Pharmacology & Pharmacy
Vol.06 No.01(2015), Article ID:53609,6 pages
10.4236/pp.2015.61005
In Vivo and in Vitro Evaluation of Permethrin, Cypermethrin or Zeta-Cypermethrin Mixed with Plant Extracts against Susceptible and Resistant (San Alfonso) Rhipicephalus (Boophilus) microplus (Acari: Ixodidae) Strains
Froylán Ibarra-Velarde*, Yazmin Alcala-Canto, Yolanda Vera-Montenegro
Department of Parasitology, Faculty of Veterinary Medicine and Zoothecnics, National University Autonomous of Mexico, Mexico City, Mexico
Email: *ibarraf@unam.mx
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 31 December 2014; accepted 26 January 2015; published 28 January 2015
ABSTRACT
Acaricide resistance is a major problem that hinders the control of the cattle tick Rhipicephalus (Boophilus) microplus in Mexico. Permethrin (P), cypermethrin (C) and zeta-cypermethrin (Z) have been used to control R. (B.) microplus, and tick populations have developed resistance to these acaricides. The aim of the present study was to evaluate the effectiveness of a mixture containing P, C, or Z mixed with plant extracts through in vitro laboratory bioassays, using susceptible and triple resistant (San Alfonso) R. microplus strains. Untreated controls received only water. Results of laboratory bioassays using larval packet tests revealed an efficacy of 100% (P), 100% (Z), and 98.03% (C) using susceptible larvae, and an efficacy of 88.67% (P), 91.51% (C), and 99.27% (Z) on triple-resistant larvae. Egg laying, larvae hatching and efficacy was assessed using ticks collected from treated and untreated animals. Product Z produced a 92.04% efficacy on engorged ticks collected from experimentally-infested cattle, whereas C and P exerted 80.66% and 20.04% efficacy, respectively. Engorged females collected exclusively from control animals were challenged in vitro with the experimental products, and efficacy was as follows: 91.37% (Z), 85.95% (C), and 13.58% (P). Adding plant extracts to a pyrethroid formulation led to dramatic increases of percent reduction of both susceptible and resistant immature ticks in contrast to untreated larvae and susceptible adults. Results from this study may lead to suggesting the adoption of an acaricide-botanical mixture strategy for tick control worldwide.
Keywords:
Rhipicephalus (Boophilus) microplus, Efficacy, Pyrethroids + Plant Extracts, In Vitro, In Vivo

1. Introduction
Ticks are ectoparasites which frequently attacks cattle located in tropical or subtropical areas worldwide. In Mexico, Boophilus microplus causes strong economical losses to cattle because they play an important role on the transmision of babesiosis and anaplasmosis. In our country, from over 30 million cattle heads, 75% of them are located in tropical or subtropical areas and therefore susceptible to suffer the mentioned diseases.
Recent data indicates that 17% of Mexican cattle exported to the USA are rejected because it is infested with ticks [1] . During decades, the most used ixodicides for tick control have been organophosphates, pyrethroids and amidins [2] . Nevertheless, treatment costs are high, the use of chemical acaricides increases the risk of leaving drug residues in meat, milk as well as the environment, and some R. (Boophilus) microplus strains have developed acaricide resistance [3] . Studies reporting the acaricidal activity of plant extracts against R. (Boophilus) microplus have encouraged the use of natural active compounds that could be used as an alternative because of their lower environmental impact and cost; e.g., the extracts of Hypericum polyanthemum [4] , limonene [5] , plants from the Meliaceae family [6] [7] , Copaifera reticulata [8] , Eucalyptus spp. [9] , Anona squamosa, Azadirachta indica [10] , among others have been evaluated for their potential efficacy against R. (Boophilus) microplus. Natural extracts, whether alcoholic or water-based, have a longstanding historic tradition in Mexico and are regarded as a danger-free approach to treat and/or control infectious diseases. Although this might be an overstatement, it may be true for some chemical groups. In the search for these control alternatives, it is believed that some plant extracts mixed with some known ixodicides could enhance the ixodicidal effect. The aim of the present study was to evaluate the effectiveness of a mixture containing Permethrin (P), cypermethrin (C) and zeta-cypermethrin (Z) mixed with plant extracts through in vitro laboratory bioassays, using susceptible and triple resistant (San Alfonso) R. microplus strains.
