Open Journal of Physical Chemistry
Vol. 3 No. 1 (2013) , Article ID: 27886 , 10 pages DOI:10.4236/ojpc.2013.31005
Influence of Solvent Polarity on the Physio-Chemical Properties and Quantitative Determinations of Tenofovir Disoproxil and Emtricitabine with Chloranilic Acid as Complexing Agent
Department of Pharmaceutical Chemistry, Faculty of Pharmaceutical Sciences, University of Jos, Jos, Nigeria
Email: *ogodaona@yahoo.co.uk
Received September 24, 2012; revised October 25, 2012; accepted November 28, 2012
Keywords: Tenofovir Disoproxil Fumarate; Emtricitabine; Physico-Chemical Studies; Charge-Transfer
ABSTRACT
Purpose: Tenofovir disoproxil fumarate (TEN) and emtricitabine (EMT) are both second generation ant-retroviral drugs used in the “treatment” of HIV/AIDS. The aim of this study is to establish the physic-chemical properties of their reaction with chloranilic acid in different solvent systems and to justify the chemical basis for simultaneous quantitative determination of these drugs in their combined formulation. Method: TEN and EMT were individually isolated from their single formulations and purified by chromatography to obtain secondary standard. Purity of the isolates were tested for by comparison with literature values. Stock solution of chloranilic acid (CA) [3.0 × 10−3 M] was prepared in the following solvents of different polarities: ethanol, acetonitrile, ethylacetate, chloroform and hexane. Equal volumes of CA and TEN [3.0 × 10−2 M] and EMT [3.0 × 10−2 M] dissolved in different solvents were mixed whereby colored products were observed. Absorption maxima were determined. Calibration curves were generated and validated. Quantitative simultaneous determination of TEN and EMT was determined by standard protocol. Stoichiometric relationships between the drugs and CA were established. Equilibrium constants were determined at different temperatures from which the Gibb’s free energies were calculated. Arrhenius equation was used to calculate the enthalpy, entropy was similarly calculated. Results: Absorption maxima of CA in different solvents are as follows: Ethanol 310 nm; Acetonitrile 330 nm; Ethyl acetate 340 nm; Chloroform 350 nm and hexane 310 nm. The complex of CA and TEN in the different solvents are: Alcohol 525 nm, Acetonitrile 500 nm; Ethyl acetate 505 nm; Chloroform 510 nm and hexane 515 nm. For EMT complex absorption maxima are: Alcohol 510 nm; Acetonitrile 515 nm’ Ethyl acetate 520 nm’ Chloroform 505 nm and hexane 530 nm. Simultaneous quantitative recovery values for TEN are: Ethanol; 97.89% ± 1.21%; Acetonitrile 101.17 V 1.51%; Ethyl acetate 96.55% ± 0.71%; Chloroform 99.11% ± 0.34% and hexane 98.03% ± 0.15%. For EMT the values are also: Ethanol: 98.92% ± 1.45%; Acetonitrile 100.471 ± 13; Ethyl acetate 97.06% ± 0.87%; Chloroform 99.31% ± 0.94% and Hexane 99.97% ± 1.63%. Stoichiometry of complexation showed a 1:1 ratio for both drugs. Equilibrium constants for TEN were highest in acetonitrile and least for Ethanol while for EMT, equilibrium constant was least for acetonitrile and highest in chloroform. Gibb’s free energy for TEN was least in ethanol and highest in acetonitrile. Gibb’s free energy for EMT was least in acetonitrile and highest in chloroform. Enthalpy for TEN was least in chloroform and highest in hexane. Similarly, the enthalpy for EMT was highest in chloroform and lowest in hexane. Conclusion: These results shows that solvent polarity influence charge transfer complexes in a non consistent fashion. The structure of the donor might have contributed to thermodynamics of complexation since orbital overlap may vary from solvent to solvent. For quantitative analysis hexane appears to be the most suitable solvent because it has the highest molar absorptivity and higher enthalpy of interactions. Molecules that can donate electrons and their stereochemistry could contribute to intensity of absorption maxima of the electronic transitions.
1. Introduction
In chemistry and indeed in organic chemistry in particular, interactions between molecules are typically described by the covalent or ionic bonding forces. Outside this classical definition are other intermediate forces like dipole-dipole, dipole-induced-dipole, van der waals, dispersive forces that are difficult to represent pictorially. Charge-transfer forces are still considered to be less than these intermediate forces. It’s therefore safe to generalize that all molecules or even matter interact with each other in some way, the majority of which may be considered non-chemical in nature. The non-chemical interaction between any two molecules may give rise to complexes of a specialized type. The most appropriate example of non-chemical complex involves the weak electronic interaction between donors and acceptors through their occupied and unoccupied orbitals [HOMO to LUMO] respectively.
