Open Journal of Clinical Diagnostics
Vol.4 No.2(2014), Article
ID:46839,7
pages
DOI:10.4236/ojcd.2014.42017
Giant Ascending Aortic Aneurysm: Are There Peculiarities in the Developing World?
Ademola J. Adekanmi1,2*, Doyin Olusunmade2
1Department of Radiology, College of Medicine, University of Ibadan, Ibadan, Nigeria
2Department of Radiology, University College Hospital, Ibadan, Nigeria
Email: *kanmiademola@gmail.com, *aadekanmi@comui.edu.ng
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 2 April 2014; revised 2 May 2014; accepted 5 June 2014
ABSTRACT
Aim: To highlight the imaging features and the peculiarities associated with a giant ascending aortic aneurysm in a typical developing world setting. Presentation of Case: A 78-year-old known hypertensive and asthmatic woman of 25 and 10 years duration respectively. She had an untreated degenerative aortic valve lesion and moderate aortic regurgitation. This case study showed the complex interplay of background untreated aortic valve disease, aortic insufficiency, chronic hypertension, chronic airway obstructive disease and ageing in a case of giant aneurysm of the entire ascending aorta. It also emphasizes the role of imaging in diagnosis and follows up of ascending aortic aneurysm particularly where prevention is key to prevent the high mortality associated with Thoracic aneurysymal rupture. Conclusion: Timely institution of appropriate treatment of aortic valve disease, prevention and adequate control of hypertension is key to prevent the development and the grave prognosis of ascending aortic aneurysm, particularly in the developing world where appropriate medical facilities, expertise and treatment are scarce.
Keywords:Ascending Thoracic, Aorta, Aneurysm, Hypertension

1. Introduction
Giant thoracic ascending aortic aneurysms are rare [1] but always bring disastrous complications. It is required to conduct emergency surgical intervention due to high risk of sudden rupture and hemorrhage and consequent high mortality [2] . Recommended imaging modalities for effective diagnosis, follow up and monitoring of treatment include Echocardiography, Multidetector computed tomography (MDCT) scan and MDCT angiography as well as magnetic resonance imaging (MRI), including MRI angiography and functional studies [3] . While chest X-ray may be helpful particularly in asymptomatic cases [4] , the definitive treatment of choice for giant aortic aneurysms is surgery, either through endovascular repair or the open surgical technique [4] . An elderly patient with aortic valve disease, aortic insufficiency, hypertension and asthma was reported in this paper. Relevant imaging tests were done to make timely diagnosis. This case has made us have a more specific understanding of this rare disease.
2. Case Presentation
A 78-year-old known hypertensive and asthmatic woman who presented with breathlessness of one year duration, which worsened in the last three weeks prior to presentation. The patient was diagnosed hypertensive and asthmatic 25 and 10 years respectively prior to presentation. The episodes of breathlessness usually developed on exertion and there was associated easy fatigability and recurrent non-productive cough. There was no history of orthopnea or paroxysmal nocturnal dyspnea. The patient however noticed recurrent swelling of her legs on sitting for some time.
She was diagnosed hypertensive on routine checks ten years ago; blood pressure at diagnosis was 150/70 mmHg. Echocardiography examination carried out then showed a calcified aortic valve with associated moderate aortic regurgitation. A diagnosis of hypertensive heart disease with a degenerative valvular lesion and moderate aortic regurgitation was made. She was placed on anti hypertensive drug (5 mg Lisinopril titrated upward to10 mg daily) but had no surgical treatment for the aortic valvular lesion.
Examination at this presentation revealed a middle aged woman, she was pale, dehydrated, and dyspneic with a respiratory rate of 32/minute but not febrile. Essential systemic findings were in the chest and cardiovascular system. Apart from the dyspnoea, her breath sounds were vesicular but reduced in both lung bases. Her pulse was regular and of good volume but had tachycardia with a rate of 124/minute. Her blood pressure was elevated, and measured 210/110 mmHg. Heart sounds, S1 and S2 only were heard with no murmur but her neck veins were distended. Radiological investigations showed giant aneurysm of the entire ascending aorta. She was commenced on Ramipril and intravenous frusemide. Surgical management was advised; she however rejected surgical intervention and died 12 days after presentation following an episode of respiratory distress.
2.1. Radiological Investigations
The chest radiograph (Postero-Anterior view) essentially showed the heart to be enlarged with a biventricular configuration and cardiac apex abutting on the lateral chest wall. The middle mediastinum was widened with a convex outer border on the right and continuous with the right cardiac margin, extending superiorly to the level of the 3rd posterior intercostal space, indicating ascending aortic enlargement. The aortic arch was also prominent (Figure 1(a)).
