Paper Menu >>
Journal Menu >>
 Open Journal of Physical Chemistry, 2011, 1, 6-10 doi:10.4236/ojpc.2011.11002 Published Online May 2011 (http://www.SciRP.org/journal/ojpc) Copyright © 2011 SciRes. OJPC Hydrothermal Synthesis and Properties of Diluted Magnetic Semiconductor Zn1-xMnxO Nanowires X. Y. Zhang1*, J. Y. Dai2, H. C. Ong3 1Department of C hemic al Eng i neering, Mo na sh U ni versi t y, Melbourne, Au st ral ia 2Department of Appl i ed Physics, Hong Kong Pol y t echnic Uni versi t y , Kowloo n, H on g K o n g, C hi n a 3Department of Physics, Chinese University of Hong Kong , Shatin, Hong Kong, China E-mail: 0Txinyi.zhang@monash.edu.au Received April 7th, 2011; revised April 28th, 2011; accepted May 11th, 2011. Abstract We report the synthesis of oriented single crystalline Mn doped ZnO nanowires through a hydrothermal method. Structural characterizations using X-ray diffraction and transmission electron microscopy revealed that the Mn was doped into the lattice structure, forming solid solution. The Mn doped ZnO nanowires pos- sess wurtzite structure with a c-axis growth orientation. The physical properties of the nanowires were inves- tigated. Mn doped ZnO nanowires were found to be ferromagnetic with Curie temperature of about 30 K. A deep level emission band at about 566 nm was observed at room temperature. Keywords: Semiconductors, Nanowire, Crystal Growth, Electron Microscopy, Curie Temperature, Photolu- minescence 1. Introduction One-dimensional nanostructures have attracted a great deal of interest not only because of their basic scientific richness, but also of their potential utilization in optical and electronic devices [1-6]. ZnO is one of the most im- portant semiconductors due to its wide direct band gap (3.37 eV) and large exciton binding energy (60 meV). To date, ZnO in bulk and thin film format has been widely used in field emission display, solar cell, chemical sensors, and other devices [7-9]. Recently, dilute magnetic semi- conductors (DMSs) have attracted a great deal of atten- tion in the past few years as enabling materials in the emerging field of “spintronics”. DMSs are semiconductor solid solutions, where a small percentage of cations are replaced by magnetic impurities such as Mn. Unusual magnetotransport and magnetooptical phenomena like large Faraday rotations, giant negative magnetoresis- tances, and magnetic field induced metalinsulator transi- tions have been observed. Among the DMSs, Mn- doped II-VI compounds have been extensively studied. However, most of the II-VI compounds studied are chalcogenides; the corresponding oxides are comparatively