Surgical Science
Vol.3 No.11(2012), Article ID:24416,7 pages DOI:10.4236/ss.2012.311105
Liver Regeneration Effect of Oncostatin M Following Hepatectomy for the Rat Cirrhotic Liver Model
Second Department of Surgery, School of Medicine, Wakayama Medical University, Wakayama, Japan
Email: *ma@wakayama-med.ac.jp
Received September 3, 2012; revised October 3, 2012; accepted October 12, 2012
Keywords: Oncostatin M; Liver Regeneration; Cirrhosis; Anti-Fibrosis
ABSTRACT
Background/Aims: Liver resection represents the treatment of choice for hepatocellular carcinoma (HCC) arising in well-compensated cirrhosis. Gene expression of the multifunctional cytokine, Oncostatin M (OSM), stimulates liver regeneration and adenoviral vector expressing OSM (AdOSM) allows a persistent expression of the gene. The aim of this study is to evaluate the benefits of the preoperative injection of AdOSM to the remnant lobes to regenerate the liver. Methods: A 70% partial hepatectomy was performed in dimethylnitrosamine-administrated cirrhotic rats with a preoperative injection of AdOSM, adenoviral vector carrying β-galactosidase (AdLacZ), or phosphate-buffered saline (PBS). The morphologic, histologic, and biochemical changes in the remnant liver and survival rates were then assessed. Results: Portal injection with clamping the portal branches of the resected lobes for 5 min made it possible to effectively transduce the adenoviral vector into the remnant lobes. The ratio of the remnant liver weight/body weight (%) was 2.3 ± 0.5 in the AdOSM group, 1.1 ± 0.3 in the AdLacZ group (p < 0.001), and 1.6 ± 0.4 in the PBS group (p = 0.02). The fibrous ratio (%) was 21.3 ± 4.6 in the AdOSM group and 35.2 ± 4.5 in the AdLacZ group on day 4 after hepatectomy and fibrous status was significantly decreased in the AdOSM group (p = 0.02). Serum hyaluronic acid which is the indicator of liver fibrosis was 215 ± 141 ng/mL in the AdOSM group and 1963 ± 1225 ng/mL in the AdLacZ group (p = 0.03). Conclusions: The OSM gene therapy may increase the possibility of hepatectomy in a cirrhotic liver by improving fibrosis, hepatic function, and hepatocyte regeneration.
1. Introduction
Although recent demonstrations of satisfactory results of nonsurgical procedures such as percutaneous ethanol injection (PEI), radiofrequency ablation (RFA), and transcatheter arterial chemoembolization (TACE), liver resection still plays an important role in treatment of Hepatocellular carcinoma (HCC) [1]. However, liver failure following hepatectomy sometimes occurs when a liver resection volume exceeds safety limits. This is because the regenerative ability and liver functions are impaired in the patients with cirrhosis. Therefore, one should consider accelerating the remnant liver regeneration to prevent postoperative liver failure [2,3].
Previously administration of hepatocyte growth factor (HGF) was aimed to support liver regeneration for the cirrhotic partial hepatectomy models and showed preferable regeneration effects [4,5]. However, HGF itself has a potential to induce cancer growth or hepatocarcinogenesis and which were the subject to be settled.
On costatin M (OSM), a pleiotropic glycoprotein cytokine that was originally purified from the conditioned media of phorbol ester stimulated U937 (histocytic lymphoma) cells, is related structurally and functionally to the subfamily of neurotrophic cytokines known as the interleukin-6 (IL-6) [6]. The OSM was originally recognized as an inhibitor for tumor cell’s proliferation. However, the OSM presents many other biological activities such as in inflammation, development, and tissue remodeling in the injured liver [7-9].
In the liver, the OSM stimulates the expression of acute phase proteins and tissue inhibitor of metalloproteinase-1 (TIMP-1) [10]. It is well known that TIMP-1 plays an important role in remodeling of extracellular matrix during liver regeneration [11]. Among the IL-6 family cytokines the OSM was the strong inducer of the TIMP-1 [12]. Therefore, administration of the OSM might help to promote the remnant liver regeneration.