These donors and acceptors are classified as either π- or σ-donors or acceptors respectively. It has been observed in many instances that these complexes exhibit electronic absorption spectra induced as a photochemical process. Mulliken [1] described these complexes as Charge-transfer complexes (CTC), a description that has received minimal criticism. The theoretical framework for donor-acceptor interaction was earlier elaborated by Weiss [2]. The molecular orbital explanation has also been offered in which an electron from a filled orbital in the donor is transferred to an empty orbital in the acceptor.
Although the concept of donor-acceptor species has been accepted, the terminology is often used with relativity because examples of single specie behaving both as donor and acceptor exist as observed with benzene and polycyclic aromatic hydrocarbons.
Evidence for the actual existence of CTC has not yet been established even though Dewar and Thompson [3] postulated that the existence of CT spectrum is not sufficient evidence for the real existence of any kind of complex between the pair. On the contrary, the hypsochromic or bathochromic shifts commonly observed in CT spectra can only be ascribed as evidence for the existence of the complex. This assumption has enabled several experiments including nuclear magnetic resonance (NMR) to be carried out to determine the thermodynamic properties of such complexes [4].
According to the current understanding, CTC generally involve integral ratio of the components except perhaps iodine complexes. The equilibrium between the donor and acceptor is usually so fast that the interaction might be considered spontaneous by classical methods of measurements. Absorptions due to complexes between donors and acceptors might still retain the absorptions due to the component molecules; but because the majority of these transitions occur at a longer wavelength and even in the visible region, absorptions due to the complex can readily be identified. The intensity of absorption band of a complex is usually determined by the molar absorptivity at the absorption maximum although intensity of absorption may be affected by dissociation of the complex when in solution. A considerable number of publications have appeared in which CTC was used as a method of analysis but systematic approach of investigating the influence of the solvents have not been so commonly investigated, hence this study.
CTC complexes have applications in Pharmaceutical analysis [5], molecular weight determinations, chromatographic separations, enantiomeric resolutions and determinations of polarity and ionization potentials of solvents.
Chemically TEN is 9-[(R)-2[[bis [[(isopropoxycarbonyl) oxy]-methoxy]phosphinyl]methoxy]propyl]adenine fumarate and EMT is 4-amino-5-fluoro-1-(2-hydroxymethyl)-1,3-oxathiolan-5-yl)-2-(1H-pyrimidone. The combination is marketed as Truvada and therefore active against subtypes 1 and 2 of HIV. TEN is a prodrug that becomes activated de novo. The pharmacokinetic studies of these drugs reveal that their bioavailability is considerably enhanced when administered with a high fat meal. Several reports have appeared on charge transfer complexes with many drugs and other organic molecules but as yet the authors have not seen any report on Tenofovir disoproxate and emtricitabine specifically. Official monographs have not carried any recommended methods of analysis hence our desire to contribute by applying charge transfer complexes using chloranilic acid acceptor.
2. Experimental
2.1. Materials
The anti retroviral drugs tenofovir (TEN) and emtricitabine (EMT) in pharmaceutical dosage forms were kindly donated by Professor JA Idoko, Chief Medical Officer, AIDS Prevention in Nigeria (APIN) clinic, University of Jos, Nigeria. The following analytical grade solvents (AnalaR: BDH) were purchased from local suppliers and were redistilled severally and further dried with anhydrous sodium sulphate: Ethanol, Acetonitrile, Ethyl acetate, Chloroform and Hexane. Chloranilic acid (Reidel de Haem, Germany) was recrystallized in acetone and dried to consistent weight. Similarly anhydrous sodium sulphate (May & Baker) was dried to a consistent weight. All other reagents were of AnalaR grade and were used without further purification.
Other materials used include melting point apparatus, thin film evaporator and glassware.
2.2. Methology
2.2.1. Extraction of the Active Drugs from Their Excipients
Tablets of tenofovir disoproxil fumarate (TEN) (10 no) containing 200 mg each were crushed in a mortar to powdered form and transferred into a beaker containing 100 mL of 96% ethanol; then transferred onto a magnetic stirrer hot plate with constant stirring for at least 20 min. The mixture was filtered using Whatman No. 1 paper. The process of extraction was repeated three more times (3 × 100 ml) .The filtrate was pooled and neutralized with sodium hydroxide solution to remove the fumarate component. The solution was further partitioned with chloroform (100 ml × 3). The chloroform fraction was evaporated to dryness under reduced pressure and the dried powder so obtained was purified by chromatography. Identity tests were carried out as described in the British Pharmacopoeia before the sample was kept for analysis .The process described above was repeated for emtricitabine (EMT), which contain 300 mg. The extracted powder was again purified by chromatography and identity tests similarly carried out and kept for the analyses. Purities of these drugs were further confirmed by melting point determinations and comparison with literature values.