2. Materials and Methods
2.1. In Vivo Test against R. (Boophilus) microplus Susceptible Larvae in Cattle
Compounds-Permetrin (P), cypermethrin (C) or Z-Cypermethrin (Z) extracts were mixed with plant extracts (Monocotyledons) at 25%. (Registration under patent regulation IK33/Power X21 initially authorized by the Federal Commission for the Protection of Sanitary Risks (Cofepris-Mexico in spanish), No. 113300CO220103/ 2012.
Animals
Animals used in this study were managed according to bioethical regulations according to the animal welfare Committee of our institution. Twenty-four european mixed race, steers with an average weight of 275 Kg, were used. They were keept on non-infested pads during the experiment. They were fed on commercial food and water was supplied ad libitum.
2.2. R. (Boophilus) microplus Strain
R. (B.) microplus susceptible and San Alfonso (resistant to amitraz, pyrethroids and organophosphates) strain larvae were kindly donated by the National Center of Parasitology (CENAPA) located in Morelos, Mexico and kept under biosecurity measures at the parasitology laboratory of our institution.
2.3. Infestation
On day 0, 1 g of 15-day-old susceptible larvae (equivalent to aproximately 20,000 larvae), were placed on the back of each animal. They remained on the experimental cattle until day 21 after infestation when ticks were engorged and fell to the floor for hatching.
2.4. Treatment
All animals were divided in 4 groups (G) of 6 animals each according to the average counts of ticks. G1 served as untreated control (T), G2 received permetrin (P), G3 was treated with cypermethrin (C) and G4 received Z-Cypermethrin (Z). Treatment was applied by aspersion using 5 litters of compound/animal. The untreated control was aspersed only with tap water using also 5 l/animal.
Twenty-four hours after treatment, engorged female ticks were collected from each group. They were placed in a sieve (100 microns mesh) and washed out with tap water. Afterwards, they were dried and placed in a Petri dish. Ticks isolated per group were transported to the parasitology laboratory and incubated at 27˚C and 80% humidity to allow continuation of the development cycle. Weight of ova and percentage of eclosion from these specimens was estimated.
The obtained data on the pre and postreatment of all experimental groups were compared with regard to the untreated control group.
2.5. Larval Immersion in Vitro Assay
The larval immersion technique proposed by [11] was used to test acaricidal efficacy in vitro. Fifteen-day old larvae from susceptible and resistant San Alfonso (Triple Resistant: Amidins, Organophosphates and Piretroids) were used. Then larvae were divided for testing in four groups: Untreated Control (T); Permethrin (P); Cypermethrin (C) and Z-cypermethrin (Z). To assess the mortality percentage, live and dead larvae were counted. Each concentration was tested in three replicates.
2.6. Obtention of Ova
Engorged R. (Boophilus) microplus were collected from the four experimental groups. They were washed and dried on absorbent paper, weighed individually in an analytical weighing scale. Four groups (T, P, C, Z) of 20 ticks each were placed in Petri dishes and incubated at 27˚C and 85% relative humidity to assess egg laying and larval hatching.
2.7. Percentage of Egg Hatching
Eggs were incubated by standard procedures at 27˚C and 85% humidity during 21 days. The eclosion percentage was estimated by counting the eggs and larvae contained in each tube with the aid of an stereoscopic microscope. The index of egg laying (IE) was calculated as follows:
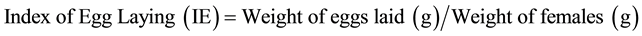
The percentage (%) inhibition of egg-laying was therefore determined with the following formulae: [12] .

The efficacy of treatment was calculated according to formulae proposed by Drummond and Whetstone (1973) where:
Estimated reproduction (ER) = [(Weight of eggs (g)/Weight of females (g)] × % eclosion × 20,000 (estimate of the number of larvae in 1 g of eggs)
Once the ER was calculated in both treated and control groups, the control % was estimated:

2.8. Challenge of Engorged Females in Vitro
Engorged adult ticks were evaluated using the immersion test according to [13] . Eighty ticks were collected from the untreated controls, washed, dried on absorbent paper and weighed individually in an analytical weighing scale. Four groups of 20 ticks with similar weights each were immersed during 5 minutes in 1 ml of water, P, C or Z diluted in 0.5 l water. Ticks from each group were recovered from the solutions, dried and placed in a 9 cm Petri dish padded with Whatmann filter paper. Petri dishes were incubated at 27˚C - 28˚C and 75% - 85% relative humidity. The effect of the experimental products on tick mortality was recorded 24 h after treatment. After 14 days, the number of females laying eggs was recorded and eggs were collected, weighed and incubated at 27˚C - 28˚C and 75% - 85% relative humidity up to the hatching of the larvae. ER was estimated as mentioned previously.
