American Journal of Analytical Chemistry
Vol. 3 No. 4 (2012) , Article ID: 18430 , 6 pages DOI:10.4236/ajac.2012.34042
Comparative Analysis and Validation Methodologies of GC and HPLC for Analysis of Cholesterol in Meat Products
1Department of Chemical Process, Federal Technologic University of Parana, Apucarana, Brazil
2Chemistry Department, Maringá State University, Maringá, Brazil
3Food Technology, Federal Technologic University of Parana, Londrina, Brazil
Email: *nesouza@utfpr.edu.br
Received February 28, 2012; revised March 24, 2012; accepted April 7, 2012
Keywords: Cholesterol Determination; Gas Chromatography; Liquid Chromatography; Method Validation; Meat Products
ABSTRACT
This study validated different extraction methodologies and compared the quantification of cholesterol by gas chromatography (GC) and high performance liquid chromatography (HPLC) in mg per 100 g of Italian-type salami and traditional bologna. The GC method used was direct saponification of the samples without derivatizations and the HPLC method was used to extract of the lipid samples. The GC limits of detection and quantification obtained for cholesterol were, respectively, 0.001 and 0.003 mg∙g–1. The HPLC values were 0.005 mg∙g–1 and 0.016 mg∙g–1. The GC recovery rate was 97.10 ± 0.13 and that of HPLC was 93.33 ± 0.22. Comparison of the cholesterol quantity found using the two chromatographic techniques shows that both are capable of quantifying cholesterol in the foods. With regard to costs, analysis time, the cost/benefit relationship was better with gas chromatography than that obtained with high performance liquid chromatography.
1. Introduction
It is noteworthy that the word “cholesterol” has great influence on the population, probably because it is associated with health problems, as an increase in the level of blood cholesterol is considered one of the main factors in cardiovascular diseases [1].
In addition, in Brazil strokes are the principal cause of death, followed by acute myocardial infarction [2]. Because of this, importance has been placed on raising awareness among the population about foods that contain cholesterol and the determination of the quantities of cholesterol in mg per 100 g of food.
Cholesterol levels in foods were first determined using gravimetric and spectrophotometric analysis. However, the values varied greatly, with studies by different authors diverging by up to 100%, mostly due to the variety of analytical procedures used [3].
In recent years, however, among all of the quantitative techniques for determining cholesterol in food, gas phase and liquid phase chromatography stand out and are in widespread use, employing a range of detection methods [4-10].
Among the proposed chromatographic methods for determining cholesterol in foods, special attention has been given to the methodology of Al-Hasani et al. [11], who applied the gas phase technique, and that of Bragagnolo et al. [12], who used liquid phase chromatogramphy, with sample saponification in both methodologies.
The studies by Dionisi et al. [13] and Toivo et al. [14] compared the sample saponification method with the method of lipid extraction followed by saponification and found that sample saponification method gave better results for determining cholesterol in foods.
Nevertheless, the objective of this study was to validate different extraction methodologies and compare the quantification of cholesterol in mg per 100 mg of food by gas phase and liquid phase chromatography in Italian-type salami and traditional bologna.
2. Experimental
2.1. Sampling
The products analysed, Italian-type salami and traditional bologna, were acquired from businesses in the city of Maringá, Paraná State, Brazil. Three brands of each product were acquired and three batches of each product were analysed individually (n = 9 for each product). The samples were ground and homogenised into one mass, vacuum packed, wrapped in aluminium foil, and stored in a freezer until submitted to the different analyses.
2.2. Materials and Methods
The standards used in the analyses were 99% pure cholesterol (C3137-Sigma Aldrich, USA), and the internal standard, 5a-cholestane (C8003-Sigma Aldrich, USA).
The method of Al-Hasani et al. [11] AL was applied in analyses by gas phase chromatography, both to the product and to the lipids extracted using the method proposed by Bligh and Dyer [15] BD.
A Shimadzu gas phase chromatograph with a flame ionisation detector, model CGS-14A, was used in the analyses, together with a Clarity Lite data processor. A Varian capillary column, model CPSil 5 CB (100 % dimethylpolysiloxane) with a length of 30 m, internal diameter of 0.25 mm, and film thickness of 0.25 m was used. The analyses were carried out with a temperature isotherm for the column oven of 280˚C for 5 min, which was defined in tests with standard cholesterol solution and 5a-cholestane.
