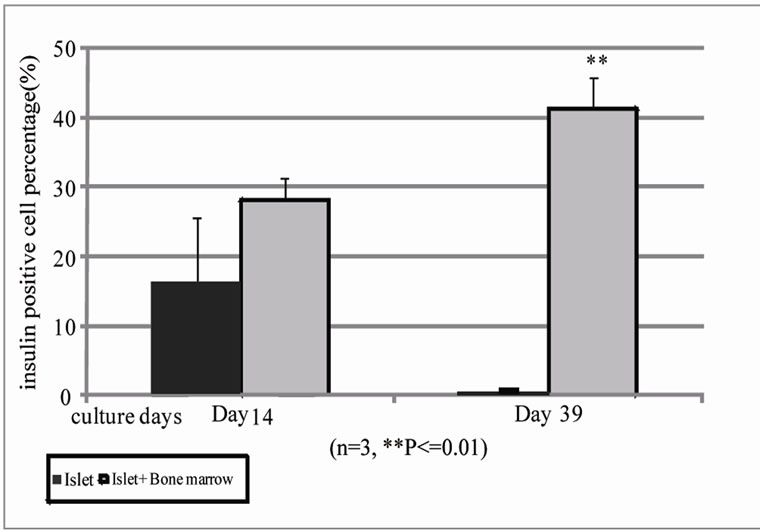
Comparison between islet with or without BM in different culture days, we found that there are significantly variation during two weeks and short period cultures, islet only groups (16.5% ± 9.19) and islet and BM coculture groups (28.15% ± 3.4%) (Figure 5(a)). However, insulin positive cells in islet with BM co-culture on Day 39 was significant increase 42.3% ± 4.5% versus 1.15% ± 0.78% for islet only cultures. The function of β-cell evaluation indicated that short term cultured human islet only insulin release is higher than BM co-cultured islet but not significance because variation (Figure 5(b)). We suspect that this increasing insulin release may resultfrom huge amount of β cell death in culture. The signifi-
 (a)
(a)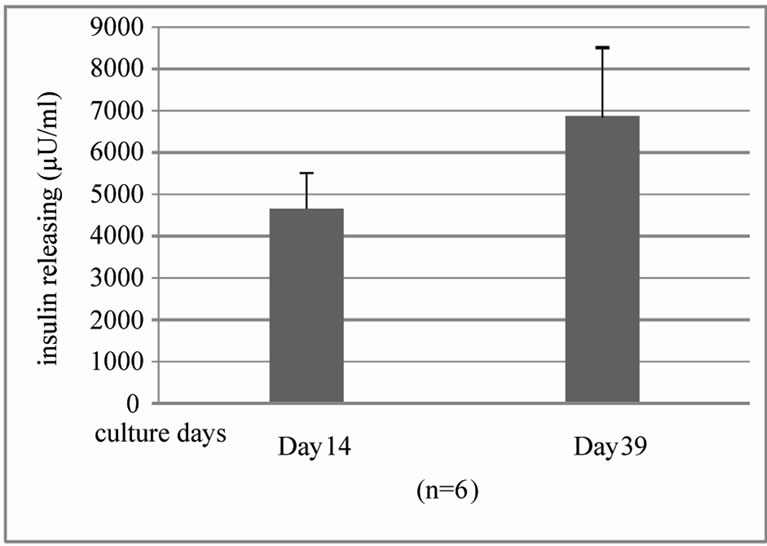 (b)
(b)
Figure 4. Quantification of insulin positive cells and insulin releasing in islet-BM co-culture between day 14 and 39 by flow cytometry and human insulin ELISA. (a). Dissociated and recovered human islet cells were fixed and stained with anti insulin antibody. Insulin positive cells were detected with flow cytometry analysis on the 14th day (left) and 39th day (right) of human islet culture (n = 3, *P < 0.05). B. Insulin release was evaluated by human insulin ELISA kit.
cant reduction of β-cell number and insulin release in continuing culture supports this notion (Figure 5).
