Computational Water, Energy, and Environmental Engineering
Vol.3 No.1(2014), Article ID:42114,5 pages DOI:10.4236/cweee.2014.31005
Synthesis and Characterization of TiO2 Porous Films for Heterogeneous Photocatalysis
1Instituto de Ingenieria, Universidad Nacional Autonóma de México, México DF, México
2Facultad de Estudios Superiores Cuautitlan, Universidad Nacional Autonoma de Mexico, México DF, México
Email: mmjc_80@yahoo.com.mx
Received November 27, 2013; revised December 27, 2013; accepted January 4, 2014
ABSTRACT
Heterogeneous photocatalysis is one of the advanced oxidation processes (AOP’s). These are very used nowadays for their implementation in tertiary water treatment with solar activation, with very good results. In this study, TiO2 porous films were synthesized by sol-gel method. Polyethylene glycol (PEG) 4000 was used as a templating reagent, which is oxidized at a lower temperature than TiO2. This allows the formation of a “skeleton” of porous TiO2 with increased surface area, resulting in higher photocatalytic activity. The film characterization was made with help of FE-SEM microscope, XRD diffractometer and scotch tape test. Finally, the oxidation experiments were performed using methylene blue as a model pollutant and they revealed an increase in the photocatalytic activity of porous films in comparison with the non-porous films.
Keywords:TiO2 Porous Films; Sol-Gel Method; Photocatalysis; PEG
1. Introduction
The world situation nowadays demands and immediate solution to problems related with contaminated water. In the last decades, the scientific research has focused its attention in this topic and has developed a wide spectrum of processes in the water treatment technologies [1,2]. This works has resulted in implementing different methods on water treatment systems: pre-treatment, and primary, secondary and tertiary treatments. Within the last set of processes, advanced oxidation processes (AOP) are found, like photocatalytic oxidation (PCO).
1.1. Principle of Heterogeneous Photocatalysis
Heterogeneous photocatalysis (which is part of the AOP) appears as an emerging technology in the domain of the environment for air and water treatment [3]. When a semiconductor catalyst SC (oxide or sulfide) is illuminated whit photons whose energy is equal to or greater than their band gap energy Egap (hν ≥ Egap), there is absorption of these photons and creation within the bulk of electron-hole pairs, which dissociate into free photoelectrons in the conduction band and photo-holes in the valence band. In the presence of a fluid phase (gas or liquid), a spontaneous adsorption occurs and according to the redox potential (or energy level) of each absorbate, an electron transfer proceeds towards acceptor molecules, whereas positive photo-holes are transferred to donor molecules. Each ion formed subsequently reacts to form the intermediate and final products [3].
1.2. Sol-Gel Process
The sol-gel process is a method very used in the development of the TiO2 films with photocatalytic purposes; its main advantage is that expensive equipment and difficult process are not necessaries to prepare the films.
The sol-gel process involves, first, the formation of a sol and, after, the formation of a gel, a solid encapsulated into a solvent. The formation of a gel is by hydrolysis and condensation [4].
The sol-gel process consists of four steps: 1) chemical solution, 2) aging, 3) drying, and 3) sintering [5].
1.3. TiO2 Films
The most used photocatalyst is Titanium dioxide (TiO2), a semiconductor with a band gap energy equal to 3.2 eV, excellent chemical stability over a wide pH range and in a large number of solvents, and economically adequate [6]. TiO2 is a material of choice for photocatalytic oxidation and several other photochemical applications and it exists in three principal crystallographic forms, named anatase, brookite and rutile. Only anatase and rutile are photocatalytically active. TiO2 can be used in PCO either as slurry or as a deposited film on suitable substrates such as silica and soda lime glasses, Si wafers and stainless steel. The latter situation avoids separation steps, what are energy demanding processes.
For the past few years, TiO2 nanostructured films/arrays have been normally obtained through modified solgel routes in the presence of templates. Among those templates, polyethylene glycol (PEG) is a traditional pore-forming one commonly used for the synthesis of porous TiO2 films [7]. The pores are generated by the evaporation of the solvent. The colloidal templates are infiltrated with the precursor solution, and subsequently removed by the calcination or the solvent extraction to create porous materials [6].
2. Experimental
2.1. Preparation of Porous TiO2 Films
Based on the works of Huang and Morales [7,8], TiO2 porous films were prepared by sol-gel method. The precursor solutions were based on titanium tetrabutoxide 0.5M (TiBu, Sigma-Aldrich), ethanol, water and acetylacetone as solvents, and hydrochloric acid as catalyst. The elements were mixed up with different molar ratios (expressed as BuTi:EtOH:AcAc:H2O:HCl).
To obtain several nano-structured surfaces, Polyetilenglycol (PEG 4000) was added in different molar concentrations (0, 0.009, 0.0012, 0.0015, 0.0018) mol/L, so there were obtained 5 different liquid precursors.
