International Journal of Clinical Medicine
Vol.4 No.3(2013), Article ID:28868,6 pages DOI:10.4236/ijcm.2013.43027
Combination Lipid Therapy on Lipid Profiles in Patients with Impaired Glucose Tolerance
![]()
Intensive Care Unit, Department of Cardiovascular Medicine, Kumamoto University, Kumamoto, Japan.
Email: *koumei@fc.kuh.kumamoto-u.ac.jp
Received January 8th, 2013; revised February 10th, 2013; accepted February 20th, 2013
Keywords: Statins; Fibrates; Dyslipidemia
ABSTRACT
Objective: This study compared the effects of combination statin and fibrate therapy with either statin or fibrate monotherapy on lipid profiles in patients with impaired glucose tolerance (IGT) and a high risk for cardiovascular disease. Methods & Patients: Forty-five patients with IGT and dyslipidemia (men 25, women 20, mean age 61.7 ± 2.4 yrs) were assigned randomly to the 3 treatment groups for a 6-month period. Results: After 6 months of treatment, low density lipoprotein levels decreased in every group, especially the statin and statin + fibrate groups. Triglyceride levels also decreased in all three groups, especially the fibrate and statin + fibrate groups. High density lipoprotein cholesterol and fasting blood glucose levels did not change in any group. The levels of remnant like cholesterol particles decreased in the fibrate and statin + fibrate groups. There was no change during the study in the levels of creatine phosphokinase, lactate dehydrogenase, or creatinine. Conclusion: Combination statin and fibrate therapy results in greater improvement in lipid profiles than monotherapy with either drug. No marked adverse effects were observed with combination therapy during the study.
1. Introduction
Patients with impaired glucose tolerance (IGT) have an increased prevalence of cardiovascular disease due to atherosclerosis [1]. This increase is attributable, in part, to associated risk factors that include hypertension and dyslipidemia. It is well known that a lot of patients with IGT have dyslipidemia without any symptoms. The dyslipidemia is characterized by elevated triglycerides (TG), low levels of high density lipoprotein (HDL) cholesterol, and high levels of low density lipoprotein (LDL) cholesterol. Statins inhibit HMG-CoA reductase and prevent the formation of mevalonate, the rate-limiting step of sterol synthesis. HMG-CoA reductase inhibition increase LDL-cholesterol clearance from plasma and decrease hepatic production of LDL. While statin therapy has beneficial effects in patients with type 2 diabetes [2], the prevalence of cardiovascular events remains elevated even in statin-treated patients with glucose intolerance [2]. In contrast, it has been reported that fibrates reduce the rate of cardiovascular events in patients with glucose intolerance [3]. The mechanism of action of fibrates involves interaction with the nuclear transcription factor PPARα that regulates the transcription of the LPL, apo CII and apo AI genes. Fibrates are able to inhibit the glucuronidation of statins. Thus, the combination of fibrates with statins may increase the risk of myotoxicity. In the present study, we investigated the effects of combination treatment with a statin and a fibrate in Japanese patients with impaired glucose tolerance and increases in both TG and LDL cholesterol levels.
2. Methods
2.1. Study Subjects
The study group comprised 45 subjects (mean age 67 +/− 11 yr) who were admitted to our hospital for examination and care of dyslipidemia. No subject had previously been diagnosed with diabetes or cardiovascular disease, with the fasting glucose level of all the subjects being < 126 mg/dl. A 75 g oral glucose tolerance test was carried out in order to assess glycaemic control and the associated risk of coronary artery disease. Both IGT and diabetes were diagnosed according to the criteria of the World Health Organization [4]. The patients who had renal dysfunction (Serum Creatinine > 1.5 mg/dL) were excluded from the present study. Three patients had diabetes [DM] and 42 had IGT. The characteristics of the patients are shown in Table 1. There were no significant
Table 1. Baseline parameters of the study participants.
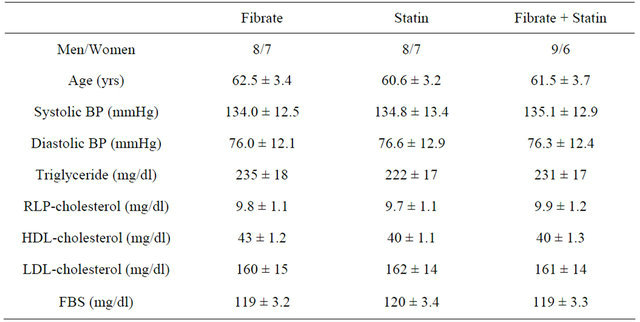
BP: Blood pressure; FBS: Fasting blood glucose; HDL: High-density lipoprotein-cholesterol; LDL: Low-density lipoprotein-cholesterol; RLP: Remnant like particles-cholesterol.
differences between the three groups. Written, informed consent was obtained from each patient before the study was commenced. The procedures used in the study were approved by the ethics committee at our institution.
