Advances in Infectious Diseases
Vol.05 No.01(2015), Article ID:54305,8 pages
10.4236/aid.2015.51005
Evaluation of Toxicity of Phenolic Compounds Using Aedes aegypti (Diptera: Culicidae) and Artemia salina
Taline Baganha Stefanello Catelan1, Eduardo José de Arruda1*, Lincoln Carlos Silva Oliveira2, Cristiano Raminelli3, Cleusa Rocha Garcia Gaban2, Isaias Cabrini1, Paulo César Vila Nova2, Ernane Simões Carbonaro1
1Science and Environmental Technology, College of Exact Sciences and Technology, Federal University of Grande Dourados (UFGD), Dourados, Brazil
2Institute of Chemistry, Federal University of Mato Grosso do Sul (UFMS), Campo Grande, Brazil
3Institute of Environmental Sciences, Chemical and Pharmaceutical, Federal University of São Paulo (UNIFESP/Diadema), São Paulo, Brazil
Email: *eduardoarruda@ufgd.edu.br
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 9 February 2015; accepted 25 February 2015; published 28 February 2015
ABSTRACT
Nowadays, dengue fever, a mosquito-borne tropical disease, has become widely distributed in Brazil. In order to prevent such disease, the population of Aedes aegypti mosquito has to be controlled. Then, the aim of this study is to assess the toxicological effects of phenolic compounds against dengue mosquitos. Hence, the larvicidal activity and toxicity of phenolic compounds against Ae. aegypti larvae and A. salina, are evaluated, respectively. The phenolic compounds 2,6-diiodophe- nol and 4-chloro-2,6-diiodophenol have different toxicity and larvicidal activity. The compound 4-chloro-2,6-diiodophenol shows the highest toxicity for larvae of Ae. aegypti, exhibiting higher toxicity than 2,6-diiodophenol. Although less toxic than Temephos® (organophosphate), the phenolic compounds evaluated by this research, are proved to be effective against Ae. aegypti larvae. This study demonstrates the importance of controlling the dengue mosquito, considering toxicological aspects of phenolic compounds to prevent environmental impacts.
Keywords:
Dengue, Ae. aegypti, Toxicological Assessment, Phenolic Compounds

1. Introduction
The mosquito Ae. aegypti is responsible for transmitting viral diseases such as dengue and yellow fever. These diseases are the major epidemiological problems for public health in tropical and subtropical areas [1] -[4] . Managing the insect population with conventional insecticides is efficient as long as it is done within the parameters of a planned management program. The use of higher concentrations for insect management increases public health risks and causes serious environmental damages by indiscriminately affecting insects and non- target organisms [5] . Thus, it is extremely important to develop strategies for controlling the mosquito, especially in the reproductive stage when the insect is most vulnerable. This study develops new larvicides that are advantageous for mosquito management throughout the insect’s reproductive cycle and have the following characteristics: high mortality, prolonged activity, increased efficiency, specificity, biodegradability, recyclability and low cost [6] , addition of low concentration, an important feature for market competitiveness.
Researches showed that phenolic compounds of plants have larvicidal and molluscicidal action against larvae of Ae. aegypti. They concluded that the ethyl acetate, hexane, and chloroform fractions obtained from the stem bark of S. brasiliensis have potential larvicide, while the chloroform and ethyl acetate fractions are toxic to B. glabrata snails. The toxicity of these active fractions as demonstrated for A. salina may be essential for obtaining more selective and biodegradable compounds to biological control the vectors for dengue and schistosomiasis [7] . The toxicity mechanism of phenolic compounds is probably due to the antioxidant properties of these compounds which is along with the damage on the epithelial membrane of gut of mosquito larvae. The present work describes the synthesis of phenolic compounds and the mechanism of their larvicida activity against Ae. aegypti and A. salina.
2. Experimental Methods
2.1. Synthesis of Phenolic Compounds
The compounds 2,6-diiodophenol (2a), 4-hydroxy-3,5-diiodoacetophenone (2b), 4-chloro-2,6-diiodophenol (2c) and 2,6-dichloro-4-iodophenol (2d) were prepared, purified and provided by Gallo et al. (2010) [9] (Scheme 1).