4. DISCUSSION
Several recent studies have suggested that adult bone marrow cells can influence beta-cell regeneration in diabetic animals [26-32]. Other reports, however, have contradicted these findings [33,34]. In this study, we addressed this question by establishing a method to quantify insulin positive cells in human islet cultured with or without bone marrow. The optimized islet culture dissociation conditions coupled with methods to label single insulin positive β cells described here allows us to quantify cultured β-cells from islet clusters.
In terms of antibodies for detecting insulin positive cells, we expected that an antibody capable of detecting both insulin and proinsulin would yield the highest sensitivity due to its wide range of binding capability for proinsulin and insulin and the results support this notion that proinsulin-only and insulin + proinsulin antibodies showed to have better results than insulin only antibody. Within the β cell granules, proinsulin is converted by a process of enzymatic cleavage to insulin and C-peptide [35,36] but over 90% of the newly generated insulin is released and only 0.5% of proinsulin is rapidly released [37]. Given this, using proinsulin antibodies for labeling β cells is optimal for flow cytometry quantification.
Adult bone marrow harbors cells to have pluripotent differentiation capacity and benefit other tissue regeneration. Pluripotent mesenchymal stem cells in adult bone marrow have the capacity to differentiate into a number of neuroectodermal [38], endothelial [39] , epithelial [40] , and muscle [41] cells. Bone marrow is capable of maintaining pancreatic islets in the absence of immunosuppression, and can constitute an immunoprivileged environment for engraftment [31,42] . Transplantation of bone marrow derived cells into diabetic mice can reducehyperglycemia through proliferation of β-cells, differentiation of pancreatic stem cells to contribute to the rege-

 (a)(b)
(a)(b)
Islet (day 14) Islet + Bone Marrow (day 14) Islet (day 39) Islet + Bone Marrow (day 39)
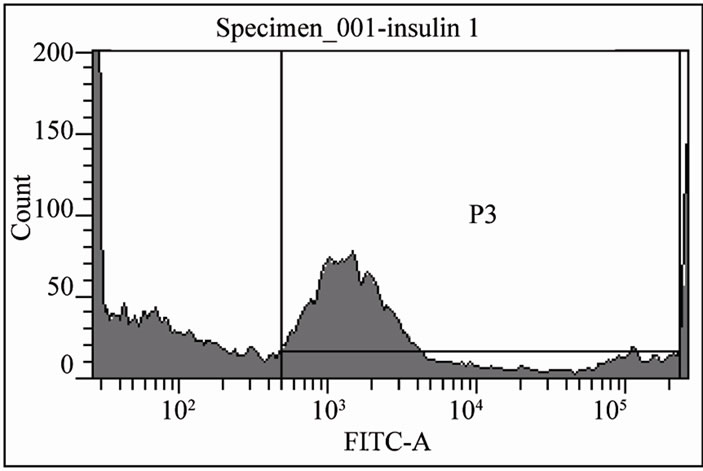
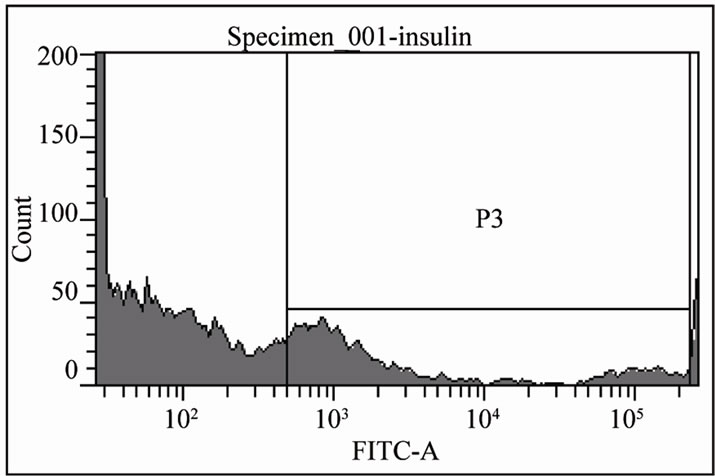
Figure 5. Comparison of human islet β cells in islet with or without BM in culture. (a) Insulin positive cells were detected with flow cytometry analysis on the 14th day (left) and 39th day (right) of human islet culture (n = 3, *p < 0.05). Top two panels indicated 14th day of islet and islet with BM flow cytometry images and bottom two lower panels are 39th day of islet and islet with BM flow cytometry images. (b) Islet Insulin release was analyzed by human insulin ELISA kit.