In general the cytokines are rapidly turned over, therefore continuous dosage are needed to keep its biological activity. To overcome the problem, several vectors including naked plasmid, liposome and adenoviral vector have been used [13].
The recombinant human adenovirus type 5 is frequently used for in vivo gene therapeutic applications. This virus infects a range of dividing and non dividing cells very efficiently and has an affinity to liver tissue in particular. Although adenovirus-related toxicity remains major concerns [14], it would still be a suitable vector for accelerating liver regeneration.
In this study, we evaluated the benefits of the preoperative injection of adenoviral vector expressing the OSM (AdOSM) to the remnant lobe to accelerate its regeneration following hepatectomy.
2. Material and Methods
2.1. Animals
During all the following experimental procedures, the animals were treated in accordance with Japanese Government Animal Protection and Management Law (No. 105), Japanese Government Notification on Feeding and Safekeeping of Animals (No. 6), the Guidelines for Animal Experiments of Wakayama Medical University, and the NIH Guide for the Care and Use of Laboratory Animals (Institute of Laboratory Animal Resources, National Research Council).
Seven-week-old male, Sprague-Dawley rats that weighed 200 to 250 g were purchased (Kiwa Jikken, Wakayama, Japan) and fed a nutritionally balanced rodent diet and water ad libitum. Liver fibrosis was induced by the intraperitoneal injections of 1% dimethylnitrosamine (DMN) (Nacalai tesque, Kyoto, Japan) dissolved in saline at 1 mL/kg body weight for 3 consecutive days per week for 3 weeks, which showed a pseudolobular formation that was similar to the characteristic pathologic changes found in liver cirrhosis in humans [15].
2.2. Replication-Defective Recombinant Adenoviral Vectors
E1-and E3-region-deleted replication defective adenoviral vectors expressing either human OSM (AdOSM) or β-galactosidase (AdLacZ) under a CA promoter (comprising a cytomegalovirus enhancer and chicken betaactin promoter) were obtained from the RIKEN Bio Resource Center (Tukuba, Japan). These vectors were propagated with 293 cells (Japan Collection of Research Bioresources, Osaka, Japan), which were human embryonic kidney cells transformed by the E1A and E1B genes. The viruses were purified twice with cesium chloride equilibrium centrifugation, then were dialyzed against phosphate buffered saline (PBS) with 10% glycerol, and titrated with 293 cells. Both viral titers of AdLacZ and AdOSM were 5.0 × 108 plaque forming unit (PFU)/mL. To confirm the expression of OSM or LacZ gene, HeLa cells (Japan Collection of Research Bioresources, Osaka, Japan) were cultured with the transduction of AdOSM or AdLacZ, as previously described [4]. Our previous study showed that the injection of high doses of AdLacZ (2 × 109 PFU) led to fetal hepatotoxicity and injection doses ranged from 5 × 108 to 1 × 109 PFU was safe in cirrhotic rats [4]. Therefore, injection dose in this study was also set to be 5 × 108 PFU.
2.3. Experimental Designs
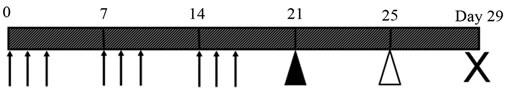
Figure 1. Experimental protocol. Intraperitoneal injection of dimethylnitrosamine (10 μg/g BW) on 3 consecutive days per week was performed for 3 weeks (arrows). Four days after the last DMN injection (Day 21), the rats were placed into 3 groups: rats were injected into the portal vain with either AdLacZ (5.0 × 108 PFU) in 0.5 mL PBS (AdLacZ group; n = 10), AdOSM (5.0 × 108 PFU) in 0.5 mL PBS (AdHGF group; n = 10) or only 0.5 mL of PBS (PBS group; n = 10) under ether anesthesia (black arrow head). These rats were performed a 70% partial hepatectomy 4 days after the portal injections (Day 25) under ether anesthesia (white arrow head). All the rats in each group were also followed and the living rats were sacrificed 4 days after hepatectomy (Day 29) to assess the liver regeneration activity (a cross mark).
