Advances in Nanoparticles
Vol.2 No.4(2013), Article ID:38365,5 pages DOI:10.4236/anp.2013.24043
Dye-Sensitized Solar Cell with Fluorinated Gel Electrolyte: Effect of TiO2 Particle Size on Performance
1Akashi National College of Technology, Akashi, Japan
2Department of Electrical and Computer Engineering, Akashi National College of Technology, Akashi, Japan
Email: ohmukai@akashi.ac.jp
Copyright © 2013 Jun Kyokane, Masato Ohmukai. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received August 27, 2013; revised September 29, 2013; accepted October 11, 2013
Keywords: Ruthenium Ion Complex; Dye-Sensitized Solar Cell; Fluorinated Oligomer; Gel Electrolyte
ABSTRACT
We fabricated dye-sensitized solar cells including fluorinated gel electrolyte and studied about the relationship between the performance of the solar cells and the aggregation state of TiO2 nano-particles on electrodes. As for the performance of the cell, the I-V characteristics were measured under irradiation. The combination of TiO2 nanoparticles with different size plays an important role in bringing unevenness to realize a large surface area, which is critical for the high performance of the cells.
1. Introduction
Dye-sensitized solar cells have attracted much attention these twenty years, since this new type of solar cells without any pn junctions was reported [1,2] in 1991 for the first time. The vast amount of efforts has been devoted to the improvement of its performance [3,4]. The conversion efficiency has reached ten percent [5] and so the commercial application is now expected strongly. In order to replace this kind of solar cells with the silicon ones, the efficiency requires the same level of silicon solar cells at least.
The other big problem of the dye-sensitized solar cells is the stability. Since the cell includes electrolyte between two electrodes, the leakage of the electrolyte inevitably limits its stability. As a preferable idea to prevent the electrolyte leakage, gelation of the electrolyte has been proposed [6,7]. Some researchers have been challenging with the cross-linking of a polymer [8]. We have been engaged on the other hand in the application of fluorinated gel electrolytes that have already been proposed to apply to a secondary battery [9]. The fluorinated gel electrolyte is cohesively derived from intermolecular force between the fluorinated alkyl groups and ionic bonding at the same time. This kind of gel does not mainly require bridging process induced by e.g. ultraviolet irradiation in order to form cross linking network, having an advantage of a simple and low-cost gelation procedure from the viewpoint of manufacturing.
Our dye-sensitized solar cells consist of two transparent electrodes, between which the gel electrolyte is put, where one of the electrode was covered with the aggregate of TiO2 nano-particles adsorbing dye. The aggregated nanoparticle TiO2 layer—often addressed mesoporous TiO2—is one of the critical part of the solar cell [5,10]. The substrates spread with TiO2 are often calcined at about 500˚C [11,12], some researcher have tried to lower the treatment temperature [13] by the irradiation of microwave [14], laser [15] or ultraviolet [16]. In addition, spray coating of TiO2 followed by pressure to form the porous TiO2 layer [17] and electrophoretic deposition of TiO2 [18] have also been challenged so far. The introduction of nano dots of CdSe [19] or PbS [20] in the porous TiO2 layer, as well as Ag particles [21], has also been studied. Other materials such as SnO2 [22-24], ZnO [25] or NiO [26] have also been investigated instead of the TiO2 single layer.
We fabricated the cells with a variety of conditions of the TiO2 layer formation and studied the influence on the performance of the cells from the point of view of I-V characteristics. The state of the TiO2 aggregate is expected to affect the contact to dye and gel electrolyte at the same time. We evaluate in this article a basic performance of the dye-sensitized solar cell including fluorinated gel electrolyte. The application of the fluorinated gel to dye-sensitized solar cells is not well studied yet. It will be able to trigger a break through to a difficult problem obstructing the development.
2. Experimental Details
We mixed 1 g of slurry including 30 wt·% of TiO2 powder (P4TH055 or P7TH055 offered by Sumitomo Titanium) with polyethylene glycol (PEG) to make paste with the help of the addition of Triton X (a nonionic surfactant) of 15 μl to increase viscosity. The difference between P4TH055 and P7TH055 is the particle size of TiO2 dispersed in the slurry. The size is not claimed clearly (50 - 70 nm) though the particle size is larger in P7TH- 055. In some cases, we added P-25 of titania nano particles (20 nm) to the paste. The paste was spread on a fluorine doped tin oxide (FTO) substrate at the thickness of 58 μm. This was then annealed at 450˚C [12] for 30 minutes to remove binders and solvent to be a photocathode. The TiO2 deposited substrate was soaked in a solution of ruthenium complex dye (Ru (4,4’-dicarboxy-2,2’bipyridine)2(NCS)2) in ethanol (1 × 104 mol/l) at 50˚C for 2 hours to adsorb the dye. The counter electrode (anode) of an FTO substrate was sputtered with a Pt target for 3 minutes using a portable sputtering apparatus of VPS- 020 by ULVAC. The thickness of Pt was confirmed to be about 10nm by means of an interference microscope.