The lateral view of the chest also confirmed biventricular cardiomegaly and the markedly dilated entire ascending aorta (Figure 1(b)).
The patient also had Computed tomography (CT) angiography of the aorta. Axial images were acquired with a 64 slice Multidetector Toshiba Aquillion computed tomography scanner. Multiplanar and 3-D reconstructions were done. The images showed cardiomegaly with a cardiothoracic ratio of 72%. A thin-walled fusiform aneurysm of the entire ascending aorta extending from the outlet of the left ventricle to the proximal aspect of the arch of the aorta was seen with the mid ascending aorta diameter of 10.2 cm at the level of the bifurcation of the pulmonary artery (Figure 2).
The Multiplanar reconstruction in the Saggital and coronal views also show previously described features. In addition, the Saggital view showed features consistent with background dorsolumbar spondylosis, as evidenced by the anterior marginal osteophytes, endplate sclerosis and some narrowed disc spaces. A focal nodular calcification was noted at the medial wall of the aortic root in the coronal view (Figure 3(a) and Figure 3(b)). No thrombus or dissection was seen, all in keeping with giant thoracic aortic aneurysm (TAA).
2.2. Discussion
An aneurysm is a focal or diffuse dilatation of an artery diameter of at least 50% or 1.5 times more than its
 (a)
(a)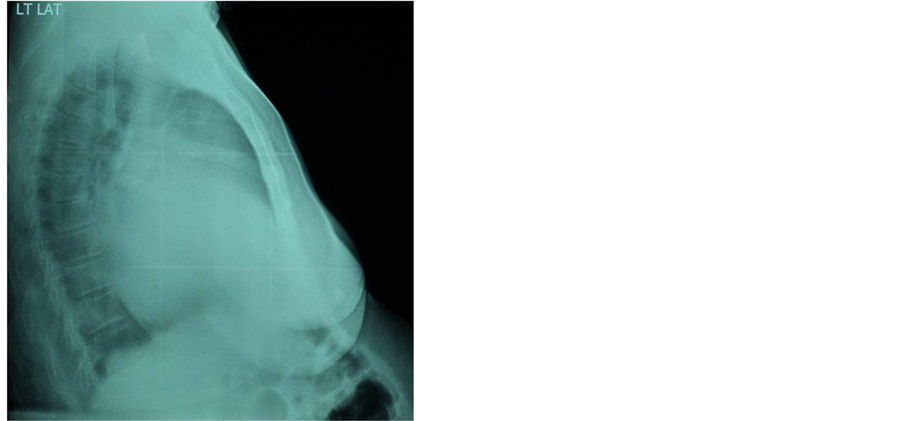 (b)
(b)
Figure 1. (a) Chest radiograph showing marked cardiomegaly with biventricular configuration, widening of the middle and superior mediastinum with a convex margin continuous with the right ventricular margin; (b) Lateral chest radiography showed cardiomegaly with bi-ventricular configuration and markedly dilated ascending aorta.

Figure 2. Contrast enhanced axial computed tomography image of the chest. This shows gross dilatation of the aortic root. Asterisk—aortic root and white arrow—left ventricle.
 (a)
(a) (b)
(b)
Figure 3. (a) MPR saggital view of CT angiography of the aorta-showing the markedly dilated ascending aorta. Background dorsal spine degenerative changes are present as evidenced by anterior dorsal spine osteophytes; (b) Multiplanar reconstructed coronal thoracic computed tomography showing fusiform, marked dilatation of the entire ascending aorta with a speck of calcification seen in the Mitral valve region.
normal size/diameter. In the thoracic aorta a maximum diameter of up to 3 cm is normal, a diameter of 4.5 cm or more is considered aneurysmal and diameters of 6 cm or more have been documented to carry significant risk of rupture [2] . While a diameter of 1.1 to 1.5 times of normal value is classified as ectasia of the thoracic aorta [1] .
Although aneurysms of the thoracic aorta are the most common vascular cause of mediastinal masses, accounting for 10% of all mediastinal masses. Thoracic Aortic Aneurysms (TAAs) are less common than abdominal aortic aneurysms accounting for 25% of all aneurysms. TAAs are commoner in males with a male to female ratio of (1.7 - 3):1 with a mean age of 65 years at presentation [2] [3] . A study carried out by Olsson et al. in Sweden in 2006 showed an incidence of 16.3/100,000 per year in men and 9.1/100,000 per year in women with a rising prevalence [5] . The incidence rate of thoracic aortic aneurysm, in studies in USA, is approximately 10.4 per 100,000 per year [6] [7] . However data on the incidence of TAAs in Nigeria is not readily available, possibly due to lack of or documentation of cases and non availability of appropriate imaging modalities in most part of the country.