Each of the precursors generated samples with tree different quantities of TiO2, 15 samples in total (Table 1). Each of the Titanium Dioxide sols that were obtained this way was applied on glass plates (76 × 26 mm), previously covered by SiO2 (by sol-gel method, tip-coating). The plates were then air dried and sintered at 605˚C for 2 h in air atmosphere.
2.2. Characterization
The surface morphology of the TiO2 films was examined by a JEOL JSM-7600F field emission scanning electron (FE-SEM), with an EDS detector (Oxford INCA XACT). The FE-SEM microscope is locates at IIM-UNAM. The titanium dioxide films were also characterized by powder X-ray diffraction (XRD) with D5MEAS equipment that uses Cu K as a source of radiation. The adherence of the samples was determined by scotch-tape test according to [9]. Finally, the sample films were tested in a solar simulator Sun-Test XLS + ATLAS, with UV radiation (300 - 400 nm) and irradiance of 30 W/m2.
3. Results and Discussion
3.1. FESEM Analysis
The results obtained are showed in Figures 1, 2 and 3, which are FESEM images obtained with a magnification of 50,000. In Figure 1 it is noted a tendency of formation of a number of pores, which is proportional to the amount of PEG added to the precursors. In A1 samples there is no evidence of porosity because the surface mor
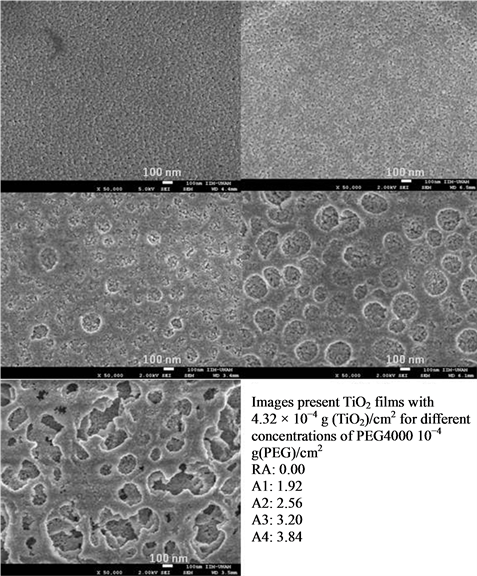
Figure 1. FE-SEM images of a film prepared with A precursor samples.
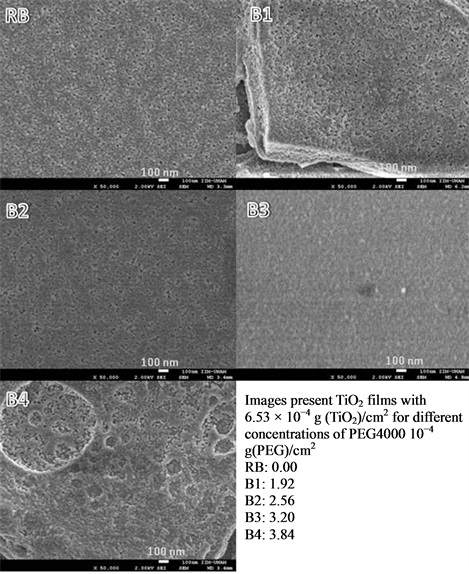
Figure 2. FE-SEM images of a film prepared with B precursor samples.
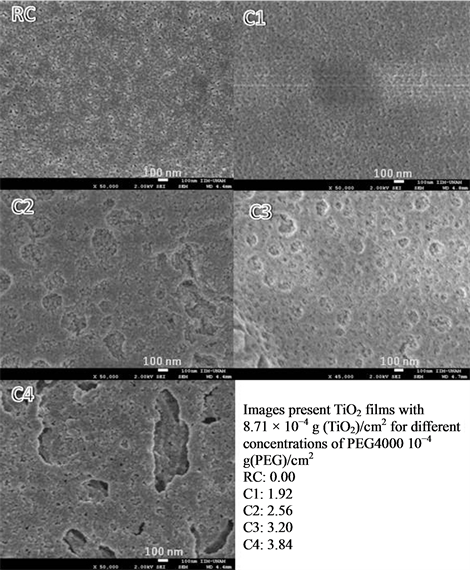
Figure 3. FE-SEM images of a film prepared with C precursor samples.
phology is very similar to RA sample (without PEG). In films A2 and A3 there is a tendency on pore size, relative to the amount of PEG. For the sample A2, it shows an average pore size of 104 ± 5 nm for the sample A3 and 208 ± 5 nm. In the sample A4, several cracks are formed, possibly due to the union of adjacent pores. In B and C samples of Figures 2 and 3, a similar effect was observed for the A1 film, where it seems to be an overlapping of pores; even more, at macroscopic scale, there is evidence of significant fractures that could affect the photocatalytic activity.
