International Journal of Clinical Medicine
Vol.1 No.2(2010), Article ID:3246,6 pages DOI:10.4236/ijcm.2010.12011
Fast Track Gynaecologic Surgery in the Overweight and Obese Patient
![]()
1Sydney Gynaecological Oncology Group, Sydney Cancer Centre, Royal Prince Alfred Hospital, Sydney, Australia; 2The University of Sydney, Sydney, Australia.
Email: jocarter@mail.usyd.edu.au
Received August 16th, 2010; revised August 23rd, 2010; accepted September 2nd, 2010.
Keywords: Fast Track Surgery, Obese, Overweight
ABSTRACT
Introduction: To review the outcomes of overweight and obese patients undergoing laparotomy and managed with a Fast Track Surgical (FTS) protocol. Methods: Between January 2008 and May 2010 patients having a laparotomy and managed on a FTS protocol were identified. They were compared to patients with a normal body mass index (BMI). Data was collected in a real time fashion and analysis undertaken in a retrospective fashion. Results: 194 patients were identified, 94 (48.4%) classified as normal BMI, 51 (26.3%) as overweight and 49 (25.3%) as obese. A vertical midline incision was performed in 170 (88%) patients. When comparing the group of overweight/obese patients (n = 100) to those with a normal BMI (n = 94) there was no significant difference in the number of benign or malignant pathologies, FIGO stage, age, insurance status, complexity of surgery, operation duration, blood loss, haemoglobin change, or need for transfusion. The proportion of patients successfully fast tracked and able to tolerate early oral feeding was similar. Patients classified as overweight or obese were significantly more likely to have a poorer performance status, have undergone vertical midline incision and to have had COX II inhibitors withheld. The median length of stay (LOS) was 3 days for the patients with a normal BMI and also 3 days for those overweight or obese. Conclusions: Overweight and obese patients undergoing a fast track surgical protocol after laparotomy for gynaecological surgery have similar outcomes when compared to patients of normal body mass index.
1. Introduction
It is an unfortunate reflection upon our society that the majority of Australians are considered overweight or obese. The health implications of this “obesity epidemic” are enormous with direct and indirect consequences including type 2 diabetes, cardiovascular disease, sleep apnoea, a higher risk of developing some cancers, hypertension and muscular-skeletal issues [1].
Obesity is most commonly measured using the Body Mass Index (BMI), which is a weight to height ratio and is calculated by dividing the body weight in kilograms by the square of the height in meters (kg/m2). Adults with a BMI of greater than 25 are considered overweight and a BMI of greater than 30 is considered obese [2].
While surgery remains the cornerstone of treatment for most gynaecologic cancers, obese and overweight patients pose additional and increased challenges making the surgery generally technically more difficult, which often necessitates a modification of standard surgical treatment [3,4] . Fast track surgery (FTS), also known as ERAS (Enhanced Recovery After Surgery) protocols have been widely adopted by most surgical groups across the globe with a slower uptake by gynaecological surgeons. In essence they combine a variety of techniques to minimize the catabolic stress induced by surgery and anaesthesia, with a resultant effect of improved outcomes and decreased length of stay [5-9] .
2. Aim
The aim of this manuscript was to review our experience of patients overweight or obese managed with FTS to determine if their outcomes were comparable to patients with normal BMI. Furthermore as laparoscopic surgical trials are being increasingly published and often compared to patients not being managed by FTS principles, we wanted to document the outcome data in this group of high risk patients, managed by laparotomy and FTS [4,10] .
3. Materials and Methods
After Ethics approval was granted, the FTS Database was searched to identify patients operated upon between January 2008 and May 2010 by laparotomy. Data was collected and entered into the database in a real time fashion and analysis undertaken in a retrospective fashion.