The iodination reaction of the phenolic compounds occurred selectively under mild reaction conditions, using water as solvent to reduce the environmental impact. Hydrogen peroxide (0.68 mL of a 30% (w/v) aqueous solution, 6 mmol) was added to a solution of the appropriate phenol (1a-d) (2 mmol) and iodine (0.76 g, 3 mmol) in distilled water (10 mL). The mixture was stirred at room temperature or at 50˚C for 24 h. an aqueous solution (10 mL) 10% (w/v) of sodium thiosulfate was then added to the mixture, which was extracted with dichloromethane or ethyl acetate (3 × 20 mL). The organic phase was dried using MgSO4. After filtration, the solvent was evaporated under reduced pressure. The residue was purified by silica gel column chromatography using a mixture of hexane/dichloromethane (1:1), unless indicated otherwise, yielding the desired product (2a-d).
2.1.1. 2,6-Diiodophenol (2a) (CAS Registry Number: 28177-54-0)
Yield 0.5744 g (83%); off-white solid; mp 64˚C - 65˚C (lit. [8] mp 66˚C - 67˚C); 1H and 13C NMR, IR and LRMS data obtained for compound 2a are in agreement with those described in the literature [8] [9] .
2.1.2. 4-Hydroxy-3,5-Diiodoacetophenone (2b) (CAS Registry Number: 7191-55-1)
The eluant used was dichloromethane; yield 0.7139 g (92%); off-white solid; mp 1757˚C - 177˚C (lit. [10] mp 173˚C); 1H and 13C NMR, IR and LRMS data obtained for compound 2b are in agreement with those in the literature [9] [10] .
2.1.3. 4-Chloro-2,6-Diiodophenol (2c) (CAS Registry Number: 15459-50-4)
Yield 0.7220 g (95%); off-white solid; mp 104˚C - 106˚C (lit. [11] mp 107˚C - 108˚C); 1H and 13C NMR, IR and LRMS data obtained for compound 2c are in agreement with those described in the literature [9] [11] .
2.1.4. 2,6-Dichloro-4-Iodophenol (2d) (CAS Registry Number: 34074-22-1)
Yield 0.5491 g (95%); off-white solid; mp 90˚C - 91˚C (lit. [12] mp 91˚C - 92˚C); 1H and 13C NMR, IR and LRMS data obtained for compound 2c are in agreement with those in the literature [9] [12] .
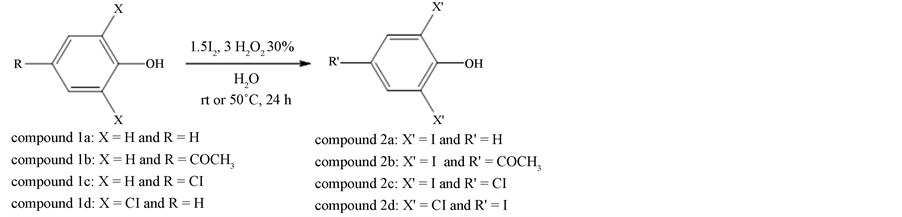
Scheme 1. Preparation of the phenolic compounds 2a-d.
2.2. Breeding and Maintenance of the Mosquito
Ae. aegyptieggs (Rockefeller strain by IBEX/Entomology/LAFICAVE/IOC/FIOCRUZ-RJ) were placed to hatch in an environmental chamber (BOD incubator) at a temperature of 27˚C ± 2˚C and relative humidity of 70% ± 5%. The larvae’s diet consisted of fish feed. Larvae were selected and allowed to grow to the adult stage. For blood supply of females used rats and as sugar food was available. The obtained Ae. aegypti eggswere used for both colony maintenance and toxicity bioassays [13] .
2.3. Bioassays of Toxicity Using Ae. aegypti Larvae
The larvicidal activity was monitored following the methodology described by the WHO-World Health Organization [14] . Ae. aegypti larvae (approximately 1000 individuals) were placed on a tray containing water and fed with fish feed (1g per liter of water) and 3rd instar larvae were selected [15] .
