Food and Nutrition Sciences
Vol.5 No.13(2014), Article
ID:47660,11
pages
DOI:10.4236/fns.2014.513129
Effects of Planting Methods and Seed Density on Vegetable Yield and Nutrient Composition of Solanum macrocarpon and Solanum scabrum in Southwest Nigeria
Mary K. Idowu1*, Durodoluwa J. Oyedele1, Ojo Kolawole Adekunle2, Oluwole Olalekan Akinremi3, Bob Eilers3
1Department of Soil Science and Land Resources Management, Faculty of Agriculture, Obafemi Awolowo University, Ile-Ife, Nigeria
2Department of Crop Production and Protection, Faculty of Agriculture, Obafemi Awolowo University, Ile-Ife, Nigeria
3Department of Soil Science, University of Manitoba, Winnipeg, Canada
Email: *marykemii@yahoo.com
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 18 April 2014; revised 23 May 2014; accepted 5 June 2014
ABSTRACT
Two field studies were carried out using Solanum macrocarpon and Solanum scabrum to investigate the effects of planting methods and seed density required for optimum biomass yield and nutrient content in southwest Nigeria. Two planting methods (drilling and broadcasting), and two seed densities (4 and 8 table spoon full) were investigated as a 2 × 2 × 3 factorial experiment in randomized complete block design and replicated three times. Four seed spoons gave 25.78 kg seeds ha−1 for S. macrocarpon and 20.67 kg·seeds·ha−1 for S. scabrum. Significant highest S. macrocarpon shoots (2.75 kg·m−2) and Ca, K, Fe and Zn contents and S. scabrum (2.95 kg·m−2) and Zn content, were obtained with drilling and four spoons of seeds compared with broadcasting and 8 spoons of seeds. This treatment gave average values for N, P, K, Mg, Ca, Na, Fe, Zn, Mn, Cu, Ca/P and Na/K ratio were 2.79%, 0.03%, 3.37%, 1.64%, 0.02%, 300 mg·kg−1, 100 mg·kg−1, 300 mg·kg−1, 8 mg·kg−1, 1.22 and 0.008 for S. macrocarpon and 2.05%, 0.27%, 5.81%, 3.27%, 2.91%, 0.09%, 280 mg·kg−1, 52.99 mg·kg−1, 359.35 mg·kg−1 and 22.42 mg·kg−1, 11.63 and 0.016 for S. scabrum. It was concluded that planting in drilling made weeding, fertilizer application, irrigation and harvesting more effective rather than by broadcasting, and four spoons of seeds per 9 m−2 produced deep green and broader leaves and balanced nutrient contents than eight spoons of seeds.
Keywords:Solanum macrocarpon, Solanum scabrum, Planting Method, Seed Density and Nutrient Contents

1. Introduction
Minimum daily intake of 400 g of fruits and vegetables has been recommended for good health and general well being [1] . Solanum macrocarpo and Solanum scabrum are among the indigenous underutilized vegetables consume by the resource poor women in the southwest Nigeria to meet daily vegetable requirements to supply minerals, vitamins and protein for women because of their inability to purchase highly nutritious food items such as egg, meat and milk [2] . Solanum macrocarpon is known as Igbagba and Solanum scabrum as Ogunmo in the region (Figure 1). Solanum scabrum leaves extract have been used to treat diarrhoea and jaundice in children. Raw fruits are also used to treat stomach ulcers when chewed and swallowed [3] . They are cooked as soups with or without melon and other ingredients. However, Production and consumption of the vegetables have been limited, primarily, due to insufficient information on the agronomic requirements, nutritional and health benefits, cooking techniques and market opportunities [4] . The two vegetables are believed to be nutritive and serve as cheap sources of minerals and vitamins. Helping women improve the productivity of more nutritious, high-value products such as vegetables will not only increases family income but also promotes ground-level nutrition by increasing the amount of healthy food available for home consumption. This crop diversification provides variety for primarily rice-based diets, which inherently lack the availability of primary, secondary and micronutrients necessary for human health [5] .
There is no information on planting methods and density for S. macrocarpon and S. scabrum in soils of southwest Nigeria. There is substantial information mainly on practices of commercial vegetable amaranth grown in the southwest Nigeria. Amaranth vegetables are grown by broadcasting large amounts of vegetable seeds in an open land, with application of unregulated amount of nitrogen fertilizer [6] . This practice often results in vegetable roots competing for space, nutrients and water [7] . Heavy doses of nitrogen fertilizer often resulted in leaching of nitrogen into water bodies. Nitrates and nitrites occur widely in our foods and drinks [8] . The contamination of water with nitrates and nitrites has potential adverse effects on communities or people who depend solely on these sources of water. Nitrite is known to be a precursor of toxic and carcinogenic N-nitrosamines [9] and induces cancer in experimental animals [10] . Vegetable Amaranth has emerged as the most cultivated vegetable in rural and urban centres of southwest Nigeria because it is a C4 species, it grows fast if in good condition: deep loose fertile soil, plenty of light and high temperature and humidity. It also use water more efficiently [11] .