Our FTS or ERAS protocol commences with preoperative patient counselling regarding the program. Patients are made aware that their anticipated LOS is 3 days, that narcotic analgesia will be limited and adequate analgesia provided by a combination of intraoperative paracoxib ± transverse abdominis plane (TAP) block [11]. Postoperatively meloxicam is prescribed for 3 days with regular paracetamol. Early oral feeding by way of oral liquids is allowed on the night of surgery and patients are commenced on a light diet on post op day 1 with rapid progression thereafter. If significant abdominal distension or vomiting develops oral feeding is ceased. Movicol or Coloxyl with Senna is commenced routinely on post op day 1 and continued post discharge. All patients receive perioperative Clexane 20-40mg SCI which is continued until discharge. Whilst undertaken on an individualized basis, extended Clexane prophylaxis post-discharge was not considered routine in all patients. Intraoperatively mechanical sequential compression devices are employed and all patients have knee high TED stockings fitted and worn peri and postoperatively for at least 1 month. In addition all patients receive intravenous ceftriaxone 1g prior to surgery unless allergic to penicillin or cephalosporins, in which case clindamycin is usually prescribed. Patients are mobilised on day 1 post surgery and catheters and IV fluids are removed on day 1 if the patient is haemodynamically stable. Patients are given an incentive Spiro-meter and encouraged to use the device 6 times per hour. Criteria for discharge include the patient 1) adequately mobilizing without assistance, 2) tolerating early oral feeding, 3) having pain and discomfort controlled by oral analgesia and 4) having adequate home supervision after discharge. Post discharge patients receive a follow up phone call from our Clinical Nurse Consultant (CNC) within 3 days of discharge.
Data elements collected relate to 1) patient characteristics, 2) hospitalization and 3) post-hospitalization details. The patient characteristics collected were: age, weight, height, body mass index (BMI), medical insurance status and performance status. Hospitalization details included the procedure performed, type of incision (transverse or midline), operating time, complexity of surgery (simple vs. complex), intraoperative estimated blood loss, whether a transfusion was required, the preoperative Hb, post operative Hb and the Hb change, whether the patient successfully completed early oral feeding and if the patient received COX Inhibitors. All inpatient complications were collected, including modified RANZCOG Quality Indicators. Date of admission and date of discharge were used to calculate LOS. Post hospitalization admissions and complications were also recorded.
Simple surgery was defined as benign low risk adnexal surgery or simple type 1 hysterectomy where formal retroperitoneal dissection or ureteric dissection was not performed. All surgeries where at least a formal pelvic sidewall dissection was undertaken were classified as “complex”. Transverse incisions were classified according to the incision in the skin, irrespective of whether it was of Maylard type or Pfannenstiel.
Patients were usually placed in dorsal supine position with both arms extended for vascular access. Skin preparation with Povidone-iodine was routinely used, the vagina also swabbed with Povidone-iodine. Apart from the initial skin incision, which was performed with a scalpel, entry was via cautery on coagulation mode for all tissue except the rectus sheath, which was incised with pure cutting current. Wounds were routinely closed in layers with the sheath approximated with a running PDS suture, subcutaneous tissue irrigated, deep dermal and subcutaneous tissue is approximated with interrupted 2/0 vicryl sutures and skin approximated with a running subcuticular 3/0 monocryl. A subcutaneous closed drain is inserted for 24 hours in obese patients.
Patients were classified on final pathological determination as either “benign” or “malignant”. Patients with proliferating or borderline ovarian tumours were classified as “benign”. Patients with malignant pathology were routinely reviewed 2 weeks postoperatively and then regularly thereafter; whilst those patients with benign pathology were reviewed 2-4 weeks post operation.
According to International Standards, a BMI of 25-29.9 was considered overweight and > 30 obese. Statistical analysis including descriptive statistics, test and ANOVA for nominal variables and chi-squared test for categorical data.
4. Results
During the study period, 194 patients had a laparotomy performed by the author for either suspected or confirmed gynaecological malignancy or complex gynaecological pathology. Ninety-four (48.4%) were classified as normal BMI, 51 (26.3%) as overweight and 49 (25.3%) as obese. Mean BMI was 27 (range 17-69). Average patient age was 54 (range 20-85 years). There were no exclusions.
One hundred and ten patients (57%) were confirmed to have an invasive malignancy and 120 patients (62%) had private medical insurance. Vertical midline incisions were performed in 170 (88%), transverse incisions in 23 (12%) and 1 patient had a modified incision for ileostomy reversal. Surgery was deemed to be “complex” in 160 (82%) patients.
During the study period 170 (88%) patients were able to tolerate the introduction of early oral feeding. COX II inhibitors were prescribed to 167 (86%) patients. Average duration of surgery was 2.36 hours (95% CI: 2.23-2.49), ranging from 1 to 10 hours with 168 (87%) surgeries lasting between 1 to 3 hours.
One hundred and forty-nine patients (77%) had zero performance status indicating they were fully active while 36 (19%) had a performance status of “1” indicating light restrictions. Nine patients (5%) had performance status of “2” or “3” indicating an inability to work or limited self-caring respectively.
