Surgical Science
Vol. 4 No. 2 (2013) , Article ID: 27864 , 4 pages DOI:10.4236/ss.2013.42027
A Case Report of Oseoid Osteoma in the Ulnar Cortex of the Distarl Radius Extending into the Distal Radioulnal Joint
—COX-2 Produced Prostaglandin E2 Possible Case of the Symptoms
Email: tanaka1041@msn.com
Received December 23, 2012; revised January 25, 2013; accepted February 4, 2013
Keywords: Osteoid Osteoma; DRUJ; COX-2; Prostaglandin E2
ABSTRACT
An 18-year-old girl presented with an osteoid osteoma in the ulnar side of the cortex of the distal radius extending into the distal radioulnar joint. The osteoid osteoma was removed from the distal radius at surgery. The patient’s symptoms were dramatically improved the day after surgery. A high concentration of prostaglandin E2 (PGE2) was seen in the joint and surrounding soft tissues, and cyclooxygenase-2 (COX-2) receptors were stained in the joint capsule and synovium inside the joint. This case report is the first to describe the concentration of PGE2 and stained COX-2 receptors in an osteoid osteoma in the ulnar cortex of the distal radius extending into the distal radioulnar joint.
1. Introduction
Osteoid osteomas are benign bone-forming neoplasms that result in progressive pain, characteristic at night, in young adults. Intra-articular osteoid osteomas within the joint capsule are rare. Makley et al. [1] and Greco et al. [2,3] demonstrated a high level of prostaglandins (PGs) in the nidus of osteoid osteoma; PGs are produced from phospholipids catalysed by cytosolic phospholipase A2 (cPLA2) and cyclooxygenase-2 (COX-2). In this case report, we describe an osteoid osteoma in the distal radioulnar joint (DRUJ), accompanied by a high concentration of PGE2 in fluid collected from the DRUJ and surrounding soft tissues. In addition, staining of the synovium of the DRUJ was positive for COX-2 receptors.
2. Case Report
The patient was an 18-year-old college girl, who was an active participant in a Japanese archery club. In October 2008, she felt pain in her right wrist joint, which gradually increased and limited her range of motion to the point that she could no longer participate in archery. By March 2009, she held her forearm in pronation and flexion; non-steroidal anti-inflammatory drugs (NSAIDs) did not improve her condition. The patient was referred to our hospital in April 2009.
Her chief complaints were right wrist pain and limitation of wrist range of motion. The right wrist was swollen on the dorsal and volar sides, but it was not warm to touch or erythematous. The range of motion of the wrist was 15 - 90 degrees of palmar flexion, and the range of motion of the forearm was 15 degrees of pronation and 0 degrees of supination. Laboratory data were within normal limits.
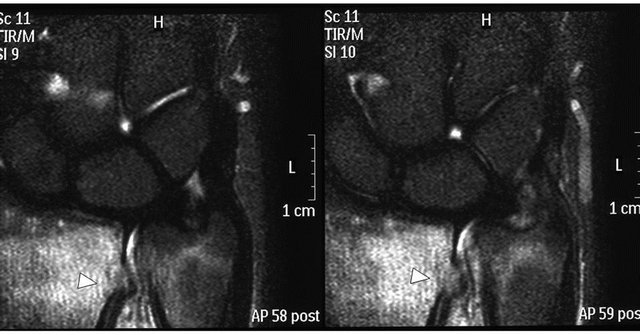
Plain radiographs showed a small round lucency of cortical bone on the ulnar side of the cortex of the distal radius and swollen soft tissue around the DRUJ. The bone surrounding the round lucency was not sclerotic (Figure 1). By Magnetic Resonance (MR) First Field Echo (FFE) imaging, which involves gradient-echo imaging sequences, a small tumor at the defect of the cortical bone was clearly seen (Figure 2(a)). By Short-TI Inversion-Recovery (STIR) imaging, by which the signal from water or fat can be suppressed, the high intensity around the tumor was found to be spread around the distal radius and ulna (Figure 2(b)); in the coronal view, the high intensity was found to be spread to the volar and dorsal sides of the wrist. By computed tomography imaging, a defect of the cortical bone at the distal radius in the DRUJ was detected.