Figure 1 showed schema of this experimental protocol. Four days after the last DMN injection, the rats were placed into 3 groups: rats were injected into the portal vain with either AdLacZ (5.0 × 108 PFU) in 0.5 mL PBS (AdLacZ group; n = 10); AdOSM (5.0 × 108 PFU) in 0.5 mL PBS (AdOSM group; n=10); or only 0.5 mL of PBS (PBS group; n = 10) under ether anesthesia. These rats were performed a 70% partial hepatectomy on day 4 after the portal injections under ether anesthesia according to the method described by our previously report [5]. All the rats in each group were also followed and the living rats were sacrificed on day 4 after hepatectomy to assess the liver regeneration activity.
2.4. Histological Examination
Paraffin-embedded liver sections were stained according to the Azan-Mallory method and then were observed microscopically to assess the fibrous status of the liver. Randomly different fields were selected from each of 50 slides (n = 5 rats in the each group) that were stained with Azan-Mallory with a light microscope at ×100 magnification. As a control, pathologic grades of liver of rats without hepatectomy were examined (20 fields of 5 rats in the each group). Hepatic damage was classified in the following manner: grade 1, hepatocellular necrosis and/ or cellular inflammatory reaction with preservation of normal architectural patterns; grade 2, increased fibrous
tissue and loss of normal architecture; grade 3, early pseudolobulization and fibrous bridging between periportal and pericentral areas (early cirrhosis); and grade 4, wide fibrous bands and extensive pseudolobulization with regenerating nodules (advanced cirrhosis) as previous report [16]. In this study, grades 3 and 4 were defined as cirrhosis.
To analyze quantitatively the fibrous status, a scionimaging software program (Scion corporation, Frederick, Md) was used to calculate the Azan-Mallory stained slides according to the ratio of the fibrosis area per unit area of the liver tissue (×40).
2.5. Biochemical Analyses
The serum obtained from blood samples was stored at –20˚C and used to measure aspartate transaminase (AST), alanine transaminase (ALT), albumin, total bilirubin, and hyaluronic acid to evaluate postoperative liver function. All parameters were measured according to standard laboratory methods.
2.6. Statistical Analysis
Data were analyzed using the Stat View J-4.5 software program (Abacus Concepts, Inc., Berkeley, CA). The relationships between categorical variables were tested using a chi-squared analysis and Fisher’s exact probability test. Survival was calculated using the Kaplan-Meier method, and comparisons of survival curves were made using the log-rank test. P-values of less than 0.05 were considered to be statistically significant.
3. Results
3.1. Prevention of DMN Inducing Liver Fibrosis by OSM Gene Therapy
Cirrhosis induced by the DMN injection was treated with PBS, AdLacZ, and AdOSM. The liver section of AzanMallory staining on day 4 after PBS, AdLacZ or AdOSM treatment were shown in Figures 2 (A)-(C). Centrilobular damage that consisted of hemorrhagic necrosis, congestion, and inflammatory cell infiltration was observed in AdLacZ and PBS treated rat livers, although these changes were scarcely found in the AdOSM treated rat livers. An image analysis indicated that the fibrosis area was 13.4% ± 3.9% in the AdOSM group, 30.1% ± 5.5% in the AdLacZ group (p < 0.001), and 26.8% ± 4.3% in the PBS group (p = 0.002). Pathological grades of 1, 2, 3, and 4 of the liver on day 4 after PBS, AdLacZ or A-dOSM treatment were shown in Table 1.