2.2. Study Design
Data for the study were collected during a 75 g oral glucose tolerance test. After the patient had fasted for 12 to 14 hours, blood samples were obtained before and 0.5, 1, and 2 hours after administration of a 75 g glucose equivalent load (Trelan-G, Takeda, Japan). Plasma glucose concentrations were determined by the glucose oxidase method using an autoanalyzer. Fasting serum total cholesterol and TG concentrations were measured enzymatically, while serum LDL and HDL-cholesterol concentrations were measured by heparin-Ca2+/Ni2+ precipitation [5,6]. Remnant lipoproteins were isolated by applying the fasting serum samples to an immunoaffinity mixed gel, which contained anti-apo-A1 and anti-apoB- 100 monoclonal antibodies (Japan Immunoresearch Laboratories) [7].
Patients were randomly allocated to receive either rosuvastatin (2.5 mg/day), bezafibrate (200 mg/day) or rosuvastatin (2.5 mg/day) plus bezafibrate (200 mg/day). The patients were also provided with dietary advice and were allowed to take medications prescribed for other conditions, with the exception of lipid-lowering drugs. A check of the patients’ condition was carried out every month and lipid profiles were measured every 3 months.
2.3. Statistical Analysis
Comparison of the data in the 3 groups was performed using one-way analysis of variance (ANOVA), followed by the Bonferroni multiple comparison test. Changes in the variables were assessed by 2-way ANOVA with repeated measures, followed by post hoc testing with Scheffe’s test. Statistical significance was defined as p < 0.05.
3. Results
The 45 patients with IGT and dyslipidemia (men 25, women 20, mean age 61.7 ± 2.4 yr) were assigned randomly to 3 groups for the 6-month treatment period (bezafibrate monotherapy: men 8, women 7, mean age 62.5 yr; rosuvastatin monotherapy: men 8, women 7, mean age 60.6 yr; bezafibrate plus rosuvastatin combined therapy: men 9, women 6, mean age 61.5 yr). As shown in Figure 1 all three therapies decreased LDL-cholesterol levels (bezafibrate monotherapy: 160 ± 15 to 149 ± 12 mg/dl, p < 0.01; rosuvastatin monotherapy: 162 ± 14 to 141 ± 14 mg/dl, p < 0.01; bezafibrate plus rosuvastatin combined therapy: 161 ± 14 to 141 ± 14g/dl, p < 0.01). Similarly, all three therapies reduced TG levels (bezafibrate monotherapy: 235 ± 18 to 179 ± 19 mg/dl, p < 0.01; rosuvastatin monotherapy: 222 ± 17 to 191 ± 19 mg/dl, p < 0.01; bezafibrate plus rosuvastatin combined therapy: 231 ± 17 to 171 ± 19 mg/dl, p < 0.01). The levels of HDL-cholesterol did not change during the study (bezafibrate monotherapy: 43 ± 1.2 to 42 ± 1.3 mg/dl; rosuvastatin monotherapy: 40 ± 1.1 to 41 ± 1.0 mg/dl; bezafibrate plus rosuvastatin therapy: 40 ± 1.3 to 40 ± 1.2 mg/dl). The levels of remnant like particles (RLP)-cholesterol decreased with bezafibrate monotherapy (9.8 ± 1.1 to 6.6 ± 1.2 mg/dl, p < 0.01) and bezafibrate and rosuvastatin combined therapy (9.9 ± 1.2 to 6.8 ± 1.4 mg/dl, p < 0.01). As shown in Figure 2, rosuvastatin monotherapy was associated with a non-significant trend of decreasing RLP-cholesterol levels (9.7 ± 1.1 to 9.4 ± 1.3 mg/dl). Fasting glucose levels also tended to decrease in

Figure 1. Changes in lipid profiles before and after 6 months of lipid-lowering therapy. *p < 0.01 vs control. HDL: highdensity lipoprotein cholesterol; LDL: low-density lipoprotein cholesterol; TG: triglycerides.

