Journal of Sustainable Bioenergy Systems
Vol.05 No.03(2015), Article ID:59941,11 pages
10.4236/jsbs.2015.53011
Lipid Production by Rhodotorula glutinis from Pulp and Paper Wastewater for Biodiesel Production
Marta Amirsadeghi1, Sara Shields-Menard2, W. Todd French1*, Rafael Hernandez3
1Dave C. Swalm School of Chemical Engineering, Mississippi State University, Starkville, Mississippi, USA
2Department of Biological Science, Mississippi State University, Starkville, Mississippi, USA
3College of Engineering, University of Louisiana at Lafayette, Lafayette, Louisiana, USA
Email: *French@che.msstate.edu
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 17 August 2015; accepted 22 September 2015; published 25 September 2015
ABSTRACT
This study investigated the potential of oleaginous yeast Rhodotorula glutinis utilizing pulp and paper wastewater effluents as cultivation media for the sustainable production of microbial lipids as biodiesel feedstock. R. glutinis is oleaginous yeast, which has the ability to produce significant quantities of intercellular lipids in the form of triacylglycerols. Yeast lipids are a promising potential feedstock for biodiesel production due to similar fatty acid composition to plant oils. The effect of various carbon sources on biomass production, lipid accumulation, substrate utilization, and fatty acid composition using R. glutinis in the pulp and paper wastewater media was studied. The pulp and paper wastewater was supplemented with glucose, xylose, and glycerol as carbon sources under nitrogen-limited conditions. The maximum lipid productions of 1.3 - 2.9 g∙L−1, which corresponded to the intracellular lipid contents of 8% - 15% cell dry weight (CDW), were obtained under various carbon substrates. A kinetic study of the batch fermentation was performed in a 3 L aerobic batch fermenter to describe the cell growth, lipid accumulation, and substrate utilization process, and the kinetic parameter was estimated. The fatty acid profile of oleaginous yeast was rich in palmitic, oleic, and linoleic acids and comparable to vegetable oils. Thus, the results of this study indicated that pulp and paper wastewater could be used to produce lipids as biodiesel feedstock.
Keywords:
Biofuels, Lipid Production, Biodiesel Feedstock, Rhodotorula glutinis, Pulp and Paper Wastewater

1. Introduction
The utilization of biodiesel has greatly increased over the past two decades due in part to concerns about the environmental consequences of fossil fuel combustion, the low lubricity quality of ultralow sulfur diesel, and the rapid rises in crude oil prices that can occur. Biodiesel, a mixture of fatty acid methyl esters (FAMEs) derived traditionally from vegetable oils and animal fats, is one of the most prominent renewable energy resources [1] . The cost of feedstock for producing biodiesel is responsible for almost 80% - 95% of the total cost of the process [2] . Oleaginous microorganisms that can accumulate 20% - 80% of their dry weight in the form of lipids are considered as alternative raw material for biodiesel production [3] . It has been shown that these microorganisms utilize glucose, xylose, glycerol, food industry waste [4] , and N-acetylglucosamine (GlcNAc) [5] for the production of lipids.
Glucose is one of the most common carbon substrates for producing microbial biomass and lipids, which has been extensively investigated for use by different oleaginous microorganisms [6] [7] . However, the use of starch or other corn sugars as a carbon source results in conflicts between the food and biofuel industries and adds significant cost to the final product. Using lignocellulosic based materials, which are representative of the largest potential of fermentable sugars, is promising as it can drastically lower the costs of microbial lipid production [8] . The depolymerization of cellulose and hemicellulose in lignocellulosic material results in a carbon source containing glucose and xylose; thusly the co-fermentation of mixed sugars must be explored for effective lipid production. Glycerol is another carbon source, which can be used as a substrate for lipid production. Since glycerol is the major by-product of biodiesel manufacturing, its usage as a carbon source for lipid accumulation may provide an additional benefit of offsetting the cost of biodiesel production. Glycerol accounts for 10% of the product output. Approximately 1 kg of glycerol could be obtained for 3.78 liters of produced biodiesel [9] .
Oleaginous microorganisms require high carbon to nitrogen ratios and the fermentation process requires high amounts of water and nutrients in the media to produce biomass with high contents of storage lipids. Various fermentation substrates have been investigated to evaluate the capability of oleaginous microorganisms in terms of growth and product formation, such as starch wastewater, molasses, wastewaters from potato, fruit juice and lettuce processing, olive oil manufacturing wastewater [10] , and municipal wastewater [11] . Wastewater with no alternative application was considered in this work instead of the wastewaters that can be used for further purposes such as animal feed.