TAAs can be classified according to the arterial wall layer, the shape and part of the ascending aorta that is involved. While true aneurysms involve all the three layers-adventitia, media and intima, false aneurysms involve only the outer layer and are contained within the adventitia [3] . TA aneurysm may also be fusiform in shape if there is circumferential involvement of the arterial wall or saccular when it involves only a portion of the wall. Fusiform and saccular aneurysms account for 80% and 20% of TAAs respectively.
Thoracic aortic aneurysms may also involve one or more segments of the thoracic aorta: the aortic root, ascending aorta, aortic arch and descending aorta. The involved segment affects the clinical presentation, natural history and treatment, and also offers clues to the etiology [8] . About 50% of thoracic aortic aneurysms involve the ascending aorta. 40% of cases occur in the descending aorta while the arch and thoracolumbar aneurysms account for 10% each [1] . Ascending aortic aneurysms may involve part or its entirety, extending as proximally as the aortic annulus and as distally as the innominate artery [4] . This is a case of a giant fusiform aneurysm of the entire ascending aorta.
Aneurysm formation results from combination of inflammation, proteolysis and disturbed survival of smooth muscle cells with elevated proteolytic matrix metalloproteinase enzymes and decrease in inhibitory enzymes resulting in elastic fiber fragmentation [1] . The pathogenesis of ascending aortic aneurysms involves a complex interplay between genetic predispositions and hemodynamic factors e.g. hypertension and aortic valvular disease [9] [10] . The commonest cause of aortic aneurysm is atherosclerosis in 80% of cases [3] . But in the ascending aorta, this is an infrequent cause [1] [11] . Cystic Medial degeneration, described as the accumulation of amorphous basophilic material in the media, with resultant elastic fibre fragmentation and smooth muscle loss, is the commonest cause of ascending aortic aneurysms. This usually occurs as an acquired degenerative condition which results to some extent from ageing but accelerated by hypertension. It may also occur in younger patients typically as part of a genetically transmitted disorder e.g. Marfan and Ehler-Danlos syndromes [1] [12] . Documented risk factors include smoking, chronic obstructive pulmonary disease (COPD), hypertension, atherosclerosis, male gender, older age, high body mass index, bicuspid or unicuspid aortic valves, genetic disorders, and positive family history [4] . Most cases however have chronic hypertension, which may contribute to the formation or the expansion of the aneurysm [4] .
Aortic valvular disorders including aortic valvular disease and bicuspid aortic valves are known causes of ascending aortic aneurysms [1] [9] [10] . In this case, the etiology was aortic valve disease, probably accelerated by hypertension. However, there is paucity of literature on cases of ascending aortic aneurysms secondary to degenerative aortic valvular lesions without associated bicuspid aortic valves. Historically, syphilis used to be a common cause of ascending aortic aneurysms but it is now an uncommon cause due to the aggressive use of antibiotics [1] . Other causes of thoracic aortic aneurysms include trauma, infection, Takayassu and Giant cell arteritis. There was no preceding history of trauma, sexually transmitted diseases or features of Takayassu and giant cell arthritis in this case.
Many patients with thoracic aortic aneurysms are without symptoms but are discovered incidentally on chest X-ray or other investigations for other ailments. In symptomatic cases, clinical symptoms are usually secondary to the presence of complications or imminence of complications. Clinical features of thoracic aortic aneurysms include breathlessness, dyspnea, dysphasia, hoarseness, chest pain and back pain due to compression of surrounding structures including the trachea, bronchi, esophagus and paralysis of the recurrent laryngeal nerve. Rarely, compression on the superior vena cava (SVC) may give rise to symptoms of SVC syndrome-distended neck veins etc. from large TAA. Aortic aneurysms involving the ascending aorta and aortic root may present with features of congestive cardiac failure due to aortic regurgitation. Large aneurysms may also rupture into the pericardial space, left pleural space, tracheobronchial tree, Superior vena cava or pulmonary artery giving rise to acute aortic syndromes [11] [13] [14] . History of breathlessness, dyspnea, distended neck veins and leg swelling from congestive cardiac failure probably secondary to the documented aortic regurgitation was present in this patient.