2.9. Probit Analysis
After susceptible and triple-resistant larvae were exposed to filter paper circles impregnated with different water-based concentrations of permethrin, cypermethrin, or Z-cypermethrin prepared from stock solutions of 10,000 ppm, results were subjected to Probit analysis using the POLO-PC software, provided by the Department of Pharmacology and Physiology of the FMVZ-UNAM. The LC50 of permethrin, cypermethrin and Z-cypermeth- rin were determined by an analysis of regression to the Probit-transformed data of death larvae with 95% confidence limits.
3. Results
3.1. In Vitro Efficacy against R. (Boophilus) microplus Susceptible Larvae
The obtained results showed that permethrin killed all larvae on the assay generating a 100% efficacy. Cipermethrin reached a 98.0% efficacy and Z-Cypermethrin generated a 100% mortality. The larvae from the untreated control remained healthy and active and no mortality was observed (Table 1).
3.2. In Vitro Efficacy against San Alfonso Triple Resistant Strain of R. (Boophilus) microplus Larvae
The average efficacy exerted by permethrin was 88.6%, cypermethrin produced a 91.5% mortality and Z-Cy- permethrin caused a 99.2% efficacy. The untreated control group remained healthy with no mortality (Table 2).
3.3. Effectiveness of Treatment
As shown in Table 3, product Z produced a 92.04% efficacy against engorged ticks collected from experimentally-infested cattle, whereas C and P exerted 80.66% and 20.04% efficacy, respectively. Regarding adult ticks challenged in vitro with the experimental products, efficacy was as follows: 91.37% (Z), 85.95% (C), and 13.58% (P) (Table 4).
Table 1. Efficacy of three compounds against larvae of a susceptible strain of Rhipicephalus (Boophilus) microplus ac- cording to the Shaw assay.
Table 2. Efficacy of three compounds against larvae of a triple-resistant (San Alfonso) strain of Rhipicephalus (Boophilus) microplus using the Shaw assay.
Table 3. Effectiveness of in vitro treatment against engorged Riphicephalus (Boophilus) microplus adult females.
Table 4. Effectiveness of in vitro treatment (Drummond assay) against engorged Riphicephalus (Boophilus) microplus adult females.
After subjecting data of the susceptible strain of Rhipicephalus (Boophilus) microplus larvae mortality to Probit analysis, the LC50 of permethrin, cypermethrin, and Z-cypermethrin were determined. The LC50 were 1015.78 ppm with 95% confidence intervals (415.82 - 1618.25) for permethrin, 436.2 (404.10 - 468.57) for cypermethrin, and 258.54 (243.76 - 273.84) por Z-cypermethrin (Table 5).
Regarding the San Alfonso strain larvae, the LC50 were 8353.7 (7956.8 - 8750.42) for permethrin, 3235.19 (3069.09 - 3419.28) for cypermethrin, and 345.08 (329.3 - 363.48) for Z-cypermethrin (Table 6).
4. Discussion
Most cattle located in the tropics, are at risk from being infested by various tick species as well as tick-borne dis- eases [14] , R. Boophilus microplus (Canestrini) is an ectoparasite of cattle that causes significant economic losses in several tropical and subtropical countries of Africa, Latin America, Northern and Eastern Australia. It represents one of the main constraints to cost-effective production due to its direct parasitic action and to the fact that it is the vector of important pathogens.
In the present study, results of in vitro bioassays using larval packet tests revealed an efficacy of 100% (P), 98.03% (C) and 100% (Z), using susceptible larvae, and an efficacy of 88.67% (P), 91.51% (C), and 99.27% (Z) on triple-resistant larvae. Data obtained strongly suggests that adding plant extracts to a pyrethroid formulation led to dramatic increases of percent reduction of both susceptible and resistant immature ticks in contrast to untreated larvae. Regarding adult females, a desirable efficacy of over 90% was assessed only with product Z. It is therefore tempting to speculate that the lower level of efficacy observed in adult ticks treated with pyrethroid-derived products in contrast to results observed in larvae, might be caused by a lower penetration of the experimental plant-derived products to the harder cuticle present in adults, or to the contribution of more developed metabolic pathways involved in pyrethroid resistance.
A variation of LC50 values for pyrethroids has been documented [15] . This variation might be due to the difference in strains and performed methodology. In this study, a significant difference in LC50 values was observed between permethrin, cypermethrin and Z-cypermethrin in biossays carried out on susceptible strains. It is therefore reasonable to suggest that the latter products have a higher efficacy than permethrin. Moreover, the resistant strain required a higher concentration to achieve the LC50 value for the three acaricides. This value was significantly higher for the experimental permethrin and cypermethrin; which prompt the need of further studies to determine the resistance to these products.