The pulp and paper industry is the fifth largest economy in the US and produces a considerable amount of wastewater. Furthermore, the pulp and paper industry can be considered as one of the largest industries in terms of water and energy utilization, consuming between 20,000 and 60,000 gallons of water per ton of product [12] . Large amounts of wastewater are produced from different steps of the pulp and paper making process as a result of high water consumption [13] . The general characteristics of the pulp and paper wastewater include high COD, high Biochemical Oxygen Demand (BOD), Suspended Solids (SS), and dark brown in coloration [14] .
The present work focuses on the production of microbial lipid to be used as biodiesel feedstock through batch aerobic fermentation of pulp and paper wastewater. Pulp and paper wastewater has been considered as a fermentation medium in order to reduce some of the cost associated with this process. The effects of the utilization of different carbon sources such as glucose, xylose, and glycerol by Rhodotorula glutinis on the lipid productivity were investigated. Kinetic models were applied to describe the processes of microbial growth, product formation, and substrate utilization, which can be further used for the process scale up and bioreactor design in a similar system.
2. Materials and Methods
2.1. Yeast Inoculum Preparation
R. glutinis (ATCC 15125) was obtained from American Type Culture Collection (ATCC). Sub-cultures of R. glutinis (ATCC 15125) was produced by adding an aliquot of R. glutinis stock, which was stored at -80˚C, into the flask of sugar broth media [1 g∙L−1 Yeast Extract, 1 g∙L−1 Na2HPO4∙12 H2O, 1 g∙L−1 KH2PO4, 0.4 g∙L−1 MgSO4∙7H2O, 1.6 g∙L−1 (NH4)2SO4, 10.0 ml Trace Mineral Solution (3.6 g∙L−1 CaCl2∙2H2O, 0.75 g∙L−1 ZnSO4∙7H2O, 0.13 g∙L−1 CuSO4∙5H2O, 0.5 g∙L−1 MnSO4∙H2O, 0.13 g∙L−1 COCl2.6 H2O, 0.17 g∙L−1 Na2MoO4.2 H2O in distilled water), 6.0 ml Iron Solution (FeSO4∙7H2O in distilled water), and 60 g∙L−1 glucose], were incubated for 4 days using a rotary shaker (New Brunswick Scientific Model I26, Edison, New Jersey) with a rotation speed of ~112 rpm at 30˚C. The media compounds were obtained from Fisher Scientific.
2.2. Wastewater Collection and Characterization
The pulp and paper wastewater was obtained from the International Paper Mill in Redwood, MS. Multiple samples of wastewater were collected and stored in plastic container. After collection, the wastewater containers were placed on ice for 24 hours prior to conducting the fermentation experiment. The wastewater was characterized prior to being used as a fermentation medium as discussed below. The pH of the wastewater was measured using a bench top pH meter (Accumet Research AR25 Dual Channel pH/Ion Meter, Fisher Scientific). The COD of the wastewater was determined according to EPA standard method [15] , and residual Ammonium-Nitrogen level was analyzed using ICS 3000 ion chromatograph (Dionex Corp, Sunnyvale, CA, USA). The initial characteristics of the wastewater and its place of origin are provided in Table 1. Based on the initial characteristics of the wastewater, low nutrients levels and moderately low COD values were recorded with this wastewater source. Since the carbon to nitrogen ratio dictates the amount of oil accumulated within these microorganisms, the wastewater was supplemented with 60 g∙L−1 of a carbon source (glucose, xylose, and glycerol), and nutrients (1 g∙L−1 K2HPO4, 1.5 g∙L−1 Na2H2PO4 and NH4Cl) prior to the experiment. The concentration of ammonium chloride as a nitrogen source was calculated to obtain a C:N ratio of 70:1.
2.3. Fermentation Experiment
The pulp and paper wastewater (2.4 L) was amended with 60 g∙L−1 carbon sources in 4 individual experiments including glucose, xylose, glycerol, and glucose-xylose (2:1) ratio and seeded with a 20% initial inoculum of R. glutinis (600 ml) in 3 L Bioflo 310 fermenter (New Brunswick Scientific, Edison, NJ) with an initial R. glutinis cell concentration of 10 g∙L−1. The pH of the amended wastewater was adjusted to 6.5 using 1M NaOH solution, and the temperature was controlled at 25˚C. The aeration rate was adjusted at 3 volume of air per volume of medium and per minute (vvm). The agitation rate was set at 300 rpm, and adjusted to maintain a Dissolved Oxygen (DO) level no lower than 60% saturation during the experiment. A polypropylene-based antifoam, 1:10 dilution of nonoil (Sigma-Aldrich, St. Louis, MO) was periodically added to prevent foaming in the vessel. Samples were obtained from the aerobic batch fermenter every 12 h interval during the first 48 h of the experiment and then every 24 hours during the 7 days of incubation time. Control experiment was conducted using R. glutinis starter cultured in sugar broth media to compare the potential lipid production by R. glutinis using pulp and paper wastewater and the optimal media components.