The normal growth rate for the ascending aorta is 0.07 - 0.2 cm/year [15] and approximately 1 mm per year [16] . Risk factors for increased aortic growth include advancing age, female sex, chronic obstructive pulmonary disease, hypertension, positive family history, aortic dissection and the initial aortic diameter. The principal predictor of the risk of rupture or dissection is the aortic size. Davies et al. carried out a study on thoracic aortic aneurysms, this showed that at aortic sizes greater than 6.0 cm, the risk of rupture was increased 27-fold and death, rupture or dissection occurred at 15.6% per year [17] . However death from rupture, we believe, will probably be higher in the developing World than the documented rate per year due to poverty hindering prompt hospital treatment with consequent late hospital presentation, scarcity of adequate diagnostic equipments and adequacy of appropriate manpower and early interventions.
2.3. Imaging
Many patients with thoracic aortic aneurysms are asymptomatic, and are incidentally discovered on chest X-ray or other investigations for other ailments [4] . Possible Chest X-ray findings that may suggest TAA on a CXR are: 1) Widened mediastinum due to prominence of the ascending aorta and 2) Presence of mass effect (e.g. deviation of the trachea) can be an indicator of an ascending aortic aneurysm. Other imaging modalities employed in the evaluation of suspected TAA include echocardiography, Multidetector computed tomography and Magnetic resonance imaging. While echocardiography, particularly the trans-thoracic route is very useful in making diagnosis and to follow up cases of TAAs involving the aortic root, particularly to access the aortic valves and left ventricular function in these patients.
The trans-oesophageal echocardiography could be used in patients with poor acoustic window to the transthoracic approach [3] . Echocardiography has the advantage of being cheap, widely available and ionizing radiation free and reproducible. MDCT and MRI offer excellent images. The high spatial resolution, ability to acquire very thin millimeter cuts and multiplanar angiographic reformation has made the MDCT an excellent modality for TAA evaluation [3] . CT is the modality of choice for identifying rupture and impending rupture of thoracic aortic aneurysms [18] . MRI is ionizing radiation free, has multiplanar capability and excellent soft tissue resolution. It excellently depicts the entire aortic anatomy and very effective in diagnosis and characterization of aneurysms in combination with MR angiography while functional MRI studies are also invaluable in the assessment of patients with aortic valve disease [3] .
The need for surgery is based on the assessment of risk of rupture, life expectancy and operative risk. Indications for surgery for ascending aortic aneurysms include an aneurysmal diameter exceeding 5.5 cm, a growth rate of more than 0.5 cm/year and symptomatic patients including those with acute aortic syndromes. Compression of surrounding organs, aortic insufficiency and ascending aortic dissection are accepted indications regardless of the size. Patients with Marfan’s syndrome and bicuspid aortic valves should undergo surgery at diameters exceeding 5.0 cm [6] [16] [19] and those with family history of aortic aneurysmal rupture or dissection should undergo surgery at diameters of 4.5 cm or higher. In this case, despite the diagnosis of aortic valve lesion ten years ago, there was no intervention to the valvular disease at that time probably due to non availability of appropriate facilities and expertise. The patient had overwhelming indications for surgery with an aneurysmal diameter of almost twice the cut off with associated symptoms and serious risk of rupture with associated symptoms but declined surgery. The unavailability of valvular interventions and appropriate surgical care for aortic aneurysm at most centers, poverty and late presentations on the part of the patients makes the already known poor prognosis of TAAs even worse and the mortality rate in the developing World higher than documented in literatures.
3. Summary
This case report highlights the multi factorial interplay of aortic valve disease, aortic regurgitation, chronic obstructive airway disease, chronic hypertension and ageing in the pathogenesis of ascending thoracic aortic aneurysm. It also described the complications of TAA, particularly rupture at diameters over 6 cm and the high mortality associated with it. There was a lack of timely intervention to the diagnosed valvular disease at diagnosis which might have slowed/stopped the development of the aneurysm due to non availability of appropriate facility, a common and serious challenge in developing nations.
Ascending aortic aneurysm is serious sequelae of aortic valve disease, valvular insufficiency and chronic hypertension particularly in the elderly. Meticulous follow up with radiological modalities, timely intervention to repair diseased valves, and control of high blood pressure could prevent development of ascending aortic aneurysm. Surgery should be done without delay when indicated to prevent aneurysmal rupture and associated high mortality.