neration of β-cells [28,29,43,44] , and improve islet graft function by promoting graft vascularization [31,45] . Bone marrow’s ability to correct hyperglycemia in vivo has been shown in cases where animals with islets transplanted into bone marrow had a higher chance to reach euglycemia than islets transplanted in the liver [46]. With the hypothesis that bone marrow supporting human islet in vitro includes increase β cells, we used our established quantification method to evaluate effects of bone marrow on the quantity of human islet β cells during culture with or without bone marrow. We found that bone marrow increases the number of insulin positive human islet β cells during long term culture on the 39th day vs the 14th day (Figure 4) and significantly increase β cell function vs. islet only in long term culture (Figure 5). We still have no answer why bone marrow cultured human islet taken so long (two or three weeks) to see the significant increase of β cell population but it may be involved bone marrow repair islet injury and islet recovery from isolation shock process which need time to recover. Islet only without bone marrow support is in slow process to loss.
In summary, we have demonstrated that human islet cultured with BM significantly increase β cells function which partially resulted from β cell population increase. Newly established method with two steps of labeling β cells coupled with flow cytometry to quantify cell samples (≥30,000 cells) in a fully objective manner is repeatable with little expense. The results indicated that BM enables supporting human islet culture by increase insulin positive human islet β cells while islet only culture lost β cells in same culture period. Further studies may be required to determine the mechanisms how bone marrow contributes to increase β cells in human islet culture.
5. CONCLUSIONS
We have demonstrated that BM supporting human islet survival and function in long term culture. It includes increase human islet β cell population evaluated by a flow cytometry-based analysis method to quick and accurate quantification of insulin-positive islet β cells from cultured human islets. Further studies for the mechanisms involved will be explored in future.
6. COMPETING INTERESTS
The authors have declared that no competing interests exist.
7. ACKNOWLEDGEMENTS
This work was funded by NIH P20RR018757 (COBRE, PI Dr. Falanga), JDRF 1-2007-180 and Roger Williams Hospital Research fund for Dr. Luo’s Research. We thank the Islet Cell Resource Centers (ICR) funded by the NIH and JDRF for distributing human islets for this project, Cell Sorting and Image Core was funded by NIH/NCRR 5P20RR018757, Dr. Camillo Ricordi’ valuable advises.