The injector and detector analysis temperature was 280˚C. The carrier gas used was hydrogen at a constant flow rate of 1.0 mL.min–1, with respective hydrogen and synthetic air flame gas flow rates of 30 mL.min–1 and 300 mL.min–1. The analyses were performed by injecting 2 mL in split mode ratio of 1/33.
The method proposed by Bragagnolo et al. [12] was followed for the high-performance liquid chromatography analyses.
A ThermoScientific liquid phase chromatograph, model Finningan Surveyor, was used with an ultraviolet and visible range detector and a Chrom Quest data processor, with a Waters reversed-phase column, model X Terra MS C18, with a length of 25 m, internal diameter of 4.6 mm, and film thickness of 5 mm. The isocratic mobile phase was acetonitrile:isopropanol (70:30) at a flow rate of 1.0 mL.min–1. All of the solvents used were filtered and degassed by ultrasound before use. The cholesterol was detected at 210 nm.
3. Results and Discussion
Table 1 shows the mean values for the retention time, the standard deviation (S) and the percent coefficient of variation (%CV) for the analytes in the prepared standard solutions. According to Verga et al. [16], the coefficient of variation obtained should be less than 5.0%, and that obtained was less than 0.4% the GC and less than 0.2% in the HPLC, which shows the good instrument repeatability with regard to the analyte retention time (tr).
With regard the study of the accuracy of the GC response, the mean rate of the area of the analyte peak to the area of the internal standards peak was calculated from the analytical curve for the solutions. The values obtained for the coefficient of variation in the study of the areas for the analytes in the five concentrations were below 3%.
For the HPLC, the coefficient of variation obtained for the mean area of the cholesterol peak was 2.19%. Values of up to 15% for the coefficient of variation are acceptable for these studies [17].
The limits of detection and quantification were determined by statistical calculations, obtained from the linear regression curve data, which gave the mean value of the correlation coefficients for the GC as 0.9996 and for the HPLC as 0.9984.
The limits of detection and quantification of cholesterol obtained in the GC were, respectively, 0.001 and 0.003 mg.g–1. The limit of detection in the HPLC was 0.005 mg.g–1 and the limit of quanti-fication was 0.016.
The mean recovery study result in the GC was 97.10 ± 0.13, while for HPLC it was 93.33 ± 0.22. These results are within the permitted limits and similar to those in a number of studies [18,19].
The cholesterol analyses were performed on both the homogenised sample and the sample lipid extract obtained by the Bligh and Dyer [15], BD, methodology. The results are given in Table 2 in mg per 100 g of food.

Table 1. Retention time repeatability of GC and HPLC.
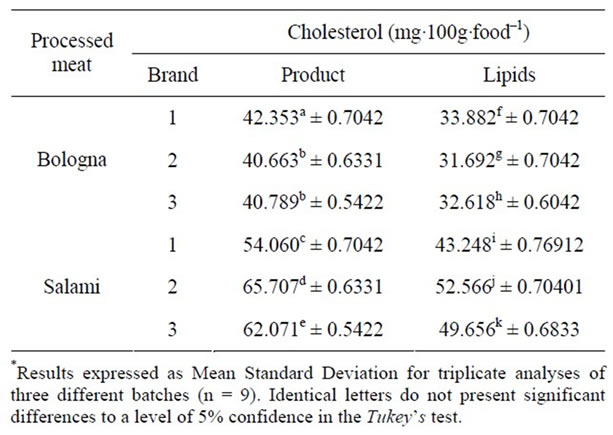
Table 2. Quantification of cholesterol in three different brands of bologna and salami.
Table 2 shows differences for the values for cholesterol in the salami sample and the lipid extract when analysed by the SAEG software, in which the Anova test (Tukey test, p < 0.05) is applied. The same is true when the results for the different brands and the results for the same brand submitted to different extraction methods are compared. The values for cholesterol in the lipids were lower than those of the direct analyses of the homogenised products.
The mean values for total lipids obtained by BD were approximately 25% for Italian-type salami and to 17% for traditional bologna. Brazilian legislation allows total lipid values of up to 35% for salami and 30% for bologna [20]. It can be seen from correlating the percentage of total lipids in the different products with the quantities of cholesterol present that these parameters correlate.
The relationship between the levels of lipid and cholesterol has been of great scientific interest for decades, and has been repeatedly studied and analysed. The discussion becomes more interesting still when intact muscles are studied, rather than processed meat products, because it is possible to change the lipid level artificially during preparation without changing the cholesterol level [21].