less investi- gated. Theoretical calculations predict that ZnO should exhibit ferromagnetism above room temperature on dop- ing with Mn [10,11]. This prediction has initiated in- creasing efforts on Mn doping of ZnO nanostructures. Diluted magnetic semiconductor (DMS) behavior has been observed in many transition-metal doped ZnO films [12-14], and Curie temperatures above room temperature were achieved [15,16]. Although the efforts aimed at un- derstanding the origin of the ferromagnetism resulted in hundreds of papers, substantial debate still remains. The available data cannot further advance our understanding of the mechanism since the data obtained so far on Mn doped ZnO are not quite consistent, for example, besides the ferromagnetic behavior, behavior of spin glass [17] and paramagnet [18] have also been observed. Therefore, more detailed works are essential to understand the be- havior of these materials. Mn doped ZnO nanowires (or nanobelts) have been obtained by doping Mn into ZnO by using various techniques, such as ion implantation [19], thermal evaporation [20-23]. These methods generally require high temperature and expensive equipments. On the other hand, wet chemical method has been proven to be a simple and versatile approach for preparing ZnO nanowires or nanorods due to its relatively low growth temperature and good potential for mass production [24- 27]. However, to the best of our knowledge, there is no report on the synthesis of Mn doped ZnO nanowires by using wet chemical method. Here, we report a simple hydrothermal method to prepare the Mn doped ZnO  X. Y. ZHANG ET AL. Copyright © 2011 SciRes. OJPC 7 nanowires. The structure and light-emitting properties are also investigated. 2. Experimental Section Materials. Zinc sulfate hydrate (ZnSO4·7H 2O), manga- nese sulfate hydrate (MnSO4·H2O), carbamide (CON2H4), sodium hydroxide (NaOH), and ethanol were of analytical grade and used without further purification. Dou- ble-distilled water was employed for all experiments. Synthesis. In a typical synthesis of Mn dopped ZnO nanowires, a solution of 50 ml 0.1M ZnSO4, 3 ml 0.1M MnSO4, 15 ml 1M carbamide (CON2H4) and 10 ml C2H5OH was placed in a sealed Teflon autoclave with the pH of the solution adjusted to 14 by NaOH. The so- lution was then heated at 180℃ for 12 hours and the re- sulting products were obtained by washing and drying the yellowish precipitates at 60℃ for 12 hours. Characterization. The microstructure and composi- tion of the nanowires were investigated using an X-ray diffractometer (XRD, PW1140/90) with Cu K radiation (25 mA and 40 kV), a scanning electron