Paper Menu >>
Journal Menu >>
 Journal of Biomaterials and Nanobiotechnology, 2011, 2, 207-215 doi:10.4236/jbnb.2011.23026 Published Online July 2011 (http://www.SciRP.org/journal/jbnb) Copyright © 2011 SciRes. JBNB 207 Study on in Vitro Degradation of Bioabsorbable Polymers Poly(Hydroxybutyrate-Co-Valerate) - (PHBV) and Poly(Caprolactone) - (PCL) Suzan Aline Casarin1*, Sônia Maria Malmonge2, Marcio Kobayashi1, José Augusto Marcondes Agnelli1 1Department of Materials Engineering, Federal University of São Carlos, São Carlos, Brazil; 2Federal University of ABC, Santo André, Brazil. Email: *sacasarin@yahoo.com.br Received March 11th, 2011; revised May 4th, 2011; accepted May 13th, 2011. ABSTRACT The increasing use of bioabsorbable polymeric materials in medicine has stimulated researchers in the materials field to search for solutions for the replacement of metallic artifacts by bioabsorbable polymers. Therefore, this study de- scribes the in vitro degradation of PHBV, PCL and the blends of these polymers, both of which are bioabsorbable polymers. The samples were prepared by extrusion followed by injection, and subjected submitted to in vitro degrada- tion in phosphate buffered saline solution with pH 7.3 and kept at 37˚C. Through the characterization of the variation of mass, molar mass, mechanical properties and morphology, the results indicated that the samples analyzed are more stable to hydrolytic degradation when compared to other bioabsorbable polymers. The materials indicate signs of deg- radation after 30 days, with a small reduction in the molar mass. After 180 days, the materials indicated a significant reduction of molar mass and reduction in the mechanical properties. Keywords: Bioabsorbable Polymers, (Poly-(Hydroxybutyrate-Co-Valerate)) - PHBV, (Poly-Caprolactone) - PCL 1. Introduction Bioabsorbable polymers are materials capable of de- grading in vivo by the action of body fluids. They are used for situations where the implant is intended to re- main in place for a predetermined period, in order to ful- fill a particular function. Its main requirements are the degradation followed by resorption and biocompatibility [1]. Over the past two decades, bioabsorbable implants have been tested and used in several orthopedic surgical procedures, including fracture fixation, bone replacement, repair of cartilage and meniscus, ligament fixation and drug vehicle. Resorbable materials have been used in the form of pins, plates and screws for orthopedic and oral and maxillo-facial surgery. Depending on the compo- nents of the polymer, these materials can be shaped to provide sufficient