Open Journal of Anesthesiology
Vol.3 No.1(2013), Article ID:27619,4 pages DOI:10.4236/ojanes.2013.31005
Airway Pressure Release Ventilation Improves Oxygenation in a Patient with Pulmonary Hypertension and Abdominal Compartment Syndrome
![]()
Naval Medical Center San Diego, San Diego, USA.
Email: Arturo.torres@med.navy.mil
Received October 28th, 2012; revised November 29th, 2012; accepted December 26th, 2012
Keywords: Airway Pressure Release Ventilation; Mechanical Ventilation; Abdominal Compartment Syndrome; Pulmonary Hypertension; Positive Pressure Ventilation
ABSTRACT
The following case describes the favorable application of airway pressure release ventilation (APRV) in a patient with pulmonary hypertension who developed respiratory failure and abdominal compartment syndrome after abdominal closure from an incarcerated umbilical hernia. A 66-year-old male with past medical history of restrictive lung disease, obstructive sleep apnea and pulmonary hypertension, presented to the operating room for an incarcerated inguinal hernia. After abdominal closure, he gradually developed decreased oxygen saturation and hypotension. APRV was initiated during post operative day 2 after inability to maintain adequate oxygen saturation with resultant hypotension on pressure control ventilation with varying degrees of positive end expiratory pressure and 100% inspired oxygen concentration. The initial set high pressure on APRV was 35 mmHg. Yet, in lieu of decreasing lung compliance, it peaked at 50 mmHg. Eventually, inhaled Nitric Oxide was initiated post operative day 3 due to increasing pulmonary arterial pressures. A bedside laparotomy was eventually performed when bladder pressures peaked to 25 mmHg. APRV gradually and temporally improved the oxygen saturation and decreased the pulmonary arterial pressures with subsequent increase in systemic blood pressures. APRV promoted alveolar recruitment and decreased the shunting associated with abdominal compartment syndrome. Better oxygen saturations lead to increases in blood pressure by decreasing the effects of hypoxic pulmonary vasoconstriction on the right ventricle (RV). In patients with decreasing lung compliance and pulmonary comorbidities, APRV appears safe and allows for improve oxygenation, after failure with conventional modes of ventilation.
1. Introduction
In the following case report, the therapeutic intervention by mechanical ventilation is demonstrated. The application of a relatively new mode of positive pressure ventilation in a patient with both pulmonary hypertension (PH) and abdominal compartment syndrome (ACS) led to temporal improvements in oxygenation. APRV is readily available in most intensive care units (ICU) ventilators and eventually into anesthesia machines. Understanding the basic science and application of this mode by general anesthesiologist allows them greater options when dealing with patients in respiratory failure.
There are few instances were specific recommendations on positive pressure ventilation (PPV) are given with patients in respiratory failure. As long as gas exchange is adequate and ventilator induce injury is prevented, any type of ventilator mode is acceptable. There are exceptions, particularly in respiratory failure due to poor lung compliance. In the following case report, a patient with pulmonary hypertension who gradually develops abdominal compartment syndrome is presented. These two disease processes can make institution of mechanical ventilation challenging. Here, a relatively new mode of PPV is presented. Why APRV proved beneficial is surmised behind clinical and physiologic data.
2. Case Report
A 66-year-old 94 kg (BMI 34) male with restrictive lung disease, pulmonary hypertension and obstructive sleep apnea (OSA) presented to the Emergency Department with an incarcerated inguinal hernia requiring urgent reduction. The patient’s functional status was moderately impaired by his pulmonary co-morbidities. With severe OSA, an Apnea/Hypopnea Index of 60 and nocturnal desaturations, he sporadically used his prescribed BIPAP. His restrictive lung disease—total lung capacity 57% of expected via pulmonary function tests (PFT), was thought to be related to body habitus (central obesity). PFTs ruled out the possibility of a neuromuscular component of his restrictive lung disease, as maximal voluntary ventilation (MVV) was normal [1]. A thoracic CT scan done during his initial evaluation failed to show any intrapulmonary process. Transthoracic echocardiogram done in 2009 revealed normal left ventricular function, moderate LV diastolic dysfunction and elevated Pulmonary Artery Pressures (PAP = 45 mmHg). Home medications included atenolol, furosemide, monopril, amlodipine, aspirin, and simvastatin.
