Neuroscience & Medicine
Vol.1 No.1(2010), Article ID:2690,5 pages DOI:10.4236/nm.2010.11005
Association of Human Herpesvirus 6 and 8 in Relapsing Remitting Multiple Sclerosis Type during Exacerbation
![]()
1Department of Neurology, Mansoura University, ElJomhoria st Mansoura University, Mansoura, Egypt; 2Department of Microbiology and Immunology; Mansoura University, Mansoura, Egypt.
Email: hassansalama@yahoo.com
Received June 28th, 2010; revised August 3rd, 2010; accepted August 10th, 2010.
Keywords: Multiple Sclerosis, HHV6, HHV8, antiviral drugs
ABSTRACT
Multiple Sclerosis (MS) is a chronic demyelinating disease of the CNS with assumed autoimmune etiology. Human herpes viruses have probable effects on relapsing-remitting MS pathogenesis presumably through molecular mimicry and/or bystander mechanisms. In this study we probed the possible contribution of the two herpes viruses, human herpesvirus 6 (HHV-6) and human herpesvirus 8 (HHV-8), in clinically definite multiple sclerosis (CDMS) patients-relapsing remitting type (RRMS) during clinical exacerbations. All patients had no history of immune modulating or suppressing drugs intake in the last 6 months. The peripheral blood samples, from CDMS patients (n = 20) (13F/7M, age (y) = 30.3 ± 3.21) and other immune mediated neurological disorders (OIND) (11F/9M, age = 25.2 ± 12.1), (Myasthenia Gravis, Guillain Barré Syndrome, ischemic stroke in adolescent and young adult with no clear risk factors), as a control group, had been enrolled within 15 months (January-2007-- March -2008). We investigated the existence of specific deoxyribonucleic acid (DNA) sequences belonging to HHV-6 and HHV-8, using polymerase chain reaction (PCR) in the isolated peripheral blood mononuclear cells (PBMCs) and in plasma. PCR demonstrated HHV-6 DNA in 7 cases (35%), HHV-8 sequences in only one cases (5%) in PBMCs from 20 relapsed CDMS patients; all HHV-6 positive cases showed positive plasma results, while the blood samples from 20 OIND patients showed negative results except one case (5%) out of 9 cases of GBS was positive for HHV-8 in PBMCs. We consequently concluded that there is considerable evidence in this study that proposed the roles of HHV-6 and HHV-8 in MS pathogenesis and clinical exacerbation.
1. Introduction
Multiple Sclerosis (MS) is a chronic demyelinating disease of the CNS with assumed autoimmune etiology that for many years have been supposed to be of potential viral triggering. Generally speaking, 85% of MS patients have relapsing remitting MS type (RRMS) in the early disease course that converted to secondary progression within 15 years in the majority of MS victims. Therefore it is considered as the most important cause of nontraumatic disability disease among young adults [1,2].
Indeed, no animal species other than humans develop MS. Even so, MS may be the consequence of a chronic virus infection, but, so far, a definitive role for HHVs in MS pathogenesis has not been established. Both HHV-6 and 8 have the same distinctiveness features (neurovirulence, neuroinvasiveness and neurotropism) of many viruses but till now there are no data that concluded in solid base the role of these viruses in development of demyelinating diseases [3-5].
Viral neuroinvasion takes place commonly in primary acquired HHV-6 and 8 infections, although demyelination has been notified as remarkable complications. The HHVs have been isolated from MS plaques that denote a potential relation with MS pathogenesis, accordingly hindering these factors could be considered as parallel management pathway in prevention or decreasing the frequency of MS relapses, as well as, long-term disability progression [6].
Several thoughts have been sophisticated to clarify the molecular mimicry or nonspecific bystander hypotheses of viruses. In agreement with this, two of the hallmarks of MS are sensitized auto-reactive T-cell and high auto-antibodies titers that are measurable in MS patients but not in healthy individuals. Nonetheless, many investigators using epidemiological observations as well as the pathological characteristics consider these viruses as participants in launching and perpetuation of the disease [7,8].
The rationale of this study was to investigate whether HHV-6 and 8 have a role in MS pathogenesis or not and their significant levels in relation to patients who have other immune mediated neurological disorders (Myasthenia Gravis, Guillain Barré Syndrome, ischemic stroke in adolescent and young adult with no clear risk factors).
