Open Journal of Psychiatry
Vol.3 No.1(2013), Article ID:27395,6 pages DOI:10.4236/ojpsych.2013.31005
Variation in the DRD2 gene affects impulsivity in intertemporal choice
![]()
1Department of Psychiatry, Sakae Seijinkai Hospital, Yokohama, Japan
2Department of Behavioral Science, Graduate School of Letters, Hokkaido University, Sapporo, Japan
3Department of Human Genetics, Graduate School of Medicine, University of Tokyo, Tokyo, Japan
4Research Center for Hepatitis and Immunology, National Center for Global Health and Medicine, Ichikawa, Japan
5Centre for Addiction and Mental Health, University of Toronto, Toronto, Canada
6Department of Neuropsychiatry, Graduate School of Medicine, University of Tokyo, Tokyo, Japan
7Division for Environment, Health and Safety, University of Tokyo, Tokyo, Japan
8Department of Physical and Health Education, Graduate School of Education, University of Tokyo, Tokyo, Japan
Email: *yoshiya-tky@umin.ac.jp
Received 10 December 2012; revised 7 January 2013; accepted 14 January 2013
Keywords: Dopamine Receptor D2 (DRD2); Impulsivity; Neuroeconomics; Single Nucleotide Polymorphism (SNP); Time-Discount Rate
ABSTRACT
Introduction: Impulsivity in intertemporal choice has been operationalized as “delay discounting”, referring to the preference for a sooner, smaller reward in neuroeconomics. It is reportedly associated with the dopaminergic systems. Dopamine receptor D2 (DRD2) is the D2 subtype of the dopamine receptor of the G-protein coupled receptor family. The aim of this study was to explore the effect of single nucleotide polymorphisms (SNPs) in DRD2 gene on delay discounting. Methods: The participants consisted of 91 healthy Japanese people (66 males and 25 females with a mean age of 40.9 ± 6.9 years). Each participant completed the Kirby’s monetary choice questionnaire (MCQ) for delayed gain and donated a whole blood sample. Two SNPs (C957T (rs6277) and TaqI A (rs1800497)) in DRD2 were genotyped by using the DigiTag2 assay. SNP linear regression analyses with 100,000 permutations were conducted for the hyperbolic time-discount rate (k). Results: The SNP C957T showed a significant association; participants with more minor alleles (T) were more impulsive in intertemporal choice for delayed gain (multiplicity-corrected P = 0.041 with a small effect size). Conclusion: The variation in the DRD2 gene is associated with impulsive decision-making. This is the first study to demonstrate an association between DRD2 and impulsivity in intertemporal choice with a multiplicitycorrected significance.
1. INTRODUCTION
Impulsivity is a core deficit in neuropsychiatric disruptions such as addiction, attention-deficit hyperactivity disorder (ADHD), and depression. In the rapidly developing field of neuroeconomics [1-4], impulsivity in intertemporal choice has been operationalized as “delay discounting”, referring to the decrease in the subjective value of a reward as the delay until its receipt increases (i.e., the preference for a sooner, smaller reward rather than a later, larger reward) [3-8]. People often prefer small but immediate rewards to large but delayed ones (temporal discounting). Behavioral and neuroeconomic studies have demonstrated that impulsivity in intertemporal choice (“impatience”) is dependent on the length of delay until receipt in human and animal discounting behavior [9-16]. In choosing between smaller sooner rewards and larger later ones, people tend to be patient in the distant future but impulsive in the near future. Impulsivity in intertemporal choice is associated with dependences (alcohol [17], tobacco [18], opiate [19], cocaine [20], and gambling [21]), antisocial personality disorder [22], and so on.
Behavioral economic, neuropsychopharmacological studies have reported that human and animal intertemporal choice behavior is better described by the hyperbolic discount model [3,5,9-11,13,14,16,23-30]. The model is expressed as V (D) = V (0)/(1 + kD), in which V (D) is the subjective value of the reward available at delay D, V (0) is the subjective value at delay D = 0, i.e., the value of an immediate reward, and the hyperbolic time-discount rate (k) is a free parameter indicating a subject’s impulsivity in intertemporal choice. Large k values correspond to more rapid (steeper) discounting, whereas small k values indicate self-control in intertemporal choice [9,13].