General anesthesia with an endotracheal tube (ETT) and PPV was initiated uneventfully. Surgical exploration revealed incarcerated bowel within the hernia. With questionable bowel viability, the abdomen was left open, with plans for re-exploration the following day; otherwise, the procedure and anesthetic were tolerated without complications. The patient was transferred to the ICU intubated on volume control ventilation. Overnight, he had decreased urine output, which responded to a combination of two 500 ml fluid boluses and furosemide. He subsequently went to the OR for an unremarkable abdominal closure, although an increase in peak inspiratory pressure coincided with closure. He remained hemodynamically stable throughout the procedure. Patient remained intubated in order to monitor respiratory compliance after abdominal closure. Back in ICU, he was mechanically ventilated on volume control. Gradually his ventilator pressure requirements began increasing along with decreasing urine output. His initial ventilator settings were volume control with a tidal volume of 600 cc, respiratory rate 18, fractional inspired oxygen concentration (FiO2) 100% and positive end-expiratory pressure (PEEP) 8. With persistent decreases in arterial oxygen saturation (SpO2) to the 80’s, no further PEEP increases or switching to pressure control improved oxygenation and led to hypotension. Chest x-ray was only remarkable for bibasilar ateclectasis, with no consolidation. Fluid management at this point was becoming difficult to assess. Overall, the patient was 5 liters positive; with a central venous pressure (CVP) ranging from 18 - 20 mm Hg, yet, urine out-put and blood pressure were decreasing. But, with existing PH and recent abdominal closure, the possibility of intravascular depletion existed. A pulmonary artery (PA) catheter revealed elevated pulmonary arterial pressures (PAP = 50 - 60 mmHg), and a normal wedge pressure. Troponin levels were normal, along with a bedside echo indicating normal LV function and ejection fraction. There was poor visualization of the RV with the TTE. Intra-abdominal pressures measured via the bladder catheter revealed Grade I ACS 15 mmHg [2]. With evolving ACS, non-surgical measure’s i.e. gastric decompression and positioning were instituted with no ventilatory improvements.
The patient continued becoming hypoxic, secondary to a combination of increasing ventilation/perfusion (V/Q) mismatch and shunting related to abdominal closure, which in turn worsened his PH. Hypotension and the SpO2 mildly responded to norepinephrine infusion and more fluid boluses. Rising PAP correlated with his clinical deterioration (Figure 1). With worsening respiratory failure on mechanical ventilation, manual bag ventilation was attempted. Aggressive manual positive pressure did not improve his SpO2, and may have caused further acute lung injury (ALI) as pink froth subsequently spewed out from his ETT and his lung compliance further decreased. At this point, with frank respiratory failure and worsening PH, APRV was instituted in hopes of increasing and maintaining maximal alveolar recruitment. With significant shunting and decreasing compliance, the high pressure (Phigh) on APRV was set at 35 mmHg. Over the following days his Phigh and length spent on high pressure (Thigh) were manipulated in order to maintain oxygenation. The patient’s hypoxemia subsequently improved, along with his hemodynamic stability. The gains were temporary, as ACS worsened, refractive to any nonsurgical interventions. The following day a bedside laparotomy was done after bladder pressures were measured at 25 mmHg. Small improvements in his hemodynamics were gradually seen, yet, his SpO2 only increased to 88% from 78%.