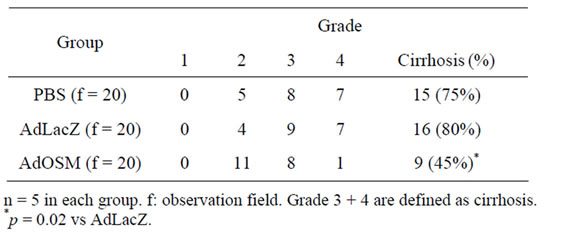
Table 1. Pathological grade of the liver on day 4 after PBS, AdLacZ or AdOSM administration.
The ratio of grade 3 + 4 (defined as liver cirrhosis) decreased in the AdOSM group, compared with the AdLacZ groups (p = 0.02). As for biochemical analyses on day 4 after PBS, AdLacZ or AdOSM treatment, serum levels of albumin and hyaluronic acid in AdOSM-treated rats were significantly improved in comparison with AdLacZ-treated rats (p = 0.03 and p = 0.01, respectively) (Table 2).
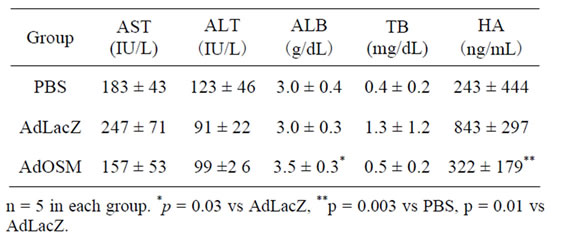
Table 2. Biochemical analyses on day 4 after PBS, AdLacZ or AdOSM administration.

Figure 2. Azan-Mallory staining (×40) of the remnant liver sections on day 4 after the adenoviral vectors injection. (A) Treated with PBS; (B) Treated with AdLacZ; (C) Treated with AdOSM. In livers treated with AdOSM, fibrotic tissue was decreased compared with the livers treated with PBS or AdLacZ.
3.2. Survival Evaluation of the Preoperative Administration of AdOSM into the Portal Vein after 70% Partial Hepatectomy
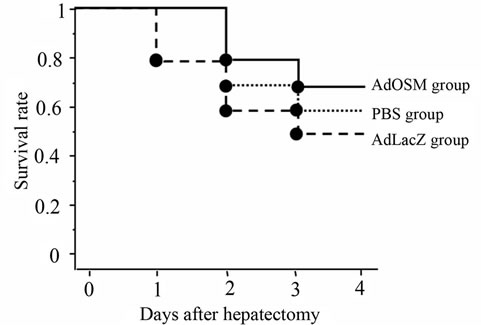
Figure 3. Survival curve after a 70% partial hepatectomy. The survival rates on day 4 after hepatectomy were 70% (7/10) in the AdOSM group, 50% (5/10) in the AdLacZ group, and 60% (6/10) in the PBS group.
Figure 3 shows the survival curves based on the KaplanMeier method after 70% hepatectomy among the three treatment groups. The survival rates on day 4 after-hepatectomy were 70% (7/10) in the AdOSM group, 50% (5/10) in the AdLacZ group, and 60% (6/10) in the PBS group. The rats that died within 4 days were autopsied, and all were found to have jaundice and extensive ascites. Macroscopic examination of the remnant liver on day 4 after hepatectomy showed that the surface and edge of the liver treated with AdOSM was smooth compared with these of AdLacZ and PBS groups (Figure 4).