There have been reports that impulsivity in intertemporal choice is associated with the dopaminergic systems [3,31] at least in part via altered temporal cognition [14]. Dopaminergic neural systems (e.g., the ventral tegmental area, the nucleus accumbens, or the ventral pallidum) are reward-processing brain regions, and recent developments in reinforcement learning theory of the systems have elucidated the importance of reward prediction [32, 33]. The dopaminergic systems have pivotal roles in temporal discounting: it has been reported that impulsive intertemporal choice between immediate and delayed money is associated with the activation of dopaminergic systems such as the ventral tegmental area [6], and that a subjective value of a delayed reward is associated with the activation of dopaminergic neural circuits [5]. Also, temporal discounting is partially attributable to risk aversion [34], which is modulated by Dopamine receptor D2 (DRD2) [35]. Regarding neuropsychiatric illnesses, a recent study reported that schizophrenic patients (who are known to have altered dopamine activities) have larger discount rates in comparison to healthy controls [36]. As for DRD2 gene polymorphisms, C957T was suggested to be associated with dysfunctional impulsivity [37]. DRD2 TaqI A polymorphism was also suggested to influence impulsivity as measured with a delay discounting task [38], and had a significant association with severe substance dependence [39].
The aim of the present study is to explore the effect of single nucleotide polymorphisms (SNPs) in the DRD2 gene on impulsivity in intertemporal choice (i.e., delay discounting) as measured by Kirby’s monetary choice questionnaire (MCQ) for delayed gain [19].
2. METHODS
2.1. Participants
All participants were recruited in 2010 from Kanagawa Prefecture, which is adjacent to Tokyo, Japan. They comprised 104 genetically unrelated, non-clinical Japanese white-collar workers in a large corporation, and they represented a high-functioning, non-clinical adult population. There were 20 ex-smokers and 17 current smokers. The participants completed the Japanese version of Kirby’s MCQ for delayed gain [19]. Trained psychiatrists and psychologists conducted a short structured diagnostic interview in accordance with the Mini-international Neuropsychiatric Interview (M.I.N.I.) [40]. This was to confirm lifetime diagnoses of affective, anxiety, and psychotic disorders, based on the criteria of the Diagnostic and Statistical Manual of Mental Disorders, 4th edition (DSM-IV) [41]. Each participant donated a whole blood sample for DNA analysis. Thirteen of the 104 participants were excluded because of a current or past DSM-IV diagnosis, degenerated DNA, or inconsistent rating on the MCQ. Four males had been diagnosed with major depressive disorder; four participants (three males and one female) were diagnosed with bipolar II disorder; DNA samples of four participants (three males and one female) were degenerated; and one male had an inconsistent rating.
After the previously described exclusions, the study participants comprised 91 high-functioning Japanese workers, which included 19 ex-smokers and 14 current smokers. (Of the 66 male participants, 19 were exsmokers and twelve were current smokers; of the 25 female participants, two were current smokers.) The mean age of the participants was 40.9 years (standard deviation [SD] = 6.9) and the age range was 28 to 56 years. The mean age of the males and females was 42.0 (SD = 6.6) and 38.0 (SD = 6.8) years, respectively, with an age range of 28 to 54 years for the males and 28 to 56 years for the females. There were no significant differences between the 91 participants and the 13 excluded participants in sex, age, smoking status, or MCQ scores.
The aim of the present study was clearly explained to all participants. They provided written, informed consent. The ethics committee of the Faculty of Medicine at the University of Tokyo approved the study, and the study conformed to the provisions of the Declaration of Helsinki.
2.2. Measurement of the Time-Discount Rate with Kirby’s Monetary Choice Questionnaire (MCQ) for Delayed Gain
The MCQ for delayed gain is a self-administered instrument for measuring time-discount rates [19]. It consists of 29 statements and features choices between two hypothetical sums of money of varying sizes and delays. The MCQ includes two validity statements to eliminate random responders (e.g. “[question 28] Would you prefer 1,000,000 yen now or 0 yen in 15 days?” and “[question 29] Would you prefer 0 yen now or 1,000,000 yen in a day?”). In the MCQ, 27 statements require choosing a small amount now or a large amount in the future; each choice helps in estimating the respondent's time-discount rate. High time-discount rates on the questionnaire are associated with self-reported impulsivity and real-life impulsive behaviors [42]. The one-year temporal stability of the MCQ has been confirmed [43].