Food production remains well below its potential in Africa. Africa has a quarter of the world’s arable land,
 (a)
(a) (b)
(b)
Figure 1. Photo of (a) Solanum macrocarpon; (b) Solanum scabrum.
but only generates 10 percent of global agricultural output. In addition, more than 75 percent of total arable land in SSA is degraded with nearly 3.3 percent of agricultural GDP lost annually because of soil and nutrient loss. Considering the increasing societal demand for food, which should be produced in a more sustainable way [12] and the adverse effects of climate change [13] . Increasing the number of vegetable diversities and the quantity of vegetable produced will increase peoples’ resilience to climate change. Agronomic requirements of S. macrocarpon and S. scabrum are different from that of the popularly grown Amaranthus spp. It is more difficult to measure the amount of fertilizer needed on vegetable farms where cultivated lands are not layout and the quantity of seed not measured. Good quality vegetable would be produced when vegetable plots are layout and appropriate and balanced fertilizer applied. Improved vegetable productivity and resources use efficiency could only be achieved, when appropriate planting technologies and seed density for the vegetables are available. Production of good quality vegetables would promote national, regional and international market opportunities for indigenous vegetables of southwest Nigeria. There is the need for an improvement in the practices of the leafy vegetable production in the region [14] .
The objective of this study was to investigate the effects of planting methods and seed density on biomass yield and nutrient composition of Solanum macrocarpon and Solanum scabrum, with a view to elucidate the best methods for producing quality vegetables.
2. Materials and Methods
2.1. Description of the Study Area
The study was carried out at the Teaching and Research Farm of the Obafemi Awolowo University, Ile-Ife, Nigeria located on latitudes 7˚31' and 7˚33'N and longitudes 4˚33'E and 4˚34'E, in the rain forest zone of southwest Nigeria. The wet and dry seasons extend from April to October and November to March, 2013, respectively. The pattern of rainfall is bimodal, with the average annual rainfall estimated to be about 1400 mm. The average monthly temperature ranges from 18.9˚C to 34.6˚C and the mean monthly relative humidity is 61% and 83% for the early and late planting seasons, respectively. The study was carried out two times. The first planting was between December, 2012 and April, 2013. The second planting was carried out between January and May, 2013.
2.2. Soil Sampling, Preparation and Analysis
Bulk surface soil samples (0 - 15 cm) were collected from unfertilized plots from Obafemi Awolowo University, Teaching and Research Farm, Ile-Ife. Soils at the sampling site have been classified as Iwo series [15] and Typic Paleustult [16] [17] . Soil samples were air dried, crushed in agate mortar, passed through a 2-mm sieve, and analyzed for following properties. Particle size distribution was determined by the modified hydrometer method [18] using 0.2 M NaOH solution as the dispersing agent. Soil pH was determined using a glass electrode pH meter in both distilled water and 0.01 M CaCl2 solution, using 1:2 soil:CaCl2 solution [19] . Soil organic carbon was determined by the chromic acid digestion method [20] . The total N concentration was determined by macro-Kjeldahl method [21] , and the available P was extracted by Bray-1 method [22] and determined using Spectrometer (Model 721 Visible Spectrophotometer, Axiom Mediral LMD. U.K.). Exchangeable K, Ca, Na and Mg were extracted with neutral (pH 7) solution of 1N NH4OAc. Potassium, Ca and Na were determined using the flame photometer (flame photometer model 2655-00 Digital Flame Analyzer, Cole-Parmer Instrument Company, Chicago, Illinois 60061) and Mg by the atomic absorption spectrophotometer (PG-900 Atomic Absorption Spectrophotometer Model, PG-instrument Ltd. United Kingdom).
2.3. Experimental Design
Experimental design was a 2 × 2 factorial arranged into a Completely Randomized Design consisting of two levels of plant methods: drilling and broadcasting, the two levels of seed density were 4 spoonful, 23.2 g 9 m−2 (25.78 kg·ha−1) for S. macrocarpon and 18.6 g 9 m−2 (20.67 kg·ha−1) for S. scabrum, and 8 table spoonful of seeds, 48.4 g 9 m−2 (53.78 kg·ha−1) for S. macrocarpon and 37.2 g 9 m−2 (41.33 kg·ha−1) for S. scabrum, it was replicated three times. Spacing was 3 m × 3 m per plot with 1 m spacing in between the plots. For planting by drilling, each plot was divided into twelve (12) rows with spacing of 0.25 m in between the rows. For four (4) spoonful seed, eight (8) spoons of dry fine sand were added to the seeds in a dry deep plastic container and for eight (8) spoons of seed, four spoons of dry fine sand were added to the seeds. Seed and sand were thoroughly mixed together and a spoon of the mixture was evenly spread per row on each plot. For planting by broadcasting, seed and sand were mixed together as stated above, and evenly spread on each plot. The vegetable seeds were obtained from the Agro dealer in Ile-Ife who purchased the seeds from vegetable farmers in Aba, Edo State, Nigeria. Sunshine organic fertilizer was obtained from the Ondo State Government, which contained 3.5%N, 1.00%P and 1.2%K was applied at 80 kg·N·ha−1 two weeks before planting. The first harvest of S. Scabrum shoot was carried out by cutting the stem at 6cm above the soil level at six weeks after planting while S. macrocarpon was harvested at eight weeks after planting. NPK 20-10-10 was applied at 80 kg·N·ha−1 immediately after the first harvesting of plant shoots. When Bemisia tabasi were observed on the vegetable plants, Neem (Azadicachta indica) leaf extract was used in controlling the insect pests. Young neem leaves were collected and rinsed with water. In the laboratory, 10 g of the neem leaves were weighed into reagent bottles each containing 100 ml of water and boiled in a water bath at 100˚C for five minute [23] . This is important to ensure that the active ingredients in the leaves were not denatured through overheating. The boiled ingredients were allowed to cool and then sieved to remove the leaves. The neem plant leaf extract was applied to the plants at 10% rate, two times at two weeks interval. The powdery colour and the pests disappeared from the plant leaves and the plant looked very healthy thereafter. The neem leaf extract was found to be very effective in controlling the insect pests. Neem leaf extract was applied starting from 14th Feb, 2013 (at seven weeks after planting) and was repeated once a week for four weeks consecutively. Plants were wetted ad libido early in the morning and in the evening starting at seed planting till March, 2013 when rains started. Weeds were removed by hand picking every week, no herbicides were used.