Bioassays for establishing the baseline were performed using the compounds 2a, 2b, 2c and 2d. These compounds were weighed and sonicated in 1mL of dimethyl sulfoxide (DMSO) 0.5% (v/v), after which unchlorinated water was added up to a volume of 20 mL to produce solutions in concentrations of 1000 mg∙L−1, 500 mg∙L−1, 250 mg∙L−1, 125 mg∙L−1, 62.5 mg∙L−1 and 31.25 mg∙L−1 (ppm). A volume of 50 mL of solution and groups of ten 3rd instar larvae were placed in plastic cups. After 24 hours of exposure, the number of dead larvae was counted, considering dead the ones that were immobile or did not respond to stimulation with a Pasteur pipette. The bioassay was performed in quadruplicate. The negative control was carried out concomitantly, using an aqueous solution of DMSO 0.5 (v/v), and the positive control was performed using Temephos®. After establishing of baseline susceptibility other bioassay was performed in quadruplicate on three different days. The concentrations used now ranged from 120 mg∙L−1 to 5 mg∙L−1. All the experiments were accompanied with negative a control using an aqueous solution of DMSO 0.5 (v/v).
2.4. Biological Assays on Ae. aegypti Eggs
The eggs were separated and selected under a binocular stereomicroscope and placed in test tubes filled with unchlorinated water. The Ae. aegypti eggs were hatched and the ovicidal bioassays performed in a BOD incubator at a temperature of 27˚C ± 2˚C, relative humidity of 70% ± 5% and a 12:12 hours light-dark cycle. Two hundred Ae. aegypti eggs approximately 15 days old were used to avoid lowering the hatching rate [16] [17] . To check the incubation period and viability, the eggs were divided into five batches of 20 eggs each, which were kept in 20 mL test tubes, with 10 mL of water and fish food added to each tube. Daily bioassays were monitored using a binocular stereomicroscope to remove larvae, count hatched eggs and to prevent the adhesion of eggs to the tube walls.
2.5. Statistical Analysis
The Probit analysis method was employed to obtain theLC10, LC50 and LC90 values of the larval stages of Ae. aegypti and A. salina with 95% confidence interval P < 0.05 [18] .
3. Results and Discussion
3.1. Bioassays of Toxicity Using Ae. aegypti Larvae
Larvae exposed 2,6-diiodophenol, 4-hydroxy-3,5-diiodoacetophenone, 4-chloro-2,6-diiodophenol and 2,6-dich- loro-4-iodophenol presented little or no movement in the initial hours of contact with the product. In contrast, after 24 hours, the larvae of the control group intensive movements, and exuviae was visible in the container, indicating that ecdysis occurred.
Table 1 shows the toxicity bioassays results for the phenolic compounds 2,6-diiodophenol and 4-chloro-2,6- diiodophenol. Despite the toxicity presented in isolated experiments with 500 mg∙L−1 and 1000 mg∙L−1 of phenolic compounds. The compounds 4-hydroxy-3,5-diiodoacetophenone and 2,6-dichloro-4-iodophenol were not used in the toxicity bioassays because of their low solubility and their poor dispersion, which rendered them unsuitable for dilution. However, they were solubilized in Tween 80®, as described in part 3.1.1.
The slopes of plotes can be used to analyze the toxicities of the compounds. Figure 1 shows that the Temephos® has the highest toxicity. The values of slopes, which were determined by analyzing the mortality curve, demonstrate that there are differences and/or heterogeneities in the larvicidal behaviours of the substances compounds, explain about the linearity of the curves. The phenolic compound 2,6-diiodophenol shows different behaviour which is due to the low solubility of this component. These differences are probably due to the low solubility of 2,6-diiodophenol, which led to high standard deviations, as indicated by the differences in larval deaths in the toxicity bioassays of the Rockefeller strain, since the linearity of the straight line requires values below 7. It can be shown thatfor 2,6-diiodophenol, this value is the tolerance limit of linearity, since the solutions of this compound contained supernatant, indicating problems of solubility and heterogeneity in the solution.
Figure 1 shows a Log-Probit mortality graph of the Rockefeller strain of Ae. aegypti exposed to 2,6-diiodo- phenol, 4-chloro-2,6-diiodophenol, based on the concentrations used in the bioassay with 3rd instar larvae of Ae. aegypti. The analysis of the curves indicated that the toxicity and larvicidal characteristic of 4-chloro-2,6-dii- odophenol is higher than those of 2,6-diiodophenol. These results are confirmed by the analysis of the LC50 for toxic and larvicidal behaviour of 4-chloro-2,6-diiodophenol, with LC50 of 11.53 mg∙L−1, that is almost eight-fold lower than 2,6-diiodophenol, with LC50 is 88.34 mg∙L−1, indicating that the toxicity of the former compound is higher than that of the latter. The slope of the straight line in the Log-Probit graph indicates the toxicity of larvicidal substances. The steeper the slope of the straight line the higher the concentration of compound required
Figure 1. Log-Probit mortality graph of the Rockefeller strain of Ae. aegypti exposed to 2,6-diiodophenol and 4-chloro-2,6- diiodophenol.