Estimated blood loss during surgery was minimal with median loss of 200ml, average of 288ml, ranging from 10ml to 3500ml. The difference in the average preoperative and postoperative Hb was 10.1g/L. Eight patients (4%) required intraoperative transfusions. One patient required transfusion of greater than 2 units of packed cells. This patient had received 3 cycles of neoadjuvant chemotherapy prior to posterior pelvic exenteration. Her preop Hb was 95 g/L and her EBL was 3500 ml.
When comparing the group of overweight/obese patients (n = 100) to those with a normal BMI (n = 94) there was no significant difference in the number of benign patients, malignant patients, FIGO stage, age less than or greater than 50, private insurance status, complexity of surgery, mean operation duration, mean or median estimated blood loss at surgery, net haemoglobin change, the number of patients transfused or requiring greater than 2 units of blood (Table 1).
Whilst not statistically significant, proportionally more overweight and obese women (39 patients) were found to have corpus tumours compared to those with normal BMI (23 patients). The proportion of patients overweight or obese (53 patients) with ovarian tumours was similar to those of normal BMI (55 patients). Fewer overweight and obese patients (6 patients) had cervical tumours compared to those of normal BMI (13 patients).
Likewise the proportion of patients successfully fast tracked and able to tolerate early oral feeding was also similar. However those patients classified as overweight or obese were significantly more likely to have a poorer performance status, have undergone vertical midline incision and to have had COX II inhibitors withheld (usually due to ACE inhibitors and abnormal renal function) (Table 2).
For patients of normal BMI, average or mean length of stay (ALOS) was 3.8 days (95% CI: 3.2-4.4) and for those classified as overweight or obese was also 3.8 days

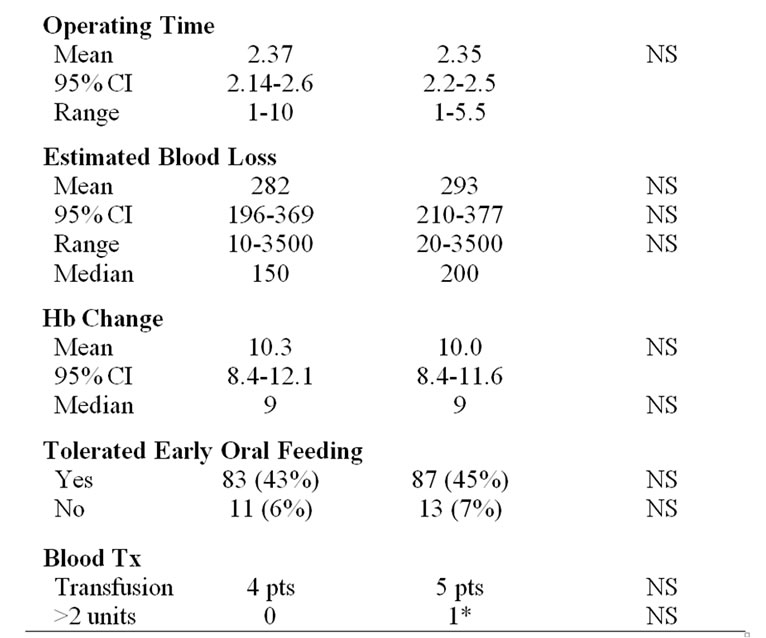
Table 1. Characteristics of normal and overweight/obese patients not statistically significant.
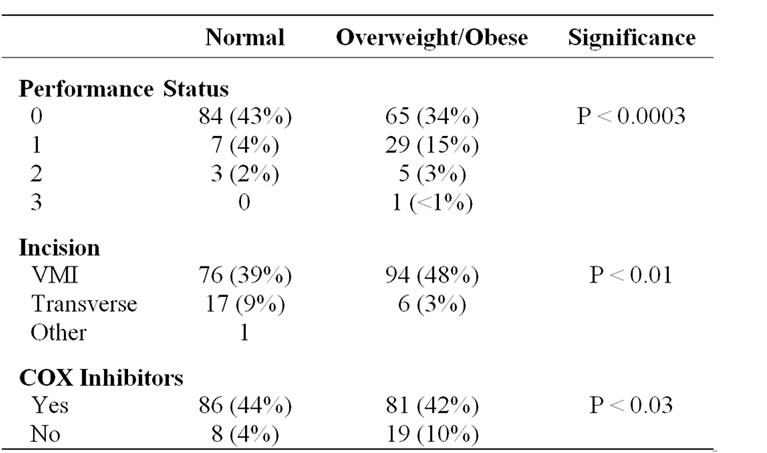
Table 2. Characteristics found to be significantly different between normal and those overweight and obese.