2.4. Data Collection
Solanum scabrum shoot was harvested at six weeks after planting (WAP) whereas Solanum macrocarpon shoot was harvested at eight WAP. The shoots of all vegetable plants on each plot were harvested by cutting stem at about 8 cm to the soil surface. Vegetables shoots were harvested at two weeks interval, the fresh weight determined and immediately transported to the Soil Science Laboratory of Obafemi Awolowo University, Ile-Ife, Nigeria. Fresh weight of harvested edible shoots was determined per plot.
2.5. Determination of Nutrient Composition
The vegetable leave samples were collected randomly from each harvested shoots. Vegetable samples were dried in a draft oven set at 70˚C in the Laboratory and 0.5 g of the vegetable tissues was digested using furnace for dry ashing according to the method [24] . The ash was extracted with 5 ml of 6N HCl and the extract was quantitatively transferred to 50 ml volumetric flask and made up to the mark. Appropriate dilutions were made and the elements analyzed against their standards. All the samples were analysed along with a bland solution. Calcium, K, Fe, Zn and Mn contents were determined using Atomic Absorption Spectrophotometer (PG-900 Atomic Absorption Spectrophotometer Model, PG-instrument Ltd. United Kingdom)equipped with an airacetylene flame and a hollow cathode lamp, and using lamps specific for each element. The device was operated under standard conditions using wavelengths and slit-widths specified for each element [25] . Plant tissue N content was determined using micro-Kjeldahl method according to [21] . Phosphorus content was determined using vanadomolybdate method [22] .
2.6. Data Analysis
Data collected were subjected to analysis of variance (ANOVA) to assess treatment effects and when it was significant means were separated using Fisher’s Least Significant Difference at 5% level of probability (LSD) [26] .
3. Results and Discussion
3.1. Characteristics of the Soil Used for the Study
Soil used in this study was a sandy loam which was slightly acidic (pH 5.27), medium organic carbon (0.93%), total N (0.15%) and available P (18.60 mg·kg−1) (Table 1). Exchangeable Ca, Mg, K and Na were 2.35, 1.44, 0.36, and 0.06 cmol·kg−1 soil, respectively, it fell within the medium fertility range with Na:K ratio of 1:6 [27] [28] .
Table 1. Properties of the soil (air dried) used for the experiment at 0 - 15 cm depth.
3.2. Effects of Planting Methods and Seed Density on Biomass Yield of S. macrocarpon and S. scabrum
The effects of planting methods and seed density on the shoot yields are shown in Figure 2(a) 1stplanting and (b) 2nd planting of S. macrocarpon and Figure 3(c) 1st planting and (d) 2nd planting of S. scabrum. S. macrocarpon shoot. Planting by drilling and at 4 table spoonful (25 g seeds) per 9 m−2 produced the highest shoot for S. scabrum. S. macrocarpon and S. scabrum have not been previously investigated with respect to the methods of planting and density of their seeds required per land area. S scabrum shoot yield increased with successive harvesting while S. macrocarpon yield decreased with increasing number of harvest. There were no significant differences when 4 table spoonful of S. macrocarpon seeds were planted either by drilling or by broadcasting. S. scabrum shoot yield obtained at 4 table spoonful when planting by drilling was about 26.09% higher than the yield when plant by broadcasting. The average cumulative shoot yield for the two vegetable types was approximately 2.0 kg·m−2 (20 ton·ha−1).