TiO2 particles are, for all samples of precursors, nanosized, with estimated particle size ranging 10 to 30 nm (based on SEM images). The analysis of XRD data for 2 theta values of 25.5, 27.5 and 37.5 degrees (which are typical double diffraction angles values for TiO2) by Scherrer Equation indicates that particles measure 30.94 nm when there is only TiO2 on the films, and average size decreases to 12.40 nm when PEG-4000 was added.
3.2. Scotch Tape Test
The main results of adhesion test based on ASTM [9], are showed in the Table2 This procedure classifies adherences from A to C, where A is excellent and C is poor. For purposes of heterogeneous photocatalysis, where the samples are often in direct contact with flowing water, 4B classification is sufficient. As C films are the only ones not meet the stated purpose.
Table 2. Adherence of TiO2 films (based on ASTM D3359-09, [9]).
3.3. X-Ray Diffraction
It was found, from the analysis of X-ray diffraction patterns for the samples without PEG-400 in the precursor (Figure 4), that there is an anatase to rutile ratio in the range of 45% to 55%. On the other hand, for samples A1, B1, C1, the average ratio was 22% - 78%. Then, despite PEG presence reduced particle size, it did not decreased the amount of anatase (Figure 5), which could be a disadvantage for TiO2 photocatalysis. According to the work of Mendoza et al. [10], a decrease of anatase might be caused by inorganic impurities in the precursors of the films.
3.4. Photocatalytic Oxidation of Methylene Blue
There was evidence of photocatalytic oxidation of aqueous methylene blue using films prepared with A precur-
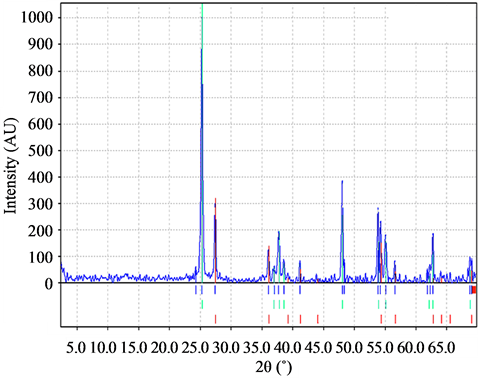
Figure 4. X-ray diffraction spectrum of the samples of TiO2 without PEG 4000.
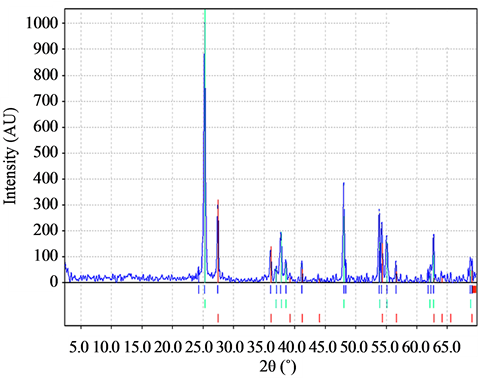
Figure 5. X-ray diffraction spectrum of the samples of TiO2 with PEG 4000.
sors as a photocatalyst (Figure 6). Because the contaminant concentration is proportional to the absorbance at a fixed wavelength (it means that this molecule fits to Lambert-Beer Law), the presence of Methylene blue and main oxidation products was monitored by UV-vis spectrophotometry. There were no evidences of detectable formation of intermediate products and absorbance peaks were used directly to obtain information presented on Figure 6.
It is observed that methylene blue is oxidized by photolysis on 52%. This fact can be contrasted with the film that has higher degradation, RA, which achieves up to 80% of oxidation efficiency. For the other cases, the film named A1 reaches up to 5% more effective degradation.
These films can be applied on the detoxification of polluted water and air, mainly based on the composition of A1 precursor for TiO2 films.
• 4. Conclusions
• It has been found that the amount of PEG as a poregenerating agent significantly affects microstructure of the obtained TiO2 nanoporous films.
• Based on the results of the scotch tape test, most of the porous films have very good adherence to glasses.
• The PEG 4000 in a precursor is an agent that generates pores on films and the amount of PEG 4000 in the precursor is proportional to pore size.
• On the surface, cracks were observed in the films A4, RB, B1, B2, B3, B4, C1, C2, C3 and C4, because pores overlapping.
• Small bumps were detected and they might be a consequence of the non-oxidized PEG-4000 during sintering. According to the work of Huang et al. [7] the presence of small cracks on the surface can be due to small clusters, consisting of PEG and TiO2 particles, when the amount of PEG is higher than a critical value on the sol. Particles on the films are nanosized and the particle size varies when PEG is added. When us-
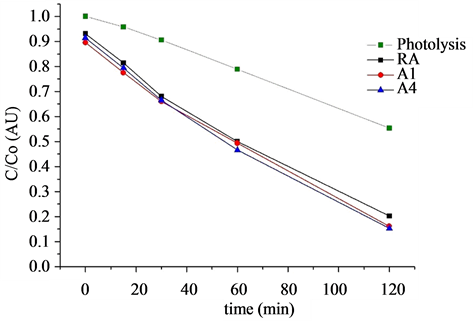
Figure 6. Photocatallytic oxidation kinetics.
ing PEG, the surface area is increased, which is in agreement with the works of Huang et al. [7], Bu et al. [13], Liu et al. [12], and Guo et al. [11].