2.2.2. Determination of Absorption Maxima for Chloranilic Acid and Its Complexes with Tenofovir Disoproxil and Emtricitabine
This procedure was adapted from an earlier report [6]. Stock solution of chloranilic acid (1%) was first prepared in 1, 4-dioxan from which 1.0 ml was transferred to test-tubes containing 5.0 mL of different solvents under investigation (ethanol, acetonitrile, ethyl acetate, hexane and chloroform). The volumes were made up to mark (10 ml volumetric flask) with different solvents and then scanned in a uv/visible spectrophotometer (Jenway, Model 6405, England). Solutions of purified tenofovir and emtricitabine (3.0 × 10−2 M each) in the different solvents were mixed in equal volumes with chloranilic acid (3.0 × 10−3 M) in 10.0 ml volumetric flasks and volume finally brought to mark with the appropriate solvent. The colors were formed spontaneously and absorbance monitored over a period of 60 minutes. It was observed that color intensity was optimal at about 30 minutes and so subsequent measurements were made after 30 minutes.
2.2.3. Construction of Calibration Curve, Its Validation and Determination of Extinction Coefficient
Tenofovir disoproxil and emtricitabine (3.0 × 10−2 M each) solutions were prepared and serial volumes (1 ml, 2 ml, to 5 ml) were quantitatively transferred to volumetric flasks in quadruplets. Chloranilic acid (1.0 ml) was added to each flask and the volumes made up to mark with appropriate solvent, then allowed to stand for at least 30 minutes before the absorbances were determined spectrophotometrically. The means of the absorbances were determined and plotted against the concentrations; the regression equation, coefficient of variation and other parameters were determined by Microsoft Excel software. Validation of these values was carried out by repeating the above procedure but with different concentrations from which errors in slope and intercept were calculated. The overall equation was used to test correspondences between absorbances and concentrations. Molar absorptivity and Sandells’ sensitivity were similarly calculated from these data.
2.2.4. Quantitative Simultaneous Determination of Tenofovir Disoproxate and Emticitabine Mixture
Ten different batches of tenofovir disoproxate and emtricitabine combination in a standard formulation or dosage form were investigated. The samples were crushed and extracted as described in Section 2.2.1, standard solutions were prepared and scanned to obtain absorption maxima for the combination. From stock solutions different concentrations in quadruplets were prepared and absorbances recorded simultaneously at the respective absorption maxima for the particular drug. Concentrations of the respective drugs were calculated by substituting into the general equation generated from the validation curves. Fresh concentrations of the drugs were again prepared and spiked with the secondary standard derived from the purification processes. Absorbances were again recorded and the derived recovery calculated. The entire procedure was repeated for each solvent employed in this study.
2.2.5. Determination of Stoichiometric Relationship in the Complexations [The Job’s Continuous Variation Plots]
Equimolar amounts of tenofovir disoproxil and emtricitabine, 3.0 × 10−2 M each, were mixed with chloranilic acid in the following ratios in 10 ml volumetric flasks: 1:9, 2:8, 3:7, 4:6, 5:5 up to 9:1. Colored complexes were formed spontaneously and allowed to stand for 30 min after which the absorbances were read on the spectrophotometer at appropriate wavelength indicated for the different solvents. The plot of absorbance against the concentration ratios [conc. of donor/conc. of donor + conc. acceptor] gives the Job’s continuous variation plot.
2.2.6. Equilibrium Constant and Free Energy Change Determinations
The dynamic changes that take place between tenofovir disoproxil and emtricitabine, both donors, and chloranilic acid (acceptor) was determined by the method earlier reported [7]. Equal concentrations of the donors (3.0 × 10−2 M) were prepared in the appropriate solvent from which 1 mL to 6 ml were transferred to different volumetric flasks in quadruplets followed by the addition of 1 ml chloranilic acid (3.0 × 10−3 M). The contents were mixed thoroughly and the volumes brought up to mark with appropriate solvent. Absorbances were recorded at the relevant absorption maximum at ambient temperature after 30 minutes. The entire procedure was repeated at 20˚C, 30˚C, 40˚C, 50˚C and 60˚C.
3. Results
The absorption maximum for chloranilic acid (CA) in the individual solvents are as follows: ethanol 310 nm; acetonitrile 330 nm; ethyl acetate 340 nm; hexane 310 nm and chloroform 350 nm. In emtricitabine the values are: ethanol 310 nm; acetonitrile 315 nm; ethyl acetate 320 nm; hexane 330 nm and chloroform 305 nm. The intensity of absorption is highest in ethanol followed by acetonitrile then ethyl acetate. The intensity of absorption was least in hexane (Figures 1 and 2). In the presence of tenoforvir (TEN) or emtricitabine (EMT), there was a bathochromic shift in the absorption maximum for each solvent (Table 1). These drugs show slightly different absorption maxima in the solvents.