Few comparative studies have quantified the cholesterol in meat derivatives. In this study, direct saponification after BD differed significantly from direct saponification alone. In a study of eggs, Beyer et al. [22] found no significant difference between direct saponification and saponification after Folch et al. [23] FO.
In addition, Fenton [24] described the great variety of methodologies applicable to the extraction of total lipids from processed foods and observed that the FO methodology obtained higher values than other proposed methods. The FO and BD methodologies are similar in that they both use chloroform and methanol, but in lower amounts and with more water in the subsequent extraction steps in BD.
The extraction residue of the BD methodology was submitted to the AL methodology and cholesterol, which had previously been ignored, was then detected and quantified. The results suggest that there is residual cholesterol of about 10% of the amount obtained when the AL method is applied directly to the homogenised product.
In addition, studies by Fenton [24], Fenton and Sim [25], Alonso et al. [26], and Bragagnolo et al. [12] suggested that saponification be applied directly to products of animal origin, for reasons of convenience.
With regard to the determinations made for the different salami products, the cholesterol levels varied from 54.060 to 65.707 mg∙100∙g–1, these values being lower than the 89 mg∙100∙g–1 of the USDA [27] and the study by Lercker and Rodriguez-Estrada [28], which varied from 100 to 300 mg∙100∙g–1 of salami.
However, in the study by Baggio and Bragagnolo [29] of salamis produced in Brazil, the values of cholesterol varied from 52.4 to 65.5 mg∙100∙g–1 of food, these results being closer to those of the present study.
With regard to the nutritional food labels, cholesterol quantity information is not compulsory under Resolution RDC No. 360 from Anvisa, the national health surveillance agency [30]. However, the label of one of the salami brands acquired indicated 24 mg of cholesterol per 40 g of product, that is, 60 mg∙100∙g–1 of salami. It is important to note that three different batches of this brand were obtained and in all of the batches, the label quantity of cholesterol was the same and close to the values obtained in this study.
The sample lipid extracts were obtained using the BD methodology for evaluating whether the cholesterol quantification results from the lipid extract were better than those obtained from the product.
As described above, the results obtained for the lipid extract were lower than those obtained for the products (Table 2). Some studies [25,31] investigated the direct saponification and saponification after lipid extraction using different proposed methodologies, the obtained results not showing significant difference between the two methods.
In this study, lipid extracts were obtained using the method proposed by BD with the intention of obtaining representative results while consuming less reagents. The results (Table 2) show that the method could not detect insignificant differences between the different methodologies proposed for quantifying the cholesterol, making the results obtained from the lipid extract unsatisfactory.
Hence, analysis by direct saponification is preferable, since it reduces the number of process steps and, consequently, the probability of errors. In addition, the potential for automation increases as the complexity of the process decreases.
Furthermore, it was found that salami has a greater quantity of cholesterol per 100 g than bologna. These results were expected, since salami has a greater quantity of lipids than bologna.
For bologna, there was no significant difference for the cholesterol obtained in the product analysis for brands 2 and 3 when statistical analysis was applied. However, differences can be seen when the Anova statistical test is applied to the different brands in the analysis of the lipids.
There was no significant difference in the statistical analysis of cholesterol levels in two samples of bologna, being between 40.663 and 40.789 mg∙100∙g–1. Only brand 1 showed significant difference when the Anova test was applied, with cholesterol values of 42.353 mg∙100∙g–1 of food.
The intra-brand variation for the same product may be related to the race, sex, age and diet of the slaughtered animals, as well as the recipe ingredients and the quality and composition of the raw materials (meat and by-products) used [4].
Higher values of cholesterol, 56 mg∙100∙g–1 are found in bologna in the study of Novelli et al. [32] which have values between 52.11 and 138 mg∙100∙g–1. However, in the study conducted by Baggio and Bragagnolo [12], the quantity of cholesterol obtained in Brazilian bologna was between 39.4 and 49.8 mg∙100∙g–1 of product.
Nevertheless, in accordance with the FDA, the foods, salami and bologna, investigated in this study can be considered to be “high cholesterol” foods, as they have more than 25 mg of cholesterol per serving [33].
The means and standard deviations determined using the BG methodology are given in Table 3. In this methodology, like that proposed by AL, direct saponification is applied. Various studies using direct saponification revealed higher recovery and accuracy than those obtained with lipid extraction followed by saponification [6,22,25, 34].