microscope (SEM, JEOL JSM-6300), transmission electron micro- scope (TEM, JEOL-2010), energy dispersive spectros- copy (EDS), and electron energy-loss spectroscopy (EELS). Quantitative EDS analysis shows the molar ratio of Mn is around 3%. The magnetic properties of the nanowires were studied using a Superconducting Quantum Interference Device (SQUID) magnetometer (MPMS- 5S). The photoluminescence (PL) measurements were performed by using a Kimmon HeCd laser (325 nm, 55 mW) in a closed cycle Oxford cryostat. The optical sig- nal was dispersed by a 0.25 m Oriel spectrometer and captured by an Andor CCD detector. 3. Results and Discussion Figure 1 shows the XRD spectrum of the Zn1-xMnxO nanowires, the reflection peaks of hexagonal structured ZnO are distinguishable, revealing that the doping of Mn does not change the wurtzite structure of ZnO. No Mn- related impurity phases were observed. Figure 2 (a) and (b) show the typical SEM and TEM images of the Zn1-xMnxO nanowires, respectively. The nanowires have uniform diameters along their entire length, the diame- ters and lengths are in the range of 10 to100 nm and 2 to 10 m, respectively. Figure 3(a) shows TEM image of a typical long Zn1-xMnxO nanowire, the right inset shows the selected-area electron diffraction (SAED) pattern taken from a single nanowire, the analysis of the SAED reveals that the Zn1-xMnxO nanowire is single crystalline and grows along the [0001] orientation. The presence of Mn in the lattice was confirmed by the EELS measure- ment, the spectrum shows that Zn and Mn coexist along with O (Figure 3(b)). The Mn-L3/L2 intensity ratio in- dicates one electron in the d-orbital suggesting Mn to be in the +2 oxidation state. The nature of single-crystal structure of the Zn1-xMnxO nanowires and the growth orientation were further confirmed by high-resolution TEM (HRTEM) image as shown in Figure 4. The HRTEM images analysis shows that the Zn1-xMnxO nanowires are structurally uniform without any signify- 30 40 50 60 70 201 112 103 110 102 101 002 100 Intensity (a.u.) 2 theta (degree) Figure 1. The XRD spectrum of the Zn1-xMnxO nanowires. Figure 2. (a) SEM; (b) TEM images of Zn1-xMnxO nanowires. 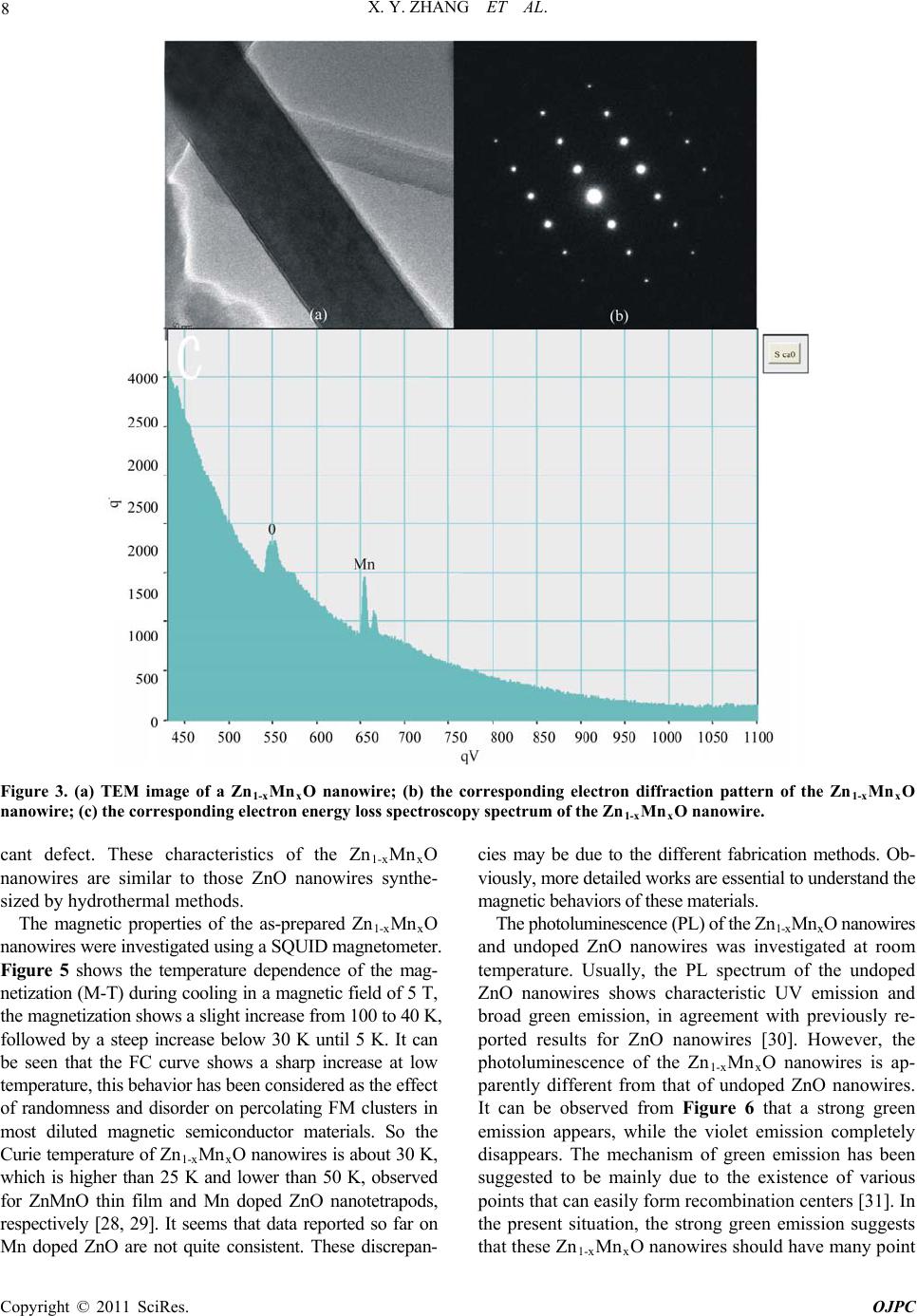 X. Y. ZHANG ET AL. Copyright © 2011 SciRes. OJPC 8 Figure 3. (a) TEM image of a Zn1-xMnxO nanowire; (b) the corresponding electron diffraction pattern of the Zn1-xMnxO nanowire; (c) the corresponding electron energy loss spectroscopy spectrum of the Zn1-xMnxO nanowire. cant defect. These characteristics of the Zn1-xMnxO nanowires are similar to those ZnO nanowires synthe- sized by hydrothermal methods. The