initial stiffness, allowing the bone to bear a certain mechanical force for a period of time, and in some cases, starts to degrade. The ideal polymer prop- erties are between the balance of mechanical, thermal and viscoelastic factors [1]. Among the main bioabsorbable and biodegradable polymers, we have the synthetic aliphatic polyesters, such as the poly (glycolic acid) - (PGA), poly (lactic acid) - PLA, poly (lactic-co-glycolic acid) - PLGA, poly (ε- ca- prolactone) - PCL, poly (hydroxybutyrate) - PHB and poly (hydroxybutyrate-co-valerate) - PHBV. Exposed to aqueous body fluids, the materials are initially hydrated. With the presence of water molecules, the degradation process takes place through the hydrolysis of esters bond s, leading to products in the form of oligomers, or mono- mers, soluble and nontoxic. The degradation then pro- ceeds by biologically active processes or by passive hy- drolytic cleavage [2,3]. Seeking an initial assessment of the behavior of these materials, the in vitro degradation tests comes out as a good alternative when compared to in vivo studies, es- sential and necessary in their evaluation as biomaterials. In the in vitro tests, the costs are lower, the process can be accelerated and the test conditions, such as tempera- ture, pH, products and byproducts of degradation, can be quantified and monitored [4]. Applied to bioresorbable polymers, the tests are, in most cases, made in phosphate buffered solution, pH 7.4, simulating the osmolarity and  Study on in Vitro Degradation of Bioabsorbable Polymers Poly(Hydroxybutyrate-Co-Valerate) - (PHBV) and Poly(Caprolactone) - (PCL) Copyright © 2011 SciRes. JBNB 208 physiological pH conditio ns [5]. The hydrolysis of hydrolytically unstable chemical groups is the predominant mechanism for the degrad ation of bioabsorbable polymers [6,7]. As a result of degrada- tion, there is loss of mass and decrease in the mechanical strength due to the decrease of mol ar mass . The poly mer chains become soluble in the extracellular fluid after reaching low values of molar mass (generally below 7000 Daltons) and, at this moment, the material has very low mechanical strength, starting the process of frag- mentation due to the local mechanical tensile [6,8]. Through the in vitro degradation, in phosphate buff- ered saline solution, with pH 7.3, of pure PHBV and PCL and from their blends in the compositions of PHBV/PCL (75/25) and PHBV/PCL (50/50), this study aimed to study the behavior of mass variation, the molar mass variation, the mechanical properties and morphology. This study was conducted aiming to obtain a bioresorb- able material with adequated mechanical properties for use in osteosynthesis. 