Due to the differential diagnosis (DD) and because NSAIDs were not effective, surgery was prescribed for the patient. A volar approach was used. The soft tissue was swollen and edematous; especially noticeable was the pronator quadratus, which was edematous and white in colour. The DRUJ was filled with an effusion, which
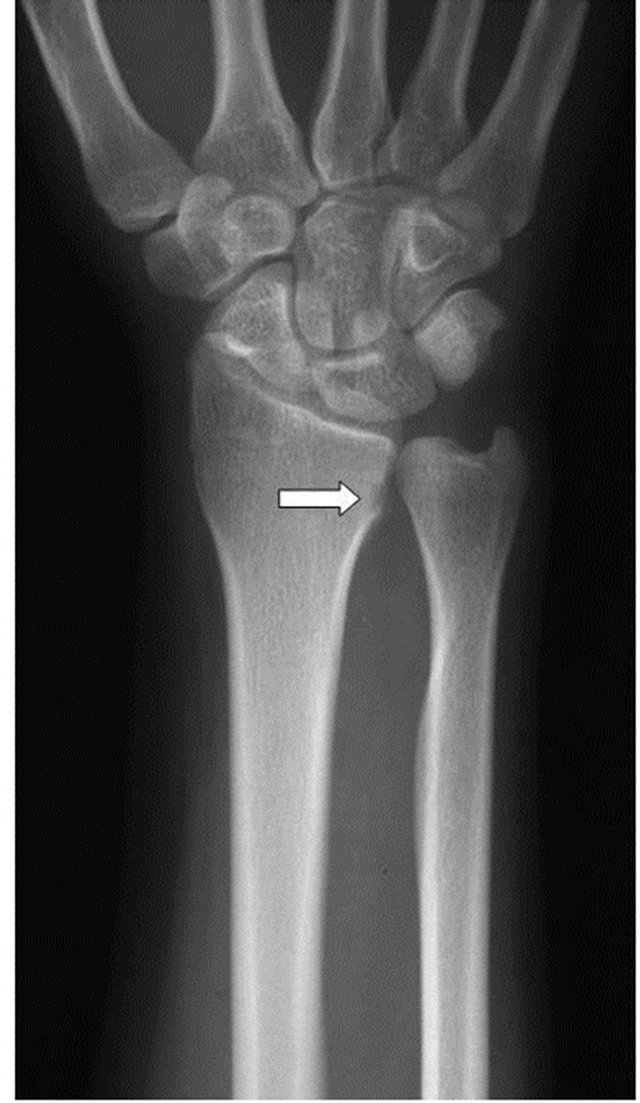
Figure 1. Plain X-ray, A-P view: a small cortical defect (white arrow) is noted at the distal radius, extending into the distal radioulnar joint.
was removed by syringe. The tumor, which showed a red colour, was exposed from the cortical bone to the DRUJ (Figure 3). A portion of the radial cortex was removed by chisel and enucleated. After irrigation, the wound was closed in layers.
Pathology of the tumor showed that the sharply delineated central nidus was composed of more or less calcified osteoid lined by plump osteoblasts and growing within highly vascularised connective tissue, without evidence of inflammation. Strong cytoplasmic staining for COX-2 was noted by anti-COX-2 immunohistochemistry stain (Immuno-Biological Laboratories Co., Ltd. Japan) (Figure 4).
The amount of PGE2 was 4600 pg/mL in the fluid of the DRUJ, 228 pg/mL in the pronator quadratus muscle, and 3 pg/mL in the serum. The measurement of concentration of PGE2 was done by SRL, Inc Japan. The amount of PGE2 was almost 1500 times higher in the DRUJ and 76 times higher in the pronator quadratus compared with that in the serum (Figure 5).
The patient’s pain and range of motion improved following surgery. Two years postoperatively, her pain has disappeared completely, and both of her wrists show the same range of motion.
3. Discussion
Osteoid osteomas generally involve the diaphysis or metaphysis of long bones, accounting for 11% of all benign bone tumor [4]. Intra-articular osteoid osteomas account for approximately 10% - 13% of all osteoid osteomas and usually present as a monoarthropathy [3,5], which may result in the misdiagnosis of arthritis. Intra-articular location of osteoid osteomas at the wrist joint is very rare, but some reports have described osteoid osteomas involving the radial styloid [6-8], which are usually observed in the carpal bone [9]. To our knowledge, only 1 report has previously described an osteoid osteoma in the DRUJ, involving the distal ulna head [9]. No other reports have previously described an osteoid osteoma in the distal radius extending to the DRUJ.
Intra-articular osteoid osteomas are associated with non-specific symptoms such as pain, joint effusion, swelling, muscle atrophy, limitation of range of motion, and advanced epiphyseal development (maturation) [10,11]. Susa et al. (2009) reported that fibrous degeneration of the pronator teres causes rotational limitation of the forearm, as a result of the PGs produced by the osteoid osteoma [12].