The quantitative relationships between the concentration of the drugs and absorbance were linear and so were quantitatively validated by standard protocol. On the
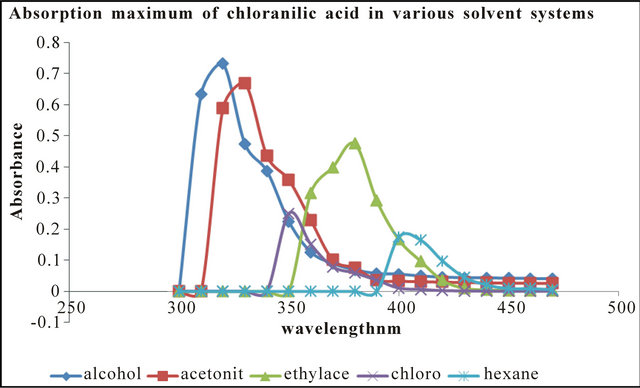
Figure 1. Absorption maxima of chloranilic acid in various solvent systems.

Figure 2. Absorption profile of TEN-CA complex in different solvent media.
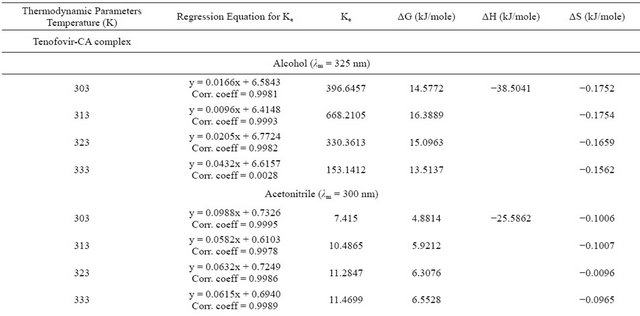
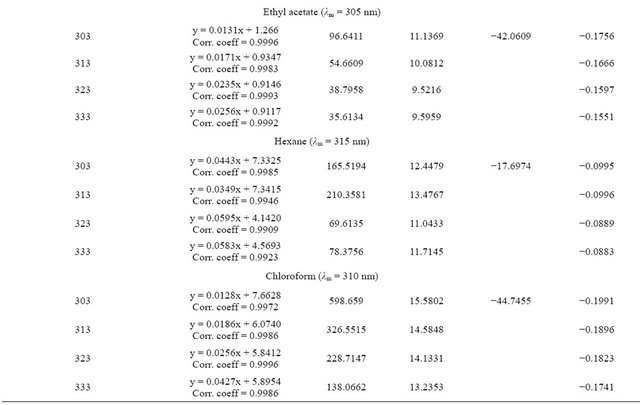
basis of linearity, the limit of detection (LOD) and limit of quantitation (LOQ) were determined and were found to range from 1.0 to 2.0 µg/mL and 5 to 25 µg/mL respectively (Tables 1 and 2). The regression equations for linearity tests are also shown in Tables 1 and 2 with corresponding standard deviations in their slopes and intercepts. The molar absorptivity of tenofovir range from 3104 l·mol−1∙cm−1 (ethyl acetate) to 11337 l·mol−1∙cm−1 (hexane) while that for emtricitabine range from 2851.5 l·mol−1∙cm−1 (ethyl acetate) to 9444.56 l·mol−1∙cm−1 (chloroform) contained in Tables 1 and 2.
The results for quantitative recovery in the different solvents are shown in Tables 1 and 2. We conclude that any of these solvents can theoretically be appropriate for quantitative assay in quality assurance but to reduce ionization effect and possible solvation effects hexane and chloroform should be preferred more especially because they have the highest molar absorptivities.
The stoichiometric ratio between CA and TEN and EMT complexes were both investigated by Job’s continuous variation plot and confirmed by molar ration experiments (not included). The attributes of Jobs plots are illustrated in Figures 1 and 2 indicating a molar ratio of 1:1. It can be observed from Figure 2 that although molar ration is considered 1:1, the plots show flexes that suggest more than one absorption maximum if this scale can be expanded further.