2. Experimental 2.1. Materials The polymer poly (hydroxybutyrate-co-valerate) - PHBV used in the development of the study was provided by PHB Industrial S/A lote FE-133 with 12% of valerate and polymer poly(caprolactone) - PCL was provided by Perstorp Caprolactones typ e CAPA 6500. 2.2. Material Processing The blends of bioabsorbable polymers PHBV/PCL were prepared in different proportions, which were 100/0, 75/25, 50/50 and 0/100. The initial preparation of the granules of polymeric mixtures was made by extrusion and the molding of the specimens was performed by in- jection molding. The materials were injected using the mold for tensile test, following standard ASTM D-638-02 [9]. 2.3. Characterization 2.3.1. In Vitro Deg radation The degradation tests were performed in compliance with the standard ASTM F1635-04a [10] and tensile speci- mens were used as specified by ASTM D 638-02 [9]. After the preparation, the samples were submitted to the in vitro degradation study in a phosphate buffered saline solution, with pH 7.3, in six replicates. The stowage of the samples was performed in test tubes, kept in an incu- bator at 37˚C. After periods of 30, 60, 90, 120, 150 and 180 days, the samples w ere removed, washed thoro ughly with water, then with distilled water, dried and charac- terized by the following techniques. 2.3.2. Mass Varia tion For the calculation of mass loss, the samples were weighed before and after the test. The calculation was performed using the final mass of dry specimen, as fol- lows: % of mass loss = [(final mass – initial mass)/ initial mass]* 100 2.3.3. Siz e -E xcl u si on Chromatograph y—SEC For the analysis, we used a Size-Exclusion Chromatog- raphy (SEC), with liquid chromatography system with high efficiency and a Waters® isocratic pump, model 1515, with Waters® refractive index detector, model 2414. 2.3.4. Tensile Test The tensile tests were performed on a Universal Testing Machine named Instron 5500R, in co mpliance with stan- dard ASTM D-638-02 [9]. The tests were performed at room temperature with a 115 mm distance between the grips, 50 mm/min speed and a 50 kN load cell. 2.3.5. Infrared Absorption Spectroscopy—FTIR Samples before and after hydrolytic degradation were analyzed in an equipment of FTIR Thermo Scientific, model Nicolet 6700, with resolution of 2 cm–1, using the technique of film the evaluation of Index of terminal carboxylic groups (IGCT) using Equation (1), 1 1 absorbance at 329 0 cm IGCT absorbance at 2970 cm (1) where 3290 cm–1 is related to the terminal carboxylic groups and 2970 cm–1 is related to vibration of axial strain C-H (abs orption band reference) [11]. 