2.4. Analytical Methods
2.4.1. Cell Mass Concentration
For each treatment, 30 ml of culture samples were taken and centrifuged at 3000 rpm, at 25˚C for 10 minutes using a Sorvall® ST 40 Centrifuge (Thermo Fisher Scientific, Waltham, MA, USA) to separate the supernatant from the cell pellets. Cell pellets were placed in the freezer at −20˚C overnight, and then freeze-dried using a Labconco Freezone 2.5 freeze drier followed by gravimetric weight measurements to determine the cell mass concentration. The supernatant was stored in the freezer at −20˚C to be used for the analysis of sugar, and nitrogen.
2.4.2. Lipid Extraction and Conversion of Lipids to Fatty Acid Methyl Esters
The intercellular lipid content of the freeze-dried samples was determined based on the modified Bligh and Dyer
Table 1. Pulp and paper wastewater initial characteristics (before treatment).
method [16] as described by Revellame et al. [17] . The lipid content of the samples was expressed by the percentage of lipid in cell dry weight. The dried lipid residues were converted into fatty acid methyl esters (FAMEs) by transesterification using sulfuric acid and methanol as catalyst and reactant, respectively [18] . The fatty acid composition in the FAMEs was analyzed using an Agilent 6890 gas chromatograph equipped with a flame ionization detector (GC-FID). Helium was used as carrier gas. The injector temperature of 260˚C was used for the injection of the 1 µL Samples. The GC oven temperature was set at 50˚C, and then ramped to 250˚C after 2 min. The FID temperature was kept at 260˚C during the analysis [19] .
2.4.3. Determination of Residual Sugars, and Ammonium-Nitrogen
The supernatant samples were analyzed using YSI 2900 Biochemistry analyzer (YSI Inc. Life Sciences, Yellow Springs, OH, USA) equipped with a glucose oxidase, xylose oxidase, and glycerol oxidase membrane probe for the measurement of glucose, xylose, and glycerol, respectively. The residual ammonium-nitrogen level was analyzed using an ICS 3000 ion chromatography (Dionex Corp, Sunnyvale, CA, USA) to monitor the point of nitrogen depletion, and nitrogen level in the wastewater. ICS is equipped with an IonPac CS16 cation exchange analytical column (250 mm × 4 mm) and CG16 guard column (50 mm × 4 mm) and a conductivity detector.
2.5. Statistical Analysis
Statistical analysis was carried out using SAS® 9.4 (Copyright © 2015, SAS Institute Inc., Cary, NC, USA). The statistical significance of differences in mean values was analyzed using one-way analysis of variance (ANOVA) with Tukey-Test at 95% confidence intervals.
3. Results and Discussion
3.1. Growth and Lipid Production by R. glutinis Using Different Carbon Substrates
It was hypothesized that the oleaginous yeast R. glutinis can produce microbial lipids from pulp and paper wastewaters amended with carbon sources, which can be further converted to biodiesel. In this study, carbon to nitrogen ratio of 70:1 with initial nitrogen and carbon source concentrations of 1.3 and 60 g∙L−1 was used, respectively. The fermentation profiles were plotted and presented in Figure 1 with the error bars that represent the standard deviation of three independent replicates. Figures 1(a)-(d) show the results of the fermentation profile over the 7 days of fermentation. From Figures 1(a)-(d), it can be seen that fat-free biomass increased significantly within the first 96 - 120 hours of cultivation and stayed constant after ~120 hours for all the carbon sources except xylose. No lag phase was observed for the growth of R. glutinis for all the carbon substrates except xylose. This is most likely due to the fact that samples were not taken during the first 12 hours of incubation when the lag phase would have occurred. In the case of xylose utilization, it can be seen from Figure 1(b) and Figure 1(c), that growth and lipid accumulation started after 24 hours lag phase. The xylose utilization was observed to lag within the first 24 hours, which could be attributed to the synthesis of enzymes by the oleaginous yeast for xylose utilization. Similar results were observed when co-fermentation of glucose and xylose was used by activated sludge microorganisms [20] . A complete depletion of ammonium-nitrogen level was observed after 48 hours of fermentation regardless of the type of substrate initially provided. In all the treatments, growth and lipid accumulation reached the maximum non-lipid biomass and lipid concentration of 12 - 17 g∙L−1 and 1.3 - 2.9 g∙L−1, respectively, which correspond to the intracellular lipid contents of 8% - 15% CDW. There were no significant differences in biomass production among the treatments using different substrates. However, glycerol grown R. glutinis was significantly higher in terms of lipid accumulation among all the treatments. A maximum lipid content of 15% ± 0.92% CDW was obtained when glycerol was used as a substrate, which could be attributed to the highest lipid accumulation. As shown in Figure 2, after glycerol, xylose grown R. glutinis cells reached the maximum lipid content of 12.6% ± 2.65% CDW.