He continued on APRV but with increasing PH (80 mmHg), inhaled nitric oxide (iNO) 20 PPM was also temporarily added and successfully discontinued after a couple of days. Over the following days his PAP returned to 40 - 50 mmHg. He remained on APRV for over 2 months then transitioned to pressure control and eventually was weaned from the ventilator. After a 3-month protracted course the patient was eventually dis-
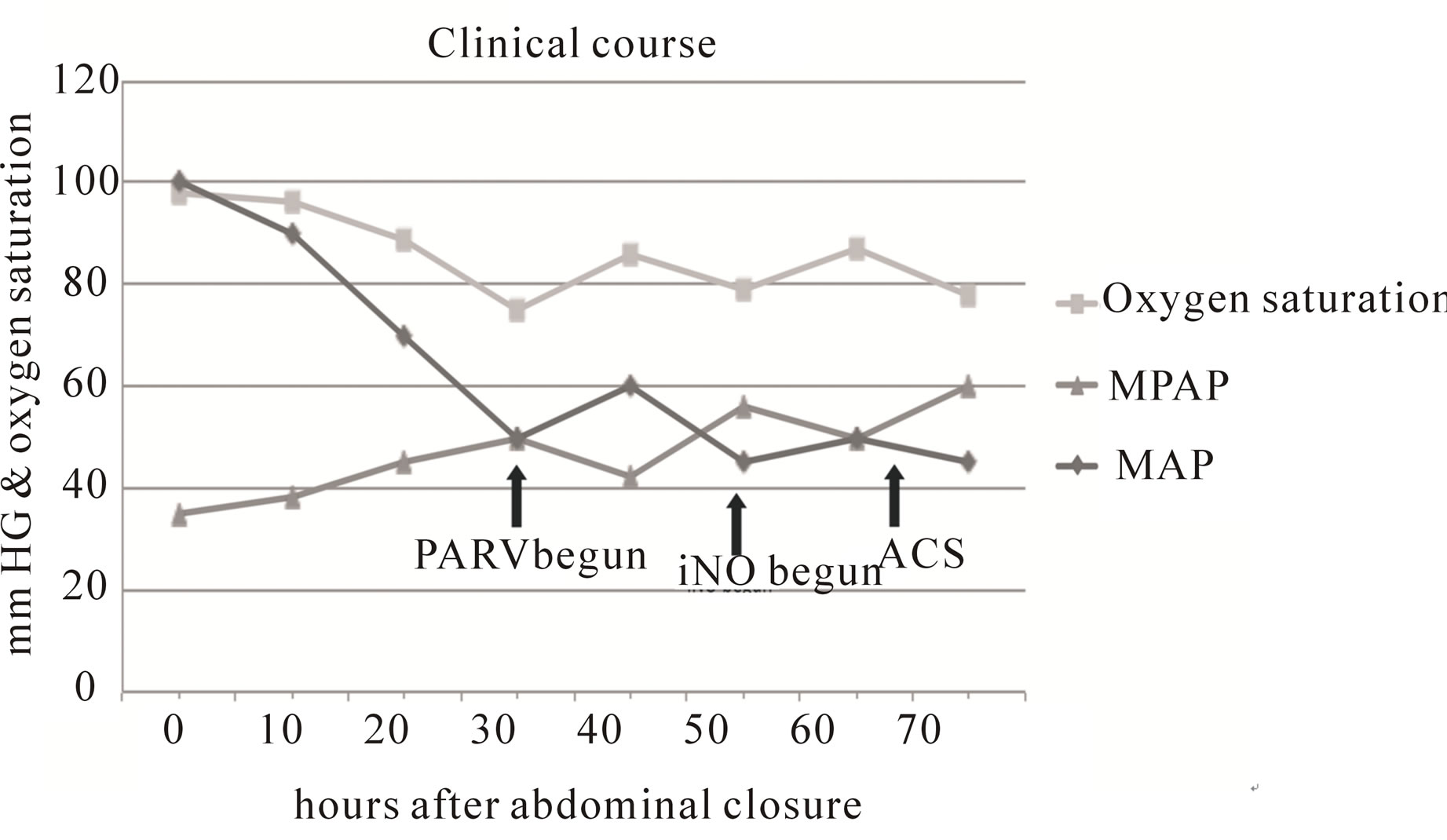
Figure 1. Graphical representation of patient’s clinical course after abdominal closure. MPAP—mean pulmonary arterial pressure; MAP—mean arterial pressure; APRV— airway pressure release ventilation; iNO—inhaled nitric oxide; ACS—acute compartment syndrome.
charged from the ICU. Subsequent follow up with Pulmonology revealed worsening diffusing capacity (DLCO) from baseline and increasing oxygen requirement. Yet, his functional activity remained unchanged from baseline.
3. Discussion
PH carries a significant risk of mortality in the perioperative period [3]. In the current case, deterioration occurred post operatively and not during induction. There have been several reports indicating the dangers associated during the induction period due to RV failure in patients with PH [4]. Hypoxemia, hypercarbia and acidosis must be especially avoided in patients with PH. The patient’s physical activity was moderately limited by PH (World Health Organization functional PH classification of [2]). His gradual decline and protracted course can be attributed to an interaction between several respiratory factors affecting his underlying PH, made worse by acute increases in intra-abdominal pressures.
PH is a syndrome caused by pathologic increases in pulmonary vascular resistance (PVR) that leads to restricted flow through the pulmonary circulation, with eventual RV failure [5]. A combination of restrictive lung disease and sleep apnea gradually led to secondary PH in this patient. OSA is known to cause nocturnal hypoxemia leading to increase PVR secondary to persistent hypoxic pulmonary vasoconstriction [6]. Overtime these two processes lead to increases in PVR and eventual PH.