REFERENCES
- Shapiro, A.M., Lakey, J.R., Ryan, E.A., et al. (2000) Islet transplantation in seven patients with type 1 diabetes mellitus using a glucocorticoid-free immunosuppressive regimen. The New England journal of medicine, 43, 230-238. doi:10.1056/NEJM200007273430401
- Shapiro, A.M., Ricordi, C., Hering, B.J., et al. (2006) International trial of the Edmonton protocol for islet transplantation. The New England journal of medicine, 355, 1318-1330. doi:10.1056/NEJMoa061267
- Shapiro, A.M.J., Ryan, E.A. and Lakey, J.R.T. (2001) Islet cell transplantation. The Lancet, 358, 07034-07039. doi:10.1016/S0140-6736(01)07034-9
- Shapiro, A.M.J., Ryan, E.A. and Lakey, J.R.T. (2001) Clinical islet transplant—State of the art. Transplantation Proceedings, 33, 3502-3503. doi:10.1016/S0041-1345(01)02416-2
- Matsumoto, S., Goel, S., Qualley, S., Strong, D.M. and Reems, J.A. (2003) A comparative evaluation of culture conditions for short-term maintenance (<24 hr) of human islets isolated using the Edmonton protocol. Cell and tissue banking, 4, 85-93. doi:10.1023/B:CATB.0000007043.15164.8a
- Paraskevas, S., Maysinger, D., Wang, R., Duguid, T.P. and Rosenberg, L. (2000) Cell loss in isolated human islets occurs by apoptosis. Pancreas, 20, 270-276. doi:10.1097/00006676-200004000-00008
- Noguchi, H. (2007) Activation of c-Jun NH2-terminal kinase during islet isolation. Endocrine Journal, 54, 169-176. doi:10.1507/endocrj.KR-87
- Iwanaga, Y., Sutherland, D.E., Harmon, J.V. and Papas, K.K. (2008) Pancreas preservation for pancreas and islet transplantation. Current opinion in organ transplantation, 13, 135-141. doi:10.1097/MOT.0b013e3282f63942
- Luo, L., Badiavas, E., Luo, J.Z. and Maizel, A. (2007) Allogeneic bone marrow supports human islet beta cell survival and function over six months. Biochemical and Biophysical Research Communications, 361, 859-864. doi:10.1016/j.bbrc.2007.07.105
- Sai, P. and Maurel, C. (1983) Enzyme-linked immunosorbent assay for quantitation of islet cell antibodies. Journal of immunological methods, 64, 189-198.
- Kuglin, B., Gries, F.A. and Kolb, H. (1988) Evidence of IgG autoantibodies against human proinsulin in patients with IDDM before insulin treatment. Diabetes, 37, 130- 132. doi:10.2337/diabetes.37.1.130
- Sodoyez, J.C., Koch, M., Lemaire, I., et al. (1991) Influence of affinity of antibodies upon their detection by liquid phase radiobinding assay and solid phase enzyme linked immunosorbent assay. Demonstration using monoclonal antibodies raised against rDNA human proinsulin. Diabetologia, 34, 463-468. doi:10.1007/BF00403281
- Hartling, S.G., Dinesen, B., Kappelgard, A.M., Faber, O.K. and Binder, C. (1986) ELISA for human proinsulin. Clinica chimica acta: International Journal of Clinical Chemistry, 156, 289-297.
- Ravazzola, M., Perrelet, A., Roth, J. and Orci, L. (1981) Insulin immunoreactive sites demonstrated in the Golgi apparatus of pancreatic B cells. Proceedings of the National Academy of Sciences of the United States of America, 78, 5661-5664. doi:10.1073/pnas.78.9.5661
- Reddy, S.N., Bibby, N.J. and Elliott, R.B. (1985) Cellular distribution of insulin, glucagon, pancreatic polypeptide hormone and somatostatin in the fetal and adult pancreas of the guinea pig: A comparative immunohistochemical study. European journal of cell biology, 38, 301-305.