magnetic properties of the as-prepared Zn1-xMnxO nanowires were investigated using a SQUID magnetometer. Figure 5 shows the temperature dependence of the mag- netization (M-T) during cooling in a magnetic field of 5 T, the magnetization shows a slight increase from 100 to 40 K, followed by a steep increase below 30 K until 5 K. It can be seen that the FC curve shows a sharp increase at low temperature, this behavior has been considered as the effect of randomness and disorder on percolating FM clusters in most diluted magnetic semiconductor materials. So the Curie temperature of Zn1-xMnxO nanowires is about 30 K, which is higher than 25 K and lower than 50 K, observed for ZnMnO thin film and Mn doped ZnO nanotetrapods, respectively [28, 29]. It seems that data reported so far on Mn doped ZnO are not quite consistent. These discrepan- cies may be due to the different fabrication methods. Ob- viously, more detailed works are essential to understand the magnetic behaviors of these materials. The photoluminescence (PL) of the Zn1-xMnxO nanowires and undoped ZnO nanowires was investigated at room temperature. Usually, the PL spectrum of the undoped ZnO nanowires shows characteristic UV emission and broad green emission, in agreement with previously re- ported results for ZnO nanowires [30]. However, the photoluminescence of the Zn1-xMnxO nanowires is ap- parently different from that of undoped ZnO nanowires. It can be observed from Figure 6 that a strong green emission appears, while the violet emission completely disappears. The mechanism of green emission has been suggested to be mainly due to the existence of various points that can easily form recombination centers [31]. In the present situation, the strong green emission suggests that these Zn1-xMnxO nanowires should have many point 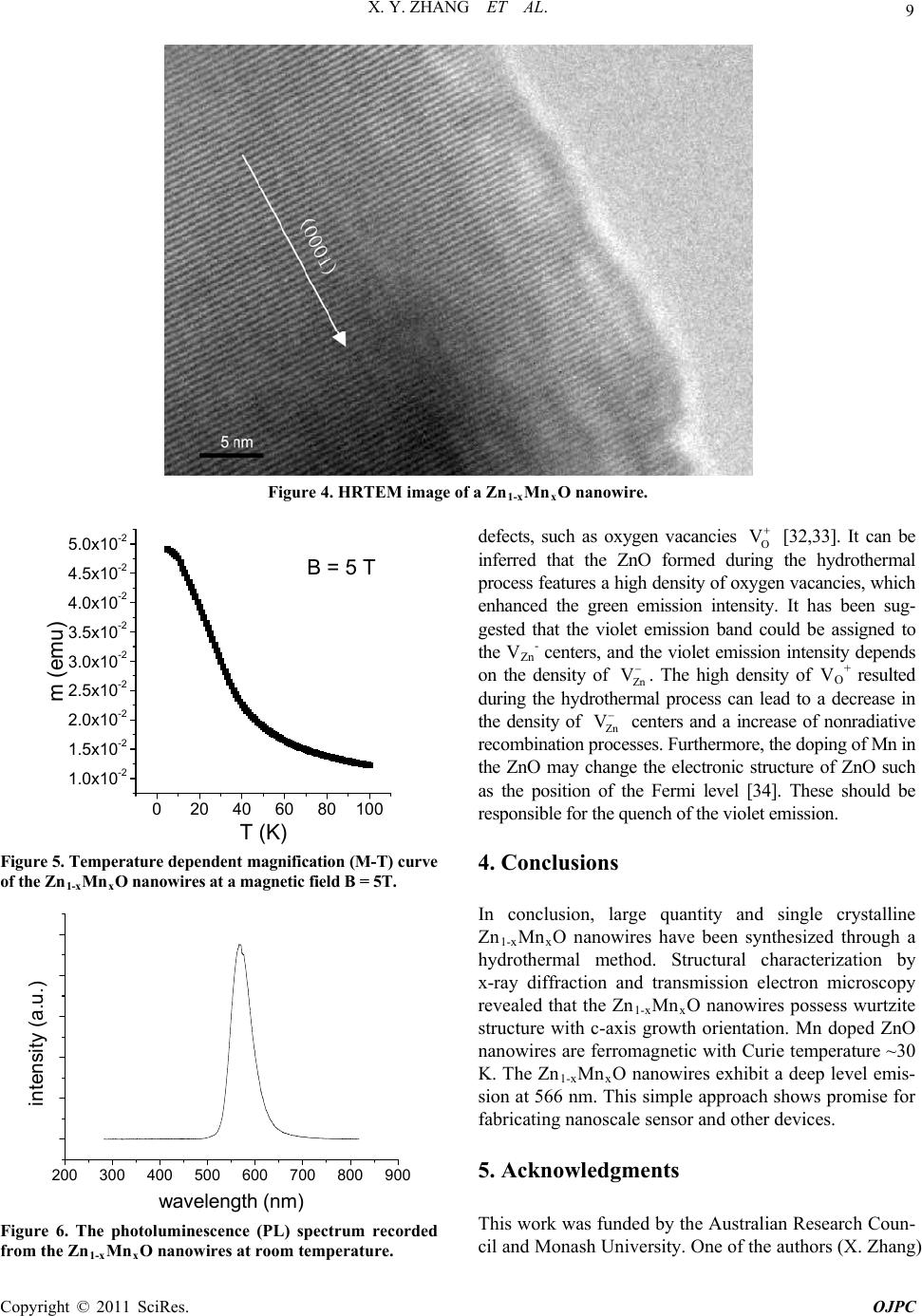 X. Y. ZHANG ET AL. Copyright © 2011 SciRes. OJPC 9 Figure 4. HRTEM image of a Zn1-xMnxO nanowire. 0 20406080100 1.0x10-2 1.5x10-2 2.0x10-2 2.5x10-2 3.0x10-2 3.5x10-2 4.0x10-2 4.5x10-2 5.0x10-2 B = 5 T T (K) m (emu) Figure 5. Temperature dependent magnific ation (M-T) curv e of the Zn1-xMnxO nanowires at a magnetic field B = 5T. 200 300 400 500600 700 800 900 intensity (a.u.) wavelength (nm) Figure 6. The photoluminescence (PL) spectrum recorded from the Zn1-xMnxO nanowires at room temperature. defects, such as oxygen vacancies O V [32,33]. It can be inferred that the ZnO formed during the hydrothermal process features a high density of oxygen vacancies, which enhanced the green emission intensity. It has been sug- gested that the violet emission band could be