The pulmonary circulatory system is the key in understanding PH. It is a high flow, low resistance circulation due to the less smooth muscle present in its arteries. The pulmonary arteries have much less vascular smooth muscle compared to systemic arteries. It also allows for greater distensibility, recruitment and greater influence by extravascular factors [3]. With increasing cardiac output, more arteries are recruited and PVR decreases under normal conditions. These anatomical arrangements allow for less intravascular pressure. Yet, pulmonary vessels are within the thorax and are subjected to both changing alveolar and intrapleural pressures. Hence, not only intravascular factors influence PVR, but also extravascular components can too. These include transmural pressure, lung volumes, body position, blood viscosity and PPV [3]. All of these so called passive influences can further modify the PVR independently. Patients already with PH can have further decompensation secondary to RV strain if these influences are not considered during the perioperative period or ICU stay.
ACS refers to organ dysfunction caused by persistent elevated abdominal pressure. The incidence varies, anywhere from 1% - 14% in ICU patients [7]. Both multiple medical and surgical conditions predispose patients to ACS. In our case, protracted bowel edema and the associated systemic inflammatory response syndrome (SIRS) after abdominal closure led to ACS. The patient’s pulmonary function was further compromise by reductions in lung compliance caused by increasing intra-abdominal pressure. Another consideration with ACS is application of PPV, since higher pressures are required in order to allow for gas exchange. With increasing ventilator pressure requirements, the risk for ventilator associated lung injury increases.
APRV provides continual positive airway pressure with intermittent releases that allow for ventilation. It also allows for spontaneous ventilation independent of the ventilator cycle via an open biphasic positive airway pressure circuit [8]. Continual mean airway pressure leads to alveoli recruitment and maintenance throughout the respiratory cycle decreasing the V/Q mismatch associated with traditional forms of PPV. Spontaneous ventilation also lessens V/Q mismatching and decreases the effects of PPV on central venous return and transmural pressure. These two conceptually physiologic advantages may both increase arterial saturation and decrease PVR in patients with conditions that lead to shunting, such as abdominal hypertension and acute respiratory distress syndrome (ARDS) [8]. There are four parameters that are manipulated in APRV. Phigh represents the continuous positive airway pressure, Thigh is the time spent on the set Phigh. The two other variables are Plow and Tlow. The former is the lower set pressure achieved with time releases and the latter is the length spent at that low pressure. Our patient’s initial Phigh begun at 35 mmHg. Yet, at one point it went up to 50 mmHg. Surprisingly, it appears he did not sustain any ventilator lung injury, although, with already existing ARDS it is difficult to assess.
He initially tolerated the induction of anesthesia and PPV without any significant alterations in hemodynamics. But, combined with existing restrictive lung disease, ARDS and further reduction in lung compliance, his status worsened. Overall, his initial deterioration presented with hypoxemia refractive to changes in PEEP and FiO2 on both volume and pressure controlled ventilation. Furthermore, PH worsened leading to hemodynamic deterioration due to RV strain. Once APRV was instituted, his SpO2 improved along with his blood pressure. The gains were temporary as he developed abdominal hypertension, and iNO was begun in order to maintain his SpO2 via pulmonary selective vasodilatation. The addition of iNO decreases the V/Q mismatch by increasing perfusion to ventilated alveoli [9].
APRV improved his respiratory status mostly by increasing and maintaining the mean inspiratory airway pressure in the face of decreasing transmural pressure from decreasing lung compliance. Yet, APRV is another form of PPV which can also affect PVR and lung compliance in either direction [8]. In APRV, the mean airway pressure, plateau pressure and Phigh are equivalent with a lengthy Thigh. High plateau pressures are associated with increased mortality in patients with ARDS [9]. Yet, in our case, by increasing the Phigh and Thigh, oxygenation improved in the face of severe V/Q mismatching, shunting and existing restrictive lung disease. Time release is an essential component in APRV that allows ventilation to occur. Ideally, a patient in APRV can spontaneously breathe, thereby optimizing ventilation and preload over other modes of PPV [8]. With ARDS and ACS, chemical paralysis was attempted in order to improve his oxygenation and abdominal compliance; thus negating his ability to ventilate spontaneously. In order to avoid hypoventilation, Tlow was increased. By stretching the Tlow, the alveolar recruitment done while in Thigh maybe countered, and lead to desaturation. Furthermore, APRV can increase PVR by prolonging the time spent at higher lung volumes than functional residual capacity. Thus, leading to compression of the alveolar vessels and outweighing the benefits of extra-alveolar vessel dilation [3]. It can be clinically inferred that Hypoxic Pulmonary Vasoconstriction influenced PVR greater than PPV, since with improvement in SpO2, his blood pressure improved regardless of the elevated mean airway pressure.