- Lukowiak, B., Vandewalle, B., Riachy, R., et al. (2001) Identification and purification of functional human betacells by a new specific zinc-fluorescent probe. Journal of Histochemistry and Cytochemistry, 49, 519-528. doi:10.1177/002215540104900412
- Fernandez, L.A., Hatch, E.W., Armann, B., et al. (2005) Validation of large particle flow cytometry for the analysis and sorting of intact pancreatic islets. Transplantation, 80, 729-737. doi:10.1097/01.tp.0000179105.95770.cd
- Jayaraman, S. (2008) A novel method for the detection of viable human pancreatic beta cells by flow cytometry using fluorophores that selectively detect labile zinc, mitochondrial membrane potential and protein thiols. Cytometry Part A, 73, 615-625. doi:10.1002/cyto.a.20560
- Dubben, S., Honscheid, A., Winkler, K., Rink, L. and Haase, H. (2010) Cellular zinc homeostasis is a regulator in monocyte differentiation of HL-60 cells by 1 alpha, 25-dihydroxyvitamin D3. Journal of leukocyte biology, 87, 833-844. doi:10.1189/jlb.0409241
- Kaltenberg, J., Plum, L.M., Ober-Blobaum, J.L., Honscheid, A., Rink, L. and Haase, H. (2010) Zinc signals promote IL-2-dependent proliferation of T cells. European journal of immunology, 40, 1496-1503. doi:10.1002/eji.200939574
- Bernard-Kargar, C., Kassis, N., Berthault, M.F., Pralong, W. and Ktorza, A. (2001) Sialylated form of the neural cell adhesion molecule (NCAM): A new tool for the identification and sorting of beta-cell subpopulations with different functional activity. Diabetes, 50, S125-S130. doi:10.2337/diabetes.50.2007.S125
- Minana, R., Sancho-Tello, M., Climent, E., Segui, J.M., Renau-Piqueras, J. and Guerri, C. (1998) Intracellular location, temporal expression, and polysialylation of neural cell adhesion molecule in astrocytes in primary culture. Glia, 24, 415-427. doi:10.1002/(SICI)1098-1136(199812)24:4<415::AID-GLIA7>3.0.CO;2-A
- Seki, T. (2002) Expression patterns of immature neuronal markers PSA-NCAM, CRMP-4 and NeuroD in the hippocampus of young adult and aged rodents. Journal of neuroscience research, 70, 327-334. doi:10.1002/jnr.10387
- Luo, L., Yano, N. and Luo, J.Z. (2006) The molecular mechanism of EGF receptor activation in pancreatic beta-cells by thyrotropin-releasing hormone. American journal of physiology, 290, E889-E899.
- Vredenburgh, J.J., Silva, O., Tyer, C., et al. (1996) A comparison of immunohistochemistry, two-color immunofluorescence, and flow cytometry with cell sorting for the detection of micrometastatic breast cancer in the bone marrow. Journal of Hematotherapy, 5, 57-62. doi:10.1089/scd.1.1996.5.57
- Li, M., Inaba, M., Guo, K.Q., Hisha, H., Abraham, N.G. and Ikehara, S. (2007) Treatment of streptozotocin-induced diabetes mellitus in mice by intra-bone marrow bone marrow transplantation plus portal vein injection of beta cells induced from bone marrow cells. International journal of hematology, 86, 438-445. doi:10.1007/BF02984002
- Chao, K.C., Chao, K.F., Fu, Y.S. and Liu, S.H. (2008) Islet-like clusters derived from mesenchymal stem cells in Wharton’s Jelly of the human umbilical cord for transplantation to control type 1 diabetes. PloS One, 3, e1451. doi:10.1371/journal.pone.0001451
- Dong, Q.Y., Chen, L., Gao, G.Q., et al. (2008) Allogeneic diabetic mesenchymal stem cells transplantation in streptozotocin-induced diabetic rat. Clinical and investigative medicine, 31, E328-E337.
- Gao, X., Song, L., Shen, K., Wang, H., Niu, W. and Qin, X. (2008) Transplantation of bone marrow derived cells promotes pancreatic islet repair in diabetic mice. Biochemical and Biophysical Research Communications, 371, 132-137. doi:10.1016/j.bbrc.2008.04.033
- Cantarelli, E., Melzi, R., Mercalli, A., et al. (2009) Bone marrow as an alternative site for islet transplantation. Blood, 114, 4566-4574. doi:10.1182/blood-2009-03-209973
- Figliuzzi, M., Cornolti, R., Perico, N., et al. (2009) Bone marrow-derived mesenchymal stem cells improve islet graft function in diabetic rats. Transplantation Proceedings, 41, 1797-1800. doi:10.1016/j.transproceed.2008.11.015
- Xie, Q.P., Huang, H., Xu, B., et al. (2009) Human bone marrow mesenchymal stem cells differentiate into insulin-producing cells upon microenvironmental manipulation in vitro. Differentiation: research in biological diversity, 77, 483-491.