In the current study, the Japanese version of the MCQ for delayed gain used the same procedure as previous studies [19,44-47] for assessing time-discount rates. As in our previous studies, One dollar was converted to 100 yen (e.g. “Would you prefer 2700 yen now or 5000 yen in three weeks?”) [45-47]. Three time-discount rates— small, medium, and large gains, and represented by k (S), k (M), and k (L), respectively—were obtained for each participant. The geometric-mean time-discount rate, k(mean), for the three rates of the different sizes was calculated by following Kirby’s procedure [19,43]. We then examined the relationship between the hyperbolic time-discount rates (i.e., k (mean)) and the variations in the DRD2 gene. Because the distribution of the timediscount rate, k, is known to be skewed, we used the log of k (i.e., ln k) in the following analysis, based on a standard analytical procedure [19,43]. The same assessment procedures for time-discount rates have recently been utilized in a structural neuroimaging study [8].
2.3. Single Nucleotide Polymorphism (SNP) Selection and Genotyping
Genomic DNA was isolated from leukocytes in whole blood by using the Wizard® genomic DNA purification kit (Promega Corporation, Madison, WI) [48]. In the present study, we selected two SNPs in the DRD2 gene: C957T (rs6277) [37] and TaqI A (rs1800497) [38], which were suggested to influence impulsivity as measured with delay discounting tasks. These SNPs were genotyped at the Human SNP-Typing Center (University of Tokyo, Tokyo, Japan) by using the DigiTag2 assay [49,50], which is designed for multiplex SNP typing. The assay was used in accordance with the manufacturer’s protocol (Olympus, Tokyo, Japan).
2.4. Statistical Analysis
The Hardy-Weinberg equilibrium (HWE) for genotype distributions was assessed via χ2 test by using PLINK version 1.07 [51]. As the SNP-based quantitative trait association analysis for an additive model, linear regression analyses using single markers for the ln k (mean) values of the MCQ were performed by using PLINK. Family-wise (i.e., SNP-wise) corrected empirical P-values were calculated on the basis of 100,000 permutations [52] under the control of the family-wise error rate (FWER) for each SNP examined [53] as a function of PLINK.
The coefficient of determination (R2) was obtained through regression analysis. R2 expresses the contribution ratio and represents the effect size in regression analysis [54,55]. R2 was categorized approximately as small (0.01 ≤ R2 < 0.1), medium (0.1 ≤ R2 < 0.3), or large (0.3 ≤ R2). Statistical power was estimated by using QUANTO version 1.2.4 [56]. Statistical analyses of mean values, SD, R2, β, χ2 tests, t tests, and regression tests for demographic data and the time-discount rates were conducted with SPSS 16.0.2 J for Windows (SPSS Incorporated, Chicago, IL) [57]. Statistical tests were two tailed and the significance level was set at P < 0.05.
3. RESULTS
Table 1 lists the two genotyped SNPs. Both SNPs had genotyping call rates greater than or equal to 0.90. Their HWE P-values were greater than or equal to 0.05, indicating that none of their genotype distribution severely deviated from the equilibrium. The minor allelic frequencies (mAFs) of both SNPs were greater than or equal to 0.05.
Table 2 lists the results of the SNP linear regression analysis. The SNP C957T (rs6277) showed an SNP-wise (i.e., multiplicity-corrected) empirically significant association with the ln k(mean) of the MCQ (P = 0.041 with a small effect size), indicating that participants having more minor alleles (T) in C957T are more impulsive in intertemporal choice for delayed gain. SNP TaqI A exhibited nonsignificant levels (P = 0.821). No significant association was observed between haplotypes comprising the two SNPs and values of ln k(mean) of the MCQ after correcting for multiple testing by the haplotypebased quantitative trait association analysis (data not presented).
The statistical power of C957T was estimated at 0.65 for n = 90, mAF = 0.050, R2 = 0.059, mean ln k (SD) = −5.71 (1.96), and two-tailed nominal P < 0.05. Furthermore, the smoking status did not affect the time-discount rate (i.e., ln k (mean)) in the present population (β = 0.095; P > 0.7). This was consistent with our previous report indicating that the intertemporal choice did not differ between mild smokers and nonsmokers in the Japanese population [27].