Figure 2. Changes in the levels of fasting blood glucose and remnant like particles cholesterol before and after 6 months of lipid-lowering therapy. *p < 0.01 vs control. FBS: fasting blood glucose; RLP: remnant like particles cholesterol.
the three groups, especially with bezafibrate monotherapy and bezafibrate and rosuvastatin combined therapy (bezafibrate monotherapy: 119 ± 3.2 to 112 ± 3.3 mg/dl; rosuvastatin monotherapy: 120 ± 3.4 to 118 ± 3.1 mg/dl; bezafibrate plus rosuvastatin combined therapy: 119 ± 3.3 to 110 ± 3.7 mg/dl). None of these changes were statistically significant. The levels of creatine phosphokinase, lactate dehydrogenase, and creatinine did not change during the study (Figure 3).
4. Discussion
In this study we showed that combination lipid therapy had beneficial effects on lipid profiles in Japanese patients with IGT patients without causing any major adverse effects. Numerous observational studies have shown an association between TG levels and increased risk of cardiovascular morbidity and mortality [8,9]. The clinical benefit of TG reduction with fibrates has been assessed in several large interventional studies [10,11].

Figure 3. Changes in the levels of CK, LDH, and Cr before and after 6 months of lipid-lowering therapy. CK: creatine phosphokinase, Cr: creatinine; LDH: Lactate dehydrogenase.
Bezafibrate is a lipid-modifying agent that has marked effects on triglyceride levels. By activating the peroxisome proliferator activator receptors, bezafibrate enhances the clearance of triglyceride-rich lipoproteins by inducing expression of lipoprotein lipase and reducing secretion of very low-density lipoprotein from the liver [12]. In the present study, significant TG reduction was noted in every patient, with the magnitude of this reduction being greater in patients treated with bezafibrate and bezafibrate plus rosuvastatin combined therapy.
Due to their TG lowering effect, fibrates offer an effective treatment strategy for achieving treatment targets, beyond a reduction in LDL cholesterol levels. In clinical practice, the beneficial effects associated with a fibrate may be maximized when used in combination with a statin. Currently, statins are usually the first class of lipid-lowering agents used in patients with coronary heart disease or in those at high risk of developing this disorder. It is possible that patients with coronary heart disease and those at high risk of atherosclerosis may benefit from combined treatment with a statin and fibrate. In the present study, the combination of a statin and fibrate was well tolerated with no major safety concerns. On the basis of these results, we consider the effects of combination therapy for cardiovascular disease should be tested prospectively in the future.
In the present study, HDL cholesterol levels tended to increase, while fasting blood glucose concentrations tended to decrease. Previous studies have also reported that HDL cholesterol levels increase and fasting blood glucose concentrations decrease after administration of lipid-lowering agents, such as statins and fibrates [11,13]. As the present study included only a small number of patients and was of relatively short duration, further studies are required to clarify the effects of either statin plus fibrate, statin alone, or fibrate alone on HDL cholesterol and fasting blood glucose levels.
The relationship between total cholesterol and LDL cholesterol and the development and progression of atherosclerosis and cardiovascular disease has been demonstrated in a number of clinical and epidemiological studies. Furthermore, the benefit of LDL cholesterol reduction is supported clearly by evidence of a significant decrease in cardiovascular events, including cardiovascular mortality, achieved by reducing LDL cholesterol with statin therapy [14-18]. The most potent triglyceridelowering agents available are fibrates, with the combination of a fibrate and statin providing complementary lipid-lowering effects. In numerous studies, this combination has been shown to control elevations in both LDL cholesterol and triglyceride levels [19-23]. However, concomitant use of a fibrate and a statin has been associated with an increased risk of myopathy. It appears that this risk may differ depending on which statin and fibrate are administered [24,25]. In the present study, we used the combination of bezafibrate (200 mg/day) and rosuvastatin (2.5 mg/day), and showed that administration of the two drugs at these dosages was both effective and safe.