Table 1. Lethal concentrations (LC, mg∙L−1) for larvae of Ae. aegypti (Rockefeller strain) exposed to 2,6-diiodophenol, 4- chloro-2,6-diiodophenol, 4-hydroxy-3,5-diiodoacetophenone and 2,6-dichloro-4-iodophenola.
aToxicity bioassays performed in quadruplicate on three different days. (−) Experiments not performed 2,6-diiodophenol and 4-chloro-2,6-diiodo- phenol.
to control the population and eliminate the mosquito larvae.
Temephos®, the insecticide/larvicide organophosphate, which is applied in the concentration 1 mg∙L−1 for control of Ae. aegypti, is normally used because of its low oral toxicity in mammals [19] . In the present study it was used as positive control. The results indicate the same confidence interval for both LC10 and LC50 which are in principle opposed to WHO (World Health Organization). The experimental results are different from the values of WHO (1992) considers that the Diagnostic Concentration (DC; 3 × LC99) or 0.012 mg∙L−1. The value of LC10 is close to the value expected for the WHO-World Health Organization CD. This value suggests pesticide resistance (WHO Tech. Rep. Ser. 818, 63p, 1992) (see Table 1 and Table 2).
Moreover, the control larvae that were subjected to the same toxicity bioassay method, showed susceptibility to insecticide. The Log-Probit mortality graph of the Rockefeller population exposed to phenolic compounds expresses the ratio of mortality caused by commercially known insecticides versus potential larvicidal actives. Note that the more distant (to the right) from the Temephos® curve, then higher the concentration of substances required to cause larval death.
3.1.1. Toxicity Bioassays with Phenolic Compounds and Tween 80®
The toxicity bioassays were conducted following the methodology described by the WHO [14] , but 0.5% Tween 80® (ethoxylated sorbitan monooleate/20 EO) was used as solubilizer. The hydrophilic nature of the polyoxyethylene chain (C64H12O26) makes Tween 80® a highly HLB (hydrophilic-lipophilic balance) surfactant that is soluble or dispersible in water or used as a dispersant and solubilizer in oils. Organic compounds are dissolved by surfactants through the formation of micelles above the critical micelle concentration (CMC). The hydrophobic cores of micelles store and transport organic molecules, increasing the apparent solubility and toxicity of organic compounds in the aqueous stage [20] .
The analysis was based on the results of the experiments performed at different concentrations and 24-hours exposure of the larvae. Replications were performed to confirm the results whenever standard deviations were high.
The phenolic compounds showed significant differences in toxicity and mortality for 3rd instar larvae of Ae. aegypti. The behaviour of these larvae during their exposure to the compounds was observed and recorded. The first changes in larval behaviour occurred after the movement or mobility in the liquid environment. The control group larvae showed intense movement in the liquid, indicated by contractions of the body and brushes, reacting to any stimuli. The larvae exposed to phenolic compounds lost their mobility early on and presented a comatose state after 24 hours of exposure.
Table 2 describes the experimental results obtained with average values of the lethal concentrations (LC) using the Probit program. The mortality rates revealed by these results indicate the toxic behavior of the phenolic compounds.
Figure 2 shows a Log-Probit mortality graph for the Rockefeller strain of Ae. aegypti larvae exposed to 2,6- diiodophenol, 4-chloro-2,6-diiodophenol, 4-hydroxy-3,5-diiodoacetophenone, 2,6-dichloro-4-iodophenol and
Table 2. Lethal concentrations (LC, mg∙L−1) of 10%, 50% and 90% to 3rd instar larvae of Ae. aegypti (Rockefeller strain) exposed to 2,6-diiodophenol, 4-chloro-2,6-diiodophenol, 4-hydroxy-3,5-diiodoacetophenone, 2,6-dichloro-4-iodophenol and Temephos®solubilizedfor 24 hoursin Tween 80®a.
aToxicity bioassays performed in quadruplicate on three different daysfollowing the methodology described by the WHO [14] .