2. Patients and Methods
This study prospectively enrolled 20 individuals who have been diagnosed RRMS with exacerbation and seen by two consultant neurologists in Mansoura university hospital throughout 15 months (January 2007, and March 2008). All MS cases showed clinical, radiological and electrophysiological (visual evoked potential (VEPs) and Brainstem Auditory evoked potentials (BAEPs)) findings that confirmed RRMS diagnosis [9,10].
The patient population was RRMS during exacerbation, the mean duration (y) of MS disease was 6.3 ± 1.86 and EDSS score was 2.88 ± 1.06. The demographic data of our RRMSpatients were 13 females and 7 males; their mean age when they included in this study was 30.3 ± 3.21years old. As regard the control groups, twenty cases with other immune mediated neurological disorders included in this study, their mean age when blood samples withdrawn was 25.2 ± 12.1 years old.
Above all, we used nested PCR (nPCR) to investigate the genomic DNA of HHV-6 and PCR for HHV-8 in peripheral blood mononuclear cells (PBMC) samples of 20 relapsing-remitting MS (RRMS) patients during exacerbation as well as control group. Two blood specimens were obtained for PCR examination from every patient. One sample was taken for PBMC separation and the second sample for plasma. Specimens from OIND were taken by the same way.
Isolated PBMCs and plasma samples were aliquoted and stored at −70℃. Blood samples from OIND patients were managed on the same technique. PBMC isolation were separated by Ficoll-Hypaque (Biotest-Germany) density gradient centrifugation and stored at −70℃. Total DNA was extracted from PBMCs and blood plasma of positive cases according to the manufacturer’s instructions (Gentra, Minneapolis, Minnesota). PCR was used for the detection of both HHV-6 (nPCR) and HHV-8 (PCR) sequences in PBMC-HHV-6 and 8-DNA and blood plasma for HHV-6-DNA.
nPCR-HHV-6, a 100 µL reaction (EzWay PCR master mix, Koma Biotech) contained 0.5µM of each primer and 0.5µg of DNA as a template. Negative controls were performed with sterile deionized water. The detection of HHV-6 DNA was carried out according to Bandobashi et al. 1997 with a nested primer set, complementary to the gene coding the major capsid protein, which recognizes of the virus. The nucleotide sequence of HHV-6 primers 5’-G C G T T T T CA GTG TGTA G TTCGGCAG-3’ and 5’- T G G C C G C A T T C G T A C A G ATACGGAGG- 3′(outer pairs); 5’-G C T A G A A C G T ATTTGCTGCAGAACG-3’ and 5’-A TC CG AAA CA A CTGTCTGACTGGCA- 3’ (inner pairs) for the first and second round of PCR, respectively [9]. The inner primers amplify a 258-bp fragment of HHV-6.
The first round of PCR was carried out at 94℃ for 3 min, followed by 30 cycles of 94℃ for 1 min, 57℃ for 1 min, and 72℃ for 1 min. Terminal extension of 72℃ for 5 min was carried out after completion of the 30 cycles. A sample (10 µl) of the first round product was used as template for the second round using the conditions described for the first round.
PCR was used to amplify 233-bp fragment of HHV-8DNA with the primers forward (AGC-CGA-AAG-GATTCC-ACC-AT) and reverse (TCC-GTG-TTG-TCT-ACGTCC-AG). A 100 µL reaction (EzWay PCR master mix, Koma Biotech) contained 0.5 µM of each primer and 0.5 µg of DNA as a template. Negative controls were performed with sterile deionized water. Reaction mixtures were subjected to 40 cycles of the following incubations: initial denaturations at 94℃ for 2 min (1 cycle), denaturation at 94℃ for 1min, annealing at 58℃ for 1 min, extension at 72℃ for 2 min (40 cycles) and final extension at 72℃ for 5 min.
A thermal cycler (Peltier-effect cycling, MJ Research, Inc.) (Watertown Mass) was used for the both PCR reactions. Detection of amplified DNA of both HHVs ten µL from the amplicon were analyzed by electrophoresis (Haefer HE33) in 2% agarose gel with 1x TBE buffer stained with ethidium bromide, visualized under UVL (Fisher Biotech, FBTIV-88) and photographed (FB-PDH1216).
All positive PBMC-HHV-6-DNA-PCR cases were analyzed again for plasma HHV-6-DNA for more confirmation. In this study we will discuss the results of PBMC samples because it is done for all patients but PCR for plasma samples were done for HHV-6-MS positive cases only.
Statistical analyses of data are given as mean ± SD. Continuous data were analyzed by the independent t test, as appropriate. P value of less than 0.05 was regarded as statistically significant.