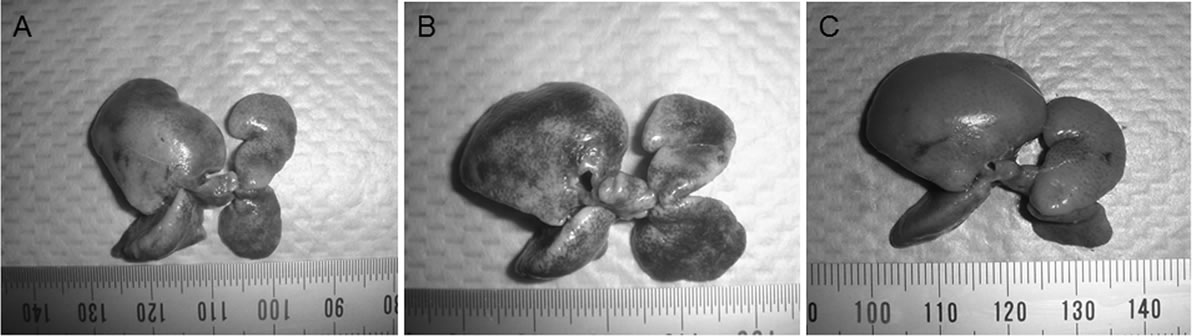
Figure 4. Macroscopic examination of the remnant liver on day 4 after hepatectomy. The surface and edge of the liver treated with PBS (A) and AdLacZ (B) were rough, whereas those were smooth in the group treated with AdOSM (C).
The ratios of the remnant liver weight/body weight (%) on day 4 after hepatectomy in the AdOSM, the AdLacZ, and the PBS groups were shown in Table 3.
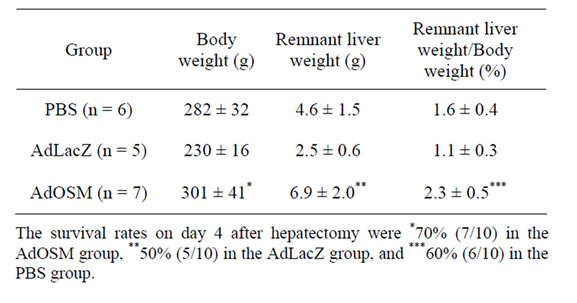
Table 3. Morphological regeneration of the remnant liver following a 70% partial hepatectomy.
A significant improvement of the ratio was found in the AdOSM group compared with the other groups.
Histological examination of the remnant liver on day 4 after hepatectomy was shown in Figure 5.

Figure 5. Azan-Mallory staining (×100) of the remnant liver on day 4 after hepatectomy. (A) Treated with PBS; (B) Treated with AdLacZ; (C) Treated with AdOSM. Advanced fibrotic changes were found in PBS and AdLacZ group.
Severe fibrotic changes with wide fibrous bands and extensive pseudolobulization were seen in the remnant liver of the
PBSand AdLacZ groups, whereas fibrosis was disappeared and slightly was seen in periportal area in the AdOSM group. An image analysis indicated that the fibrosis area was 21.3% ± 4.6% in the AdOSM group, 35.2% ± 4.5% in the AdLacZ group (p < 0.001), and 31.5% ± 5.2% In the PBS group (p = 0.003). Pathological grade of the remnant liver on day 4 after 70% hepatectomy were shown in Table 4.
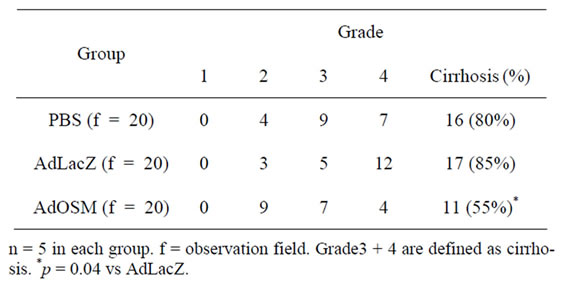
Table 4. Pathological grade of the remnant liver on day 4 after 70% hepatectomy.
The ratio of grade 3 + 4 (defined as liver cirrhosis) decreased in the AdOSM group, compared with the AdLacZ groups (p = 0.04). As for biochemical analyses on day 4 after 70% partial hepatectomy, serum levels of AST, ALT, total bilirubin and hyaluronic acid were significantly improved compared with AdLacZ-treated rats (p < 0.001, p = 0.002, p = 0.02 and p = 0.03, respectively) (Table 5).