We describe here the preparation of gel electrolyte. Dimethyl sulfoxide was mixed with LiI (10 mmol/g) for 20 minutes with the help of ultrasonic agitation. We then added 2-acrylamid-2-methyl-propanesulfonic acid (AMPS) to this solution and mixed for 200 minutes further. The AMPS is one of fluorinated oligomers [9] that has the structure shown in Figure 1. The amount of AMPS added for gelation should be roughly larger than 330 g/l; the critical gelation density that was obtained experimentally. If the density of the AMPS is too low, spontaneous cohesion is possibly hard to occur owing to a large distance between each oligomer. The image of the gelation state of AMPS oligomers are shown in Figure 2. We finally formed a solar cell by putting the gel electrolyte between a photocathode and an anode and sealed the environs of the cell with epoxy resin.
We evaluated the I-V characteristics of the solar cells by measuring the current and voltage appeared at the cell

Figure 1. Structure of AMPS; a kind of fluorinated oligomers. The RF in the figure stands for a fluoroalkyl group that acts as intra-group cohesion.
when a variable resistive load was connected to the cell that was irradiated by white light (AM 1.5) from a filtered xenon lamp (UXL500SX by Ushio) at the intensity of 55 mW/cm2. The resistive load was varied between 0 and 100 kΩ.
3. Results and Discussion
We show I-V characteristics in Figures 3 and 4 corresponding to the slurry of P4TH055 and P7TH055, respectively. The each figure shows the three kinds of I-V characteristics at the PEG amount of 10, 20 and 30 wt·%. The both figures show typical electric generating properties as a solar cell. The open-circuit voltage is 0.42 or 0.44 V in Figure 3 and 0.5 in Figure 4. The open-circuit voltage is not dependent of the PEG concentration. On the other hand, the voltage is larger with the larger particle size of the TiO2, which is clear from the comparison of the two figures. If the size of TiO2 particle is small, the excited electrons at a dye adsorbed at the TiO2 particle surface experience the strong image force from the TiO2, though it cannot be quantitatively discussed here. The idea is consistent with the fact that the open-circuit voltage is independence of the PEG concentration.

Figure 2. The image of the gelation state of AMPS oligomers.
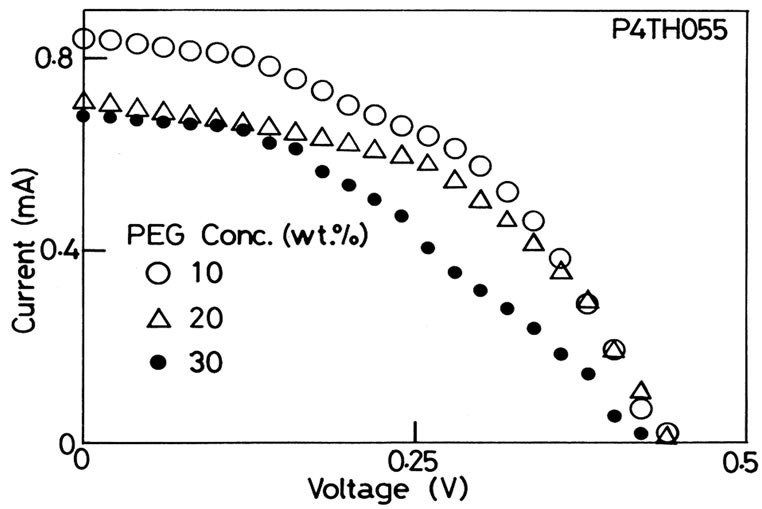
Figure 3. The I-V characteristics when slurry of P4TH055 was used. The open-circuit voltage was not varied regardless of the concentration of PEG. The shunt-circuit current, on the other hand, was largest when the concentration of PEG was 10 wt·%.