However, in the control experiment under similar fermentation conditions, R. glutinis accumulated lipids up to 27% ± 4.6% on a cellular biomass basis with 21 ± 3.12 gL−1 biomass production within 6 days, which was significantly higher than all the treatments in terms of biomass and lipid production. Lower lipid content of R. glutinis cultivated in pulp and paper wastewater could be due to the presence of organic material and lignin breakdown products in the wastewater, which acts as inhibitory compounds on lipid synthesis. However, in the control experiment, a combination of (NH4)2SO4 and yeast extract was used as a nitrogen source, but in the


Figure 1. Fermentation profile of R. glutinis grown on pulp and paper wastewater using different substrates: (a) Pure glucose; (b) Pure xylose; (c) Glucose-xylose (2:1) ratio; and (d) Pure glycerol.
Figure 2. Percentage of total lipid extract and FAMEs yield based on cell dry weight (% CDW).
experiment using wastewater as a medium, NH4Cl was replaced as a nitrogen source to discard the negative effects of sulfate on the final wastewater. It is reported that organic nitrogenous compounds such as yeast extract and peptone have a positive effect on lipid production, but negatively affect the cell growth; conversely, inorganic nitrogen sources like (NH4)2SO4 and NH4Cl are effective for cell growth, but not favorable for lipid production [21] . Since nutrient composition can have different effects on the growth and lipid accumulation, the lower lipid content could be as a result of using a different nitrogen source. Thus, further investigations need to be done to achieve a more detailed knowledge about the effects of nutrient compound on the cell growth and lipid accumulation.
3.2. Utilization of Different Substrates by R. glutinis
R. glutinis was able to completely deplete the supplied carbon sources in 7 days of fermentation except in the treatments where xylose was supplied (Figure 1). It was observed that considerable amounts of xylose remained in the media after 7 days of cultivation when pure xylose and also xylose in combination with glucose was used. Approximately, 22% of the initial xylose sugar remained in the fermenter when xylose was used as a sole substrate. When substrate combination of glucose-xylose was used, the residual xylose remaining was about 46%. The sequential utilization of glucose followed by xylose was observed (Figure 1(d)), which indicates that the consumption of glucose was favored by R. glutinis. The sequential utilization of glucose and xylose could be due to synthesizing enzymes by R. glutinis in order to metabolize xylose [20] . Recent studies indicated that oleaginous yeasts tend to sequentially convert the mixed sugars. The sequential substrate consumption pattern was reported for the yeast Lipomyces starkeyi [22] . However, a simultaneous utilization of glucose and xylose rather than sequential pattern, no diauxic growth behavior, and no presence of lag phase was reported when oleaginous yeast strain Trichosporon cutaneum was used in a stirred-tank bioreactor [23] . In this study, it was apparent from the data that R. glutinis is following a classic diauxic growth curve.
3.3. Kinetic Modeling
Monod or logistic equations can be used to explain the growth process of most microorganisms [6] [24] [25] . The logistic equation was selected due to its simplicity for calculating fermentation kinetic parameters. Fat-free biomass was characterized using the logistic model and presented as:
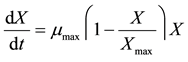 (1)
(1)
where
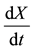 is the rate of cell growth;
is the rate of cell growth;
 (h−1) is the maximum specific growth rate; X (g×L−1) is the fat-free
(h−1) is the maximum specific growth rate; X (g×L−1) is the fat-free
biomass concentration at any time t (h);
 is the maximum value of fat-free biomass in the medium. Assuming that at the beginning of the fermentation (t = 0), the fat-free biomass was given by its initial value
is the maximum value of fat-free biomass in the medium. Assuming that at the beginning of the fermentation (t = 0), the fat-free biomass was given by its initial value , integrating Equation (1) becomes:
, integrating Equation (1) becomes:
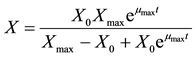 (2)
(2)
Product formation was modeled by a Luedeking-Piret equation [26] . The rate of lipid accumulation depends
on the instantaneous fat-free biomass (X) and fat-free biomass rate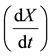 :
:
 (3)
(3)
where
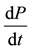 is the lipid accumulation rate; n and m are empirical constants stand for the non-growth correlation
is the lipid accumulation rate; n and m are empirical constants stand for the non-growth correlation
coefficient and growth correlation coefficient. After substituting
 by Equation (2) in Equation (3), and in-
by Equation (2) in Equation (3), and in-
tegrating assuming at t = 0, the concentration of lipid is equal to its initial concentration

The substrate was utilized primarily for cell growth, along with product formation and cell maintenance. The substrate utilization was described by Luedeking-Piret equation [26] , expressing the rate of substrate utilization
as a function of instantaneous fat-free biomass rate

coefficient term:

where



and



where

After substituting

pressing the substrate concentration:

where S0 is the initial substrate concentration at the beginning of the fermentation (t = 0).