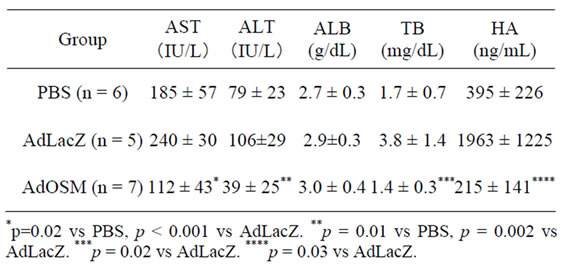
Table 5. Biochemical analyses on day 4 after 70% partial hepatectomy.
4. Discussion
In this study, the oncostatin M showed degradation of the fibrosis induced by DMN and accelerated liver regeneration after partial hepatectomy although in cirrhotic situation.
Hepatectomy can transiently increase the concentration of endogenous cytokines such as HGF and OSM, however, the cirrhotic liver impairs the response to endogenous these cytokines changes. Our previous study showed over expression of HGF recovered rat cirrhotic model to a clearly less cirrhotic condition and achieved a favorable regeneration [5]. It means that HGF played important role to liver growth, repair and regeneration following hepatectomy. However, the HGF effect on HCC or carcinogenesis must be considered [16]. On the other hand, carcinogenic effect is not noted OSM although a liver regeneration effect is accepted same as HGF. It suggested that HGF and OSM also play different roles in the hepatic regeneration, and HGF are essential for the initiation of hepatic differentiation including car cinogenesis, while OSM is essential for the maturation of hepatocytes [9].
OSM is able to induce the synthesis of acute phase proteins in hepatocytes and it is involved in the development, function, and regeneration of the liver such as following hepatectomy. For example, OSM-deficient mice exhibit defects in liver regeneration after partial hepatictomy and CCl4 liver injury [17]. In this study, we demonstrate that DMN administrated rat exhibit impaired liver regeneration, i.e., delayed hepatocyte proliferation, persistent liver fibrosis, and increased tissue destruction. We also show that the injection of AdLacZ in rats failed to liver regeneration after 70% hepatectomy and that AdOSM-administrated rats recovered the regenerative liver response, suggesting that OSM is a key downstream mediator of IL-6 in liver regeneration. We first show that consistent with these observations, restoration of liver mass after 70% hepatectomy was rapid in AdOSM livers. Especially, the recovery rate during the initial 4 days was markedly increased in AdOSM-treated rats, while it was comparable to AdLacZ-treated rats 4 days after hepatectomy. Although we did not examine the direct effects of OSM on the cell cycle progression, our results suggest that OSM mediated signaling is required for the early proliferative response of hepatocytes to hepatectomy.
As for the mechanism of hepatocyte regeneration with OSM after hepatectomy, it is known to enhance hepatic regeneration of hepatoblasts by inducing the activation of the signal transducer and activator of transcription 3 (STAT3) pathways [18]. STAT3 mediates the OSM signaling for functional maturation of hepatocytes into hepatectomized liver cells and morphological changes including the formation of E-cadherin-based adherence junctions [19]. OSM, IL-6, and leukemia-inhibitory factor share STAT3 signaling cascades, and OSM exploit the distinct hepatocytic differentiation signaling in an OSM receptor (OSMR) [20]. Although the regulatory mechanisms of OSMR are still unclear, it is estimated that OSMR expression is regulated by a signaling pathway activated during the process of hepatogenesis. The regulation of OSM signaling might be largely dependent on the expression status of OSMR in residual liver tissues after hepatectomy, because this functional redundancy can be explained by the shared use of the receptor subunit gp130 [21].
5. Conclusion
The preoperative portal injection of the OSM gene promoted liver regeneration and attenuated liver fibrosis after hepatectomy in cirrhotic rats without hepatotoxicity. This system might be appropriate for clinical trials consisting of the preoperative percutaneous transhepatic portal injection system under ultrasonographic guidance to prevent the postoperative liver failure.