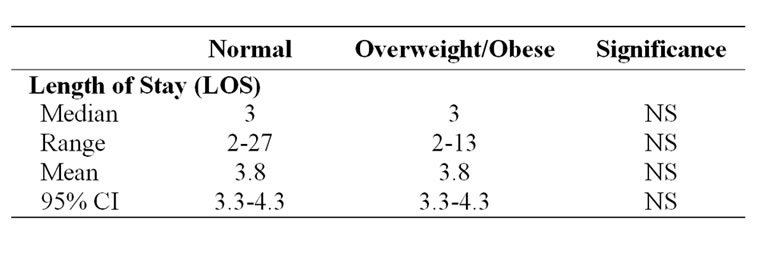
Table 3. Length of stay data for normal and overweight and obese.
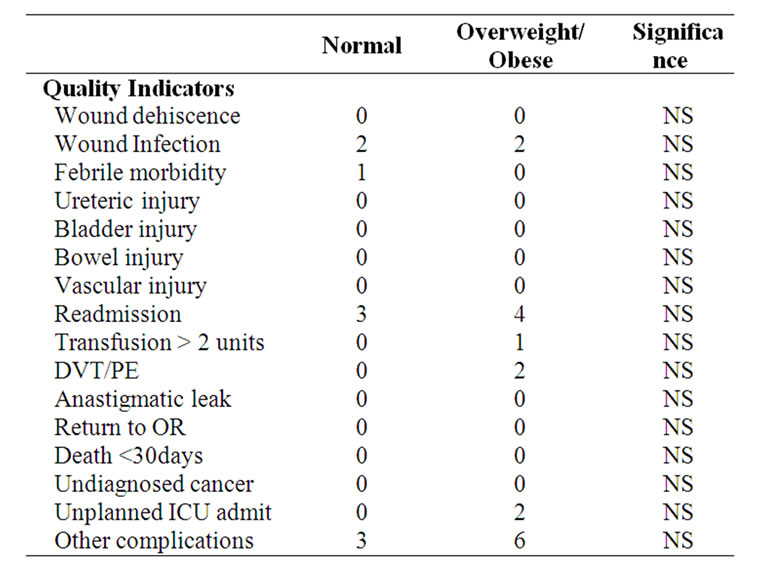
Table 4. SGOG modified ranzcog quality indicators.
(95% CI: 3.4-4.2). Average length of stay (ALOS) is often used as the marker of central tendency but is affected by extreme variables. The median LOS was 3 days for both the patients with normal BMI (range 2-27) and for those overweight or obese (range 2-13)(Table 3).
The perioperative morbidity was not considered excessive as shown by the RANZCOG quality indicators with no difference between those with normal BMI and those obese or overweight (Table 4).
5. Discussion
It is a poor reflection on our society that over half of our population is considered overweight and much of our nutrition is derived from supermarkets in prepackaged containers; that many individuals do not exercise, often lead a sedentary lifestyle and are more likely to drive a car down the street to run an errand rather than walk. The data presented here supports these statistics, with over half the patients referred for complex gynaecological or gynaeoncology surgery being overweight or obese.
Whilst alternatives to surgery should always be considered when discussing management options with women with benign gynaecological conditions, this is not usually appropriate for the gynaecological oncologist, where surgery is the cornerstone for the treatment of most cancers [4] . “Women should not be denied appropriate surgery for their cancers because of weight alone” [12] however surgery in this cohort of patients is not without its challenges.
Fast track surgery involves extensive preoperative evaluation, optimization of medical status and counseling patients of what we expect from them in the perioperative period. During this preoperative evaluation, there may be an opportunity to attempt weight loss or consider bariatric surgery such as gastric banding, or other non surgical options. With urgent surgical indications, such as a diagnosis of cancer, these options are often not available or appropriate. The anaesthetist should review the patient preoperatively to discuss not only the risks of surgery but also to make considered decisions regarding such issues as patient placement on the operating table, vascular access, airway management, ventilation requirements, provision of adequate oxygenation, safe extubation and possible postoperative ventilatory support in the intensive care unit (as the incidence of failure of early extubation is significantly higher than in non-obese patients) [13].