3.3. Effects of Planting Methods and Seed Density on Nutrient Contents of S. macrocarpon and S. scabrum
The effects of planting methods and seed density on nitrogen and phosphorus content of S. macrocarpon and S. scabrum leaves are illustrated in Figure 4(a) & Figure 4(b). Average values of 2.70% and 2.05% for S. macrocarpon and S. scabrum, respectively were obtained. Planting in drilling gave phosphorus content of 0.42% while broadcasting gave 0.12% for S. scabrum whereas values of 0.29 for drilling and 0.30 for broadcasting were obtained for S. macrocarpon. Seed density had no significant effect on N and P leave contents. Planting by drilling increases Ca, K, Fe and Zn of S. macrocarpon shoot and only Zn of S. Scabrum shoot significantly (Figure 5(a) to Figure 5(f)).This result is in agreement with previous finding that Solanum scabrum is a good source of zinc [29] . The average Ca (0.4%), Mg (1.6%), K (3.4%), Na (0.02%), Fe (300.8 mg·kg−1), Zn (103.5 mg·kg−1), Mn (300.7 mg·kg−1), and Cu (8.9 mg·kg−1) for S. macrocarpon and Ca (2.9%), Mg (3.0%), K (5.8%), Na (0.09%), Fe (280.5 mg·kg−1), Zn (53.0 mg·kg−1), Mn (359.4 mg·kg−1), and Cu (22.42 mg·kg−1) for S scabrum were obtained for the current study (Table 2). Depending on crop species, previous studies have established that plant nutrient composition requirements for optimal plant growth are between 2% and 5% for N, 0.3 and 0.5% for P, 0.1 and >5.0% for Ca and 2 and 5% for K [30] . Critical deficiency contents for Fe is 50 to 150 mg·kg−1, toxicity contents for Fe are above 500 mg·kg−1, deficiency contents for Manganese in plants are between 10 and 20 mg·kg−1 and toxicity varies widely between 200 and 5300 mg·kg−1. Critical toxicity contents for Cu concentration in plant leaves are between 20 and 30 mg·kg−1 [30] . Results of our study showed that the levels of nutrients in S. scabrum and S. macrocarpon fell within sufficiency levels for optimal growth and are less than the toxicity levels. S. scabrum and S. macrocarpon therefore, could serve as cheaper sources of macro and micro nutrients
 (a)
(a)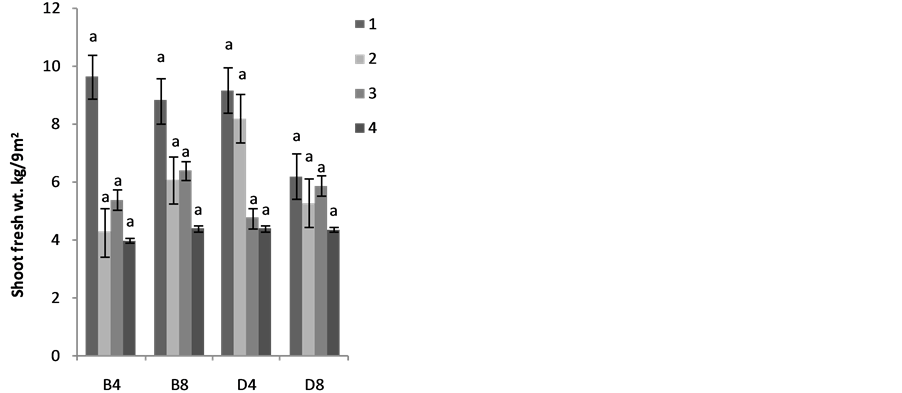 (b)
(b)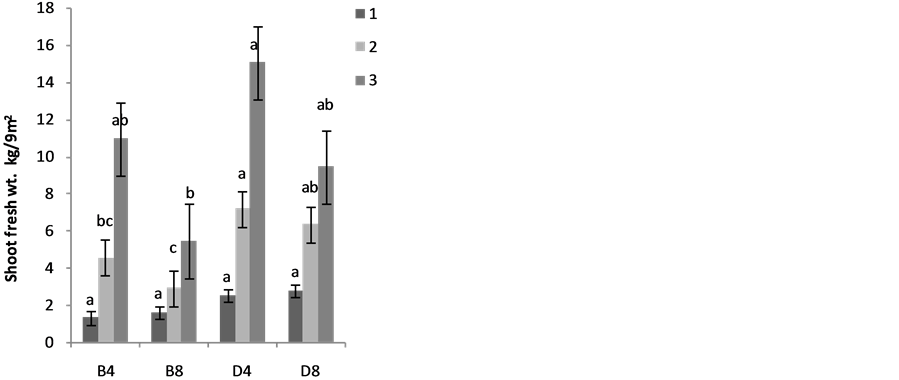 (a)
(a) (b)
(b)
Figure 2. Effects of planting method and seed density on (a) first planting and (b) second planiting of S. macrocarpon shoot yeild, and (c) first planting and (d) second planiting of S. scabrum shoot yeild. In this figure and subsequent one, B4 mean broadcast with 4 table spoonful of seed; B8 = broadcast with 8 table spoonful of seed; D4—drilling with 4 table spoonful of seed and D8 = drilling with 8 table spoonful of seed.
especially, Fe, Zn, and Mn for women and children in Nigeria and beyond. Calcium and phosphorus are important in bone, teeth and muscle metabolism [31] . Food is considered “good “if the Ca/P ratio is >1 but poor if <0.5. In our study, S. scabrum had higher Ca/P ratio compared with S. macrocarpon, and plant in drilling at 4 table spoonful seeds gave the highest value and was the best treatment for the vegetables (Table 3).