6. Acknowledgements
Authors thank for RIKEN Bio Resource Center for providing E1-and E3-region-deleted replication defective adenoviral vectors expressing human OSM and β-galactosidase.
REFERENCES
- K. Uchiyama, M. Ueno, T. Hama, M. Kawai, M. Tani, H. Terasawa, et al., “Recurrence of Primary Hepatocellular Carcinoma after Hepatectomy—Differences Related to Underlying Hepatitis Virus Species,” Hepatogastroenterology, Vol. 52, No. 62, 2005, pp. 591-595.
- K. Uchiyama, K. Mori, K. Tabuse, M. Ueno, S. Ozawa, T. Nakase, et al., “Assessment of Liver Function for Successful Hepatectomy in Patients with Hepatocellular Carcinoma with Impaired Hepatic Function,” Journal of Hepatobiliary Pancreatic Surgery, Vol. 15, No. 6, 2008, pp. 596-602 doi:10.1007/s00534-007-1326-2
- M. Ueno, K. Uchiyama, S. Ozawa, S. Hayami, S. Kiriyama and H. Yamaue, “A New Prediction Model of Postoperative Complications after Major Hepatectomy for Hepatocellular Carcinoma,” Digestive Surgery, Vol. 26, No. 5, 2009, pp. 392-329. doi:10.1159/000229973
- S. Ozawa, K. Uchiyama, M. Nakamori, K. Ueda, M. Iwahashi, H. Ueno, et al., “Combination Gene Therapy of HGF and Truncated Type II TGF-Beta Receptor for Rat Liver Cirrhosis after Partial Hepatectomy,” Surgery, Vol. 139, No. 4, 2006, pp. 563-573. doi:10.1016/j.surg.2005.10.003
- M. Ueno, K. Uchiyama, M. Nakamori, K. Ueda, M. Iwahashi, S. Ozawa, et al., “Adenoviral Vector Expressing Hepatocyte Growth Factor Promotes Liver Regeneration by Preoperative Injection: The Advantages of Performing Selective Injection to the Remnant Lobe,” Surgery, Vol. 141, No. 4, 2007, pp. 511-519. doi:10.1016/j.surg.2006.10.006
- T. Hamada, A. Sato, T. Hirano, T. Yamamoto, G. Son, M. Torii, et al., “Oncostatin M Gene Therapy Attenuates Liver Damage Induced by Dimethylnitrosamine in Rats,” American Journal of Pathology, Vol. 171, No. 3, 2007, pp. 872-881. doi:10.2353/ajpath.2007.060972
- K. Nakamura, H. Nonaka, H. Saito, M. Tanaka and A. Miyajima, “Hepatocyte Proliferation and Tissue Remodeling Is Impaired after Liver Injury in Oncostatin M Receptor Knockout Mice,” Hepatology, Vol. 39, No. 3, 2004, pp. 635-644. doi:10.1002/hep.20086
- I. Znoyko, N. Sohara, S. S. Spicer, M. Trojanowska and A. Reuben, “Expression of Oncostatin M and Its Receptors in Normal and Cirrhotic Human Liver,” Journal of Hepatology, Vol. 43, No. 5, 2005, pp. 893-900. doi:10.1016/j.jhep.2005.04.020
- X. J. Dong, H. Zhang, R. L. Pan, L. X. Xiang and J. Z. Shao, “Identification of Cytokines Involved in Hepatic Differentiation of mBM-MSCs under Liver-Injury Conditions,” World Journal of Gastroenterology, Vol. 16, No. 26, 2010, pp. 3267-3278. doi:10.3748/wjg.v16.i26.3267
- C. D. Richards, M. Shoyab, T. J. Brown and J. Gauldie, “Selective Regulation of Metalloproteinase Inhibitor (TIMP-1) by Oncostatin M in Fibroblasts in Culture,” Journal of Immunology, Vol. 150, No. 12, 1993, pp. 5596- 5603.