2.3.6. Analysis by Scanning Electron Microscopy—SEM The morphological analyses were performed in a Philips XL 30 FEG Scanning Electron Microscope, for the sam- ples degraded by 90 and 180 days, for comparison pur- poses, it was also performed on samples that were not de- graded. All the analyses were performed on the samples surface. 3. Results and Discussion The mass variation is commonly used to characterize the hydrolytic and/or enzimatic degradation of bioresorbable polymers [12,13]. The mass normalized due to the im- mersion time indicated a distinct behavior between the two polymers studied, and the two polymers proved to be more stable to hydrolytic degradation when compared to other bioabsorbable polymers, such as PLLA [14] and  Study on in Vitro Degradation of Bioabsorbable Polymers Poly(Hydroxybutyrate-Co-Valerate) - (PHBV) and Poly(Caprolactone) - (PCL) Copyright © 2011 SciRes. JBNB 209 PLGA 50 [13]. The PHBV samples mass increased along the time in contact with saline solution, probably becau se they absorbed water, since the PCL samples mass de- creased, being the period of 90 days considered as the period in which the material mass decrease was maxi- mum, as sh own in Tabl e 1 and Figure 1. For the blends, it was observed that the PHBV/PCL (75/25) blend had a behavior similar to that of pure PHBV polymer, while the PHBV/PCL (50/50) blend had a behavior similar to the PCL polymer. The evaluation of the molar mass variation was moni- tored through the SEC (Size-Exclusion Chromatography) analysis, where we observed a significant reduction in the molar mass of pure PHBV and PHBV in the blends. For the pure PCL, the molar mass remained constant throughout the hydrolytic degradation, as shown in Ta- ble 2 and Figure 2. By comparing t he mass loss results with the molar mass results, it can be noticed that the PHBV p olym er doe s no t lose mass along the 180 days in contact with saline solu- tion at 37˚C, but loses approximately 40% of molar mass, which can be explained by the fact that PHBV absorbs water along the time and consequently the polymer chains in the bulk sample breaks up, causing a molar mass re- duction. The opposite occurs in the case of pure PCL polymer, the polymer degrades mainly in the sample surface by erosion process with no splitting of the poly- mer chains, thus the molar mass remained constant throughout th e test and there was a mass loss. With regard to the mechanical performance, in this case, the most important proper ty is the Modulu s of Elas- ticity under Tens ile or