Figure 2. Log-Probit mortality graph of the Rockefeller strain of Ae. aegypti exposed to 2,6-diiodophenol, 4-chloro-2,6-diiodophenol, 4-hydroxy-3,5-dii- odoacetophenone, 2,6-dichloro-4-iodophenol and Temephos® solubilized with 0.5% Tween 80®.
Temephos® solubilized with 0.5% Tween 80®, indicating the concentrations used in the bioassay on Ae. aegypti larvae. The analysis of this graph show that the toxicity of these compounds follows a decreasing order: Temephos® > 4-chloro-2,6-diiodophenol > 2,6-dichloro-4-iodophenol > 2,6-diiodophenol > 4-hydroxy-3,5-diiodoa- cetophenone. These results are confirmed by an analysis of the compounds’ toxicity and larvicidal behaviour, which indicates LC50 of 6.35 mg∙L−1 for 4-chloro-2,6-diiodophenol, 69.94 mg∙L−1 for 2,6-dichloro-4-iodophenol, 94.88 mg∙L−1 for 2,6-diiodophenol, 109.64 mg∙L−1 for 4-hydroxy-3,5-diiodoacetophenone and 0.017 mg∙L−1 for Temephos® (Table 2).
Because of their solubility, different concentrations of phenolic compounds led to more pronounced changes in larval behaviour, such as a gradual decrease of movement on the surface of the aqueous solution, indicating the presence of supernatant, which was confirmed by direct visual observation. The presence of supernatant indicated the low solubility of the phenolic compounds in water, so they were dissolved with a suitable surfactant, thereby decreasing the liquid surface tension. This enabled additional information to be obtained about their toxicity and larvicidal activity, and the effects of their toxicity in non-target organisms (A. salina) to be analyzed.
Figure 2 shows a Log-Probit mortality graph for the Rockefeller strain of Ae. aegypti larvae exposed to 2,6-diiodophenol, 4-chloro-2,6-diiodophenol, 4-hydroxy-3,5-diiodoacetophenone and 2,6-dichloro-4-iodophenol, indicating the concentrations used in the bioassay on Ae. aegypti larvae.The analysis of this graph show that the toxicity of these compounds follows a decreasing order: 4-chloro-2,6-diiodophenol > 2,6-dichloro-4-iodophenol > 2,6-diiodophenol > 4-hydroxy-3,5-diiodoacetophenone. These results are confirmed by an analysis of the compounds’ toxicity and larvicidal behaviour, which indicates LC50 of 6.35 mg∙L−1 for 4-chloro-2,6-diiodophenol, 69.94 mg∙L−1 for 2,6-dichloro-4-iodophenol, 94.88 mg∙L−1 for 2,6-diiodophenol, and 109.64 mg∙L−1 for 4-hy- droxy-3,5-diiodoacetophenone.The addition of Tween 80® (polyoxyethylene (20) sorbitan monooleate/ICI Americas, Inc.) increased the compound’s solubility in the aqueous medium, improving the homogeneity of the dilutions and the reproducibility of the results.
It is possible to analyze and correlate the slope of the phenolic compounds directly through the Log-Probit plot, with attention to the toxicity of each compound and target organism. This graph completes the results discussed earlier herein, leading to the conclusion that the steeper the slope of the straight line the higher the concentration of compound required to control the population and eliminate the mosquito larvae. The toxicity values obtained from the mortality curve indicate that the phenolic compounds are less heterogeneous for the Rockefeller strain of Ae. aegypti than the insecticide Temephos® (organophosphate) (Figure 2). The values of the angular coefficient “slope” for the phenolic compounds 2,6-diiodophenol, 4-hydroxy-3,5-diiodoacetophenone, 4-chloro-2,6-diiodophenol and 2,6-dichloro-4-iodophenol, were, respectively, 6.20, 1.15, 3.49 and 6.97. The larvicidal effect of the phenolic compounds was heterogeneous. Because the slope of the mortality line was lower than 7.