We next pay attention to the shunt-circuit current in Figures 3 and 4. Figure 4 shows the larger shunt-circuit current by about 50% compared with those in Figure 3. With the larger particle size, it needs the less number of contacts between the TiO2 particles. We surmise that the bigger particles are more preferable to build continuous paths from the dye to the photocathode of FTO. The increase of PEG amount suppresses the shunt-circuit current in both cases. As the PEG concetration increases, the particles tend to disperse in the slurry so far away not to gather together, which results in the low density of contacting of the TiO2 particles. This obstructs the forming of path between the dye and the photocathode. The fact supposedly affects the density of contact between the TiO2 particles. The maximum generating electric power is shown in Figure 5 as a function of PEG concentration. We can conclude that P7TH055 with 10 wt·% of PEG provided the highest generating electric power in these experiments.
We performed the similar experiments that we fixed

Figure 4. The I-V characteristics when slurry of P7TH055 was used. It shows a similar tendency to Figure 1, except for the fact that the values of the open-circuit voltage and the shunt-circuit current are larger than those shown in Figure 1.

Figure 5. The optimum electric generating power as a function of the concentration of PEG.
the ratio of PEG to be 10 wt·% and added P-25 (10 - 100 wt·% of the TiO2 in the slurry) and obtained the interesting results shown in Figures 6 (P4TH055) and 7 (P7TH- 055). The open-circuit voltages fitted in the region between 0.4 and 0.5 V in the both cases, but were scat-tered in the region. The variation of the open-circuit voltage is the effect of addition of the nano particles with a different size. The surface morphology of the TiO2 where the dye is adsorbed becomes irregular by adding the different-size TiO2 particles. We found the prominent feature in the shunt-circuit current in the both cases. The shuntcircuit current is about 4 times as large by adding 100 wt·% of P-25 compared with that of the 10 wt·% addition of P-25. Figure 8 shows the maximum electric generating power as a function of the concentration of P-25. The P-25 seems to play a role to be filled up in the void and to help to form a path easily between the dye and the photocathode. It appears the mixing of nano TiO2 particles of different sizes is critical to enhance the current. The difference in size between P4TH055 and P-25 is

Figure 6. The I-V characteristics when slurry of P4TH055 was used. It shows the characteristics depends noticeably on the concentration of P-25.
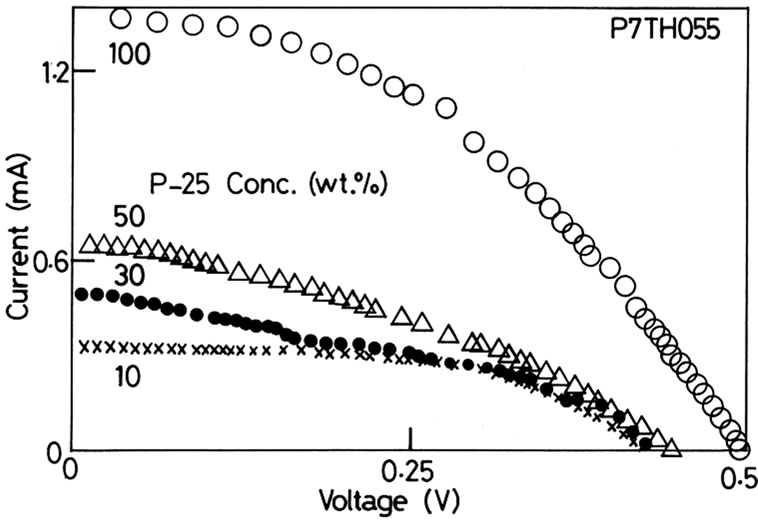
Figure 7. The I-V characteristics when slurry of P7TH055 was used. It shows the similar tendency to Figure 4.
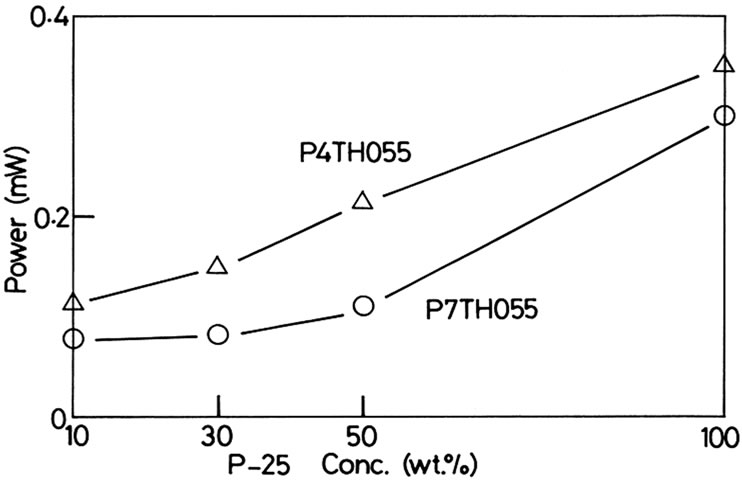
Figure 8. The optimum electric generating power as a function of concentration of P-25.
larger than that between P7TH055 and P-25. From the fact that the generation power became the smaller when P7TH055 was used, too large difference in size of the mixed particles lowers the generating power.