In general, the quantity of cholesterol was higher in salami than in bologna, which is attributable to the different recipes of these processed meats. The sample results obtained by the AL methodologies by gas chromatography and those determined by the BG methodology with HPLC are equivalent, with no significant difference when the Anova test is applied (p < 0.05). This was expected from the good validation results given by both methodologies obtained at the beginning of the study.
While effective, the chromatography methods are costly. Comparing the chromatographic instruments used in this study shows that the chromatographic run with the GC is approximately three times faster than that with the HPLC.
With regard to the costs of the mobile phase, that for the HPLC, which requires adequate preparation to use the instrument (filters, degassing, and labour), was higher than that for the GC in this study.
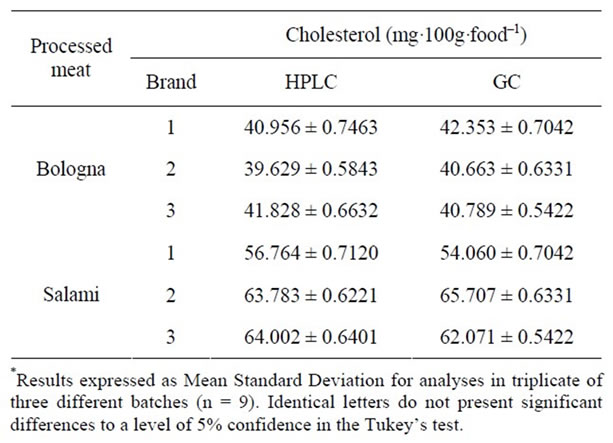
Table 3. Quantification of cholesterol by HPLC, with the BG methodology, and GC by AL.
The AL extraction methodology applied in the GC analyses required longer handling time on the part of the analyst than that required in the analyses using the HPLC method developed by BG. An advantage of the BG methodology is its ability to quantify both cholesterol and its oxides, leading to its use when determining cholesterol in foods, due to its relatively low temperatures (30˚C), which prevents cholesterol oxidation.
HPLC with ultraviolet range detector has limited application in the analysis of lipids, as some compounds do not absorb ultraviolet light. However, the presence of unsaturations and polar groups on the lipid molecules makes it possible to analyse cholesterol and its oxides [35].
4. Conclusion
The extraction of the cholesterol in meat products in this study led to good results, using either gas phase or liquid phase chromatography instruments. The GC and HPLC methodologies were satisfactorily validated, giving values validated by the literature for the extraction of the cholesterol in Italian-type salami and traditional bologna. The cholesterol values in the GC obtained from the lipids extracted by BD were lower than the values obtained by the direct analysis of the homogenised products, as shown by extracting the cholesterol in the residue of the product mass from the BD methodology. GC is comparatively a more practical choice with regard to the instrument operation, the chromatographic run speed, and the costs, whereas HPLC is the best option to simultaneously quantify cholesterol and its oxides.
5. Acknowledgements
The authors are grateful to CAPES and CNPq for financial support.
REFERENCES
- M. Careri, A. Mangia, M. Musci and G. Parolari, “Development of Gas Chromatographic Method for Determination of Cholesterol Oxides in Meat Products,” Chromatographia, Vol. 47, 1998, pp. 42-48.
- Ministry of Health, “Manual Treatment of Hereditary Coagulation Disorders (Series A. Technical Standards and Manuals),” Ministry of Health, Brasília, 2006.