• There is an important reduction of the amount of anatase on the films when PEG is added to precursors.
• Films are photocatalytically active despite the fact that photolysis is observed in parallel.
• The sample RA (without PEG) could degrade the 80% of the methylene blue, with the film A1 obtaining a degradation of 85%, 5% more than the sample without PEG.
• From the above it can be determined that there are two agents involved in photocatalytic degradation, the surface area, determined by the number of pores and anatase to rutile ratio.
• When compared with non-porous films, photocatalytic activity was lower, despite higher porosity.
Acknowledgements
Authors acknowledge to UNAM, Instituto de Ingenieria de la DGAPA-UNAM (PAPIIT IN107913 project) and ICYT (PICSO11-13 project) for supporting this work. In addition, the authors are grateful to UNAM, and to Department of Mechanical and Solar Energy Group, both from Instituto de Ingenieria de la UNAM.
REFERENCES
- O. M. Alfano, D. Bahnemann, A. E. Cassano, R. Dillert and R. Goslich, “Photocatalysis in Water Environments Using Artificial and Solar Light,” Catalysis Today, Vol. 58, No. 2-3, 2000, pp. 199-230. http://dx.doi.org/10.1016/S0920-5861(00)00252-2
- S. E. Manaham, “Environmental Chemistry,” 9th Edition, CRC Press, USA, 2010.
- J. M. Herrmann, “Heterogeneous Photocatalysis: An Emerging Discipline Involving Multiphase Systems,” Catalysis Today, Vol. 24, No. 1-2, 1995, pp. 157-164. http://dx.doi.org/10.1016/0920-5861(95)00005-Z
- C. Estrada, J. Blanco and S. Malato, “Purificacion de aguas por Fotocatalisis Heterogenea: Estado del arte,” CIEM 2011 AT, Spain.
- K. Ertl, “Handbook of Heterogeneous Catalysis,” Vol. 1, VHC, USA, 1997. http://dx.doi.org/10.1111/j.1551-2916.2010.04205.x
- J. Yao, Y. Bai, N. Chen, M. Takahashi and T. Yoko, “Sol-Gel Preparation, Characterization, and Photocatalytic Activity of Macroporous TiO2 Thin Films,” Journal of the American Ceramic Society, Vol. 94, No. 4, 2011, pp. 1191-1197. http://dx.doi.org/10.1111/j.1551-2916.2010.04205.x
- W. Huang, M. Lei, H. Huang, J. Chen and H. Chen, “Effect of Polyethylene Glycol on Hydrophilic TiO2 Films: Porosity-Driven Superhydrophilicity,” Surface & Coatings Technology, Vol. 204, No. 24, 2010, pp. 3954-3961. http://dx.doi.org/10.1016/j.surfcoat.2010.03.030
- J. C. Morales-Mejia, “Uso de Colectores Solares para la Remoción Fotocatalítica de Disruptores Endócrinos Presentes en agua Contaminada,” PhD Thesis, Programa de Maestria y Doctorado en Ingenieria, UNAM, Mexico, 2013.
- ASTM, “Standard Test Methods Measuring Adhesion by Tape Test,” ASTM International, 2012.
- A. Mendoza, “Caracterización Microestructural y Morfologia de TiO2 para Aplicaciones Termoluminiscentes,” Revista Mexicana de Fisica, Suplemento I, 2003.
- B. Guo, Z. Liu, L. Hong, H. Jiang and J. Yang Lee, “Photocatalytic Effect of the Sol-Gel Derived Nanoporous TiO2 Transparent Thin Films,” Thin Solid Films, Vol. 479, No. 1-2, 2005, pp. 310-315. http://dx.doi.org/10.1016/j.tsf.2004.11.197
- C. Liu, R. Li and W. Zhang, “Adsorption Property and Photocatalytic Activity of Sol-Gel Prepared Porous TiO2 Using PEG1000 as Template,” Applied Mechanics and Materials, Vol. 48-49, 2011, pp. 153-156.
- S. Bu, X. Liu, Z. Jin, L. Yang and Z. Cheng, “Fabrication of TiO2 Porous Thin Films Using PEG Templates and Chemistry of the Process,” Materials Chemistry and Physics, Vol. 88, No. 2-3, 2004, pp. 273-279. http://dx.doi.org/10.1016/j.matchemphys.2004.03.033