- Kang, E.M., Zickler, P.P., Burns, S., et al. (2005) Hematopoietic stem cell transplantation prevents diabetes in NOD mice but does not contribute to significant islet cell regeneration once disease is established. Experimental Hematology, 33, 699-705. doi:10.1016/j.exphem.2005.03.008
- Akashi, T., Shigematsu, H., Hamamoto, Y., et al. (2008) Bone marrow or foetal liver cells fail to induce islet regeneration in diabetic Akita mice. Diabetes/Metabolism Research and Reviews, 24, 585-590. doi:10.1002/dmrr.884
- Bennett, D.L., Bailyes, E.M., Nielsen, E., et al. (1992) Identification of the type 2 proinsulin processing endopeptidase as PC2, a member of the eukaryote subtilisin family. The Journal of biological chemistry, 267, 15229- 15236.
- Smeekens, S.P., Montag, A.G., Thomas, G., et al. (1992) Proinsulin processing by the subtilisin-related proprotein convertases furin, PC2, and PC3. Proceedings of the National Academy of Sciences of the United States of America, 89, 8822-8826. doi:10.1073/pnas.89.18.8822
- Rhodes, C.J. and Halban, P.A. (1987) Newly synthesized proinsulin/insulin and stored insulin are released from pancreatic B cells predominantly via a regulated, rather than a constitutive, pathway. The Journal of cell biology, 105, 145-153. doi:10.1083/jcb.105.1.145
- Rice, C.M. and Scolding, N.J. (2008) Autologous bone marrow stem cells—Properties and advantages. Journal of the neurological sciences, 265, 59-62. doi:10.1016/j.jns.2007.06.011
- Lee, R.H., Seo, M.J., Reger, R.L., et al. (2006) Multipotent stromal cells from human marrow home to and promote repair of pancreatic islets and renal glomeruli in diabetic NOD/scid mice. Proceedings of the National Academy of Sciences of the United States of America, 103, 17438-17443. doi:10.1073/pnas.0608249103
- Kataoka, K., Medina, R.J., Kageyama, T., et al. (2003) Participation of adult mouse bone marrow cells in reconstitution of skin. The American journal of pathology, 163, 1227-1231. doi:10.1016/S0002-9440(10)63482-7
- Abedi, M., Foster, B.M., Wood, K.D., et al. (2007) Haematopoietic stem cells participate in muscle regeneration. British journal of haematology, 138, 792-801. doi:10.1111/j.1365-2141.2007.06720.x
- Cantarelli, E., Melzi, R., Mercalli, A., et al. (2009) Bone marrow as an alternative site for islet transplantation. Blood, 114, 4566-4574.
- Hasegawa, Y., Ogihara, T., Yamada, T., et al. (2007) Bone marrow (BM) transplantation promotes beta-cell regeneration after acute injury through BM cell mobilization. Endocrinology, 148, 2006-2015. doi:10.1210/en.2006-1351
- Hess, D., Li, L., Martin, M., et al. (2003) Bone marrow-derived stem cells initiate pancreatic regeneration. Nature Biotechnology, 21, 763-770. doi:10.1038/nbt841
- Johansson, U., Rasmusson, I., Niclou, S.P., et al. (2008) Formation of composite endothelial cell-mesenchymal stem cell islets: A novel approach to promote islet revascularization. Diabetes, 57, 2393-2401. doi:10.2337/db07-0981
- Salazar-Banuelos, A., Wright, J., Sigalet, D. and BenitezBribiesca, L. (2008) The bone marrow as a potential receptor site for pancreatic islet grafts. Archives of medical research, 39, 139-141. doi:10.1016/j.arcmed.2007.09.004