- N. Bragagnolo and D. B. Rodriguez-Amaya, “Teores de Colesterol, Lipídios Totais e Ácidos Graxos em Cortes de Carne Suína,” Ciência e Tecnologia de Alimentos, Vol. 22, No. 1, 2002, pp. 98-104. doi:10.1590/S0101-20612002000100018
- N. M. W. Pratiwi, P. J. Murray and D. G. Taylor, “Total Cholesterol Concentrations of the Muscles in Castrated Boer Goats,” Small Ruminant Research, Vol. 64, No. 1-2, 2006, pp. 77-81. doi:10.1016/j.smallrumres.2005.04.016
- J. Dong, W. Chen, S. Wang, J. Zhang, H. Li, H. Guo, Y. Man and B. Chen, “Jones Oxidation and High Performance Liquid Chromatographic Analysis of Cholesterol in Biological Samples,” Journal of Chromatography B, Vol. 858, No. 1-2, 2007, pp. 239-246. doi:10.1016/j.jchromb.2007.08.038
- T. T. N. Dinh, J. R. Blanton Jr., J. C. Brooks, M. F. Miller and L. D. Thompson, “A Simplified Method for Cholesterol Determination in Meat and Meat Products,” Journal of Food Composition and Analysis, Vol. 21, No. 4, 2008, pp. 306-314. doi:10.1016/j.jfca.2008.02.001
- A. Daneshfar, T. Khezeli and H. J. Lotfi, “Determination of Cholesterol in Food Samples Using Dispersive Liquid-Liquid Microextraction Followed by HPLC-UV,” Journal of Chromatography B, Vol. 877, No. 4, 2009, pp. 456-460. doi:10.1016/j.jchromb.2008.12.050
- H. Farwanah, J. Wirtz, T. Kolter, K. Raith, R. H. H. Neubert and K. Sandhoff, “Normal Phase Liquid Chromatography Coupled to Quadrupole Time of Flight Atmospheric Pressure Chemical Ionization Mass Spectrometry for Separation, Detection and Mass Spectrometric Profiling of Neutral Sphingolipids and Cholesterol,” Journal of Chromatogry B, Vol. 877, No. 27, 2009, pp. 2976-2982. doi:10.1016/j.jchromb.2009.07.008
- W. J. Griffiths and Y. Wang, “Analysis of Neurosterols by GC-MS and LC-MS/MS,” Journal of Chromatography B, Vol. 877, No. 26, 2009, pp. 2778-2805. doi:10.1016/j.jchromb.2009.05.017
- V. A. Isidorov and L. Szczepaniak, “Gas Chromatographic Retention Indices of Biologically and Environmentally Important Organic Compounds on Capillary Columns with Low-Polar Stationary Phases,” Journal of Chromatography A, Vol. 1216, No. 51, 2009, pp. 8998- 9007. doi:10.1016/j.chroma.2009.10.079
- S. M. Al-Hasani, J. Hlavac and M. W. Carpenter, “Rapid Determination of Cholesterol in Single and Multicomponent Prepared Foods,” Journal of AOAC International, Vol. 76, No. 4, 1993, pp. 902-906.
- N. Bragagnolo, T. Saldanha and M. R. Mazalli, “Avaliação Comparativa Entre dois Métodos Para Determinação do Colesterol em Carnes e Leite,” Ciência e Tecnologia de Alimentos, Vol. 24, No. 1, 2004, pp. 109-113. doi:10.1590/S0101-20612004000100013
- F. Dionisi, P. A. Golay, J. M. Aeschlimann and L. B. Fay, “Determination of Cholesterol Oxidation Products in Milk Powder: Methods Comparison and Validation,” Journal of Agricultural and Food Chemistry, Vol. 46, No. 6, 1998, pp. 2227-2233. doi:10.1021/jf971060
- J. Toivo, K. Phillips, A. M. Lampi and V. Piironen, “Determination of Sterols in Foods: Recovery of Free, Esterified, Glycosidic Sterols,” Journal of Food Composition and Analysis, Vol. 14, No. 6, 2001, pp. 631-643. doi:10.1006/jfca.2001.1019
- E. G. Bligh and E. J. Dyer, “A Rapid Method of Total Lipid Extraction and Purification,” Canadian Journal of Biochemistry and Physiology, Vol. 37, No. 8, 1959, pp. 911-917. doi:10.1139/o59-099
- A. F. Verga, N. M. Saito, Y. S. Hirata and C. Barros, “Validação de Métodos Analíticos Para Fins de Certificação de Laboratórios,” Expo-Labor, Vol. 15, No. 3, 2009, pp. 115-128.
- M. A. Campanero, “Development and Validation of a HPLC Method for Determination of Amphotericin B in Plasma and Sputum Involving Solid Phase Extraction,” Chromatographia, Vol. 46, No. 11-12, 1997, pp. 641-647. doi:10.1007/BF02490525
- C. M. Bandeira, J. M. Ferreira, N. Bragagnolo and L. R. B. Mariutti, “Desenvolvimento de Metodologia Analítica Para Determinação de Colesterol em Ração Para Ruminantes Através de Planejamento Experimental Fatorial,” Química Nova, Vol. 31, No. 6, 2008, pp. 1422-1426. doi:10.1590/S0100-40422008000600027
- S. L. Nixdorf, M. Matsushita and N. E. Souza, “Colesterol em Refeições Comerciais Consumidas na Cidade de Londrina (PR), Brasil,” Boletim CEPPA, Vol. 15, 1997, pp. 149-158.