The Snarr hypothesis stipulates that a specific activating substance may induce inflammation of the synovium in intra-articular osteoid osteomas [13], thereby increasing PG synthesis, particularly PGE2, as initially described by Makley and Dunn (1982); subsequent reports have shown that osteoid osteomas produce PGE2, F2, and I2 [1]. Especially, the synthesis of PG in the nidus was found to be 26 - 33 times greater than that in fragments of normal bone [1]. PGs are produced from phospholipids catalysed by cPLA2 and COX-2 through arachidonic acid metabolism. The 2 known forms of COX (commonly referred to as COX-1 and COX-2) are both non-selectively inhibited by traditional NSAIDs. COX-2 is induced rapidly and transiently when stimulated by inflammatory cytokines, and, more recently, has been shown to be up-regulated in various carcinomas and play a central role in tumor genesis. Some investigators have suggested that COX-2 expressed primarily by osteoblasts in the nidus play a major role in activating arachidonic acid metabolism, resulting in secondary synovitis of the involved joint in intra-articular osteoid osteomas. The expression and location of COX-2 in osteoid osteomas has previously been described [14,15]; however, the
 (a) (b)
(a) (b)
Figure 2. (a) Magnetic resonance (MR) imaging, coronal view (fast field echo). The white arrowhead indicates the bone tumor, which shows a high-intensity area; (b) MR imaging, coronal view (short-TI inversion-recovery), of the distal radioulnar joint, showing pooling of the effusion and oedema around the bone tumor (white arrowhead) extending to the distal ulna.
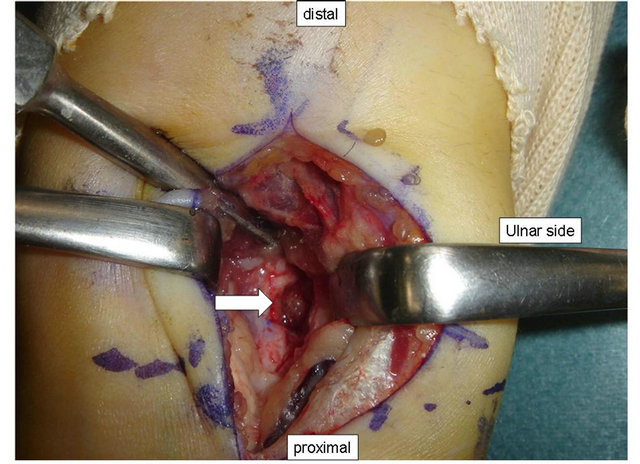
Figure 3. In the distal radius, the tumor (white arrow) became visible after part of the radial cortex was surgically removed.
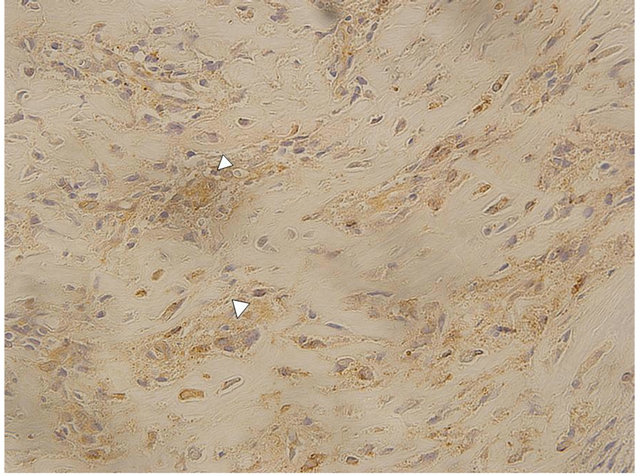
Figure 4. White arrowheads indicate the strong cyclooxygenase-2 staining in osteoblasts in the tumor (400× magnification).