In this study, the kinetic models expressing cell growth, lipid accumulation, and substrate utilization were fitted to the experimental data for estimation of the model kinetic parameters. The kinetic equations were solved using a nonlinear regression method. Matlab (MATLAB Release 2014a, The MathWorks, Inc., Natick, Massachusetts, United States) was used for data analysis using nonlinear least squares method (Levenberg-Marquardt algorithm) for minimizing residual sum of square of errors (RSSE). The kinetic parameters related to biomass growth (





The values of maximum specific growth rate


Table 2. The values of kinetic parameters for R. glutinis cultivated in pulp and paper wastewater supplemented with different substrate sources.
3.4. FAMEs Yield and Fatty Acid Profile of Oleaginous Yeast
The fatty acid composition of oleaginous yeasts was greatly dependent on the type of medium used and the growth condition. Some cultivation conditions such as temperature, pH of the medium, the type of the substrate, C:N ratio of the culture, and available oxygen for microorganisms can affect the accumulated lipids and derived fatty acids profile. The results of the characterization of total extracted lipid and FAMEs yield by transesterification of the lipids within 7 days of fermentation were shown in Figure 2. The transesterification yield was in the range of 40% - 60% (w/w) based on extracted lipid for all the treatments.
In this investigation, the fatty acid profile of R. glutinis was determined using FAMEs analysis in order to evaluate the potential of using this oleaginous yeast as biodiesel feedstock. Figure 3 shows the composition of the produced FAMEs from transesterified R. glutinis lipids. According to Figure 3, palmitic acid (C16:0), oleic acid (C18:1), and linoleic acid (C18:2) were the main intracellular fatty acids, which accounted for approximately 22% - 25%, 26% - 35%, and 23% - 27% of the total FAMEs for all the substrates, respectively. The palmitic acid (C16:0) level, which was the majority of the saturated fatty acids, did not change considerably by the type of carbon sources and remained in the order of 22% - 25%. Similar results were observed when other types of oleaginous microorganisms such as Mortierella isabellina and Cunninghamella echinulata [27] [28] , and Lipomyces starkeyi [22] were used. The maximum level of monounsaturated fatty acid was achieved in glucose grown R. glutinis, which was 34% ± 0.85%.
The composition of the FAMEs was comparable to the fatty acid profile of the soybean and rapeseed oil, which are the commonly used biodiesel feedstocks in the US and the EU, respectively. Comparing the fatty acid composition of R. glutinis in this study with soybean and rapeseed oil indicates that the FAMEs derived from transesterification of lipids from R. glutinis were more saturated (Table 3). Increased levels of saturated fatty acids improve the quality of the biodiesel such as increasing the cetane number, decreasing the level of nitrogen
Figure 3. Fatty acid composition of transesterified lipids produced by R. glutinis grown in pulp and paper wastewater.
Table 3. Comparison of the saturated, monounsaturated, and polyunsaturated fatty acids and their corresponded cetane number, iodine value, saponification value, and higher heating value for the common vegetable oils biodiesel feedstock with R. glutinis lipids.
oxide, and increasing oxidative stability. On the other hand, the unsaturated fatty acids are responsible for improving cold flow property of the biodiesel [29] . Thus, the quality of a biodiesel produced from this feedstock could have improved in terms of oxidative stability, cetane number (CN), and nitrogen oxide level (decreased NOx), which are attributed to the higher levels of saturated fatty acids and lower levels of polyunsaturated fatty acids.