There is evidence that triglyceride-rich lipoproteins are atherogenic and are a strong risk factor for cardiovascular disease. We and others have shown that among the triglyceride-rich lipoproteins, remnant lipoproteins, especially from very-rich lipoproteins (VLDL), have a strong atherogenic effect [26-32]. In the past it has been difficult to assay levels of remnant lipoprotein due to their heterogeneous properties. However, a simple and reliable technique for measuring levels of remnant like lipoprotein particles (RLP) cholesterol using an immunoseparation method has now been developed. It has been shown that this technique isolates mainly remnants of VLDL from fasting serum, with high RLP cholesterol levels predicting future coronary events in patients with coronary artery disease [13]. It has also been shown that high levels of RLP cholesterol are an independent risk for cardiovascular events in patients with glucose intolerance [32]. Previous studies reported a significant association between reduced RLP cholesterol levels and a decrease in cardiovascular events [13,32]. The present study showed that bezafibrate treatment effectively reduced RLP cholesterol levels, while treatment with either rosuvastatin alone or bezafibrate plus rosuvastatin also resulted in a reduction in RLP cholesterol levels. However, the magnitude of the reduction in RLP cholesterol levels was greater with bezafibrate plus rosuvastatin than rosuvastatin alone. In the present study, the majority of patients had insulin resistance. Fibrate therapy may have a tendency to improve insulin sensitivity in addition to modulating TG levels, thereby leading to a reduction in RLP cholesterol levels.
In conclusion, the combination of bezafibrate and rosuvastatin has beneficial effects in patients with mixed dyslipidemia who are at high risk of further atherosclerosis complications. However, the present results need to be confirmed by further studies on a larger number of patients.
REFERENCES
- T. Almdal, H. Scharling, J. S. Jensen and H. Vestergaad, “The Independent Effect of Type 2 Diabetes Mellitus on Ischemic Heart Disease, Stroke, and Death. A PopulationBased Study of 13000 Men and Women with 20 Years of Follow-Up,” Archives of Internal Medicine, Vol. 164, No. 13, 2004, pp. 1422-1426. doi:10.1001/archinte.164.13.1422
- R. Collins, J. Armitage, S. Parish, P. Sleigh and R. Peto, “MRC/BHF Heart Protection Study of Cholesterol-Lowering with Simvastatin in 5963 People with Diabetes: A Randomized Placebo-Controlled Trial,” Lancet, Vol. 361, No. 9374, 2003, pp. 2005-2016. doi:10.1016/S0140-6736(03)13636-7
- H. B. Rubins, S. J. Robins, D. Collins, et al., “Diabetes, Plasma Insulin, and Cardiovascular Disease: Subgroup Analysis from the Department of Veterans Affairs HighDensity Lipoprotein Intervention Trial (VA-HIT),” Archives of Internal Medicine, Vol. 162, No. 22, 2002, pp. 2597-2604. doi:10.1001/archinte.162.22.2597
- World Health Organization, “Report of a WHO Study Group. Prevention of Diabetes Mellitus: World Health Org.,” Tech. Rep. Ser., No. 844, Geneva, 1994.
- A. Noma, H. Okabe, K. Netsu-Nakayama, Y. Ueno and H. Shinohara, “Improved Method for Simultaneous Determination of Cholesterol in Highand Low-Density Lipoproteins,” Clinical Chemistry, Vol. 25, No. 8, 1979, pp. 1480-1481.
- H. Kawano, T. Motoyama, N. Hirai, K. Kugiyama, H. Yasue and H. Ogawa, “Endothelial Dysfunction in Hypercholesterolemia Is Improved by L-Arginine Administration: Possible Role of Oxidative Stress,” Atherosclerosis, Vol. 161, No. 2, 2002, pp. 375-380. doi:10.1016/S0021-9150(01)00671-2