Based on the assumed stoichiometry of 1:1, we proceeded to determine the equilibrium constants and other thermodynamic parameters of the complexation employing the theoretical formulation of Benesi-Hidelbrand [8] reproduced as follows:
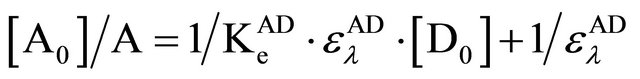 (1)
(1)
Here [Ao] and [Do] are initial concentrations of the donor and acceptor species. A is absorbance of the complex at λm while 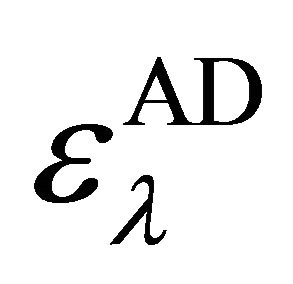 is the molar absorptivity,
is the molar absorptivity, 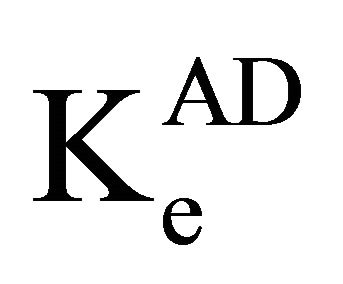 is the equilibrium constant. From Equation 1, the plot of [Ao]/A against 1/[Do] yielded linear graphs from which
is the equilibrium constant. From Equation 1, the plot of [Ao]/A against 1/[Do] yielded linear graphs from which 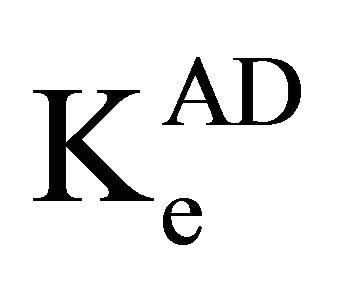 and
and 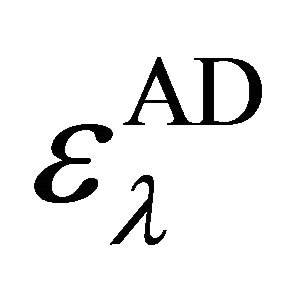 were calculated from slope and intercept respectively. Tables 3 and 4 show details of regression equations for the plots of Ke at different temperatures. The other physicochemical properties like ΔG, ΔH and ΔS under different solvent systems are also indicated. ΔG was calculated from the equation
were calculated from slope and intercept respectively. Tables 3 and 4 show details of regression equations for the plots of Ke at different temperatures. The other physicochemical properties like ΔG, ΔH and ΔS under different solvent systems are also indicated. ΔG was calculated from the equation
 (2)
(2)
while ΔH was calculated from Arrhenius equation, i.e.
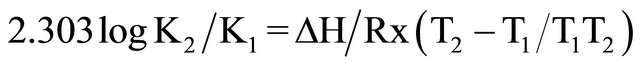 (3)
(3)
Generally, the equilibrium constants determined in these investigations did not follow a regular decline with temperature. The equilibrium constants for TEN in ethanol and chloroform appear to be of the same magnitude
Table 1. Summary of spectral properties of tenofovir disoproxil/CA in different solvent systems.
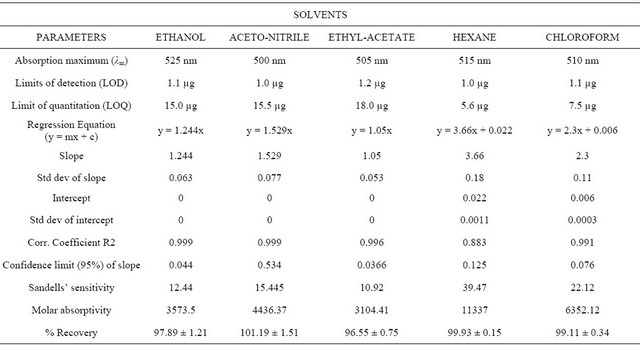
Table 2. Summary of spectral properties of emtricitabine/CA in different solvent systems.

(Tables 3 and 4) but lower than observed with other solvents. The highest equilibrium constants were recorded for acetonitrile that is over 300× over that of ethanol and 260× higher than in chloroform.
For EMT, the equilibrium constant was least for acetonitrile followed by ethyl acetate and highest for ethanol. The influence of solvent on equilibrium constant did not follow any specific pattern although a decline of Ke with increase in temperature was more noticeable than observed for TEN.
The Gibb’s free energy ΔG of complexation calculated for TEN in ethanol and chloroform were of the same magnitude while that in acetonitrile and ethyl acetate were also of the same magnitude. The figures for hexane
Table 3. Summary of thermodynamic properties for tenofovir disoproxil/CA complex at different temperatures and solvent systems.
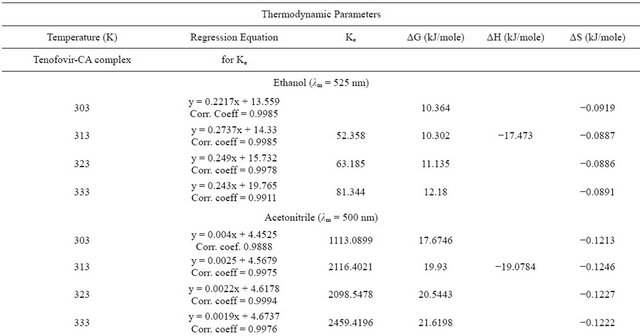
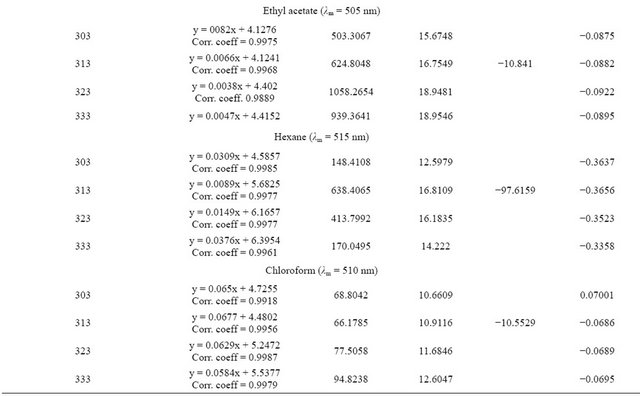
were of intermediate value. For EMT, the ΔG was least for acetonitrile followed by ethyl acetate whereas the figures for ethanol and chloroform are of the same magnitude. The enthalpy of complexation of TEN in hexane was exceptionally high being almost 10× higher than in other solvents. Generally the enthalpies measured for TEN were lower than those for EMT. In fact the highest enthalpy for EMT was obtained in chloroform. The entropies for TEN and EMT in all the solvents show similarity in magnitude.