assigned to the VZnP - centers, and the violet emission intensity depends on the density of Zn V . The high density of VOP + resulted during the hydrothermal process can lead to a decrease in the density of Zn V centers and a increase of nonradiative recombination processes. Furthermore, the doping of Mn in the ZnO may change the electronic structure of ZnO such as the position of the Fermi level [34]. These should be responsible for the quench of the violet emission. 4. Conclusions In conclusion, large quantity and single crystalline Zn1-xMnxO nanowires have been synthesized through a hydrothermal method. Structural characterization by x-ray diffraction and transmission electron microscopy revealed that the Zn1-xMnxO nanowires possess wurtzite structure with c-axis growth orientation. Mn doped ZnO nanowires are ferromagnetic with Curie temperature ~30 K. The Zn1-xMnxO nanowires exhibit a deep level emis- sion at 566 nm. This simple approach shows promise for fabricating nanoscale sensor and other devices. 5. Acknowledgments This work was funded by the Australian Research Coun- cil and Monash University. One of the authors (X. Zhang)  X. Y. ZHANG ET AL. Copyright © 2011 SciRes. OJPC 10 thanks the Australian Research Council for the Austra- lian Research Fellowship. 6. References [1] S. Iijima, Nature Vol. 354, No. 6348, 1991, pp. 56-58. [2] M. H. Devoret, D. Esteve, and C. Urbina, Nature Vol. 360, No. 6404, 1992, pp. 547-553. [3] H. J. Dai, E. W. Wong, Y. Z. Lu, S. Fan, and C. M. Lie- ber, Nature Vol. 375, No. 6534, 1995, pp. 769-772. [4] A. P. Alivisatos, Science Vol. 271, No. 5251, 1996, pp. 933-937. [5] M. H. Huang, S. Mao, H. Feick, H. Yan, Y. Wu, H. Kind, E. Weber, R. Russo, and P. Yang, Science Vol. 292, 2001, pp. 1897-1899. [6] A. M. Morales and C. M. Lieber, Science Vol. 279, No. 5348, 1998, pp. 208-211. [7] N. Wang, Y. F. Zhang, Y. H. Tang, C. S. Lee, and S. T. Lee, Appl. Phys. Lett. Vol. 73, No. 26, 1998, pp. 3902- 3904. [8] D. C. Look, Mater. Sci. Eng. B Vol. 80, No. 1-3, 2001, pp. 383-387. [9] Z. K. Tang, G. K. L. Wong, P. Yu, M. Kawasaki, A. Oh- tomo, H. Koinuma, and Y. Segawa, Appl. Phys. Lett. Vol. 72, No. 25, 1998, pp. 3270-3272. [10] T. Dietl, H. Ohno, F. Matsukura, J. Cibert, and D. ferrand, Science Vol. 287, No. 5455, 2000, pp. 1019-1022. [11] T. Dietl, Semicond. Sci. Technol. Vol. 17, No. 4, 2002, pp. 377. [12] Y. W. Heo, M. P. Lvill, K. Ip, D. P. Norton, S. J. Pearton, J. G. Kelly, R. Rairigh, A. F. Hebard, and T. Steiner, Appl. Phys. Lett. Vol. 84, No.13, 2004, pp. 2292-2294. [13] W. Prellier, A. Foucheta, and B. Mercey, J. Phys. C Vol. 15, No. 37, 