A comparison of the results of the phenolic compounds with and without the addition of Tween 80® shows a difference in the LC10, LC50 and LC90. The compound 4-chloro-2,6-diiodophenol dissolved in Tween 80® produces in an evenly dispersed solution, resulting in a decrease in the lethal concentrations, i.e., LC10 from 2.67 mg∙L−1 to 2.19 mg∙L−1, LC50 from 11.53 mg∙L−1 to 6.35 mg∙L−1, and LC90 from 49.71 mg∙L−1 to 18.36 mg∙L−1, thus increasing the toxicity and/or larvicidal potential (see Table 1 and Table 2). However, the compound 2,6- diiodophenol showed a slight increase in lethal concentrations. The addition of Tween 80® to the water-inso- luble compounds 2,6-dichloro-4-iodophenol and 4-hydroxy-3,5-diiodoacetophenone enabled the determination of their lethal concentrations (LC). The lethal concentrations of 2,6-dichloro-4-iodophenol were LC10: 46.66 mg∙L−1, LC50: 69.94 mg∙L−1, and LC90: 104.84 mg∙L−1, while those of 4-hydroxy-3,5-diiodoacetophenone were LC10: 70.19 mg∙L−1, LC50: 109.64 mg∙L−1, and LC90: 171.28 mg∙L−1.
3.1.2. Toxicity for Ae. aegypti Eggs
According to the larvicidal analysis, bioassays were performed with Ae. aegypti eggs to verify their viability after exposure to the phenolic compounds. This analysis indicated that the phenolic compounds showed very similar results. On the fifth day of the analysis, 10% of the eggs hatched after exposure to a concentration of 120 mg∙L−1 of the compound 2,6-diiodophenol. In contrast, hatching was 100% inhibited by exposure to the compounds 4-chloro-2,6-diiodophenol, 4-hydroxy-3,5-diiodoacetophenone and 2,6-dichloro-4-iodophenol.
3.2. Evaluation of Toxicity to Artemia salina
The analysis of mortality of simple organisms such as the marine microcrustacean Artemia Salina Leach enables a comprehensive evaluation of toxicity and is considered a preliminary bioassay in the study of assets with potential biological activity [21] .
The first step in this assessment was the cultivation of A. salina to ensure a high hatching rate of cysts and attain a survival rate of at least 95% under the conditions of the bioassay. With adequate lighting, temperature and oxygenation, it was possible to reproduce the optimum environmental conditions for A. salina, obtaining high hatching and survival rates. Brine Shrimp Lethality Test (BSL) was held from eggs of A. salina Miramar-high hatching in a sea salt solution (38 g∙L−1) were used for the toxicity test, while quinidine sulfate served as the positive control, and DMSO 0.5 (v/v) as the negative control. Initially, the dilutions were made with unchlorinated tap water, but after 24 hours the mortality rate was 100%. Therefore, the dilutions were made with water in which the eggs had hatched, thus ensuring the viability of 100% of the Artemia Salina nauplii population for the analysis.
Dolabella (1997) evaluated solvents that are toxic and non-toxic to A. salina and found that water and Tween 80® were non-toxic (100% survival), DMSO was moderately toxic, and ethane followed by methanol were highly toxic, causing 100% mortality at a concentration of 5% (v/v) [22] . Table 3 shows the lethal concentrations to A. salineexposed to phenolic compoundssolubilized in Tween 80®.
Figure 3 shows a Log-Probit mortality graph for theA. salinaexposed to 2,6-diiodophenol, 4-chloro-2,6-dii- odophenol, 4-hydroxy-3,5-diiodoacetophenone and2,6-dichloro-4-iodophenolsolubilized with 0.5% Tween 80®. The values of the angular coefficient “slope” for the phenolic compounds 2,6-diiodophenol, 4-hydroxy-3,5-dii- odoacetophenone and 2,6-dichloro-4-iodophenol weresimilar to those found for Ae. aegypti larvae, except for4-chloro-2,6-diiodophenol.
A comparison of larvicidal activity and toxicity indicated that all the compounds under study are toxic to A. salinaand Ae. aegypti. The Lethal Concentration (LC) of the compound 2,6-diiodophenol which caused a mortality rate of 10% of the population of A. salina was 56.97 mg∙L−1, while the lethal concentration of this compound that resulted in 10% mortality of the population of Ae. aegypti larvae was 54.12 mg∙L−1 (LC10) and most of the LCs values were lower for A. salina (LC10, LC50 and LC90) than Ae. aegypti larvae.
Analysis of Table 2 and Table 3 reveals that lower concentrations of all the phenolic compounds are needed
Table 3. Lethal concentrations (LC) of 10%, 50% and 90% to A. salina 48 hours after hatchinga*.
a*oxicity bioassays were performed in quadruplicate on three different days.