Table 4. Summary of thermodynamic properties for emtricitabine/CA complex at different temperatures and solvent systems.
4. Discussion
Charge transfer complexes (CTC) between donors and acceptors, particularly of the type π to π usually give rise to intense colors in solutions. The electronic transition that gives rise to these colors have been explained by molecular orbital theory as involving transfer of electron(s) between the highest occupied molecular orbital (HOMO) of the donor and the lowest unoccupied molecular orbital (LUMO) of the acceptor. Color formation is the only evidence of charge-transfer interaction. The intensities of colors are greatly influenced by the nature of the solvent and the energy difference between HOMO and LUMO as demonstrated in this investigation. The absorption maxima for CA in these solvents and their intensities clearly shows that solvents play a crucial part in the electronic transitions as some of the solvents are either σ- (as with chloroform) or π- donors (as with acetonitrile). The non-bonding electrons in ethyl acetate and ethanol did not seem to contribute to the intensity of absorption. The intense purple colors that characterize the interactions of the drugs with chloranilic acid suggest that if complexation were not complete, as evidenced by equilibrium constants, the original color due to chloranilic acid alone would still be present in the mixture. Because the bathochromic shift is large in these interacttions, interference would absolutely be absent at the wavelength of measurement.
In this investigation, intensity of absorption for tenofovir was highest in chloroform, ethanol, then acetonitrile followed by ethyl acetate and least in hexane. Similar trends were observed for emtricitabine. The enhanced intensity in chloroform might be ascribed to stabilizing effect of the solvent molecules due to σ-donor property while the presence of electron pairs in alcohol and acetonitrile similarly stabilized the complex formed by hydrogen bonding. Hexane molecules don’t have comparable stabilizing effect, hence the reduced intensity. It might be reasonable to suppose that in the equilibrium condition in hexane, solvent molecules favor dissociation of the CT complex hence its low intensity. The intensity of absorption in ethyl acetate is intermediate between hexane and acetonitrile. The structural features of the absorption curves for tenofovir in chloroform and ethanol show flexes that suggest multiple peaks. This could be explained as due to local excitations within the CT complex structure and possible ionization/hydrogen bonding within the complex. The absorption curves for emtricitabine show smooth curves without such flexes. This difference may be due to the structure of the molecules and the effectiveness of the overlapping orbitals in the complex.
Very few reports are available correlating the influence of solvents and their dielectric constants on electronic excitation energies and by extension absorption maxima. In this study, the order of absorption maxima for tenofovir in relation to dielectric constant is as follows: hexane, 515 nm; chloroform, 510 nm; ethyl acetate, 505 nm; acetonitrile, 500 nm and ethanol, 525 nm. Except for ethanol there appears to be slight decline in absorption maxima with decrease in dielectric constant. For emtricitabine, it’s as follows: hexane 530 nm; chloroform 505 nm; ethyl acetate 520 nm; acetonitrile 515 nm and ethanol 510 nm. The differences between absorption maxima for emtricitabine might be due to the smaller size of the molecule.
Molar absorptivity usually measures the intensity of absorption band of the species involved therefore hexane and chloroform exhibit a more favorable property for quantitative analysis.
The Job’s continuous variation plots (Figures 3 and 4) confirms that stoichiometry of the CTC are in a ratio of 1:1 confirming that ionization, ter-molecular complexes, solvation or hydrogen bonding inter-reaction did not appear to take place in these solvents to any significant extent. Similar stoichiometry has been reported severally in the literature [6,8-11].
On the basis of these stoichiometries for tenofovir and emtricitabine, Benesi-Hildebrand formulation [12] was adapted to determine the physic-chemical properties i.e. equilibrium constants, Gibbs free energy change, enthalpy and entropy, of these complexes in different solvents. The figures are presented in Tables 3 and 4.