4. DISCUSSION
Our study is to demonstrate that a genetic variation in the DRD2 gene is associated with impulsivity in intertemporal choice (i.e., delay discounting). We observed that a minor allele (T) in C957T (rs6277) was positively associated with impulsivity in intertemporal choice for delayed gain with a multiplicity-corrected significance: participants having more minor alleles were more impulsive in intertemporal choice. Colzato et al. [37] reported that the minor homozygotes had higher scores on selfreported dysfunctional impulsivity with a nominal significance. Their results are consistent with ours.
In this study, no significant association was observed between TaqI A (rs1800497) and impulsivity in intertemporal choice. The SNP was originally assigned in DRD2, but was later found to be located within exon 8 of the adjacent gene, ankyrin repeat and kinase domain containing 1 (ANKK1). The protein encoded by ANKK1 gene belongs to the Ser/Thr protein kinase superfamily
Table 1. Results of DRD2 SNP Genotyping.
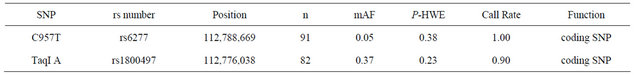
DRD2, Dopamine receptor D2; mAF, minor allelic frequency; P-HWE, P-value of the Hardy-Weinberg equilibrium; SNP, single nucleotide polymorphism.
Table 2. Results of SNP linear regression analysis for the hyperbolic time-discount rate.

The SNP C957T (rs6277) shows a family-wise-corrected significant association with the hyperbolic time-discount rate, ln k (mean); β, regression coefficient; 95% C.I., 95 percent confidence interval of β; R2, coefficient of determination; SNP, single nucleotide polymorphism; PEMP1, empirical P-value on the basis of 100,000 permutations; PEMP2, family-wise (SNP-wise) corrected PEMP1.
involved in signal transduction pathways. Eisenberg et al. [38] reported that major homozygotes of TaqI A showed a nominal significance in their delay discounting task [58] which was similar to the MCQ we used. It might suggest that a certain haplotype including TaqI A in strong linkage disequilibrium (LD) with DRD2 polymorphisms or another unknown variant had an effect on impulsivity in intertemporal choice. In our study, the smaller sample size might have led to a nonsignificant association between TaqI A and impulsivity.
The pallidum is one of sub-cortical structures rich in dopamine and DRD2, and reportedly provides expected reward value signals that are used to facilitate or inhibit motor actions [59]. Therefore, DRD2 polymorphisms might affect the signal levels in the pallidum, which lead to modifying intertemporal choice behaviors. Another possibility is that variations in DRD2 affects impulsivity in intertemporal choice via altered temporal cognition [14].
We now address the limitations of the present study. The first limitation is that the study participants were employees of a major corporation and therefore were not necessarily representative of the general community. Our study participants likely represent a high-functioning segment of the population and the score distribution of Kirby’s MCQ could be different in the general population. The second limitation is that the significance has a small effect size because of the small sample size. A larger sample will be needed to show a large effect size in future molecular neuroeconomic studies.
In conclusion, we have demonstrated that variation in the DRD2 gene is associated with impulsive decisionmaking. By utilizing neuroeconomic theory, future psychiatric studies could investigate the role of the DRD2 gene in impulsive problem behavior observed in addiction and anti-social personality disorder. This is the first study to demonstrate an association between DRD2 and impulsivity in intertemporal choice with a multiplicitycorrected significance.
5. ACKNOWLEDGEMENTS
This study was supported by Grants-in-Aid for Scientific Research (number 17019029 and number 21300242) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
REFERENCES
- Glimcher, P.W. and Rustichini, A. (2004) Neuroeconomics: The consilience of brain and decision. Science, 306, 447-452. doi:10.1126/science.1102566
- Camerer, C., Loewenstein, G.F. and Prelec, D. (2005) Neuroeconomics: How neuroscience can inform economics. Journal of Economic Literature, 43, 9-64. doi:10.1257/0022051053737843
- Takahashi, T. (2009) Theoretical frameworks for neuroeconomics of intertemporal choice. Journal of Neuroscience, Psychology, and Economics, 2, 75-90. doi:10.1037/a0015463
- Hasler, G. (2012) Can the neuroeconomics revolution revolutionize psychiatry? Neuroscience and Biobehavioral Reviews, 36, 64-78. doi:10.1016/j.neubiorev.2011.04.011
- Kable, J.W. and Glimcher, P.W. (2007) The neural correlates of subjective value during intertemporal choice. Nature Neuroscience, 10, 1625-1633. doi:10.1038/nn2007
- McClure, S.M., Laibson, D.I., Loewenstein, G. and Cohen, J.D. (2004) Separate neural systems value immediate and delayed monetary rewards. Science, 306, 503-507. doi:10.1126/science.1100907
- McClure, S.M., Ericson, K.M., Laibson, D.I., Loewenstein, G. and Cohen, J.D. (2007) Time discounting for primary rewards. Journal of Neuroscience, 27, 5796-5804. doi:10.1523/JNEUROSCI.4246-06.2007
- Yu, R. (2012) Regional white matter volumes correlate with delay discounting. PLoS One, 7, e32595. doi:10.1371/journal.pone.0032595
- Mazur, J.E. (1987) An adjusting procedure for studying delayed reinforcement. In: Commons, M. L., Mazur, J. E., Nevin, J. A. and Rachlin, H., Eds., The Effect of Delay and of Intervening Events on Reinforcement Value, Quantitative Analyses of Behavior, Erlbaum, Hillsdale, New Jersey, 55-73.