3.5. Prediction of the Quality of the Biodiesel from Fatty Acid Composition
To evaluate the quality of the biodiesel, which can be produced from R. glutinis lipids, some fuel properties such as cetane number, iodine value, saponification value, and higher heating value were estimated using available published models. Bamgboye and Hansen showed that the cetane number can be predicted by developing an equation, which relates the cetane number of biodiesel fuels with FAMEs composition of the feedstock used for biodiesel production [30] . One of the properties of the biodiesel that affect the long-term storage of the fuel is oxidative stability of the fuel. The most common method to compare the oxidative stability of the biodiesel fuels is to determine the iodine number or iodine value, which is the indication of a higher numbers of double bonds. Although iodine value may not be the best measurement of oxidative stability since it does not take into consideration the double bond positions for oxidation in fatty acid chain, it can be considered as a rough estimate for evaluating the biodiesel stability properties. A theoretical model was developed to predict the iodine value and saponification value of biodiesel fuels from the fatty acid profile of the produced FAMEs, so by using the suggested models the properties of the target biodiesel can be estimated without any need of testing these properties [31] . The developed model for prediction of cetane number, iodine value, and saponification value is provided in equations 9 [30] , 10 [31] , and 11 [31] , respectively. To predict the higher heating value of the biodiesel, which characterizes the energy content of it, a model was developed by Demirbag [32] that uses the iodine value and the saponification value of the biodiesel. The proposed model is given as equation 12 [32] . Table 3 lists the cetane number, iodine value, saponification value, and the higher heating value of the biodiesels produced from different vegetable oil feedstocks [33] . These biodiesel values were also determined for the R. glutinis lipid source, and presented in Table 3. Some variations on the iodine value of the R. glutinis lipid were observed with a range of 90.4 - 102.2 g iodine/100g of oil using different substrate. The cetane number, saponification value, and higher heating value were quite similar using different substrates. Comparing these four biodiesel properties of the R. glutinis lipid raw material with the most common plant oils used as biodiesel feedstock indicates that these chemical values were in the range of those observed for the vegetable oils (soybean, rapeseed, palm, and sunflower).



where,

4. Conclusion
Cultivation of oleaginous yeast R. glutinis in pulp and paper wastewater indicated that this oleaginous microorganism is able to grow in pulp and paper wastewater for producing microbial lipid. The oleaginous yeast used in this study was able to convert pure glycerol and the mixture of glucose and xylose very efficiently, demonstrating the potential utilization of industrial waste glycerol and xylose containing wastes such as lignocellulosic wastes. The fatty acid composition of the oleaginous yeast was similar to the fatty acid profile of vegetable oils which makes its chemical characteristic similar to plant oils suitable for biodiesel production. The cetane number, saponification value, and higher heating value of the derived biodiesel (FAMEs) were quite similar to those observed for the biodiesel produced from common vegetable oils with some improved quality. The applied mathematical model was able to successfully simulate the growth and lipid accumulation process.
Acknowledgements
This work was financially supported by the US department of energy, Office of Energy Efficiency and Renewable Energy (Grant No: DE-FG36-06GO86025). The authors thank Magan Green for the technical assistance. The authors are grateful to the International Paper Company for their assistance.
Cite this paper
MartaAmirsadeghi,SaraShields-Menard,W. ToddFrench,RafaelHernandez, (2015) Lipid Production by Rhodotorula glutinis from Pulp and Paper Wastewater for Biodiesel Production. Journal of Sustainable Bioenergy Systems,05,114-125. doi: 10.4236/jsbs.2015.53011
References
- 1. Nigam, P.S. and Singh, A. (2011) Production of Liquid Biofuels from Renewable Resources. Progress in Energy and Combustion Science, 37, 52-68.
http://dx.doi.org/10.1016/j.pecs.2010.01.003 - 2. Yuste, A.J. and Dorado, M.P. (2006) A Neural Network Approach to Simulate Biodiesel Production from Waste Olive Oil. Energy & Fuels, 20, 399-402.
http://dx.doi.org/10.1021/ef050226t - 3. Ratledge, C. and Wynn, J.P. (2002) The Biochemistry and Molecular Biology of Lipid Accumulation in Oleaginous Microorganisms. Advances in Applied Microbiology, 51, 1-51.
http://dx.doi.org/10.1016/S0065-2164(02)51000-5 - 4. Huang, C., Chen, X.F., Xiong, L., Chen, X.D., Ma, L.L. and Chen, Y. (2013) Single Cell Oil Production from Low-Cost Substrates: The Possibility and Potential of Its Industrialization. Biotechnology Advances, 31, 129-139.