From literature data [5,13,14], the equilibrium constant
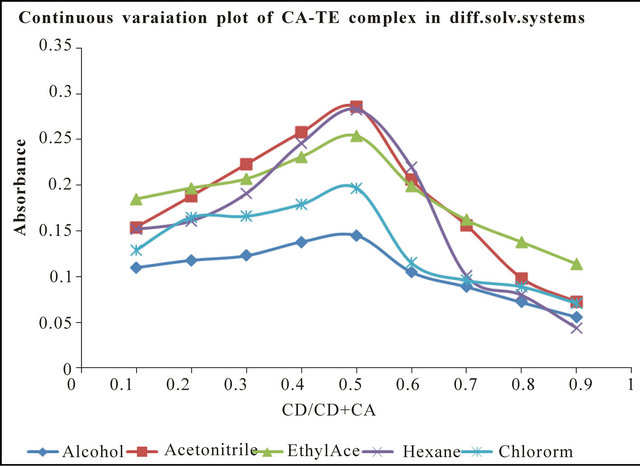
Figure 3. Jobs continuous variation plot for TEN-CA complex in different solvent systems.
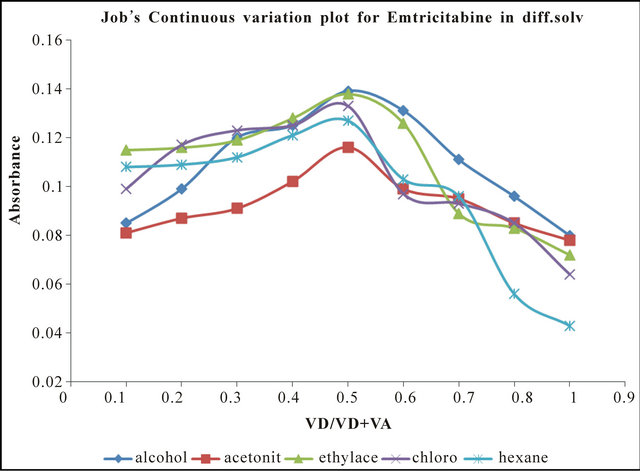
Figure 4. Jobs continuous variation plot for EMT-CA complex in different solvent systems.
or association constant is seriously influenced by the nature of the solvent employed. Equilibrium constant is a measure of stability of the CTC formed. The equilibrium constant Ke in this report was carried out by graphical method and regression equations are shown in Tables 3 and 4. Our observations presented in this report shows similar behavior except that no consistent pattern has been established on the basis of polarity. Tenofovir and chloranilic acid shows equilibrium constant (Tables 3 and 4) in the following order: acetonitrile > ethyl acetate > hexane > chloroform and > ethanol. For emtricitabine, the order is chloroform > ethanol > hexane > ethyl acetate > acetonitrile.
These inconsistencies can be explained on the basis of the structure of the molecules since CTC formation depends on the energy barrier between the donor and acceptor orbitals. For example, tenofovir has phosphonate groups that can interact with solvent molecules and also stabilize the donor-acceptor complex. It is also conceivable that there could be competition between the solvent molecules and the donor or acceptor molecules leading to enhance stability. Also other mechanistic influences, such as hydrogen bonding as with ethanol, could contribute to stability as observed for emtricitabine.
On the basis of stoichiometry, we’re tempted to conclude that solvent effects are truly minimal more so when the solvents were thoroughly dehydrated prior to these measurement. Another factor that influences the value and position of equilibrium constant is temperature. From theoretical considerations, it’s expected that equilibrium values should decrease with increase in temperature. Since our study covered temperatures of 293 K to 333 K, we observed inconsistencies in the order of equilibrium values (Tables 3 and 4). We can only speculate that increases in temperature might have affected the structure of solvent molecules.
It has been reported [5] that equilibrium constant values obtained by optical method from which the BenesiHildebrand equation is derived are characterized by large errors. Inspire of this limitation, the technique has wide appeal because of its adaptability. Indeed various equations have been developed from the original BenesiHildebrand equation. More recent reports in which nuclear magnetic resonance chemical shift methods are employed give more accurate values of equilibrium constants [14].
Most determinations of ΔG, ΔH and ΔS are based on the determination of equilibrium constant Ke by spectrophotometric methods which inherently carries along the error from Ke values even when extreme care had been taken during experimentation. Our observation that the Gibbs free energy for tenofovir and emtricitabine are within the range of 10 KJ/mol and 20 KJ/mol suggest that the degree of overlap within the CTC supra-structure are the same and might have experienced the same environmental factors. These results are also in agreement with earlier reports [7,15,16].
The enthalpies for these complexities were carried out by both graphical methods where log Ke was plotted against 1/T and by Arrhenius equation. The Arrhenius equation gave a better estimate of ΔH, therefore the graphical method was discarded in this presentation. The enthalpy ΔH, of formation of the complex are lower for tenofovir than for emtricitabine (Tables 3 and 4) suggesting that emtricitabinechloranilic acid complex is more stable than that for tenofovir. The architecture of the molecules probably explains this difference.