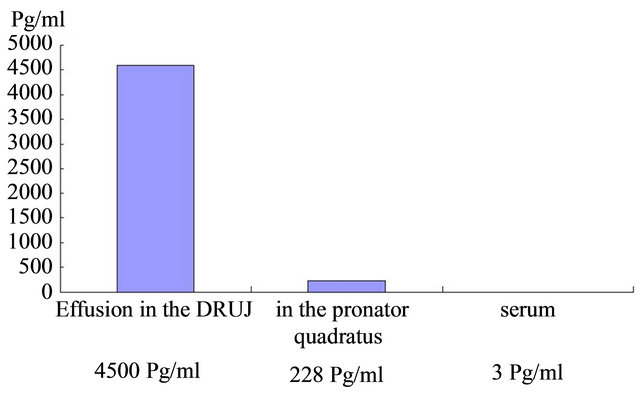
Figure 5. The concentration of prostaglandin E2 in the effusion of the distal radioulnar joint, pronator quadratus, and serum.
concentration of PGE2 in osteoid osteomas has not previously been measured and associated clinical information has not previously been reported, thereby making the present case unique. The osteoid osteomas might be COX-2 produced benign tumor.
REFERENCES
- J. T. Makley and M. J. Dunn, “Prostaglandin Synthesis by Osteoid Osteoma,” Lancet, Vol. 2, No. 8288, 1982, p. 42. doi:10.1016/S0140-6736(82)91174-6
- F. Greco, F. Tamburrelli and G. Ciabattoni, “Prostaglandins in Osteoid Osteoma,” International Orthopaedics, Vol. 15, No. 1, 1991, pp. 35-37. doi:10.1007/BF00210531
- F. Greco, F. Tamburrelli and G. Ciabattoni, “Intra-Articular Osteoid Osteoma,” Journal of Orthopaedic Traumatology, Vol. 18, No. 1, 1992, pp. 63-69.
- Y. Kawaguchi, et al., “Mechanism of Intramedullary High Intensity Area on T2-Weighted Magnetic Resonance Imaging in Osteoid Osteoma: A Possible Role of COX-2 Expression,” Pathology International, Vol. 51, No. 12, 2001, pp. 933-937. doi:10.1046/j.1440-1827.2001.01305.x
- S. D. Allen and A. Saifuddin, “Imaging of Intra-Articular Osteoid Osteoma,” Clinical Radiology, Vol. 58, No. 11, 2003, pp. 845-852. doi:10.1016/S0009-9260(03)00213-7
- G. D. Chloros, et al., “Osteoid Osteoma of the Radial Styloid Mimicking de Quervain Tenosynovitis,” Southern Medical Journal, Vol. 100, No. 10, 2007, pp. 1045-1047. doi:10.1097/SMJ.0b013e3181514ad5
- B. Fromm, A. Martini and E. Schmidt, “Osteoid Osteoma of the Radial Styloid Mimicking Stenosing Tenosynovitis. A Case Report.” The Journal of Hand Surgery, Vo1. 7, No. 2, 1992, pp. 236-238. doi:10.1016/0266-7681(92)90100-G
- L. De Smet, “Synovitis of the Wrist Joint Caused by an Intra-Articular Perforation of an Osteoid Osteoma of the Radial Styloid,” Clinical Rheumatology, Vol. 19, No. 3, 2000, pp. 229-230. doi:10.1007/s100670050163
- M. J. Kransdorf, et al., “Osteoid Osteoma,” Radiographics, Vol. 11, No. 4, 1991, pp. 671-696.
- T. W. Bauer, et al., “Juxta-Articular Osteoid Osteoma,” The American Journal of Surgical Pathology, Vol. 15, No. 4, 1991, pp. 381-387. doi:10.1097/00000478-199104000-00007
- K. Brabants, S. Geens and B. van Damme, “Subperiosteal Juxta-Articular Osteoid Osteoma,” Journal of Bone and Joint Surgery, Vol. 68, No. 2, 1986, pp. 320-324.
- M. Susa, et al., “Rotational Limitation of the Forearm Secondary to Osteoid Osteoma of the Radius,” The Journal of Rheumatology, Vol. 36, No. 9, 2009, pp. 2131- 2132. doi:10.3899/jrheum.090169
- J. W. Snarr, M. R. Abell and W. Martel, “Lymphofollicular Synovitis with Osteoid Osteoma,” Radiology, Vol. 106, No. 3, 1973, pp. 557-560.
- Y. Kawaguchi, et al., “Intraarticular Osteoid Osteoma Associated with Synovitis: A Possible Role of Cyclooxygenase-2 Expression by Osteoblasts in the Nidus,” Modern Pathology, Vol. 13, No. 10, 2000, pp. 1086-1091. doi:10.1038/modpathol.3880202
- D. V. Mungo, et al., “COX-1 and COX-2 Expression in Osteoid Osteomas,” Journal of Orthopaedic Research, Vol. 20, No. 1, 2002, pp. 159-162. doi:10.1016/S0736-0266(01)00065-1