3. Results
Between January 2007, and March 2008, 40 patients’ blood samples were collected in Neurology department, Mansoura University, There were 20 patients who had been classified as CDMS according to published Poser and McDonald criteria of MS diagnosis, all of them are RRMS type and investigated during relapse. The control group included twenty patients who had other immune related neurological diseases.
Indeed, this prospective study significantly estimated the proportion of HHV-DNA sequences positive cases in comparison with prospectively classified OIND. The OIND spectrum comprised 9 cases with Guillain Barré Syndrome, 5 for Myasthenia Gravis, and 6 patients had ischemic stroke in adolescent and young adult with no clear risk factor. The clinical characteristics of each group in our study were compatible with so-called immune based neurological disorder, either B-cell or T-cell mediated, just to exclude the possibility of cross immunity, dominating one type of immune reaction or B-T cell conversion immunoprocess.
Among RRMS cases there were seven patients (35%) showed positive results for HHVs-DNA sequences. Pin pointing to the type of HHVs, The study implicated the relatively high frequency of HHV-6 DNA sequence in 7 cases (35%), HHV-8 sequence in only one case (5%) in PBMCs that drawn from 20 RRMS patients throughout exacerbation; all patients had no history of immunomodulatory or immunosuppressive treatments intake 6 months prior to the last attack (Table 1).
There was one female patient who showed positive DNA sequence for both HHV-6 and 8 and her EDSS score was 5.5. This patient also experienced high frequency of MS exacerbation.
The PCR of the blood samples that had been drawn from twenty OIND patients showed no positive DNA sequence for HHV-6 or 8 except one case out of 9 cases of GBS (11.1%) was positive for HHV-8 that represent 5% of the total OIND patients (Table 1). There was no signifi
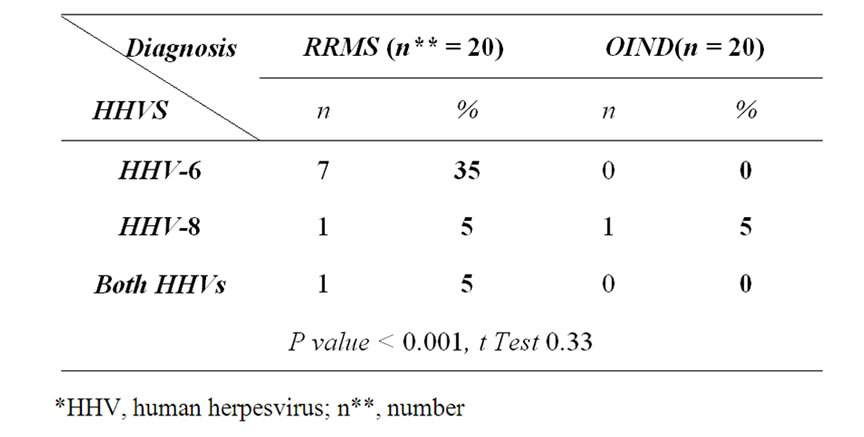
Table 1. Comparison between positive PCR-HHVs* cases.
cant predilection to females. Three out of the seven patients (42.8%) who had positive HHV-6-DNA sequences as well had high EDSS scores (more than 4).
4. Discussion
Indeed, this prospective study significantly estimated the proportion of HHV-DNA sequences positive MS cases in comparison with prospectively classified OIND. It was detected significantly (35%) in blood samples of RRMS patients more than control group (%5) (p < 0.05). This is indicating that viral agents could contribute to demyelination plaques and MS pathogenesis [5,6,10-12].
Moreover there is significant increase in frequency of relapses and severity of MS disease among HHVs positive patients (42.8%) (p < 0.05) who showed relative high EDSS. The percentage of cases with positive HHV-6 is higher thanHHV-8 [5,13].
In harmony with our results, Simmons 2001 and others stated that HHV-6 DNA correlates with active HHV-6 infection and is not found in healthy individuals. In this distinctive study, IgM titers showed a considerable raise to the p41/38 early antigen in patients with RRMS, in comparison to healthy controls and individuals with other neurological disease [14-16].
Two further studies have established the existence of increased IgM responses to HHV-6 in patients with MS, Additionally, HHV-6 DNA was identified in 30% of MS patients (15 of 50) and in 0% of 47 controls consisting of healthy individuals, patients with other inflammatory diseases, and patients with other neurological diseases, despite the fact that no correlation was demonstrated in Enbom, 2001 study [17-19].