Surgical planning is also crucial, paying special attention to the three main hazards of the operation: blood loss, infection and thromboembolism. The site and type of the incision must be carefully considered. Many morbidly obese patients have associated medical conditions, particularly diabetes mellitus, which can contribute to delay wound healing. Although there are no hard and fast rules, in general, there is a lot to be said for an incision not buried under the pannus. Excess body fat increases tension at wound edges, hence wound separation is always a concern. Surgical considerations include type of wound retractor, access to long and super long surgical instruments and consideration of modification of the planned procedure if poor access precludes safe surgery. In overweight and obese women, there is an increased likelihood of inability to complete surgery, smaller surgical margins, reduced lymph node yield, increased surgical time, and greater blood loss. Care with homeostasis, prophylactic antibiotics and measures to reduce the risk of thromboembolism should of course be employed [14].
The operating suite also needs advance warning, particularly with the morbidly obese patients, as alternate trolleys, mechanical patient lifters, additional staff to move the patient and use of bariatric beds all need to be considered.
Postoperatively caring for obese surgical patients can be a challenge for nurses and multiple co-morbidities can compound the difficulty in caring for these patients. A FTS program decreases the amount of time a patient is in bed, which in turn lessens the physical stress on nurses as the patient does not require rigorous skin, wound or pressure care, which would often need to be carried out by several nurses. Nutrition, mobility, wound care and pain management is all nursing issues which require extra consideration preoperatively in an obese patient.
An obese patient’s lean body mass can be depleted while recovering from surgery, while maintaining body fat. This can be detrimental to recovery as it can result in a negative nitrogen balance [15]. The FTS program’s early oral feeding protocol provides a great benefit to obese patients and assists in optimal wound healing.
Postoperative analgesia also requires careful consideration in overweight and obese patients. Research shows that unrelieved pain can inhibit the immune system, decrease gastrointestinal motility and lead to respiratory dysfunction by increasing oxygen demand [16]. Postoperative pain management can be challenging in obese patients, as some drug’s pharmacokinetics can be changed in adipose tissue [17]. As early mobility is an essential component of the FTS program, analgesia must allow mobilization and participation in recovery by the patient.
Using a well documented, multimodal approach known as fast track surgery or enhanced surgical recovery we have demonstrated that patient outcomes are improved when compared to patients managed by conventional surgical principles [18]. Such programs attempt to reduce the surgical stress response by a variety of mechanisms to allow a more rapid return of the patient’s normal function and activity resulting in a reduced LOS, without an increase in complications or readmissions. This review has demonstrated that in overweight and obese women undergoing either complex gynaecological surgery or gynaeoncology surgery that outcomes are comparable to those of normal BMI.
In gynaecological oncology patients, obesity has been shown to be an independent predictor of an adverse event (AE) [19]. It is reassuring that our adverse event rate was not excessive and indeed comparable to the non-obese patients. Frumovitz and colleagues have shown a higher EBL in obese patients undergoing surgery for cervical cancer [20] however we found no increased blood loss or need for transfusion.
It would appear from the data presented here that the morbidity is low in the population reported. This may be due to the fact, that as patients are discharged early, some of the morbidities are not captured. However the converse is also plausible, that the morbidity is indeed low as patients are well managed surgically, discharged from hospital early and have a lower rate of nosocomial infections and other complications that may be developed as a consequence of longer hospital stay.
In summary, obese and overweight patients are more likely to have a poorer performance status, have a greater likelihood of having a vertical midline incision than a transverse incision and due to associated comorbidities, are less likely to receive intraoperative or postoperative COX inhibitors for analgesia. It would appear from our experience that patients classified as either overweight or obese undergoing a FTS protocol with an experienced surgeon, have similar outcomes after laparotomy when compared to patients of normal BMI. Surgery is more challenging but equally good outcomes can be achieved with a FTS or ESR program in overweight and obese patients having gynaecological surgery.
REFERENCES
- Commonwealth of Australia, “Australia: The Healthiest Country by 2020,” Technical Report 1: Obesity in Australia: A need for Urgent Action, Vol. 4, July 2009, pp. 21-128.
- WHO, “Obesity and Overweight,” World Health Organisation, Fact Sheet, No. 311, September 2006.
- B. Anderson, J. P. Connor, J. I. Andrews, C. S. Davis, R. E. Buller, J. I. Sorosky, et al., “Obesity and Prognosis In Endometrial Cancer,” American Journal of Obstetrics and Gynecology, Vol. 174, No. 4, April 1996, pp. 1171- 1178.
- A. Pellegrino, M. Signorelli, R. Fruscio, A. Villa, A. Buda, P. Beretta, et al., “Feasibility And Morbidity of Total Laparoscopic Radical Hysterectomy with or without Pelvic Lymphadenectomy in Obese Women with Stage I Endometrial Cancer,” Archives of Gynecology and Obstetrics, Vol. 279, No. 5, May 2009, pp. 655-660.