http://dx.doi.org/10.1016/j.biotechadv.2012.08.010 - 5. Zhang, G., French, W.T., Hernandez, R., Hall, J., Sparks, D. and Holmes, W.E. (2011) Microbial Lipid Production as Biodiesel Feedstock from N-Acetylglucosamine by Oleaginous Microorganisms. Journal of Chemical Technology and Biotechnology, 86, 642-650.
http://dx.doi.org/10.1002/jctb.2592 - 6. Mondala, A.H., Hernandez, R., French, T., Mcfarland, L., Domingo, J.W.S., Meckes, M., Ryu, H. and Iker, B. (2012) Enhanced Lipid and Biodiesel Production from Glucose-Fed Activated Sludge: Kinetics and Microbial Community Analysis. AIChE Journal, 58, 1279-1290.
http://dx.doi.org/10.1002/aic.12655 - 7. Shields-Menard, S.A., Amirsadeghi, M., Sukhbaatar, B., Revellame, E., Hernandez, R., Donaldson, J.R. and French, W.T. (2015) Lipid Accumulation by Rhodococcus rhodochrous Grown on Glucose. Journal of Industrial Microbiology and Biotechnology, 42, 693-699.
http://dx.doi.org/10.1007/s10295-014-1564-7 - 8. Taherzadeh, M.J. and Karimi, K. (2007) Acid-Based Hydrolysis Processes for Ethanol from Lignocellulosic Materials: A Review. BioResources, 2, 472-499.
- 9. Papanikolaou, S. and Aggelis, G. (2002) Lipid Production by Yarrowia lipolytica Growing on Industrial Glycerol in a Single-Stage Continuous Culture. Bioresource Technology, 82, 43-49.
http://dx.doi.org/10.1016/S0960-8524(01)00149-3 - 10. Schneider, T., Graeff-Hönninger, S., French, W.T., Hernandez, R., Claupein, W., Holmes, W.E. and Merkt, N. (2012) Screening of Industrial Wastewaters as Feedstock for the Microbial Production of Oils for Biodiesel Production and High-Quality Pigments. Journal of Combustion, 2012, Article ID: 153410.
http://dx.doi.org/10.1155/2012/153410 - 11. Hall, J., Hetrick, M., French, T., Hernandez, R., Donaldson, J., Mondala, A. and Holmes, W. (2011) Oil Production by a Consortium of Oleaginous Microorganisms Grown on Primary Effluent Wastewater. Journal of Chemical Technology and Biotechnology, 86, 54-60.
http://dx.doi.org/10.1002/jctb.2506 - 12. Ince, B.K., Ince, O. and Cetecioglu, Z. (2011) Pollution Prevention in the Pulp and Paper Industries. INTECH Open Access Publisher.
- 13. Poudel, S.R., Marufuzzaman, M., Ekşioglu, S.D., Amir Sadeghi, M. and French, T. (2015) Supply Chain Network Model for Biodiesel Production via Wastewaters from Paper and Pulp Companies. In: Poudel, S.R., Marufuzzaman, M., Ekşioglu, S.D., AmirSadeghi, M. and French, T., Eds., Handbook of Bioenergy, Springer International Publishing, Berlin, 143-162.
http://dx.doi.org/10.1007/978-3-319-20092-7_6 - 14. Pokhrel, D. and Viraraghavan, T. (2004) Treatment of Pulp and Paper Mill Wastewater—A Review. Science of The Total Environment, 333, 37-58.
http://dx.doi.org/10.1016/j.scitotenv.2004.05.017 - 15. EPA Standard Method 5220 D, Standard Methods for the Examination of Water and Wastewater, 20th Edition.
- 16. Bligh, E.G. and Dyer, W.J. (1959) A Rapid Method for Total Lipid Extraction and Purification. Canadian Journal of Biochemistry and Physiology, 37, 911-917.
http://dx.doi.org/10.1139/o59-099 - 17. Revellame, E.D., Hernandez, R., French, W., Holmes, W.E., Benson, T.J., Pham, P.J., Forks, A. and Callahan II, R. (2012) Lipid Storage Compounds in Raw Activated Sludge Microorganisms for Biofuels and Oleochemicals Production. RSC Advances, 2, 2015-2031.
http://dx.doi.org/10.1039/c2ra01078j - 18. Christie, W.W. (1993) Preparation of Ester Derivatives of Fatty Acids for Chromatographic Analysis. Advances in Lipid Methodology, 2, 69-111.