- Ministry of Agriculture, Livestock and Supply, “Annex II. Technical Regulations for Food Labeling,” Official Gazette, Instruction No. 4, 31 March 2000.
- F. Jimenez-Colmenero, J. Carballo and S. Cofrades, “Healthier Meat and Meat Products: Their Role as Functional Foods,” Meat Science, Vol. 59, No. 1, 2001, pp. 5-13. doi:10.1016/S0309-1740(01)00053-5
- J. D. Beyer, F. X. Milani, M. J. Dutelle and R. L. Bradley Jr., “Gas Chromatographic Determination of Cholesterol in Egg Products,” Journal of Association of Official Analytical Chemists, Vol. 72, No. 5, 1989, pp. 746-748.
- J. Folch, M. Less and S. Stanley, “A Simple Method for the Isolation and Purification of Total Lipids from Animal Tissues,” Journal of Biological Chemistry, Vol. 226, No. 1, 1957, pp. 497-509.
- M. Fenton, “Review: Chromatographic Separation of Cholesterol in Foods,” Journal of Chromatography, Vol. 624, No. 1-2, 1992, pp. 369-388. doi:10.1016/0021-9673(92)85689-Q
- M. Fenton and J. S. Sim, “Determination of Egg Yolk Cholesterol Content by On-Column Capillary Gás Chromatography,” Journal of Chromatography, Vol. 540, 1991, pp. 323-329. doi:10.1016/S0021-9673(01)88819-1
- L. Alonso, L. Lozada, J. Fontecha and M. Juárez, “Determination of Cholesterol in Milk Fat by Gas Chromatography with Direct Injection and Sample Saponification,” Chromatographia, Vol. 41, No. 1-2, 1995, pp. 23- 28. doi:10.1007/BF02274190
- United States Department of Agriculture, Agricultural Research Service, 2006. USDA Nutrient Database for Standart Reference (Release 15). Nutriente Data Laboratory Home Page, 2006. http://www.nal.usda.gov/fnic/foodcomp/Data/SR15/reports/sr15page.htm
- G. Lercker and M. T. Rodriguez-Estrada, “Cholesterol Oxidation: Presence of 7-Ketocholesterol in Different Food Products,” Journal of Food Composition and Analysis, Vol. 13, No. 4, 2000, pp. 625-631. doi:10.1006/jfca.2000.0901
- S. R. Baggio and N. Bragagnolo, “Formação de Óxidos de Colesterol e Alterações dos Ácidos Graxos em Produtos Cárneos,” Revista do Instituto Adolfo Lutz, Vol. 66, 2007, pp. 10-17.
- ANVISA, “National Agency of Sanitary Surveillance,” Official Gazette, RDC-Resolution No. 360, 23 December 2003.
- C. Maraschielo, I. Diaz and J. A. G. Regueiro, “Determination of Cholesterol in Fat and Muscle of Pig by HPLC and Capillary Gás Chromatography with Solvent Venting Injection,” Journal of High Resolution Chromatography, Vol. 19, No. 3, 1996, pp. 165-168.
- E. Novelli, E. Zanardi, G. P. Ghiretti, G. Campanini, G. Dazzi, G. Madarena and R. Chizzolini, “Lipid and Cholesterol Oxidation in Frozen Stored Pork, Salame Milano and Mortadella,” Meat Science, Vol. 48, No. 1-2, 1998, pp. 29-40. doi:10.1016/S0309-1740(97)00072-7
- Food and Drug Administration, “Food Labeling: Nutrient Content Claims, General Principles, Petitions, Definition of Terms; Definitions of Nutrient Content Claims for the Fat, Fatty Acid, Cholesterol Content of Food,” Federal Register, Vol. 58, No. 3, pp. 2302-2426.
- M. R. Mazalli, A. C. H. F. Sawaya, M. N. Eberlin and N. Bragagnolo, “HPLC Method for Quantification and Characterization of Cholesterol and Its Oxidation Products in Eggs,” Lipids, Vol. 41, No. 6, 2006, pp. 615-622. doi:10.1007/s11745-006-5010-0
- K. B. Choudhari, S. Jayanthi, R. B. Murty and R. R. Matharu, “A High-Performance Liquid Chromatographic Method for the Analysis of Lipids from Lyophilized Formulations,” Journal of Chromatography A, Vol. 724, No. 1-2, 1996, pp. 343-347. doi:10.1016/0021-9673(95)00933-7
NOTES
*Corresponding author.