Furthermore, sodium and potassium are important intracellular and extracellular cations. The Na/K ratio is important in determining the health status of an individual. A ratio of less than one has been recommended to prevent high blood pressure [32] . In Table 3, our vegetables produced Na/K ratio less than one, which is in consonance with recommended ratio. Comparing S. marcroarpon and A. Scabrum with Amaranthus hybridus, a popular vegetable widely cultivated for commercial basis in peri-urban of Nigeria. Analyzsis of the chemical composition of three leafy vegetables in Imo State, Nigeria using undefatted and defatted methods [33] , undefatted Amaranthus hybridus contained 64.60 mg·g−1 Na, 11.05 mg·g−1 K, 13.68 mg·g−1 Mg, 28.80 mg·g−1 Ca, 87.50 mg·g−1 Fe and 0.99 mg·g−1 P, which gave a Na/K of 5.85. While defatted A. hybridus gave 24.80 mg·g−1 Na, 8.65 mg·g−1 K, 12.48 mg·g−1 Mg, 26.40 mg·g−1 Ca, 0.81 mg·g−1 Fe and 8.57 mg·g−1 P, which gave a Na/K of 2.87. The results of our study showed that S. macrocarpon and S. scabrum contained less sodium than A.
 (a)
(a)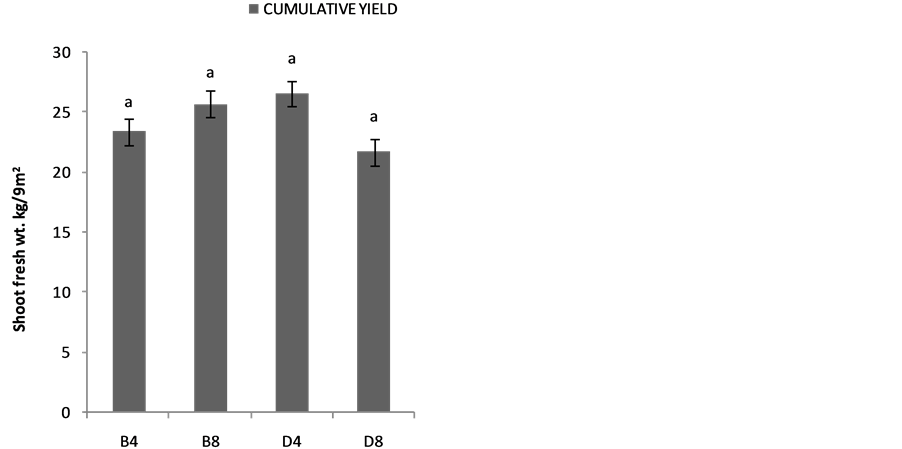 (b)
(b)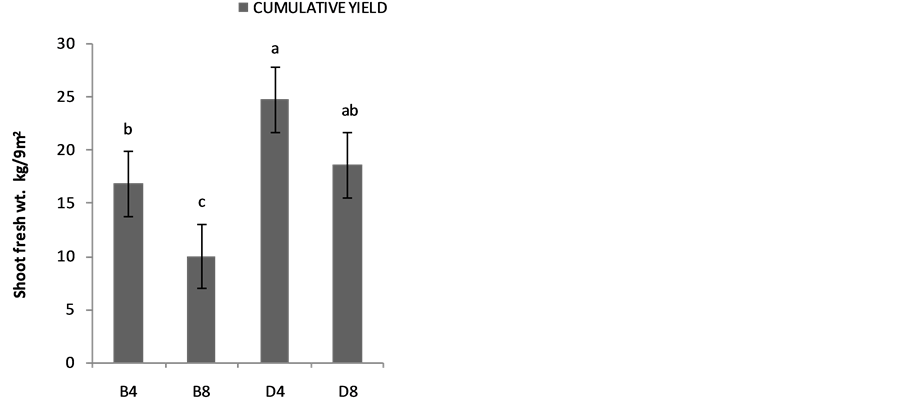 (c)
(c)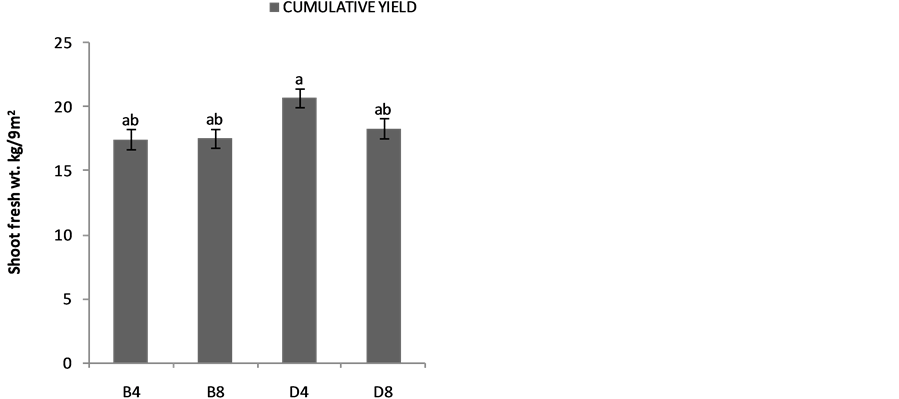 (d)
(d)
Figure 3. Effects of planting method and seed density on cummulative shoot yield (a) first planting and (b) second planting of S. Macrocarpon and (c) first planting and (d) second planting of S. Scabrum.
hybridus which is widely consumed in Nigeria.