- H. Fukushima, K. Kugiyama, S. Sugiyama, O. Honda, S. Koide, S. Nakamura, H. Kawano, H. Soejima, S. Miyamoto, K. Kugiyama, M. Yoshimura, T. Sakamoto and H. Ogawa, “Comparison of Remnant-Like Lipoprotein Particles in Postmenopausal Women with and without Coronary Artery Disease and with Men with Coronary Artery Disease,” American Journal of Cardiology, Vol. 88, No. 12, 2001, pp. 1370-1373. doi:10.1016/S0002-9149(01)02115-4
- M. H. Criqui, G. Heiss, R. Cohn, L. D. Cowan, C. M. Suchindran, S. Bangdiwala, et al., “Plasma Triglyceride Level and Mortality from Coronary Heart Disease,” New England Journal of Medicine, Vol. 328, 1993, pp. 1220- 1225. doi:10.1056/NEJM199304293281702
- P. Cullen, “Evidence That Triglycerides Are an Independent Coronary Heart Disease Risk Factor,” American Journal of Cardiology, Vol. 86, No. 9, 2000, pp. 943-949. doi:10.1016/S0002-9149(00)01127-9
- H. B. Rubins, S. J. Robins, D. Collins, C. L. Fye, J. W. Anderson, M. B. Elam, et al., “Gemfibrozil for the Secondary Prevention of Coronary Heart Disease in Men with Low Levels of High-Density Lipoprotein Cholesterol,” New England Journal of Medicine, Vol. 341, 1999, pp. 410-418. doi:10.1056/NEJM199908053410604
- The BIP Study Group, “Secondary Prevention by Raising HDL Cholesterol and Reducing Triglycerides in Patients with Coronary Artery Disease: The Bezafibrate Infarction Prevention (BIP) Study,” Circulation, Vol. 102, 2000, pp. 21-27. doi:10.1161/01.CIR.102.1.21
- K. L. Goa, L. B. Barradell and G. L. Plosker, “Bezafibrate. An Update of Its Pharmacology and Use in the Management of Dyslipidaemia,” Drugs, Vol. 52, No. 5, 1996, pp. 725-753. doi:10.2165/00003495-199652050-00008
- K. Sano, T. Nakamura, M. Hirano, Y. Kitta, T. Kobayashi, D. Fujioka, Y. Saito, T. Yano, K. Watanabe, Y. Watanabe, H. Mishina, J. Obata, K. Kawabata and K. Kugiyama, “Comparative Study of Bezafibrate and Pravastatin in Patients with Coronary Artery Disease and High Levels of Remnant Lipoprotein,” Circulation Journal, Vol. 74, No. 8, 2010, pp. 1644-1650.
- J. Shepherd, S. M. Cobbe and I. Ford (For the West of Scotland Coronary Prevention Study Group), “Prevention of Coronary Heart Disease with Pravastatin in Men with Hypercholesterolemia,” New England Journal of Medicine, Vol. 333, 1995, pp. 1301-1307. doi:10.1056/NEJM199511163332001
- The Long-term Intervention with Pravastatin in Ischaemic Disease (LIPID) Study Group, “Prevention of Cardiovascular Events and Death with Pravastatin in Patients with Coronary Heart Disease and a Broad Range of Initial Cholesterol Levels,” New England Journal of Medicine, Vol. 339, No. 19, 1998, pp. 1349-1357. doi:10.1056/NEJM199811053391902
- F. M. Sacks, M. A. Pfeffer and L. A. Moye (For the Cholesterol and Recurrent Events Trial Investigators), “The Effect of Pravastatin on Coronary Events after Myocardial Infarction in Patients with Average Cholesterol Levels,” New England Journal of Medicine, Vol. 335, 1996, pp. 1001-1009. doi:10.1056/NEJM199610033351401
- Scandinavian Simvastatin Survival Study Group, “Randomized Trial of Cholesterol Lowering in 4444 Patients with Coronary Heart Disease: The Scandinavian Simvastatin Survival Study (4S),” Lancet, Vol. 344, No. 8934, 1994, pp. 1383-1389.
- J. R. Downs, M. Clearfield, S. Weis, et al., “Primary Prevention of Acute Coronary Events with Lovastatin in Men and Women with Average Cholesterol Levels: Results of AFCAPS/TexCAPS: Air Force/Texas Coronary Atherosclerosis Prevention Study,” Journal of the American Medical Association, Vol. 279, No. 20, 1998, pp. 1615-1622. doi:10.1001/jama.279.20.1615
- O. Eliav, D. Schurr, P. Pfister, Y. Friedlander and E. Leitersdorf, “High-Dose Fluvastatin and Bezafibrate Combination Treatment for Heterozygous Familial Hypercholesterolemia,” American Journal of Cardiology, Vol. 76, No. 1, 1995, pp. 76A-79A. doi:10.1016/S0002-9149(05)80023-2