- 19. Revellame, E., Hernandez, R., French, W., Holmes, W. and Alley, E. (2010) Biodiesel from Activated Sludge through in Situ Transesterification. Journal of Chemical Technology and Biotechnology, 85, 614-620.
http://dx.doi.org/10.1002/jctb.2317 - 20. Mondala, A., Hernandez, R., Holmes, W., French, T., Mcfarland, L., Sparks, D. and Haque, M. (2013) Enhanced Microbial Oil Production by Activated Sludge Microorganisms via Co-Fermentation of Glucose and Xylose. AIChE Journal, 59, 4036-4044.
http://dx.doi.org/10.1002/aic.14169 - 21. Huang, J., Shi, Q., Zhou, X., Lin, Y., Xie, B. and Wu, S. (1998) Studies on The Breeding of Mortierella isabellina Mutant High Producing Lipid and Its Fermentation Conditions. Microbiology, 4, 1-5.
- 22. Zhao, X., Kong, X., Hua, Y., Feng, B. and Zhao, Z. (Kent) (2008) Medium Optimization for Lipid Production through Co-Fermentation of Glucose and Xylose by the Oleaginous Yeast Lipomyces starkeyi. European Journal of Lipid Science and Technology, 110, 405-412.
http://dx.doi.org/10.1002/ejlt.200700224 - 23. Hu, C., Wu, S., Wang, Q., Jin, G., Shen, H. and Zhao, Z.K. (2011) Simultaneous Utilization of Glucose and Xylose for Lipid Production by Trichosporon cutaneum. Biotechnology for Biofuels, 4, 25.
http://dx.doi.org/10.1186/1754-6834-4-25 - 24. Zeng, J.J., Zheng, Y.B., Yu, X.C., Yu, L., Gao, D.F. and Chen, S.L. (2013) Lignocellulosic Biomass as a Carbohydrate Source for Lipid Production by Mortierella isabellina. Bioresource Technology, 128, 385-391.
http://dx.doi.org/10.1016/j.biortech.2012.10.079 - 25. Yang, J., Rasa, E., Tantayotai, P., Scow, K.M., Yuan, H. and Hristova, K.R. (2011) Mathematical Model of Chlorella minutissima UTEX2341 Growth and Lipid Production under Photoheterotrophic Fermentation Conditions. Bioresource Technology, 102, 3077-3082.
http://dx.doi.org/10.1016/j.biortech.2010.10.049 - 26. Luedeking, R. and Piret, E.L. (1959) A Kinetic Study of the Lactic Acid Fermentation. Batch Process at Controlled pH. Journal of Biochemical and Microbiological Technology and Engineering, 1, 393-412.
http://dx.doi.org/10.1002/jbmte.390010406 - 27. Ruan, Z., Zanotti, M., Wang, X., Ducey, C. and Liu, Y. (2012) Evaluation of Lipid Accumulation from Lignocellulosic Sugars by Mortierella isabellina for Biodiesel Production. Bioresource Technology, 110, 198-205.
http://dx.doi.org/10.1016/j.biortech.2012.01.053 - 28. Papanikolaou, S., Galiotou-Panayotou, M., Fakas, S., Komaitis, M. and Aggelis, G. (2007) Lipid Production by Oleaginous Mucorales Cultivated on Renewable Carbon Sources. European Journal of Lipid Science and Technology, 109, 1060-1070.
http://dx.doi.org/10.1002/ejlt.200700169 - 29. Sanford, S.D., White, J.M., Shah, P.S., Wee, C., Valverde, M.A. and Meier, G.R. (2009) Feedstock and Biodiesel Cha-racteristics Report. Ames.
- 30. Bamgboye, A.I. and Hansen, A.C. (2008) Prediction of Cetane Number of Biodiesel Fuel from the Fatty Acid Methyl Ester (FAME) Composition. International Agrophysics, 22, 21-29.
- 31. Gopinath, A., Puhan, S. and Nagarajan, G. (2009) Theoretical Modeling of Iodine Value and Saponification Value of Biodiesel Fuels from Their Fatty Acid Composition. Renew Energy, 34, 1806-1811.
http://dx.doi.org/10.1016/j.renene.2008.11.023 - 32. Demirbas, A. (1998) Fuel Properties and Calculation of Higher Heating Values of Vegetable Oils. Fuel, 77, 1117-1120.
http://dx.doi.org/10.1016/S0016-2361(97)00289-5 - 33. Ramos, M.J., Fernández, C.M., Casas, A., Rodríguez, L. and Pérez, á. (2009) Influence of Fatty Acid Composition of Raw Materials on Biodiesel Properties. Bioresource Technology, 100, 261-268.
http://dx.doi.org/10.1016/j.biortech.2008.06.039
NOTES
*Corresponding author.