Figure 3. Log-Probit mortality graph for the A salina exposed to 2,6-diiodo- phenol, 4-chloro-2,6-diiodophenol, 4-hydroxy-3,5-diiodoacetophenone and 2,6-dichloro-4-iodophenolsolubilized with 0.5% Tween 80®.
to kill A. salina than to kill Ae. aegypti larvae, clearly indicating that these phenolic compounds have higher larvicidal activity against A. salina than Ae. aegypti larvae. These results demonstrate the importance of using this crustacean for the detection of bioactive substances. Studies involving new actives of different origins with potential biological behaviour are increasing, so the analysis of bioindicators of lethality is imperative. The bioassay with A. salina proved to be an excellent indicator of toxicity, and is a simple and low cost method of analysis.
The phenolic compounds 2,6-diiodophenol (2a) and 4-chloro-2,6-diiodophenol (2c) have different toxicity and larvicidal behaviour. The compound 4-chloro-2,6-diiodophenol (2c) showed the highest toxicity for larvae of Ae. aegypti and A. salina, exhibiting higher toxicity than 2,6-diiodophenol. However, because the solubility of compounds 2,6-diiodophenol and 4-chloro-2,6-diiodophenol is low, these compounds had to be dissolved with Tween 80® (surfactant) to reduce the problems of homogeneity in toxicity bioassays. The use of Tween 80® augmented the larvicidal action of the phenolic compounds. LCs, in general, have been reduced.
4. Conclusion
The toxicological properties of phenolic compounds are different for the target organisms: Ae. aegypti larvae and non target A. salina. The results suggest that the toxicity mechanisms are different and the solubility is an important parameter for toxicity. However, further studies are needed to elucidate them, and should be fully understood. The ovicidal analysis indicated that the phenolic compounds inhibited the hatching of Ae. aegypti eggs by 100% up to the fifth day of analysis and were toxic to larvae of Ae. aegypti and A. salina. The compound 4- chloro-2,6-diiodophenol (2c) was the most toxic.
Acknowledgements
The authors acknowledge the Brazilian funding agencies CAPES, CNPq/PROCAD 2008, and FUNDECT for financial and fellowship support.
References
- WHO―World Health Organization (2007) Dengue/DHF. Situation of Dengue/Dengue Haemorrhagic Fever in the South-East Asia Region. Variable Endemicity for DF/DHF in countries of SEA Region. http://www.searo.who.int/en/Section10/Section332_1100.htm
- WHO―World Health Organization (2007) Impact of Dengue. http://www.who.int/csr/disease/dengue/impact/en/index.html
- WHO―World Health Organization (2008) Fiebre Amarilla. http://www.who.int/topics/yellow_fever/es/
- WHO―World Health Organization (2006) Report on Dengue. http://www.who.int/tdr/publications/publications/swg_dengue_2.htm
- WHO―World Health Organization (2001) Dengue/Dengue Haemorrhagic Fever in the Americas. Summary of Research and Control in the Americas. TDR News, 64. http://www.who.int/tdr/publications/tdrnews/news64/dengue.htm
- Autran, E.S., Neves, I.A., da Silva, C.S.B., Santos, G.K.N., da Câmara, C.A.G. and Navarro, D.M.A.F. (2009) Chemical Composition, Oviposition Deterrent and Larvicidal Activities against Aedes aegypti of Essential Oils from Piper marginatum Jacq. Bioresource Technology, 100, 2284-2288. http://dx.doi.org/10.1016/j.biortech.2008.10.055
- Santos, C.C.S., Araujo, S.S., Santos, A.L.L.M., Almeida, E.C.V., Diasa, A.S., Damascena, N.P., Santos, D.M., Santos, M.I.S., Junior, K.A.L.R., Pereira, C.K.B., Lima, A.C.B., Shana, A.Y.K.V., Sant’ana, A.E.G., Estevam, C.S. and Araujo, B.S. (2014) Evaluation of the Toxicity and Molluscicidal and Larvicidal Activities of Schinopsis brasiliensis Stem Bark Extract and Its Fractions. Revista Brasileira de Farmacognosia, 24, 298-303. http://dx.doi.org/10.1016/j.bjp.2014.07.006
- Horiuchi, C.A. and Satoh, J.Y. (1984) A Convenient Procedure for Iodination of Electron-Rich Aromatic Compounds Using Iodine-Copper(II) Acetate. Bulletin of the Chemical Society of Japan, 57, 2691. http://dx.doi.org/10.1246/bcsj.57.2691
- Gallo, R.D.C., Gebara, K.S., Muzzi, R.M. and Raminelli, C. (2010) Efficient and Selective Iodination of Phenols Promoted by Iodine and Hydrogen Peroxide in Water. Journal of the Brazilian Chemical Society, 21, 770-774. http://dx.doi.org/10.1590/S0103-50532010000400026
- Banker, W., Sansbury, H. and Simmonds, W.H.C. (1943) The Preparation of Iodine-Containing x-Ray Contrast Substances. III. The Structure of “Choloselectan”. Journal of Chemical Technology and Biotechnology, 62, 193-194.