To summarize the results, the slurry of P7TH055 (the larger size) realize the higher electric generating power than that of P4TH055 and both slurry tended to show that the increase of PEG concentration reduced the power. In case of adding P-25 titanium powder, the power was suppressed compared with the case without P-25, but the power increases drastically with the addition of P-25 until 100 wt·%. With the P-25 addition, the power is larger when we used the slurry of P4TH055. We consider that the balance of the mixing of TiO2 particles with a different size affect the performance of the solar cell elaborately. In all cases, the open-circuit voltage was not varied especially when P-25 was not added. Only the shunt-circuit current was varied noticeably. It is surely because the TiO2 aggregation state determines the number of dyes covered on TiO2 and the number of path between the dye and an FTO electrode at the same time. It is desirable to enhance the both factors at the same time, but this is a kind of antinomy.
Y. Saito et al. reported [27] the interesting results of the correlation between morphology and solar cell characteristics. They concluded that roughness factor of the films is desired to be large until 1400 to obtain the larger coverage of the adsorbed dye with the larger photocurrent. This did not depend on the nano particle size according to their results. It appears that our results do not agree with theirs at a glance. It should be noticed, however, that the mixing condition is quite different as follows. In our experimental conditions, the amount of P-25 is under 50% of the total TiO2; the fine particles of P-25 were imbedded interstitially. Their experiments were contrary performed under the condition that large particles bigger than 100 nm were dispersed in the medium of fine particles such as P-25. We mixed the particles in different size, but the sizes are in the same degree.
4. Conclusion
We fabricated dye-sensitized solar cells with a fluorinated gel electrolyte, and investigated the effect of aggregation state of TiO2 particles from the point of view of I-V characteristics. The increase in the PEG amount lowered the generating power. This suggested that the too much PEG obstructs the formation of electric path between the adsorbed dye and an FTO electrode. The slurry of P7TH055 is more favorable, which means that the larger particles are easier to form continuous paths. The addition of P-25 about 100 wt·% drastically increased the generating power. The mixing or the particles in different size noticeably affected the cell performance. The difference of the two kinds of particles is too large, and the mixing effect is less effective.
5. Acknowledgements
The authors wish to thank Prof. Hideo Sawada for the preparation of fluorinated oligomer of AMPS used in our experiments.
REFERENCES
- B. O’Regan and M. Grtäzel, “A Low-Cost, High-Efficiency Solar Cell Based on Dye-Sensitized Colloidal TiO2 Films,” Nature, Vol. 353, No. 1991, pp. 737-740. http://dx.doi.org/10.1038/353737a0
- M. K. Nazeeruddin, A. Kay, I. Rodicio, R. H. Baker, E. Mueller, P. Liska, N. Vlachopoulos and M. Grtäzel, “Conversion of Light to Electricity by Cis-X2bis(2,2'- bipyridyl-4,4'-dicarboxylate)ruthenium(II) Charge-Transfer Sensitizers (X = Cl-, Br-, I-, CN-, and SCN-) on Nanocrystalline Titanium Dioxide Electrodes,” Journal of the American Chemistry Society, Vol. 115, No. 14, 1993, pp. 6382-6390. http://dx.doi.org/10.1021/ja00067a063
