W. LANE ET AL.
13
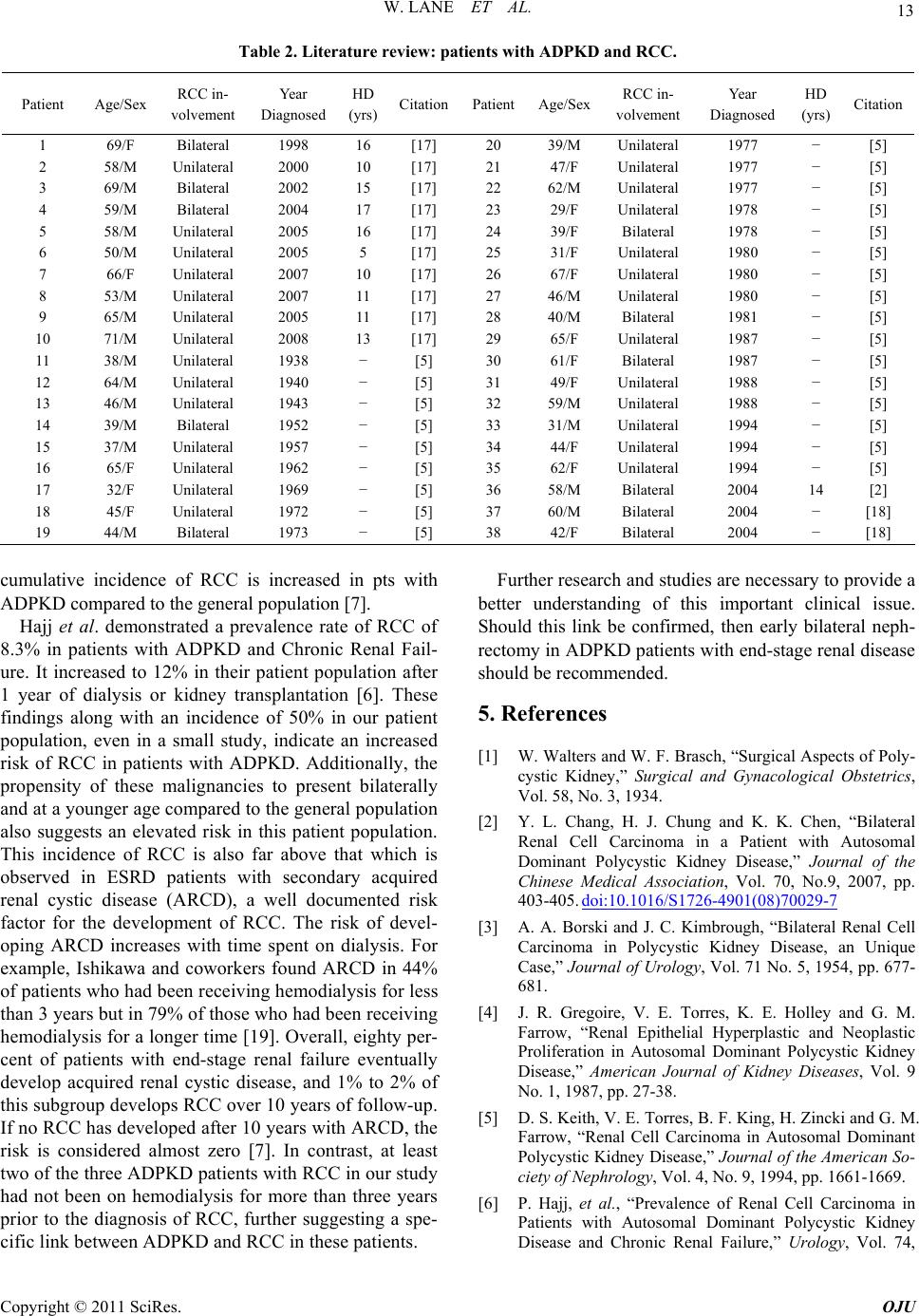
atie
Patient Age/Sex RCC in-
volvement
Year
Diagnosed
HD
(yrs) Citation
Table 2. Literature review: pnts with ADPKD and RCC.