In addition efficacy on larvae of the experimental pyrethroids used in the present study was similar to pyrethroid-based commercial products, which has been determined as 100% [16] . In contrast, the same authors reported a higher efficacy (92% - 99%) on engorged females than the one determined in the present study. Thus, studies are required to improve the pharmacological design of these compounds in order to enhance their efficacy on adult ticks.
Willadsen and Kemp [17] have regrettably discussed the impact that decades of scientific advances have had on tick control for farming and animal husbandry practices. In Australia, for example, adoption of acaricide-based tick control programs among cattle producers ranged from 19% to as low as 6% of farms surveyed. Contributing factors include the complexity of the livestock-tick interactions, the cost of implementation, and the limited success rates of the various tick control strategies available. Nari and Hansen [18] mentioned that the cost of development of a new ixodicide can be in excess of 100 to 230 million US dollars and that this process might take more than 10 years from the discovery of a candidate to the launching on the market. Furthermore, selection of resistance genes in ticks against current ixodicides is an increasing problem [19] . Therefore, new
Table 5. Lethal concentration of permethrin, cypermethrin and Z-cypermethrin on a suceptible strain of Riphicephalus (Boophilus) microplus larvae.
Table 6. Lethal concentration of permethrin, cypermethrin and Z-cypermethrin on San Alfonso strain of Riphicephalus (Boophilus) microplus larvae.
strategies for tick control should be undertaken to optimize the use of available drugs. The success of some experimental acaricides obtained from natural extracts has prompted research on active phytochemicals that might have efficacy against ticks. There are many studies on the activity of plants against engorged females and larvae most of which show the reduction in the egg-laying capacity of ticks exposed to different natural extracts [8] - [20] .
The present findings highly suggest to undertake broader bioassays in order to elucidate the role of natural compounds combined with current ixodicides in the larvicidal and anti-fertility outcomes described in the present paper and to improve acaricidal effectiveness, particularly in resistant strains. Results from this study may lead to suggest the adoption of an acaricide-botanical mixture strategy for the control of triple-resistant R. microplus in Mexico and elsewhere.
5. Conclusion
The mixture of three pyrethroids with plant extracts exerted higher acaricidal efficacy under in vitro and in vivo conditions.
Acknowledgements
The authors are indebted to Ing. Alejandro Canales-Farías from Laboratorios Shark, S.A. de C.V for kind donation of the compounds.
References
- Bram, R.A., George, J.E., Reichar, R.E. and Tabaciinic, W.J. (2002) Threat of Foreign Arthropod-Borne Pathogens to Livestock in the United States. Journal of Medical Entomology, 39, 405-416. http://dx.doi.org/10.1603/0022-2585-39.3.405
- Rodriguez-Vivas, I., Rivas, A.L., Chowell, G., Fragoso, S.H., Rosario, C.R., Garcia, Z., Smith, S.D., Williams, J.J. and Schwager, S.J. (2007) Spatial Distribution of Acaricide Profiles (Boophilus microplus Strains Susceptible or Resistant to Acaricides) in Southeastern Mexico. Veterinary Parasitology, 146, 158-169. http://dx.doi.org/10.1016/j.vetpar.2007.01.016
- Miller, R.J., Davey, R.B. and George, J.E. (2002) Modification of the Food and Agriculture Organization Larval Packet Test to Measure Amitraz Susceptibility against Ixodidae. Journal of Medical Entomology, 39, 645-651. http://dx.doi.org/10.1603/0022-2585-39.4.645
- Ribeiro, V.L.S., Toigo, E., Bordignon, S.A.L., Gonçalves, K. and von Poser, G. (2007) Acaricidal Properties of Extracts from the Aerial Parts of Hypericum polyanthemum on the Cattle Tick Boophilus microplus. Veterinary Parasi- tology, 147:199-203. http://dx.doi.org/10.1016/j.vetpar.2007.03.027
- Ferrarini, S.R., Oliveira, D.M., da Rosa, R.G., Rolim, V., Eifler-Lima, V.L., von Poser, G. and Ribeiro, V.L.S. (2008) Acaricidal Activity of Limonene, Limonene Oxide And β-Amino Alcohol Derivatives on Rhipicephalus (Boophilus) microplus. Veterinary Parasitology, 157, 149-153. http://dx.doi.org/10.1016/j.vetpar.2008.07.006
- Mulla, M.S. and Su, T. (1999) Activity of Biological Effects of Neem Products against Arthropods of Medical and Veterinary Importance. American Mosquito Control Association, 15, 133-152.