- Bickel, W.K. and Marsch, L.A. (2001) Toward a behavioral economic understanding of drug dependence: Delay discounting processes. Addiction, 96, 73-86. doi:10.1046/j.1360-0443.2001.961736.x
- Frederick, S., Loewenstein, G. and O’Donoghue, T. (2002) Time discounting and time preference: A critical review. Journal of Economic Literature, 40, 351-401. doi:10.1257/002205102320161311
- Prelec, D. (2004) Decreasing impatience: A criterion for non-stationary time preference and “hyperbolic” discounting. Scandinavian Journal of Economics, 106, 511- 532. doi:10.1111/j.0347-0520.2004.00375.x
- Ainslie, G. (2005) Precis of breakdown of will. Behavioral and Brain Sciences, 28, 635-650. doi:10.1017/S0140525X05000117
- Takahashi, T. (2005) Loss of self-control in intertemporal choice may be attributable to logarithmic time-perception. Medical Hypotheses, 65, 691-693. doi:10.1016/j.mehy.2005.04.040
- Bickel, W.K., Miller, M.L., Yi, R., Kowal, B.P., Lindquist, D.M. and Pitcock, J.A. (2006) Behavioral and neuroeconomics of drug addiction: Competing neural systems and temporal discounting processes. Drug and Alcohol Dependence, 90, S85-S91.
- Reynolds, B. (2006) A review of delay-discounting research with humans: Relations to drug use and gambling. Behavioural Pharmacology, 17, 651-667. doi:10.1097/FBP.0b013e3280115f99
- Petry, N.M. (2002) Discounting of delayed rewards in substance abusers: Relationship to antisocial personality disorder. Psychopharmacology, 162, 425-432. doi:10.1007/s00213-002-1115-1
- Bickel, W.K., Odum, A.L. and Madden, G.J. (1999) Impulsivity and cigarette smoking: Delay discounting in current, never, and ex-smokers. Psychopharmacology, 146, 447-454. doi:10.1007/PL00005490
- Kirby, K.N., Petry, N.M. and Bickel, W.K. (1999) Heroin addicts have higher discount rates for delayed rewards than non-drug-using controls. Journal of Experimental Psychology. General, 128, 78-87. doi:10.1037/0096-3445.128.1.78
- Coffey, S.F., Gudleski, G.D., Saladin, M.E. and Brady, K.T. (2003) Impulsivity and rapid discounting of delayed hypothetical rewards in cocaine-dependent individuals. Experimental and Clinical Psychopharmacology, 11, 18- 25. doi:10.1037/1064-1297.11.1.18
- MacKillop, J., Anderson, E.J., Castelda, B.A., Mattson, R.E. and Donovick, P.J. (2006) Divergent validity of measures of cognitive distortions, impulsivity, and time perspective in pathological gambling. Journal of Gambling Studies, 22, 339-354. doi:10.1007/s10899-006-9021-9
- Petry, N.M., Kirby, K.N. and Kranzler, H.R. (2002) Effects of gender and family history of alcohol dependence on a behavioral task of impulsivity in healthy subjects. Journal of Studies on Alcohol, 63, 83-90.