Knox et al., 1998, stated that HHV-6 may host CNS of MS patients and activated, as measured by DNA in plasma and CSF, from time to time. Anyhow, HHV-6 A variant was detected in the PBMC of MS patients but not in controls [20].
It has been proposed that disagreements in these results may be caused by variations in reagents, procedures, racial and patient selection. Further serological, cellular/immune response, molecular, and clinical studies are required to explain the task of HHV-6 and 8 in MS pathogenesis [5,6,12,17,21].
Clearly, there is one patient who had HHV-8 (5%) with high EDSS that did not show significant differences with control group, which denotes the modest role of HHV-8 in disease progression and relapses frequency more than MS pathogenesis [22].
However, it is of interest that, when other investigators studied HHV-6 and 8 in RRMS, they gave attention to virus immunoglobulin titers and DNA but they did not correlate both of them with exacerbation, [23,24] therefore our distinctive study focus on the role of HHVs during relapses, this higher percentage of positive cases in relation to other studies may denote a considerable relation between exacerbation and HHVs, in addition to HHVs potential task in MS pathogenesis, plus all cases in this study had not receive any of beta-interferon drugs that have potential antiviral effects. As well racial factor may play also an important role in this high percentage [25,26].
5. Conclusions
We concluded that there is sizeable evidence in this study that put forward the association of HHV-6 and HHV-8 with RRMS during exacerbation. At the same time we have to be watchful to explain this mysterious disease very simply on the base of chronic viral infection, but we support extremely Egyptian therapeutic trials which put forward the role of antiviral drugs (acyclovir or valacyclovir) in treatment of MS exacerbation via evaluation of viral serology, radiological as well as immunological outcomes and the effect of these drugs on long term disease disability.
REFERENCES
- D. E. McFarlin and H. F. McFarland, “Multiple Sclerosis,” The New England Journal of Medicine, Vol. 307, 1982, pp. 1183-1188.
- J. E. Friedman, M. J. Lyons, G. Cu, D. V. Ablashl, J. E. Whitman, M. Edgar, M. Koskiniemi, A. Vaheri and J. B. Zabriskie, “The Association of the Human Herpesvirus-6 and MS,” Multiple Sclerrosis, Vol. 5, No. 5, pp. 355-362.
- H. Lassmann, “Comparative Neuropathology of Chronic Experimental Allergic Encephalomyelitis and Multiple Sclerosis,” Springer Verlag, Berlin, 1983.
- J. F. Kurtzke, “Epidemiological Evidence for Multiple Sclerosis as an Infection,” Clinical Microbiology Review, Vol. 6, No. 4, 1993, pp. 382-427.
- S. S. Solden, R. Berti, N. Salem, et al., “Association of Human Herpes Virus 6 (HHV-6) with Multiple Sclerosis: Increased Igm Response to HHV-6 Early Antigen and Detection of Plasma HHV-6 DNA,” Nature Medicine, Vol. 3, 1997, pp. 1394-1397.
- S. F. Hunterand D. A. Hafler, “Ubiquitous Pathogens. Links between Infection and Autoimmunity in MS,” Neurology, Vol. 55, No. 2, 2000, pp. 164-165.
- A. K. Abbas, K. M. Murphy and A. Sher, “Functional Diversity of Helper T Lymphocytes,” Nature, Vol. 383, No. 6603, 1996, pp. 787-793.
- M. Allegretta, J. A. Nicklas and S. Sriram, R. J. Albertini and O. T. Abramsky, “Cells Responsive to Myelin Basic Protein in Patients with Multiple Sclerosis,” Science, Vol. 247, No. 4943, 1990, pp. 718-721.
- W. L. McDonald, D. A. S. Compston, G. Edan, D. E. Goodkin, H. P. Hartung, F. D. Lublin, et al., “Diagnostic Criteria for MS: Guidelines from the International Panel on the Diagnosis of MS,” Annals of Neurology, Vol. 50, No. 1, 2001, pp. 121-127.
- C. M. Poser, D. W. Paty, L. Scheinberg, W. I. McDonald, F. A. Davis, G. C. Ebers, et al., “New Diagnostic Criteria for Multiple Sclerosis: Guidelines for Research Protocols,” Annals Neurology, Vol. 13, No. 2, pp. 227-231.
- K. Bandobashi, M. Daibata, M. Kamioka, Y. Tanaka, I. Kubonishi, H. Taguchi, Y. Ohtsuki and I. Miyoshi, “Human Herpesvirus 6 (HHV-6)-Positive Burkitt’s Lymphoma: Establishment of a Novel Cell Line Infected with HHV-6,” Blood, Vol. 90, No. 3, 1997, pp. 1200-1207.