4. Conclusion
The study revealed that planting methods and seed density affect yield and quality of edible shoots of S macrocarpon and S. scabrum. Average Fe (300.8 mg·kg−1), Zn (103.5 mg·kg−1), Mn (300.7 mg·kg−1), and Cu (8.9 mg·kg−1) for S. macrocarpon and Fe (280.5 mg·kg−1), Zn (53.0 mg·kg−1), Mn (359.4mg·kg−1), and Cu (22.42 mg·kg−1) for S scabrum were observed. S. macrocarpon and S. scabrum. The nutrient concentrations of the vegetables fell within the recommended levels for optimal plant growth and for human consumption. S. macrocarpon and S. scabrum are cheap sources of Ca, K, Mg, Fe and Zn. Planting at the seed rate of 4 table spoonfuls 9 m−2 planted by drilling. The study concluded that planting by drilling at 4 spoons, 23.2 g 9 m−2 (25.78 kg·ha−1) for S. macrocarpon and 18.6 g 9 m−2 (20.67 kg·ha−1) for S. scabrum was the optimum seed population. Planting by drilling would be more beneficial because fertilizer application, weeding, pests control, water application and harvesting would be carried out more efficiently compared with planting by broadcasting.
 (a)
(a)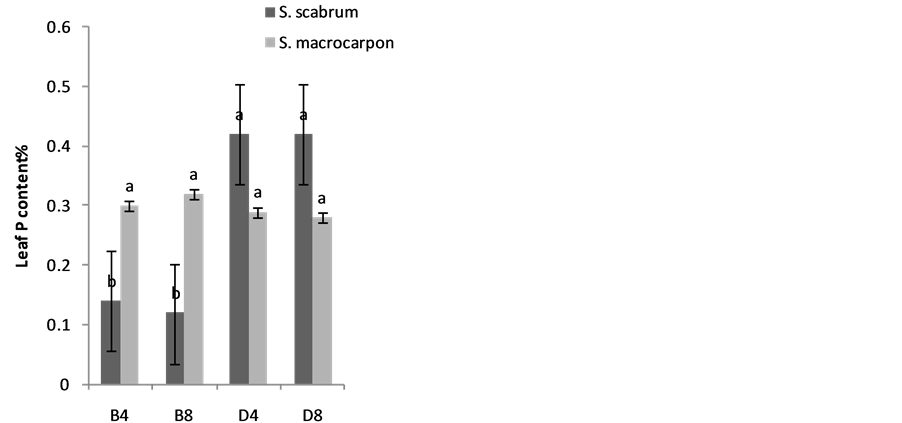 (b)
(b)
Figure 4. Effects of planting method and seed density on (a) N and (b) P contents of S. macrocarpon and S. scabrum Plant Shoot.
 (a)
(a)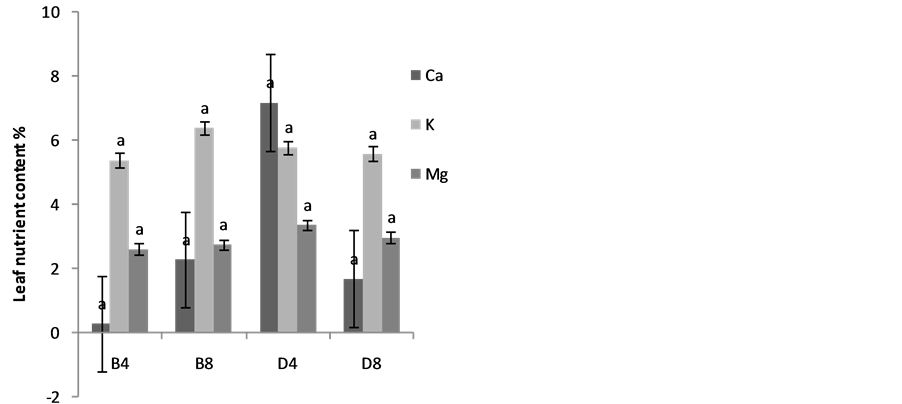 (b)
(b)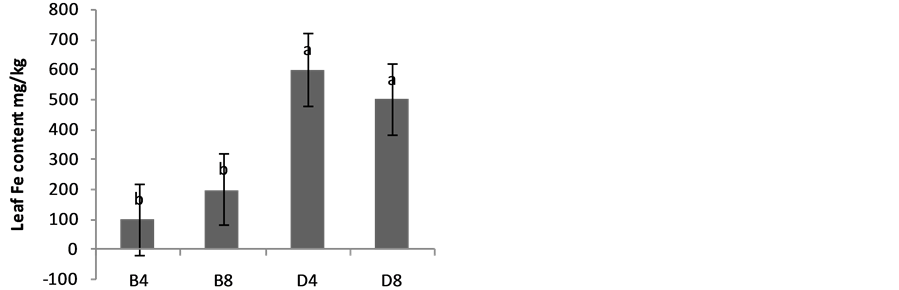 (c)
(c)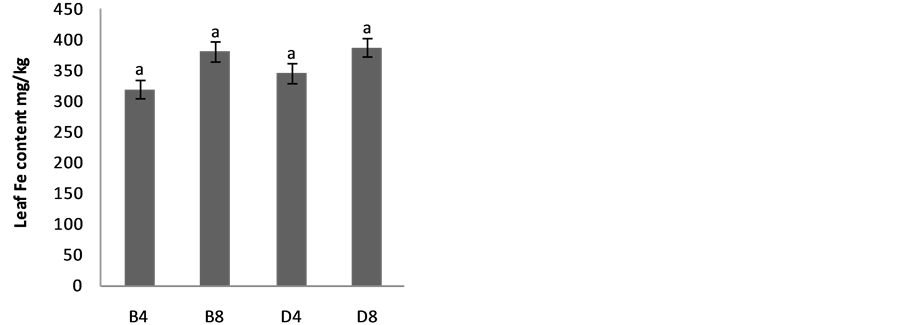 (d)
(d) (e)
(e) (f)
(f)
Figure 5. Effects of Planting method and seed density on Ca, K and Mg contents of (a) S. macrocarpon and (b) S. scabrum plant shoot, Fe, Zn, Mn & Cu content of (c & e) S. macrocarpon and (d & f) S. scabrum plant shoots.
Table 2. Average nutrient compositions of Solanum macrocarpon and Solanum scabrum.
Table 3. Effects of planting method and seed density on CA/P and NA/K ratios of S. scabrum and S. macrocarpon plant SHOOT.
PM = planting method; SD = seed density; In this table asterisk along the same column means a significant different “p < 0.05”.
Acknowledgements
The authors are grateful to the Foreign Affairs, Trade and Development Canada (DFATD) and International Development Research Centre (IDRC) for the project research grant number 106511. This study was a portion of the Nigeria-Canadian Vegetable (NI-CAN VEG) Research.
References
- Nishida, C., Uauy, R., Rumanyika, S. and Shetty, P. (2004) The Joint WHO / FAO Expert Consultation on Diet, Nutrition and the Prevention of Chronic Disease, Process, Product and Policy Implications. Public Health Nutrition, 7, 245-250. http://dx.doi.org/10.1079/PHN2003592