- B. Li, L. Wang, B. Kang, P. Wang and Y. Qiu, “Review of Recent Progress in Solid-Satate Dye-Sensitized Solar Cells,” Solar Energy Materials and Solar Cells, Vol. 90, No. 5, 2006, pp. 549-573. http://dx.doi.org/10.1016/j.solmat.2005.04.039
- M. Grtäzel, “Recent Advances in Sensitized Mesoscopic Solar Cells,” Accounts of Chemical Research, Vol. 42, No. 11, 2009, pp. 1788-1798. http://dx.doi.org/10.1021/ar900141y
- Y .Chiba, A. Islam, Y. Watanabe, R. Komiya, N. Koide and L. Han, “Dye-Sensitized Solar Cells with Conversion Efficiency of 11.1%,” Japanese Journal of Applied Physics, Vol. 45, 2006, pp. L638-L640. http://dx.doi.org/10.1143/JJAP.45.L638
- Y. H. Lai, C. Y. Lin, J. G. Chen, C. C. Wang, K. C. Huang, K. Y. Liu, K. F. Lin, J. J. Lin and K. C. Ho, “Enhancing the Performance of Dye-Sensitized Solar Cells by Incorporating Nanomica Platelets in Gel Electrolyte,” Solar Energy Materials and Solar Cells, Vol. 94, No. 4, 2010, pp. 668-674. http://dx.doi.org/10.1016/j.solmat.2009.11.027
- S. Murai, S. Mikoshiba and S. Hayase, “Influence of Alkyl Dihalide Gelators on Solidification of Dye-Sensitized Solar Cells,” Solar Energy Materials and Solar Cells, Vol. 91, No. 18, 2007, pp. 1707-1712. http://dx.doi.org/10.1016/j.solmat.2006.12.018
- T. C. Wei, C. C. Wan and Y. Y. Wang, “Preparation and Characterization of a Micro-Porous Polymer Electrolyte with Cross-Linking Network Structure for Dye-Sensitized Solar Cell,” Solar Energy Materials and Solar Cells, Vol. 91, No. 20, 2007, pp. 1892-1897. http://dx.doi.org/10.1016/j.solmat.2007.07.005
- J. Kyokane, K. Shima and H. Sawada, “Electrical Properties of Fluorinated Gel Electrolytes Using High Ionic Conducting Solution and Its Application to Secondary Battery,” Thin Solid Films, Vol. 438-439, 2003, pp. 257- 261. http://dx.doi.org/10.1016/S0040-6090(03)00794-6
- X. Wu, L. Wang and Y. Qiu, “In Situ Synthesis of Mesoporous TiO2 Anatase Films and Their Photovoltaic Performance in Dye Sensitized Solar Cells,” Japanese Journal of Applied Physics, Vol. 45, 2006, pp. L1149- L1151. http://dx.doi.org/10.1143/JJAP.45.L1149
- T. V. Nguyen, H. C. Lee and O. B. Yang, “The Effect of Pre-Thermal Treatment of TiO2 Nano-Particles on the Performances of Dye-Sensitized Solar Cells,” Solar Energy Materials and Solar Cells, Vol. 90, No. 7-8, 2006, pp. 967-981. http://dx.doi.org/10.1016/j.solmat.2005.06.001
- K. M. Lee, V. Suryanarayanan and K. C. Ho, “A Study on the Electron Transport Properties of TiO2 Electrodes in Dye-Sensitized Solar Cells,” Solar Energy Materials and Solar Cells, Vol. 91, No. 15-16, 2007, pp. 1416-1420. http://dx.doi.org/10.1016/j.solmat.2007.03.007
- M. Murayama, E. Yamazaki, N. Nishikawa, N. Hashimoto and T. Mori, “Low-Temperature Fabrication of TiO2 Necking Electrode by Sol-Gel Method and Its Application to Dye-Sensitized Solar Cell,” Japanese Journal of Applied Physics, Vol. 45, 2006, pp. 7917-7921. http://dx.doi.org/10.1143/JJAP.45.7917
- S. Uchida, M. Tomiha, N. Masaki, A. Miyazawa and H. Takizawa, “Preparation of TiO2 Nanocrystalline Electrode for Dye-Sensitized Solar Cells by 28GHz Microwave Irradiation,” Solar Energy Materials and Solar Cells, Vol. 81, No. 1, 2004, pp. 135-139. http://dx.doi.org/10.1016/j.solmat.2003.08.020
- G. Mincuzzi, L. Vesce, A. Reale, A. D. Carlo and T. M. Brown, “Efficient Sintering of Nanocrystalline Titanium Dioxide Films for Dye Solar Cells via Raster Scanning Laser,” Applied Physics Letters, Vol. 95, No. 10, 2009, Article ID: 103312. http://dx.doi.org/10.1063/1.3222915