- S. Beer, K. M. Rösler and C. W. Hess, “Diagnostic Value of Paraclinical Tests in Multiple Sclerosis: Relative Sensitivities and Specificities for Reclassification According to the Poser Committee Criteria,” Journal of Neurology Neurosurgery Psychiatry, Vol. 59, No. 2, pp. 152-159.
- M. L. Opsahl and P. G. Kennedy, “Investigating the Presence of Human Herpesvirus 7 and 8 in Multiple Sclerosis and Normal Control Brain Tissue,” Journal of the Neurological Sciences, Vol. 240, No. 1-2, pp. 37-44.
- A. Simmons, “Herpesvirus and Multiple Sclerosis,” Herpes, Vol. 8, No. 3, pp. 60-63.
- E. Norrby, H. Link and J. E. Olsson. “Comparison of Antibodies against Different Viruses in Cerebrospinal Fluid and Plasma Samples from Patients with Multiple Sclerosis,” Infect Inmunology, Vol. 10, 1974, p. 688.
- R. T. Johnson, “The Virology of Demyelinating Diseases,” Annals Neurology, Vol. 36, 1994, pp. 54-60.
- C. Martin, M. Enbom, M. Söderström, S. Fredrikson, H. Dahl, J. Lycke, et al., “Absence of Seven Human Herpesviruses, Including HHV-6, by Polymerase Chain Reaction in CSF and Blood from Patients with Multiple Sclerosis and Optic Neuritis,” Acta Neurological Scandinavica, Vol. 95, No. 5, 1997, pp. 280-283.
- M. Enbom, F. Z. Wang, S. Fredrikson, C. Martin, H. Dahl and A. Linde, “Similar Humoral and Cellular Immunological Reactivities to Human Herpesvirus 6 in Patients with Multiple Sclerosis and Controls,” Clinical Diagnostic Lab Immunology, Vol. 6, No. 4, 1999, pp. 545-549.
- M. Enbom, “Multiple Sclerosis and Kaposi’s SacomaChronic Diseases Associated with New Human Herpesviruses?” Scandinavial Journal Infectious Diseases, Vol. 33, No. 9, 2001, pp. 648-658.
- K. K. Knox, D. Harrington and D. R. Carrigan, “Active Human Herpesvirus Type 6 Infections are Present in the Central Nervous System, Lymphoid Tissues and Peripheral Blood of Patients with Multiple Sclerosis,” Annals Neurology, Vol. 44, 1998, p. 485.
- M. Mayne, J. Krishnan, L. Metz, et al., “Infrequent Detection of Human Herpesvirus 6 DNA in Peripheral Blood Mononuclear Cells from Multiple Sclerosis Patients,” Annals Neurology, Vol. 44, No. 3, 1998, pp. 391- 394.
- E. Merelli, R. Bedin, P. Sola, P. Barozzi, G. L. Mancardi, G. Ficarra, et al., “Human Herpes Virus 6 and Human Herpes Virus 8 DNA Sequences in Brains of Multiple Sclerosis Patients, Normal Adults and Children,” Journal Neurology, Vol. 244, No. 7, 1997, pp. 450-454.
- K. Felgenhauer and H. Reiber, “The Diagnostic Significance of Antibody Specificity Indices in Multiple Sclerosis and Herpesvirus Induced Diseases of the Nervous System,” The Journal of Clinical Investigation, Vol. 70, No. 1, 1992, pp. 28-37.
- A. Rotola, E. Cassai, M. R. Tola, E. Granieri and D. Di Luca, “Human Herpesvirus 6 is Latent in Peripheral Blood of Patients with Relapsing-Remitting Multiple Sclerosis,” Journal of Neurology Neurosurgery Psychiatry, Vol. 67, No. 4, pp. 529-531.
- J. Lycke, B. Svennerholm, E. Hjelmquist, et al., “Acyclovir Treatment of Relapsing Remitting Multiple Sclerosis. Randomized Placebo-Controlled, Double-Blind Study,” Journal Neurology, Vol. 243, No. 3, pp. 214-224.
- K. R. Beutner, “Valacyclovir: A Review of Its Antiviral Activity, Pharmacokinetic Properties, and Clinical Efficacy,” Antiviral Research, Vol. 28, No. 4, 1995, pp. 281- 290.