Any patient suffering from decrease pulmonary compliance and worsening gas exchange can be a candidate for APRV. Many physicians equate APRV to inverse inspiratory to expiratory i.e. ventilation. Essentially, by increasing the inspiratory time, the mean airway pressure is extended allowing for increase recruitment of alveoli, similarly to APRV. Yet, in APRV, the physician has greater control on the time and pressure being delivered by the ventilator. Prior to implementation, physicians must ensure that they understand the various adjustable settings.
4. Conclusions
The conceptual benefits of APRV in patients with respiratory failure have yet to be proven with large scale studies. But in our case, improved oxygenation with APRV led to temporal improvements in both hemodynamics and PH in a patient with evolving ACS. Identification of all the intra and extra-thoracic factors influencing PVR in patients with PH aids in bedside management. Overall, this case highlights the complexities of taking care of patients with underlying PH, restrictive lung disease, evolving ACS and demonstrates the incorporation of APRV in this scenario.
APRV is available in most ICUs. With further advances in anesthesia machines, this mode will also be available to anesthesiologists. This case illustrates a setting were the application and physiology behind APRV benefited the patient.
REFERENCES
- J. Steier, S. Kaul, J. Seymour, et al., “The Value of Multiple Tests of Respiratory Muscle Strength,” Thorax, Vol. 62, No. 11, 2007, pp. 975-980. doi:10.1136/thx.2006.072884
- M. L. Malbrain, M. L. Cheatham, A. Kirkpatrick, et al., “Results from the International Conference of Experts on Intra-Abdominal Hypertension and Abdominal Compartment Syndrome. I. Definitions,” Intensive Care Medicine, Vol. 32, No. 11, 2006, pp. 1722-1732.
- J. Strumpher and E. Jacobsohn, “Pulmonary Hypertension and Right Ventricular Dysfunction: Physiology and Perioperative Management,” Journal of Cardiothoracic and Vascular Anesthesia, Vol. 10, No. 6, 2011, pp. 687-704.
- P. S. Myles, J. L. Hall, C. B. Berry, et al., “Primary Pulmonary Hypertension: Prolonged Cardiac Arrest and Successful Resuscitation Following Induction of Anesthesia for Heart-Lung Transplantation,” Journal of Cardiothoracic and Vascular Anesthesia, Vol. 8, No. 6, 1994, pp. 678-681. doi:10.1016/1053-0770(94)90203-8
- D. B. Badesch, H. C. Champion, M. A. Sanchez, et al., “Diagnosis and Assessment of Pulmonary Arterial Hypertension,” Journal of the American College of Cardiology, Vol. 54, No. 1, 2009, pp. S55-S66. doi:10.1016/j.jacc.2009.04.011
- R. Dallari, G. Barozzi, G. Pinelli, et al., “Predictors of Survival in Subjects with Chronic Obstructive Pulmonary Disease Treated with Long-Term Oxygen Therapy,” Respiration, Vol. 61, No. 1, 1994, pp. 8-13. doi:10.1159/000196296
- D. J. Dries and J. J. Marini, “Airway Pressure Release Ventilation,” Journal of Burn Care & Research, Vol. 10, No. 6, 2009, pp. 929-936.
- F. Ichinose, J. D. Roberts Jr. and W. M. Zapol, “Inhaled Nitric Oxide: A Selective Pulmonary Vasodilator: Current Uses and Therapeutic Potential,” Circulation, Vol. 109, 2004, pp. 3106-3111. doi:10.1161/01.CIR.0000134595.80170.62
- “Ventilation with Lower Tidal Volumes as Compared with Traditional Tidal Volumes for Acute Lung Injury and the Acute Respiratory Distress Syndrome. The Acute Respiratory Distress Syndrome Network,” The New England Journal of Medicine, Vol. 342, No. 18, 2000, pp. 1301-1308.