Young’s Modulus. This is defin ed as the ratio between the nominal tensile stress and the corresponding elongation, below the limit of proportion- ality of the material and is expressed in force per area unit (MPa) [15]. Table 3 and Figure 3 show the results of the values of the Modulus of Elasticity along the hydrolytic degrada- tion. It is possible to observe that, by comparing the Mo- dulus of Elasticity values of the polymers and its blends samples before and after immersion in the salin e solution, there was an increase in the Modulus of Elasticity in ten- sion throughout the time, as shown in Table 3 and Fi- gure 3. It can be noticed in Table 4 and Figure 4 that there was an increase in the tension at break for pure PHBV and for the blends. In the PCL analysis, it can be noticed that the behavior is hardly different. Regarding the elon- gation at break (Table 5 and Figure 5) there was a de- crease in values, in the case of PHBV, reaching 44% in samples degraded for 180 days. The results are con sistent with those obtain ed by SEC, Table 1. Mass loss. 30 days60 days90 days 120 days 150 days180 days PHBV 0.11%0.24%0.04% 0.10% 0.21%0.28% PHBV/PCL (75/25) –0.07% 0.21%0.04% 0.11% 0.19%0.35% PHBV/PCL (50/50) –0.24%–0.12%–0.31% –0.29% –0.21%–0.28% PCL –0.53%–0.55%–0.59% –0.51% –0.44%–0.41% Figure 1. Loss of mass due to period. Table 2. Loss of molar mass. PHBV PHBV/PCL (75/25) PHBV/PCL (50/50) PCL 30 days0.8% 7.6% 1.7% 0.0% 60 days14.8% 9.8% 7.0% 0.0% 90 days20.0% 16.9% 8.8% 0.3% 120 days28.4% 22.5% 10.0% 0.1% 150 days36.8% 27.6% 17.1% 0.0% 180 days39.7% 34.1% 17.1% 0.0% Figure 2. % of molar mass loss due to period. 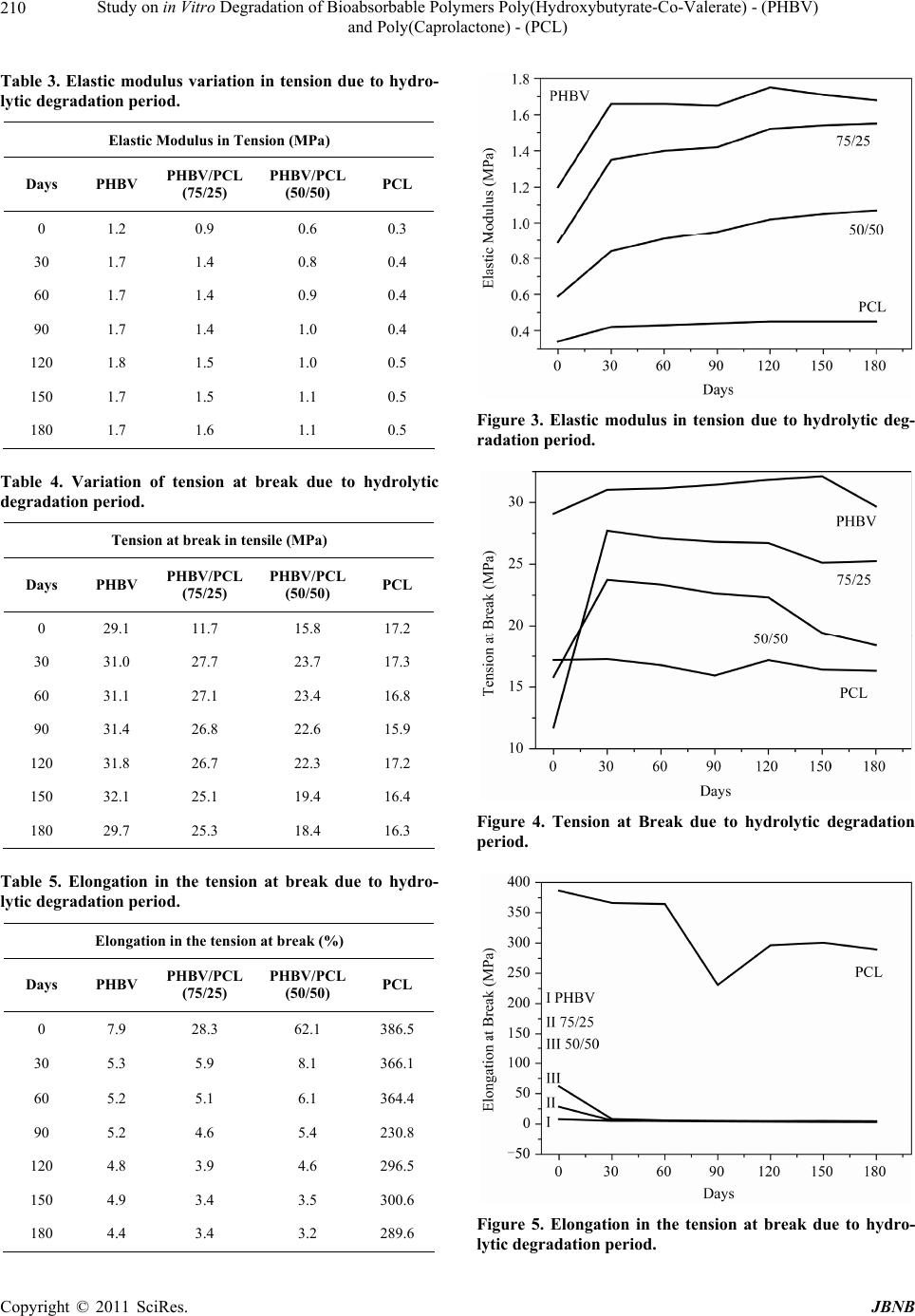 Study on in Vitro Degradation of Bioabsorbable Polymers Poly(Hydroxybutyrate-Co-Valerate) - (PHBV) and Poly(Caprolactone) - (PCL) Copyright © 2011 SciRes. JBNB 210 Table 3. Elastic modulus variation in tension due to hydro- lytic degradation period. Elastic Modulus in Tension (MPa) Days PHBV PHBV/PCL (75/25) PHBV/PCL (50/50) PCL 0 1.2 0.9 0.6 0.3 30 1.7 1.4 0.8 0.4 60 1.7 1.4 0.9 0.4 90 1.7 1.4 1.0 0.4 120 1.8 1.5 1.0 0.5 150 1.7 1.5 1.1 0.5 180 1.7 1.6 1.1 0.5 Table 4. Variation of tension at break due to hydrolytic degradation period. Tension at break in tensile (MPa) Days PHBV PHBV/PCL (75/25) PHBV/PCL (50/50) PCL 0 29.1 11.7 15.8 17.2 30 31.0 27.7 23.7 17.3 60 31.1 27.1 23.4 16.8 90 31.4 26.8 22.6 15.9 120 31.8 26.7 22.3 17.2 150 32.1 25.1 19.4 16.4 180 29.7 25.3 18.4 16.3 Table 5. Elongation in the tension at break due to hydro- lytic degradation period. Elongation in the tension at br eak ( % ) Days PHBV PHBV/PCL (75/25) PHBV/PCL (50/50) PCL 0 7.9 28.3 62.1 386.5 30 5.3 5.9 8.1 366.1 60 5.2 5.1 6.1 364.4 90 5.2 4.6 5.4 230.8 120 4.8 3.9 4.6 296.5 150 4.9 3.4 3.5 300.6 180 4.4 3.4 3.2 289.6 Figure 3. Elastic modulus in tension due to hydrolytic deg- radation period. Figure 4. Tension at Break due to hydrolytic degradation period. Figure 5. Elongation in the tension at break due to hydro- lytic degradation period. 