- Sozou, P.D. (1998) On hyperbolic discounting and uncertain hazard rates. Proceedings of the Royal Society of London. Series B, 265, 2015-2020. doi:10.1098/rspb.1998.0534
- Crean, J., Richards, J.B. and de Wit, H. (2002) Effect of tryptophan depletion on impulsive behavior in men with or without a family history of alcoholism. Behavioural Brain Research, 136, 349-357. doi:10.1016/S0166-4328(02)00132-8
- Reynolds, B., Karraker, K., Horn, K. and Richards, J.B. (2003) Delay and probability discounting as related to different stages of adolescent smoking and non-smoking. Behavioural Processes, 64, 333-344. doi:10.1016/S0376-6357(03)00168-2
- Reynolds, B. and Schiffbauer, R. (2004) Measuring state changes in human delay discounting: An experiential discounting task. Behavioural Processes, 67, 343-356.
- Ohmura, Y., Takahashi, T. and Kitamura, N. (2005) Discounting delayed and probabilistic monetary gains and losses by smokers of cigarettes. Psychopharmacology, 182, 508-515. doi:10.1007/s00213-005-0110-8
- Ohmura, Y., Takahashi, T., Kitamura, N. and Wehr, P. (2006) Three-month stability of delay and probability discounting measures. Experimental and Clinical Psychopharmacology, 14, 318-328. doi:10.1037/1064-1297.14.3.318
- Estle, S.J., Green, L., Myerson, J. and Holt, D.D. (2007) Discounting of monetary and directly consumable rewards. Psychological Science, 18, 58-63. doi:10.1111/j.1467-9280.2007.01849.x
- Takahashi, T., Oono, H. and Radford, M.H.B. (2007) Empirical estimation of consistency parameter in intertemporal choice based on Tsallis’ statistics. Physica A, 381, 338-342. doi:10.1016/j.physa.2007.03.038
- Sellitto, M., Ciaramelli, E. and di Pellegrino, G. (2011) The neurobiology of intertemporal choice: Insight from imaging and lesion studies. Reviews in the Neurosciences, 22, 565-574. doi:10.1515/RNS.2011.046
- Dayan, P. and Abbott, L.F. (2001) Theoretical Neuroscience. MIT Press, Cambridge.
- Schultz, W. (2004) Neural coding of basic reward terms of animal learning theory, game theory, microeconomics and behavioural ecology. Current Opinion in Neurobiology, 14, 139-147. doi:10.1016/j.conb.2004.03.017
- Takahashi, T., Ikeda, K. and Hasegawa, T. (2007) A hyperbolic decay of subjective probability of obtaining delayed rewards. Behavioral and Brain Functions, 25, 3-52.
- Simon, N.W., Montgomery, K.S., Beas, B.S., Mitchell, M.R., LaSarge, C.L., Mendez, I.A., Bañuelos, C., Vokes, C.M., Taylor, A.B., Haberman, R.P., Bizon, J.L. and Setlow, B. (2011) Dopaminergic modulation of risky decision-making. Journal of Neuroscience, 31, 17460-17470. doi:10.1523/JNEUROSCI.3772-11.2011
- Heerey, E.A., Robinson, B.M., McMahon, R.P. and Gold, J.M. (2007) Delay discounting in schizophrenia. Cognitive Neuropsychiatry, 12, 213-221. doi:10.1080/13546800601005900
- Colzato, L.S., van den Wildenberg, W.P., Van der Does, A.J. and Hommel, B. (2010) Genetic markers of striatal dopamine predict individual differences in dysfunctional, but not functional impulsivity. Neuroscience, 170, 782- 788. doi:10.1016/j.neuroscience.2010.07.050
- Eisenberg, D.T.A., MacKillop, J., Modi, M., Beauchemin, J., Dang, D., Lisman, S.A., Lum, J.K. and Wilson, D.S. (2007) Examining impulsivity as an endophenotype using a behavioral approach: A DRD2 TaqI A and DRD4 48-bp VNTR association study. Behavioral and Brain Functions, 3, 2. doi:10.1186/1744-9081-3-2
- Young, R.M., Lawford, B.R., Nutting, A. and Noble, E.P. (2004) Advances in molecular genetics and the prevention and treatment of substance misuse: Implications of association studies of the A1 allele of the D2 dopamine receptor gene. Addictive Behaviours, 29, 1275-1294. doi:10.1016/j.addbeh.2004.06.012
- Sheehan, D.V., Lecrubier, Y., Sheehan, K.H., Amorim, P., Janavs, J., Weiller, E., Hergueta, T., Baker, R. and Dunbar, G.C. (1998) The Mini-International Neuropsychiatric Interview (M.I.N.I.): The development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. Journal of Clinical Psychiatry, 59, 22-57.