- http://nicanveg.org/sites/default/files/factsheets/NICANVEG_igbagba_factsheet_1.pdf
- Musyimi, D.M., Chemisto, J.K. and Buyela, D.K. (2012) Growth and Physiological Response of African Nightshades (Solanum scabrum Mill) to Sodium Chloride Salinity Stress. Global Advanced Research Journal of Food Science and Technology, 1, 066-073.
- Smith, F. and Eyzaguirre, P. (2007) African Leafy Vegetables: Their Role in the World Health Organization’s Global Fruit and Vegetables Initiative. African Journal of Food, Agriculture, Nutrition and Development, 7, 1-9.
- IFDC (2013) IFDC and Partners Empower Women in Bangladesh. Report, 38, 1-3.
- Idowu, M.K. (2008) Soil Fertility Management Strategies for Peri-Urban Vegetable Production in the Southwestern Nigeria. In: Adeboye, O.C., Taiwo, K.A. and Fatufe, A.A., Eds., Food, Health and Environmental Issues in Developing Countries: The Nigerian Situation, Alexander vonHumboldt Foundation, Cuvillier Verlag Gottinger, 20-28.
- Mwai, G.N., Onyango, M.O.A., Onyango, J.G.M., Chadha, L. and Oluoch, M.O. (2009b) Effect of Planting Density on Yield Components of Nightshade. Acta Horticulturae, 806, 733-739.
- Okafor, P.N. and Ogbona, U.I. (2003) Nitrate and Nitrite Contamination of Water Sources and Fruit Juices Marketed in South-Western Nigeria. Journal of Food Composition and Analysis, 16, 213-218.
- Bassir, O. and Maduagwu, E.N. (1978) Occurrence of Nitrate, Nitrite, Dimethylamine and Dimethylnitrosamines in Some Fertilized Nigerian Beverages. Journal of Agricultural and Food Chemistry, 26, 200-203. http://dx.doi.org/10.1021/jf60215a052
- Sen, N.P. and Baddon, P. A. (1997) Baddon Trends in the Levels of Residual Nitrite in Canadian Cured Meat Products over the Past 25 Years. Journal of Agriculture and Food Chemistry, 45, 4714-4718. http://dx.doi.org/10.1021/Jf9706641
- Olufolaji, A.O., Adeleye, F.O. and Ojo, O.O. (2013) Effect of Soil Moisture Stress on the Emergence, Establishment and Productivity of (Amaranthus cruentus L.). Agriculture and Biology Journal of North America. http://www.scihub.org/ABJNA
- Zingore, S. (2013) Site-specific nutrient management practices for smallholder farming systems. The R4 Nutrient Stewardship. International Plant Nutrition Institute (IPNI). Seminar Presented at the IPNI Scholar Award at the Obafemi Awolowo University, Ile-Ife, Nigeria on 11 December, 2013.