- C. Kerr, C. Langdon, F. Graham, J. Gauldie, T. Hara and C. D. Richards, “Adenovirus Vector Expressing Mouse Oncostatin M Induces Acute-Phase Proteins and TIMP-1 Expression in Vivo in Mice,” Journal of Interferon & Cytokine Research, Vol. 19, No. 10, 1999, pp. 1195-1205. doi:10.1089/107999099313145
- F. M. Botelho, D. R. Edwards and C. D. Richards, “Oncostatin M Stimulates c-Fos to Bind a Transcriptionally Responsive AP-1 Element within the Tissue Inhibitor of Metalloproteinase-1 Promoter,” Journal of Biological Chemistry, Vol. 273, No. 9, 1998, pp. 5211-5218. doi:10.1074/jbc.273.9.5211
- F. Xue, T. Takahara, Y. Yata, Y. Kuwabara, E. Shinno, K. Nonome, et al., “Hepatocyte Growth Factor Gene Therapy Accelerates Regeneration in Cirrhotic Mouse Livers after Hepatectomy,” Gut, Vol. 52, No. 5, 2003, pp. 694- 700. doi:10.1136/gut.52.5.694
- S. L. Brody and R. G. Crystal, “Adenovirus-Mediated on Vivo Gene Transfer,” Annals of the New York Academy of Sciences, Vol. 716, 1994, pp. 90-101. doi:10.1111/j.1749-6632.1994.tb21705.x
- D. J. Pritchard and W. H. Butler, “Apoptosis—The Mechanism of Cell Death in Dimethylnitrosamine-Induced Hepatotoxicity,” Journal of Pathology, Vol. 158, No. 3, 1989, pp. 253-260. doi:10.1002/path.1711580314
- L. Zender and S. Kubicka, “Molecular Pathogenesis and Targeted Therapy of Hepatocellular Carcinoma,” Onkologie, Vol. 31, 2008, pp. 550-555. doi:10.1159/000151586
- D. E. Cressman, L. E. Greenbaum, R. A. DeAngelis, G. Ciliberto, E. E. Furth, V. Poli, et al., “Liver Failure and Defective Hepatocyte Regeneration in Interleukin-6-Deficient Mice,” Science, Vol. 274, No. 5291, 1996, pp. 1379-1383. doi:10.1126/science.274.5291.1379
- A. Kamiya, T. Kinoshita, Y. Ito, T. Matsui, Y. Morikawa, K. Nakashima, et al., “Fetal Liver Development Requires a Paracrine Action of Oncostatin M through the gp130 Signal Transducer,” EMBO Journal, Vol. 18, No. 8, 1999, pp. 2127-2136. doi:10.1093/emboj/18.8.2127
- Y. Ito, T. Matsui, A. Kamiya, T. Kinoshita and A. Miyajima, “Retroviral Gene Transfer of Signaling Molecules into Murine Fetal Hepatocytes Defines Distinct Roles for the STAT3 and Ras Pathways during Hepatic Development,” Hepatology, Vol. 32, No. 6, 2000, pp. 1370-1376. doi:10.1053/jhep.2000.19815
- T. Kinoshita and A. Miyajima, “Cytokine Regulation of Liver Development,” Biochimica et Biophysica Acta, Vol. 1592, No. 3, 2002, pp. 303-312. doi:10.1016/S0167-4889(02)00323-3
- T. Yamashita, M. Honda, K. Nio, Y. Nakamoto, T. Yamashita, H. Takamura, et al., “Oncostatin m Renders Epithelial Cell Adhesion Molecule-Positive Liver Cancer Stem Cells Sensitive to 5-Fluorouracil by Inducing Hepatocytic Differentiation,” Cancer Research, Vol. 70, No. 11, 2010, pp. 4687-4697. doi:10.1158/0008-5472.CAN-09-4210
NOTES
*Corresponding author