- L. N. Lewis, J. L. Spivack, S. Gasaway, E. D. Williams, J. Y. Gui, V. Manivannan and O. P. Siclovan, “A Novel UV-Mediated Low-Temperature Sintering of TiO2 for Dye-Sensitized Solar Cells,” Solar Energy Materials and Solar Cells, Vol. 90, No. 7-8, 2006, pp. 1041-1051. http://dx.doi.org/10.1016/j.solmat.2005.05.019
- J. Halme, J. Saarinen and P. Lund, “Spray Deposition and Compression of TiO2 Nanoparticle Films for Dye-Sensitized Solar Cells on Plastic Substrates,” Solar Energy Materials and Solar Cells, Vol. 90, No. 7-8, 2006, pp. 887-899. http://dx.doi.org/10.1016/j.solmat.2005.05.013
- S. M. A. Fateminia, R. Y. Rad, T. Ebadzadeh and S. Ghashghai, “Effect of Dispersing Media on Microstructure of Electrophoretically Deposited TiO2 Nanoparticles in Dye-Sensitized Solar Cells,” Applied Surface Science, Vol. 257, No. 20, 2011, pp. 8500-8505. http://dx.doi.org/10.1016/j.apsusc.2011.05.002
- L. J. Diguna, M. Murakami, A. Sato, Y. Kumagai, T. Ishihara, N. Kobayashi, Q. Shen and T. Toyoda, “Photoacoustic and Photoelectrochemical Characterization of Inverse Opal TiO2 Sensitized with CdSe Quantum Dots,” Japanese Journal of Applied Physics, Vol. 45, 2006, pp. 5563-5568. http://dx.doi.org/10.1143/JJAP.45.5563
- B. Ma, F. Luo, L. Wang, X. Wu, C. Zhan and Y. Qiu, “Structure and Morphology of Nanosized PbS on TiO2 Nanoporous Film in Dye-Sensitized Solar Cells,” Japanese Journal of Applied Physics, Vol. 46, 2007, pp. 7745-7748. http://dx.doi.org/10.1143/JJAP.46.7745
- C. Wen, K. Ishikawa, M. Kishima and K. Yamada, “Effects of Silver Particles on the Photovoltaic Properties of Dye-Sensitized TiO2 Thin Films,” Solar Energy Materials and Solar Cells, Vol. 61, No. 4, 2000, pp. 339-351. http://dx.doi.org/10.1016/S0927-0248(99)00117-8
- S. Chappel and A. Zaban, “Nanoporous SnO2 Electrodes for Dye-Sensitized Solar Cells Improved Cell Performance by the Synthesis of 18 nm SnO2 Colloids,” Solar Energy Materials and Solar Cells, Vol. 71, No. 2, 2002, pp. 141-152. http://dx.doi.org/10.1016/S0927-0248(01)00050-2
- W. P. Tai, K. Inoue and J. H. Oh, “Ruthenium Dye-Sensitized SnO2/TiO2 Coupled Solar Cells,” Solar Energy Materials and Solar Cells, Vol. 71, No. 4, 2002, pp. 553-557. http://dx.doi.org/10.1016/S0927-0248(01)00140-4
- W. P. Tai, “Photoelectrochemical Properties of Ruthenium Dye-Sensitized Nanocrystalline SnO2:TiO2 Solar Cells,” Solar Energy Materials and Solar Cells, Vol. 76, No. 1, 2003, pp. 65-73. http://dx.doi.org/10.1016/S0927-0248(02)00250-7
- S. Rani, P. Suri, P. K. Shishodia and R. M. Mehra, “Synthesis of Nanocrystalline ZnO Powder via Sol-Gel Route for Dye-Sensitized Solar Cells,” Solar Energy Materials and Solar Cells, Vol. 92, No. 12, 2008, pp. 1639-1645. http://dx.doi.org/10.1016/j.solmat.2008.07.015
- F. Vera, R. Schrebler, E. Munoz, C. Suarez, P. Cury, H. Gomez, R. Cordova, R. E. Marotti and E. A. Dalchiele, “Preparation and Characterization of Eosin Band Erythrosine J-Sensitized Nanostructured NiO Thin Film Photocathodes,” Thin Solid Films, Vol. 490, No. 2, 2005, pp. 182-188. http://dx.doi.org/10.1016/j.tsf.2005.04.052
- Y, Saito, S. Kambe, T. Kitamura, Y. Wada and S. Yanagida, “Morphology Control of Mesoporous TiO2 Nanocrystalline Films for Performance of Dye-Sensitized Solar Cells,” Solar Energy Materials and Solar Cells, Vol. 83, No. 1, 2004, pp. 1-13. http://dx.doi.org/10.1016/j.solmat.2004.02.010