- Baker, J.L. (2013) Climate Change, Disaster Risk, and the Urban Poor Cities Building Resilience for a Changing World. Urban Development Series. The World Bank. Washington DC, 322 p. https://openknowledge.worldbank.org/bitstream/handle/10986/6018/683580PUB0EP-I0067869B09780821388457.pdf?sequence=1
- Agong, S.G. and Makinde, P.W. (2003) Improvement of the African Leafy Vegetables. Research Report Submitted to AURDC, Arusha.
- Smyth, A.J. and Mortgomery, R.F. (1962) The Soil and Land Use of Central Western Nigeria. The Government Printer, Ibadan, 265 p.
- Soil Survey Staff (1975) Soil Taxonomy. A Basic System of Soil Classification for Making and Interpreting Soil Surveys. U.S.D.A. Agricultural Handbook, 436.
- Okusami, T.A. and Oyediran, G.O. (1985) Slope-Soil Relationships on an Aberrant Toposeqence in Ife Area of Southwestern Nigeria: Variabilities in Soil Properties. Ife Journal of Agriculture, 7, 1-15.
- Bouyoucous, G.H. (1962) Hydrometer Method Improved for Making Particle Size Analysis of Soils. Agronomy Journal, 54, 464-465. http://dx.doi.org/10.2134/agronj1962.00021962005400050028x
- Thomas, G.W. (1996) Soil pH and Soil Acidity. In: Sparks, D.L., Ed., Methods of Soil Analysis, Part 3, Chemical Methods, SSSA and ASA, Madison, 475-490.
- Walkley, A. and Black, I.A. (1934) An Examination of the Degtareff Method for Determining Soil Organic Matter and a Proposed Modification of the Chromic Acid Titration Method. Soil Science, 37, 29-38.http://dx.doi.org/10.1097/00010694-193401000-00003
- Bremner, J.M. (1996) Total Nitrogen. In: Sparks, D.L., Ed., Methods of Soil Analysis, Part 3, Chemical Methods, SSSA and ASA, Madison, 1123-1184.
- Kuo, S. (1996) Phosphorus. In: Sparks, D.L., Ed., Methods of Soil Analysis, Part 3, Chemical Methods, SSSA and ASA, Madison, 869-920.
- Adekunle, O.K. and Fawole, B. (2003) Comparison of Effects of Extracts of Siam Weed, Neem and Carbofuran on Generation Time and Reproduction of Meloidogyne incognita Race 2 on Tomato. Environment and Ecology, 21, 720-726.
- Piper, C.S. (1944) Dry Ashing. Soil and Plant Analysis. Interscience, Publishers Inc., New York, 221-222.
- Association of Official Analytical Chemist (AOAC) (1990) Official Methods of Analysis of the Association of Official Analytical Chemist. 15th Edition, Association of Official Analytical Chemists, Washington DC.
- SAS, 9.1 Version 2002-2003.
- Idowu, M.K. and Aduayi, E.A. (2006) Effects of Sodium and Potassium Application on Water Content and Yield of Tomato in Southwestern Nigeria. Journal of Plant Nutrition, 29, 2131-2145. http://dx.doi.org/10.1080/01904160600972761
- Enwezor, W.O., Udo, E.J., Ayotade, K.A., Adepetu, J.A. and Chude, V.Q. (1990) A Review of Soil and Fertilizer Use Research in Nigeria. Federal Ministry of Agriculture, Water Resources and Rural Development, 109-200.
- Musyimi, D.M., Chemisto, J.K. and Buyela, D.K. (2012) Growth and Physiological Response of African Nightshades (Solanum scabrum Mill) to Sodium Chloride Salinity Stress. Global Advanced Research Journal of Food Science and Technology, 1, 066-073.
- Marschner, H. (1998) Functions of Mineral Nutrients. In: Marschner, H., Ed., Mineral Nutrition of Higher Plant, 2nd Edition, Academic Press, New York, 229-396.
- Akubagwo, I.E., Obasi, N.A. Chinyere, G.C. and Ugbogu, A.E. (2008) Amaranthus hybridus L and Solanum nigrum L Subjected to Different Processing Methods. African Journal of Biochemistry Research, 2, 040-044.
- F.N.D. (2002) Food and Nutrition Board, Institute of Medicine. National Academy of Sciences. Dietary Reference Intake for Energy, Carbohydrate, Fibre, Fat, Fatty Acids, Cholesterol, Protein and Amino Acid (Micronutrients).
- Iheanacho, K.M.E. and Udebuani, A.C. (2009) Nutritional Composition of Some Leafy Vegetables Consumed in Imo State, Nigeria. Journal of Applied Science and Environmental Management, 13, 35-38.
NOTES

*Corresponding author.