Year
Diagnosed
HD
(yrs) Citation Patient Age/SexRCC in-
volvement
1 69/F [17] 20 39/M [5] Bilateral 1998 16 Unilateral 1977 −
2 58/M Unilateral 2000 10 [17] 21 47/F Unilateral 1977 − [5]
3
4
69/M
59/M
Bilateral 2002
Bilateral
15
17
[17]
[17]
22
23
62/M
29/F
Unilateral 1977
Unilateral
− [5]
−
[5] 2004 1978
5 58/M Unilateral 2005 16 [17] 24 39/F Bilateral 1978 − [5]
6 50/M Unilateral 2005 5 [17] 25 31/F Unilateral 1980 − [5]
7 66/F Unilateral 2007 10 [17] 26 67/F Unilateral 1980 − [5]
8 53/M Unilateral 2007 11 [17] 27 46/M Unilateral 1980 − [5]
9 65/M Unilateral 2005 11 [17] 28 40/M Bilateral 1981 − [5]
10 71/M Unilateral 2008 13 [17] 29 65/F Unilateral 1987 − [5]
11 38/M Unilateral 1938 − [5] 30 61/F Bilateral 1987 − [5]
12 64/M Unilateral 1940 − [5] 31 49/F Unilateral 1988 − [5]
13 46/M Unilateral 1943 − [5] 32 59/M Unilateral 1988 − [5]
14 39/M Bilateral 1952 − [5] 33 31/M Unilateral 1994 − [5]
15 37/M Unilateral 1957 − [5] 34 44/F Unilateral 1994 − [5]
16 65/F Unilateral 1962 − [5] 35 62/F Unilateral 1994 − [5]
17 32/F Unilateral 1969 − [5] 36 58/M Bilateral 2004 14 [2]
18 45/F Unilateral 1972 − [5] 37 60/M Bilateral 2004 − [18]
19 44/M Bilateral 1973 − [5] 38 42/F Bilateral 2004 − [18]
cumive ice is ied its with
DP com
rthearchdies aessary to provide a
beter untandihis imant cnical i
and K. K. Chen, “Bilateral
arcinoma in a Patient with Autosomal
Dominant Polycystic Kidney Disease,” Journal of the
ulat
KD
nciden
pared to t
of RCC
he general population [7].
ncreasn p
A
Hajj et al. demonstrated a prevalence rate of RCC of
8.3% in patients with ADPKD and Chronic Renal Fail-
ure. It increased to 12% in their patient population after
1 year of dialysis or kidney transplantation [6]. These
findings along with an incidence of 50% in our patient
population, even in a small study, indicate an increased
risk of RCC in patients with ADPKD. Additionally, the
propensity of these malignancies to present bilaterally
and at a younger age compared to the general population
also suggests an elevated risk in this patient population.
This incidence of RCC is also far above that which is
observed in ESRD patients with secondary acquired
renal cystic disease (ARCD), a well documented risk
factor for the development of RCC. The risk of devel-
oping ARCD increases with time spent on dialysis. For
example, Ishikawa and coworkers found ARCD in 44%
of patients who had been receiving hemodialysis for less
than 3 years but in 79% of those who had been receiving
hemodialysis for a longer time [19]. Overall, eighty per-
cent of patients with end-stage renal failure eventually
develop acquired renal cystic disease, and 1% to 2% of
this subgroup develops RCC over 10 years of follow-up.
If no RCC has developed after 10 years with ARCD, the
risk is considered almost zero [7]. In contrast, at least
two of the three ADPKD patients with RCC in our study
had not been on hemodialysis for more than three years
prior to the diagnosis of RCC, further suggesting a spe-
cific link between ADPKD and RCC in these patients.
Should this link be confirmed, then early bilateral neph-
rectomy in ADPKD patients with end-stage renal disease
should be recommended.
Fu
t
r rese
ders
and stu
ng of t
re nec
port lissue.
5. References
[1] W. Walters and W. F. Brasch, “Surgical Aspects of Poly-
cystic Kidney,” Surgical and Gynacological Obstetrics,
Vol. 58, No. 3, 1934.
[2] Y. L. Chang, H. J. Chung
Renal Cell C
Chinese Medical Association, Vol. 70, No.9, 2007, pp.
403-405. doi:10.1016/S1726-4901(08)70029-7
[3] A. A. Borski and J. C. Kimbrough, “Bilateral Renal Cell
eoplastic
V. E. Torres, B. F. King, H. Zincki and G. M.
l Dominant Polycystic Kidney
Carcinoma in Polycystic Kidney Disease, an Unique
Case,” Journal of Urology, Vol. 71 No. 5, 1954, pp. 677-
681.
[4] J. R. Gregoire, V. E. Torres, K. E. Holley and G. M.
Farrow, “Renal Epithelial Hyperplastic and N
Proliferation in Autosomal Dominant Polycystic Kidney
Disease,” American Journal of Kidney Diseases, Vol. 9
No. 1, 1987, pp. 27-38.
[5] D. S. Keith,
Farrow, “Renal Cell Carcinoma in Autosomal Dominant
Polycystic Kidney Disease,” Journal of the American So-
ciety of Nephrology, Vol. 4, No. 9, 1994, pp. 1661-1669.
[6] P. Hajj, et al., “Prevalence of Renal Cell Carcinoma in
Patients with Autosoma
Disease and Chronic Renal Failure,” Urology, Vol. 74,
Copyright © 2011 SciRes. OJU