5. Conclusion
There is no dramatic difference between the solvents on the physic-chemical properties of Tenofovirand emtricitabinechloranilic acid complexes. Our study reveals however that hexane and chloroform are better solvents as medium for charge-transfer complex formation as these solvents gave very high reproducibility and molar absorbtivity during the simultaneous quantitative analysis.
6. Acknowledgements
The Authors are grateful to the Department of Pharmaceutical Chemistry for material assistance.
REFERENCES
- R. S. Mulliken, “Molecular Complexes,” Journal of the American Chemical Society, Vol. 74, No. 3, 1952, pp. 811-813. doi:10.1021/ja01123a067
- J. Weiss, “The Formation and Structure of Some Organic Compounds,” Journal of the Chemical Society, Vol. 65, 1942, pp. 245-247. doi:10.1039/jr9420000245
- M. J. S. Dewar and C. C. Thompson, “Pi-Molecular Complexes III. A Critique of Charge-Transfer and Stability Constants for Some Tetracynoethylene-Hydrocarbon Complexes,” Tetrahedron, Vol. 22, Suppl. 7, 1966, pp. 97-114. doi:10.1016/S0040-4020(01)99099-4
- S. Moon and Park, “Charge-Transfer Complexes of Photochemically Interesting Organic Systems,” Texas Technical University, 1972, pp. 39-79.
- R. Foster, “Organic Transfer Complexes,” Academic Press, London, 1969.
- J. O. Onah and U. Ajima, “Quantitative Analysis of Tenofovir by Titrimetry, Extractive Ion-Pair Spectrophotometry and Charge-Transfer Complexation Methods,” Tropical Journal of Pharmaceutical Research, Vol. 10, No. 1, 2011, pp. 89-96.
- M. U. Adikwu, K. C. Ofokansi and A. A. Attama, “Thermodynamic Studies of the Charge-Transfer Interaction of Chloranilic Acid with Moclobemide and Promethazine Hydrochloride,” Biological & Pharmaceutical Bulletin, Vol. 21, No. 12, 1998, pp. 1243-1246. doi:10.1248/bpb.21.1243
- M. M. Ayad, S. F. Belal, M. S. El Adl and A. A. Al Kheir, “Spectrophotometric Determination of Some Corticosteroid Drugs through Charge-Transfer Complexation,” Analyst, Vol. 109, No. 11, 1984, pp. 1417-1422. doi:10.1039/an9840901417
- F. A. Ibrahim, M. S. Rizk and F. Belal, “A Spectrophotometric Method for the Determination of Some Pharmaceutically Important Hydrazines and Pyrozoline Derivatives,” Analyst, Vol. 111, No. 11, 1986, pp. 1285-1288. doi:10.1039/an9861101285
- M. U. Adikwu and K. C. Ofokansi, “Spectrophotometric Determination of Moclobemide by Charge-Transfer Complexation,” Journal of Pharmaceutical and Biomedical Analysis, Vol. 16, No. 3, 1997, pp. 529-532. doi:10.1016/S0731-7085(97)00086-1
- J. O. Onah and J. E. Odeiani, “Physico-Chemical Studies on the Charge-Transfer Complex Formed between Sulphadoxine and Pyrimethamine with Chloranilic Acid,” Journal of Pharmaceutical and Biomedical Analysis, Vol. 29, No. 4, 2002, pp. 639-647. doi:10.1016/S0731-7085(02)00102-4
- B. A. Benesi and J. Hildebrand, “Ultraviolet Absorption Bands of Iodine and Aromatic Hydrocarbons,” Journal of the American Chemical Society, Vol. 71, No. 8, 1949, pp. 1832-1833. doi:10.1021/ja01176a030
- J. Grundnes, S. D. Christian, V. Cheam and S. B. Farnham, “Solvent Effects of Strong Charge-Transfer Complexes IV. Trimethylamine and Sulphur Dioxide in the Vapour Phase,” Journal of the American Chemical Society, Vol. 93, No. 1, 1971, pp. 20-23. doi:10.1021/ja00730a003
- M. S. Subhani, N. K. Bhatti, M. Mohammad and A. Y. Khan, “Spectrophotometric Studies of Charge-Transfer Complexes of 2,3-Dichloro-5.6-dcyano-pbenzoquinone,” Turkish Journal of Chemistry, Vol. 24, 2000, pp. 223- 230.
- J. O. Onah, “Thermodynamic and Analytical Studies on the Charge-Transfer Complex Formed between Chloranilic Acid and Derivative of Metronidazole,” Global Journal of Pure and Applied Sciences, Vol. 10, 2004, pp. 125-131.
- L. A. Walker, S. Pullen, B. Donovan and J. Sension, “On the Structure of Iodine Charge-Transfer Complexes in Solution,” The Journal of Physical Chemistry Letters, Vol. 242, No. 1-2, 2000, pp. 177-183.
NOTES
*Corresponding author.