- M. Farnier and S. Dejager (The French Fluvastatin Study Group), “Effect of Combined Fluvastatin-Fenofibrate Therapy Compared with Fenofibrate Monotherapy in Severe Primary Hypercholesterolemia,” American Journal of Cardiology, Vol. 85, No. 1, 2000, pp. 53-57. doi:10.1016/S0002-9149(99)00606-2
- L. E. Spieker, G. Noll, M. Hannak and T. F. Luscher, “Efficacy and Tolerability of Fluvastatin and Bezafibrate in Patients with Hyperlipidemia and Persistently High Triglyceride Levels,” Journal of Cardiovascular Pharmacology, Vol. 35, No. 3, 2000, pp. 361-365. doi:10.1097/00005344-200003000-00003
- E. Leitersdorf, E. N. Muratti, O. Eliav, V. Meiner, S. Eisenberg, E. J. Dann, E. Sehayek, T. K. Peters and Y. Stein, “Efficacy and Safety of a Combination FluvastatinBezafibrate Treatment for Familial Hypercholesterolemia: Comparative Analysis with a Fluvastatin-Cholestyramine Combination,” American Journal of Medicine, Vol. 96, No. 5, 1994, pp. 401-407. doi:10.1016/0002-9343(94)90165-1
- P. Pauciullo, C. Borgnino, R. Paoletti, M. Mariani and M. Mancini, “Efficacy and Safety of a Combination of Fluvastatin and Bezafibrate in Patients with Mixed Hyperlipidaemia (FACT Study),” Atherosclerosis, Vol. 150, No. 2, 2000, pp. 429-436. doi:10.1016/S0021-9150(00)00379-8
- A. Shek and M. J. Ferrill, “Statin-Fibrate Combination Therapy,” Annals of Pharmacotherapy, Vol. 35, No. 7-8, 2001, pp. 908-917. doi:10.1345/aph.10315
- T. Prueksaritanont, C. Tang, Y. Qui, L. Mu, R. Subramanian and J. H. Lin, “Effects of Fibrates on Metabolism of Statins in Human Hepatocytes,” Drug Metabolism and Disposition, Vol. 30, No. 11, 2002, pp. 1280-1287. doi:10.1124/dmd.30.11.1280
- N. R. Phillips, D. Waters and R. J. Havel, “Plasma Lipoproteins and Progression of Coronary Artery Disease Evaluated by Angiography and Clinical Events,” Circulation, Vol. 88, 1993, pp. 2762-2770. doi:10.1161/01.CIR.88.6.2762
- H. N. Hodis, W. J. Mack, S. P. Azen, P. Alaupovic, J. M. Pogoda, L. LaBree, et al., “Triglycerideand CholesterolRich Lipoproteins Have a Differential Effect on Mid/ Moderate and Severe Lesion Progression as Assessed by Quantitative Coronary Angiography in a Controlled Trial of Lovastatin,” Circulation, Vol. 90, 1994, pp. 42-49. doi:10.1161/01.CIR.90.1.42
- J. R. McNamara, P. K. Shah, K. Nakajima, L. A. Cupples, P. W. Wilson, J. M. Ordovas, et al., “Remnant-Like Particle (RLP) Cholesterol Is an Independent Cardiovascular Disease Risk Factor in Women: Results from the Framingham Heart Study,” Atherosclerosis, Vol. 154, No. 1, 2001, pp. 229-236. doi:10.1016/S0021-9150(00)00484-6
- K. Kugiyama, H. Doi, T. Motoyama, H. Soejima, K. Misumi, H. Kawano, et al., “Association of Remnant Lipoprotein Levels with Impairment of EndotheliumDependent Vasomotor Function in Human Coronary Arteries,” Circulation, Vol. 97, 1998, pp. 2519-2526. doi:10.1161/01.CIR.97.25.2519
- K. Kugiyama, H. Doi, K. Takazoe, H. Kawano, H. Soejima, Y. Mizuno, et al., “Remnant Lipoprotein Levels in Fasting Serum Predict Coronary Events in Patients with Coronary Artery Disease,” Circulation, Vol. 99, 1999, pp. 2858-2860. doi:10.1161/01.CIR.99.22.2858
- S. M. Grundy, “Hypertriglyceridemia, Atherogenic Dyslipidemia, and the Metabolic Syndrome,” American Journal of Cardiology, Vol. 81, No. 4, 1998, pp. 18B-25B. doi:10.1016/S0002-9149(98)00033-2
- T. Nakamura, H. Takano, K. Umetani, K. Kawabata, J. E. Obata, Y. Kitta, et al., “Remnant Lipoproteinemia Is a Risk Factor for Endothelial Vasomotor Dysfunction and Coronary Artery Disease in Metabolic Syndrome,” Atherosclerosis, Vol. 181, No. 2, 2005, pp. 321-327. doi:10.1016/j.atherosclerosis.2005.01.012
NOTES
*Corresponding author.