2003, pp. R1583-R1601. [14] S. J. Han, J. W. Song, C. H. Yang, S. H. Park, Y. H. Jeong, and K. W. Rhie, Appl. Phys. Lett. Vol. 81, No. 22, 2002, pp. 2412-2414. [15] P. Sharma, A. Gupta, K. V. Rao, F. J. Owens, R. Sharma, R. Ahuja, J. M. O. Guillen, B. Johansson, and G. A. Ge- hring, Nature Mater. Vol. 2, 2003, pp. 673-677. [16] D. P. Norton, S. J. Pearton, A. F. Hebard, N. Theodoro- poulou, L. A. Boatner, and R. G. Wilson, Appl. Phys. Lett. Vol. 82, No. 26, 2003, pp. 239-241. [17] T. Fukumura, Z. Jin, M. Kawasaki, T. Shono, T. Hase- gawa, and H. Koinuma, Appl. Phys. Lett. Vol. 78, No. 7, 2001, pp. 958-960. [18] X. M. Cheng, and C. L. Chien, J. Appl. Phys. Vol. 93, No. 10, 2003, pp. 7876-7878. [19] C. Ronning, P. X. Gao, Y. Ding, Z. L. Wang, and D. Schwen, Appl. Phys. Lett. Vol. 84, No. 5, 2004, pp. 783-785. [20] M. H. Huang, Y. W. Henning Feick, N. Tran, E. Weber, and P. Yang, Adv. Mater. Vol. 13, No. 2, 2001, pp. 113-116. [21] Z. R. Dai, Z. W. Pan, and Z. L. Wang, Adv. Funct. Mater. Vol. 13, No. 1, 2003, pp. 9-24. [22] V. A. L. Roy, A. B. Djurišić, H. Liu, X. X. Zhang, Y. H. Leung, M. H. Xie, J. Gao, H. F. Lui, and C. Surya, Appl. Phys. Lett. Vol. 84, No. 5, 2004, pp. 756-758. [23] Y. Q. Chang, D. B. Wang, X. H. Luo, X. Y. Xu, X. H. Chen, L. Li, C. P. Chen, R. M. Wang, J. Xu, D. P. Yu, Appl. Phys. Lett. Vol. 83, No. 19, 2003, pp. 2292-2294. [24] L. Vayssieres, K. Keis, S. –E. Lindquist, and A. Hagfeldt, J. Phys. Chem. B Vol. 105, No. 17, 2001, pp. 3350-3352. [25] L. Vayssieres, K. Keis, A. Hagfeldt, and S. –E. Lindquist, Chem. Mater. Vol. 13, No. 12, 2001, pp. 4395-4398. [26] L. E. Greene, M. Law, J. Goldberger, F. Kim, J. C. John- son, Y. F. Zhang, R. J. Saykally, and P. D. Yang, Angew. Chem. Int. Ed. Vol. 42, No. 26, 2003, pp. 3031-3034. [27] B. Liu, and H. C. Zeng, J. Am. Chem. Soc. Vol. 125, No. 15, 2003, pp. 4430-4431. [28] S. W. jung, S. J. An, G. C. Yi, C. U. Jung, S. I. Lee, S. Cho, Appl. Phys. Lett. Vol. 80, No. 24, 2002, pp 4561- 563. [29] V. A. L. Roy, A. B. Djurisic, H. Liu, X. X. Zhang, Y. H. leung, M. H. Xie, J. Gao, H. F. Liu, C. Surya, Appl. Phys. Lett. Vol. 84, No. 5, 2004, pp 756-758. [30] B. D. Yao, L. Feng, C. Cheng, M. M. T. Loy, N. Wang, Appl. Phys. Lett. Vol. 96, No. 22, 2010, pp. 223105 (1-3). [31] D. Dingle, Physics Review Letters, Vol. 23, No. 11, 1969, pp. 579-581. [32] B. Q. Cao, F. Q. Sun, W. P. Cai, Electrochem. Solid-State Letters. Vol. 8, No. 9, 2005, pp. 237-G240. [33] Y. Li, G. W. Meng, L. D. Zhang, Appl. Phys. Lett. Vol. 76, No. 15, 2000, pp. 2011-2013. [34] A. L. He, X. Q. Wang, Y. Q. Fan, and Y. P. Feng, J. Appl. Phys. Vol. 108, No. 8, 2010, pp. 084308(1-5). |