- American Psychiatric Association (2000) Diagnostic and statistical manual of mental disorders, 4th Edition, text revision: DSM-IV-TR. American Psychiatric Press, Washington DC.
- Hirsh, J.B., Morisano, D. and Peterson, J.B. (2008) Delay discounting: Interactions between personality and cognitive ability. Journal of Research in Personality, 42, 1646- 1650. doi:10.1016/j.jrp.2008.07.005
- Kirby, K.N. (2009). One-year temporal stability of delaydiscount rates. Psychonomic Bulletin & Review, 16, 457- 462. doi:10.3758/PBR.16.3.457
- Takahashi, T., Sakaguchi, K., Oki, M., Homma, S. and Hasegawa, T. (2006) Testosterone levels and discounting delayed monetary gains and losses in male humans. Neuro Endocrinology Letters, 27, 439-444.
- Takahashi, T., Ikeda, K., Fukushima, H. and Hasegawa, T. (2007) Salivary-alpha amylase levels and hyperbolic discounting in male human. Neuro Endocrinology Letters, 28, 101-104.
- Takahashi, T., Shinada, M., Inukai, K., Tanida, S., Takahashi, C., Mifune, N., Takagishi, H., Horita, Y., Hashimoto, H., Yokota, K., Kameda, T. and Yamagishi, T. (2010) Stress hormones predict hyperbolic time-discount rates six months later in adults. Neuro Endocrinology Letters, 31, 616-621.
- Kawamura, Y., Takahashi, T., Liu, X., Nishida, N., Tokunaga, K., Ukawa, K., Noda, Y., Yoshikawa, A., Shimada, T., Umekage, T. and Sasaki, T. (In Press) DNA polymorphism in the FKBP5 gene affects impulsivity in intertemporal choice. Asia-Pacific Psychiatry. doi:10.1111/appy.12009
- Promega Corporation (2005) Technical manual: Wizard® genomic DNA purification kit: Instructions for use of products A1120, A1123, A1125 and A1620. Promega Corp., Madison.
- Nishida, N., Tanabe, T., Hashido, K., Hirayasu, K., Takasu, M., Suyama, A. and Tokunaga, K. (2005) DigiTag assay for multiplex single nucleotide polymorphism typing with high success rate. Analytical Biochemistry, 346, 281-288. doi:10.1016/j.ab.2005.08.007
- Nishida, N., Tanabe, T., Takasu, M., Suyama, A. and Tokunaga, K. (2007) Further development of multiplex single nucleotide polymorphism typing method, the DigiTag2 assay. Analytical Biochemistry, 364, 78-85. doi:10.1016/j.ab.2007.02.005
- Purcell, S., Neale, B., Todd-Brown, K., Thomas, L., Ferreira, M.A., Bender, D., Maller, J., Sklar, P., de Bakker, P.I., Daly, M.J. and Sham, P.C. (2007) PLINK: A tool set for whole-genome association and populationbased linkage analyses. American Journal of Human Genetics, 81, 559-575. doi:10.1086/519795
- Good, P. (2000) Permutation tests: A practical guide to resampling methods for testing hypothesis. Second Edition, Springer-Verlag, New York.
- Hochberg, Y. and Tamhane, A.C. (1987) Multiple comparison procedures: Probability and mathematical statistics. Wiley-Interscience, New York. doi:10.1002/9780470316672
- Cohen, J. (1988) Statistical power analysis for the behaviorral sciences. Second Edition, Lawrence Erlbaum, Hillsdale.
- Field, A. (2005) Discovering statistics using SPSS. Second Edition, Sage Publications, London.
- Gauderman, W.J. (2002) Sample size requirements for matched case-control studies of gene-environment interaction. Statistics in Medicine, 21, 35-50. doi:10.1002/sim.973
- SPSS Inc. (2007) SPSS base 16.0 user’s guide. SPSS Inc., Chicago.
- DeVona, J. (2005) Delay discounting task. Binghamton, New York.
- Tachibana, Y. and Hikosaka O. (2012) The primate ventral pallidum encodes expected reward value and regulates motor action. Neuron, 76, 826-837. doi:10.1016/j.neuron.2012.09.030
NOTES
*Corresponding author.