- C. P. Delaney, M. Zutshi, A. J. Senagore, F.H. Remzi, J. Hammel and V. W. Fazio, “Prospective, Randomized, Controlled Trial between a Pathway of Controlled Rehabilitation with Early Ambulation and Diet and Traditional Postoperative Care after Laparotomy and Intestinal Resection,” Disease of Colon Rectum, Vol. 46, No. 7, July 2003, pp. 851-859.
- C. Eskicioglu, S. S. Forbes, M. A. Aarts, A. Okrainec and R. S. McLeod, “Enhanced Recovery after Surgery (ERAS) Programs for Patients Having Colorectal Surgery: A Meta-Analysis of Randomized Trials,” Journal of Gastrointestional Surgery, Vol. 13, No. 12, December 2009, pp. 2321-2329.
- H. Kehlet, “Principles of Fast Track Surgery. Multimodal Perioperative Therapy Programme,” Chirurg, Vol. 80, No. 8, August 2009, pp. 687-689.
- H. Kehlet and D. W. Wilmore, “Evidence-Based Surgical Care and the Evolution of Fast-Track Surgery,” Annals Surgery, Vol. 248, No. 2, August 2008, pp. 189-198.
- D. Wilmore and H. Kehlet, “Management of Patients in Fast Track Surgery,” British Medical Journal, Vol. 322, 2001, pp. 473-476.
- N. Chopin, J. M. Malaret, M. C. Lafay-Pillet, A. Fotso, H. Foulot and C. Chapron, “Total Laparoscopic Hysterectomy for Benign Uterine Pathologies: Obesity does not Increase the Risk of Complications,” Human Reproduction, Vol. 24, No. 12, December 2009, pp. 3057-3062.
- J. G. McDonnell, B. O. Donnell, G. Curley, A. Heffernan, C. Power and J. G. Laffey, “The Analgesic Efficacy of Transversus Abdominis Plane Block after Abdominal Surgery: A Prospective Randomized Controlled Trial,” Anesthesia and Analgesia, Vol. 104, No. 1, January 2007, pp. 193-197.
- A. L. Tan and S. Mohiuddin, “Obesity and Surgery in Gynaecology Oncology,” Oil & Gas Magazine, Vol. 10, No. 4, December 2008, pp. 32-33.
- J. L. Parlow, R. Ahn and B. Milne, “Obesity is a Risk Factor for Failure of ‘Fast Track’ Extubation Following Coronary Artery Bypass Surgery,” Canadian Journal of Anaesthesia, Vol. 53, No. 3, March 2006, pp. 288-294.
- C. I. Alexander and W. Liston, “Operating on the Obese Woman-a Review,” An International Journal of Obstetrics and Gynaecology, Vol. 113, No. 10, September, 2006, pp. 1167-1172.
- J. Davidson, M. Kruse, D. Cox and R. Duncan, “Critical Care of the Morbidly Obese,” Critical Care Nursing Quarterly, Vol. 26, No. 2, 2003, pp. 105-116.
- S. Gallager and J. Gates, “Obesity, Panniculitis, Panniculectomy, and Wound Care: Understanding the Challenges,” Journal of Wound, Ostomy and Continence Nursing, Vol. 30, No. 6, 2003, pp. 334-341.
- N. Baugh, H. Zuelzer, J. Meador and J. Blankenship, “Wounds in Surgical Patients Who are Obese,” The American Journal of Nursing, Vol. 106, No. 6, 2007, pp. 40-50.
- J. Carter, R. Szabo, W. Sim, S. Pather S. Philp, K. Nattress, et al., “Fast Track Surgery in Gynaecological Oncology. A Clinical Audit,” Australia and New Zealand Journal of Obstetrics and Gynaecology, Vol. 50, 2010, pp. 159-163.
- S. Kondalsamy-Chennakesavan, C. Bouman, S. De Jong, K. Sanday, J. Nicklin, R. Land, et al., “Clinical Audit in Gynecological Cancer Surgery: Development of A Risk Scoring System to Predict Adverse Events,” Gynecologic Oncology, Vol. 115, No. 3, December 2009, pp. 329-333.
- M. Frumovitz, C. C. Sun, A. Jhingran, K. M. Schmeler, R. Dos Reis, M. R. Milam, et al., “Radical Hysterectomy in Obese and Morbidly Obese Women with Cervical Cancer,” Obstetrics & Gynecology, Vol. 112, No. 4, October 2008, pp. 899-905.