- Borges, M.F., Ferri, P.H., Silva, W.J., Silva, W.C. and Silva, J.G. (2003) In Vitro Efficacy of Extracts of Melia azedarach against the Tick Boophilus microplus. Medical and Veterinary Entomology, 17, 228-231. http://dx.doi.org/10.1046/j.1365-2915.2003.00426.x
- Fernandes, F.F. and Freitas, E.P.S. (2007) Acaricidal Activity of an Oleoresinous Extract from Copaifera reticulata (Leguminosae: Caesalpinioideae) against Larvae of the Southern Cattle Tick, Rhipicephalus (Boophilus) microplus (Acari: Ixodidae). Veterinary Parasitology, 147, 150-154. http://dx.doi.org/10.1016/jj.vetpar.2007.02.035
- Chagas, A.C.S., Passos, M.W.M., Prates, H.T., Leite, R.C., Furlong, J. and Fortes, I.C.P. (2002) Acaricidal Effect of Essential Oils and Emulsion Concentrates of Eucalyptus spp on Boophilus microplus. Brazilian Journal of Veterinary Research and Animal Science, 39, 247-253. http://dx.doi.org/10.1590/S1413-95962002000500006
- Magadum, S., Mondal, D.B. and Ghosh, S. (2009) Comparative Efficacy of Annona squamosa and Azadirachta indica Extracts against Boophilus microplus Izatnagar Isolate. Parasitology Research, 105, 1085-1091. http://dx.doi.org/10.1007/s00436-009-1529-3
- Shaw, R.D. (1966) Culture of an Organophosphorus-Resistant Strain of Boophilus microplus (Can.) and an Assessment of Its Resistance Spectrum. Bulletin of Entomological Research, 56, 389-405.
- Sardá Ribeiro, V., Avancini, C., Goncalves, K., Toigo, E. and Von Poser, G. (2008) Acaricidal Activity of Calea serrata (Asteraceae) on Boophilus microplus and Rhipicephalus sanguineus. Veterinary Parasitology, 151, 351-354.
- Drummond, R.O. and Whetstone, T.M. (1973) Lone Star Tick: Laboratory Tests of Acaricides. Journal of Economic Entomology, 66, 1274-1276. http://dx.doi.org/10.1093/jee/66.6.1274
- Bock, R., Jackson, L., De Vos, A. and Jorgensen, W. (2004) Babesiosis of Cattle. Parasitology, 129, S247-S269.
- Sharma, A.K., Kumar, R., Kumar, S., Nagar, G., Kumar, S.N., Sing Rawat, S., Dhakad, M.L., Rawat, A.K.S., Ray, D.D. and Ghosh, S. (2012) Deltamethrin and Cypermethrin Resistance Status of Rhipicephalus (Boophilus) microplus Collected from Six Agro-Climatic Regions of India. Veterinary Parasitology, 188, 337-345. http://dx.doi.org/10.1016/j.vetpar.2012.03.050
- Fragoso, H., Martinez, I., Ortiz, N. and Osorio, M. (2006) Comparison of the Efficacy of Organofosfates, Piretroids and Amidines Ixodicides against a Boophilus microplus Ticks Reinfestation in Naturally Infested Cattle. XXX National Congress of Buiatrícs, Mexico.
- Willadsen, P. and Kemp, D.H. (1988) Vaccination with Concealed Antigens for Tick Control. Parasitology Today, 4, 196-l98. http://dx.doi.org/10.1016/0169-4758(88)90084-1
- Nari, A. and Hansen, H.J. (1999) Resistance of the Ecto and Endoparasites: Actual and Future Solutions. 67th General Session, International Organization of Epizootias, París.
- Fernandes, F.F. (2001) Toxicological Effects and Resistance to Pyretroids in Boophilus microplus from Goiás, Brasil. Arquivo Brasileiro de Medicina Veterinária e Zootecnia, 53, 548-552. http://dx.doi.org/10.1590/S0102-09352001000500004
- Fernandes, F.F., Freitas, E.P.S., Costa, A.C. and Silva, I.G. (2005) Larvicidal Potential of Sapindus saponaria to Control the Cattle Tick Boophilus microplus. Pesquisa Agropecuária Brasileira, 40, 1243-1245. http://dx.doi.org/10.1590/S0100-204X2005001200013
NOTES

*Corresponding author.