- Hunter, W.H. and Joyce, F.E. (1917) A Catalytic Decomposition of Certain Phenol Silver Salts. II. Thermal Decompositions: The Formula of the Unpolymerized Residues. Journal of the American Chemical Society, 39, 2640-2646. http://dx.doi.org/10.1021/ja02257a016
- Brazier, S.A. and McCombie, H. (1912) The Condensation of α-Keto-β-Anilino-α, β-Diphenylethane and Its Homologues with Phenylcarbimide and with Phenylthiocarbimide. Journal of the Chemical Society, Transactions, 101, 2352- 2358. http://dx.doi.org/10.1039/ct9120102352
- Hartberg, W.K. and Craig Jr., G.B. (1970) Reproductive Isolation in Stegomyia Mosquitoes. II. Hybrid Breakdown between Aedes aegypti and A. mascarensis. Evolution, 24, 692-703. http://www.jstor.org/discover/2406550?sid=21105479139143&uid=2&uid=3738880&uid=2134&uid=70&uid=4 http://dx.doi.org/10.2307/2406550
- WHO-World Health Organization (1985) Instruction for Determining the Susceptibility or Resistance of Mosquito Larvae to Insecticides. WHO-VBC 81-807, Geneva, 1-6.
- Gadelha, D.P. and Toda, A.T. (1985) Biologia e comportamento do Aedes aegypti. Revista Brasileira de Malariología e Doenças Tropicais, 37, 29-36.
- Forattini, O.P. (2002) Culicidologia médica: Identificação, biologia e epidemiologia. Vol. 2, EDUSP, São Paulo, 864 p.
- Monteiro, L.C.C., Souza, J.R.B. and Albuquerque, C.M.R. (2007) Eclosion Rate, Development and Survivorship of Aedes albopictus (Skuse) (Diptera: Culicidae) under Different Water Temperatures. Neotropical Entomology, 36, 966- 971. http://dx.doi.org/10.1590/S1519-566X2007000600021
- Finney, D.J. (1972) Probit Analysis: A Statistical Treatment of the Sigmoid Response Curve. 7th Edition, Cambridge University Press, Cambridge, 33.
- Sa, R.A., de Lima Santos, N.D., da Silva, C.S.B., Napoleao, T.H., Gomes, F.S., Cavada, B.S., Coelho, L.C.B.B., do Amaral Ferraz Navarro, D.M., Bieber, L.W. and Paiva, P.M.G. (2009) Larvicidal Activity of Lectins from Myracrodruon urundeuva on Aedes aegypti. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology, 149, 300-306. http://dx.doi.org/10.1016/j.cbpc.2008.08.004
- de Souza, E.F., Peres, M.R. and de Moraes, S.B. (2010) Avaliação do desempenho de surfactantes para a solubilização de fases líquidas não aquosas em meio aquoso. Química Nova, 33, 532-538. http://dx.doi.org/10.1590/S0100-40422010000300008
- Maciel, M.A., Pinto, A.C. and Veiga, V.F. (2002) Plantas medicinais: A necessidade de estudos multidisciplinares. Química Nova, 25, 429-438. http://qnesc.yordan.com/qn/qnol/2002/vol25n3/15.pdf http://dx.doi.org/10.1590/S0100-40422002000300016
- Dolabela, M.F. (1997) Triagem in vitro para atividade antitumoral e anti Trypanossoma cruzi de extratos vegetais, produtos naturais e substâncias sintéticas. Dissertação (Mestrado), Instituto de Ciências Biológicas, Universidade Federal de Minas Gerais-UFMG, Belo Horizonte, 128 p.
NOTES
*Corresponding author.