Acknowledgements
We thank all the staff of the Computed tomography unit of the Department of radiology University College Hospital, Ibadan, Nigeria for their support.
References
- Evangelista, A. (2010) Aneurysm of the Ascending Aorta. Heart, 96, e979-e985.
- Canon, C. (2009) Radiology: McGraw-Hill Specialty Board Review. McGraw-Hill Education.
- Dahnert, W. (2011) Radiology Review Manual. 7th Edition, Williams & Wilkins, Lippincott.
- Olsson, C., Thelin, S., Stahle, E., Ekbom, A. and Granath, F. (2006) Thoracic Aortic Aneurysm and Dissection: Increasing Prevalence and Improved Outcomes Reported in a Nationwide Population-Based Study of More than 14,000 Cases from 1987 to 2002. Circulation, 114, 2611-2618.
- Ramanath, V.S., Oh, J.K., Sundt, T.M. and Eagle, K.A. (2009) Acute Aortic Syndromes and Thoracic Aortic Aneurysm. Mayo Clinic Proceedings, 84, 465-481. http://dx.doi.org/10.1016/S0025-6196(11)60566-1
- Clouse, W.D., Hallett, J.W., Schaff, H.V., et al. (1998) Improved Prognosis of Thoracic Aortic Aneurysms. A Population-Based Study. JAMA, 280, 1926-1929.
- Daly, C. and Mulcahy, D. (2000) Thoracic Aortic Aneurysms. Irish Heart Foundation, 3, 34-36.
- Tseng, E. Thoracic Aortic Aneurysm. http://emedicine.medscape.com/article/424904-overview
- Kuivaniemi, H., Platsoucas, C.D. and Tilson, D. (2008) Aortic Aneurysms. An Immune Disease with a Strong Genetic Component. Circulation, 117, 242-252.
- Patel, H.J. and Deeb, M. (2008) Ascending and Arch Aorta. Pathology, Natural History and Treatment. Circulation, 118, 188-195.
- Isselbacher, E.M. (2005) Thoracic and Abdominal Aortic Aneurysms. Circulation, 111, 816-828. http://dx.doi.org/10.1161/01.CIR.0000154569.08857.7A
- Cury, M., Zeidan, F. and Lobato, A.C. (2013) Aortic Disease in the Young: Genetic Aneurysm Syndromes, Connective Tissue Disorders, and Familial Aortic Aneurysms and Dissections. International Journal of Vascular Medicine, 2013, Article ID: 267215.
- Woo, Y. and Mohler, E. (2010) Clinical Features and Diagnosis of Thoracic Aortic Aneurysm. Up ToDate. http://46.4.230.144/web/UpToDate.v19.2/contents/f26/19/27232.htm
- Srivastava, V., AlHadid, K., Saravanan, P., Zacharias, J. and Bittar, M.N. (2011) Giant Aneurysm of the Ascending Aorta. BMJ Case Reports, 2011, 4-7.
- Cozijnsen, L., Braam, R.L., Waalewijn, R.A., Schepens, M.A.M., Loeys, B.L., Van Oosterhout, M.F.M., et al. (2011) What Is New in Dilatation of the Ascending Aorta? Review of Current Literature and Practical Advice for the Cardiologist. Circulation, 123, 924-928. http://dx.doi.org/10.1161/CIRCULATIONAHA.110.949131
- Elefteriades, J.A. (2002) Natural History of Thoracic Aortic Aneurysms: Indications for Surgery and Surgical versus Nonsurgical Risks. The Annals of Thoracic Surgery, 74, S1877-S1880.
- Davies, R.R., Goldstein, L.J., Coady, M.A., Tittle, S.L., Rizzo, J.A., Kopf, G.S., et al. (2002) Yearly Rupture or Dissection Rates for Thoracic Aortic Aneurysms: Simple Prediction Based on Size. The Annals of Thoracic Surgery, 73, 17-28.
- Prescott-Focht, J.A., Martinez-Jimenez, S., Hurwitz, L.M., Hoang, J.K., Christensen, J.D., Ghoshhajra, B.B., et al. (2013) Ascending Thoracic Aorta: Postoperative Imaging Evaluation. Radiographics?: A Review Publication of the Radiological Society of North America, Inc., 33, 73-85.�
- Akhtar, J., Siddiqui, M.A., Khan, N.A. and Alam, M.A. (2013) Right Phrenic Nerve Palsy: A Rare Presentation of Thoracic Aortic Aneurysm. Malaysian Journal of Medical Sciences, 20, 98-101.
NOTES

*Corresponding author.