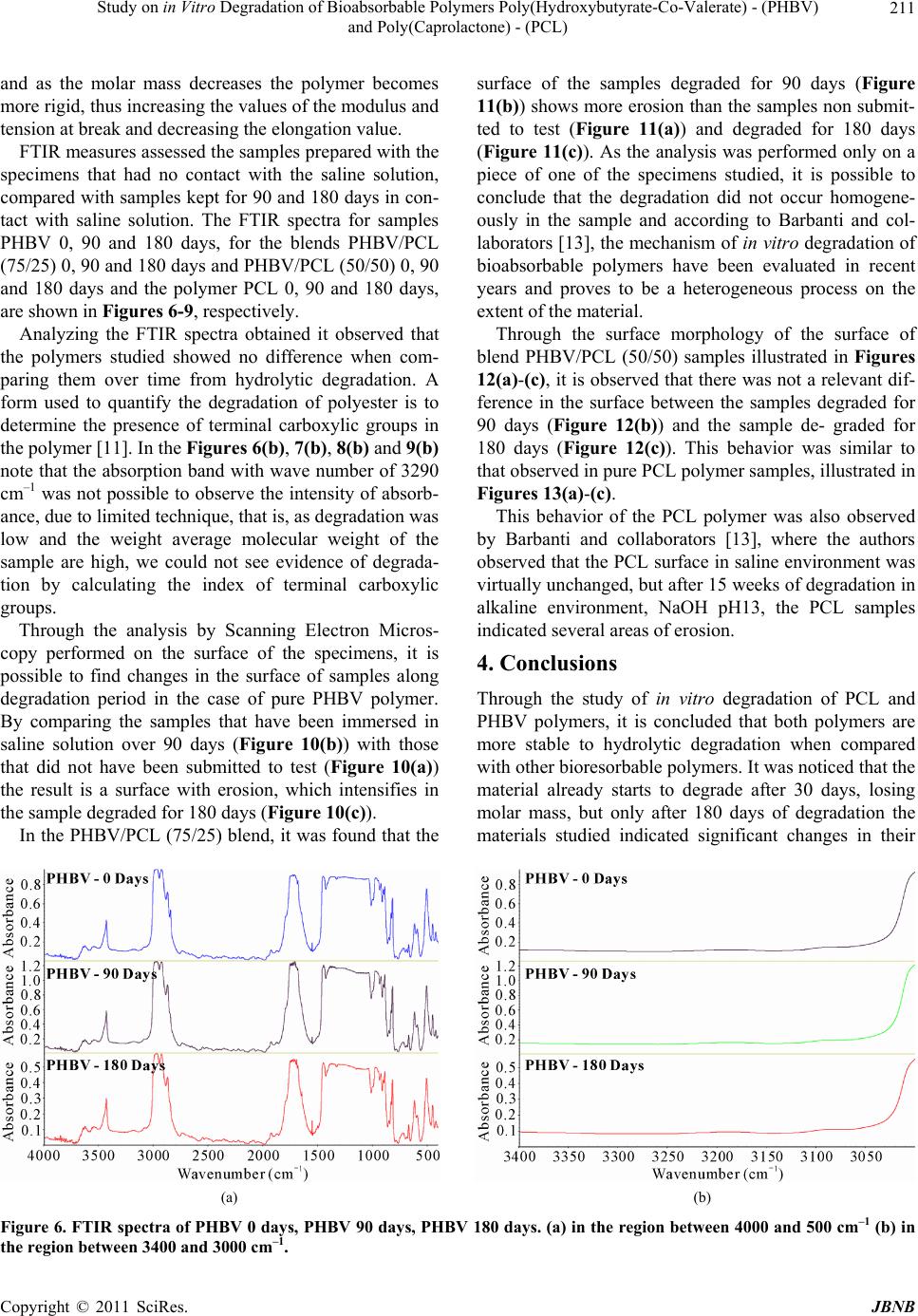 Study on in Vitro Degradation of Bioabsorbable Polymers Poly(Hydroxybutyrate-Co-Valerate) - (PHBV) and Poly(Caprolactone) - (PCL) Copyright © 2011 SciRes. JBNB 211 and as the molar mass decreases the polymer becomes more rigid, thus increasing the values of the modulus and tension at break and decreasing the elongation value. FTIR measures assessed the samples prepared with the specimens that had no contact with the saline solution, compared with samples kept for 90 and 180 days in con- tact with saline solution. The FTIR spectra for samples PHBV 0, 90 and 180 days, for the blends PHBV/PCL (75/25) 0, 90 and 180 days and PHBV/PCL (50/50) 0, 90 and 180 days and the polymer PCL 0, 90 and 180 days, are shown in Figures 6- 9 , respectively. Analyzing the FTIR spectra obtained it observed that the polymers studied showed no difference when com- paring them over time from hydrolytic degradation. A form used to quantify the degradation of polyester is to determine the presence of terminal carboxylic groups in the polymer [11]. In the Figures 6(b), 7(b), 8(b) and 9(b) note that the absorption band with wave number of 3290 cm–1 was not possible to observe the intensity of absorb- ance, due to limited technique, that is, as degradation was low and the weight average molecular weight of the sample are high, we could not see evidence of degrada- tion by calculating the index of terminal carboxylic groups. Through the analysis by Scanning Electron Micros- copy performed on the surface of the specimens, it is possible to find changes in the surface of samples along degradation period in the case of pure PHBV polymer. By comparing the samples that have been immersed in saline solution over 90 days (Figure 10(b)) with those that did not have been submitted to test (Figure 10(a)) the result is a surface with erosion, which intensifies in the sample degraded for 180 days (Figure 10(c)). In the PHBV/PCL (75/25) blend, it was found that the surface of the samples degraded for 90 days (Figure 11(b)) shows more erosion than the samples non submit- ted to test (Figure 11(a)) and degraded for 180 days (Figure 11(c)). As the analysis was performed only on a piece of one of the specimens studied, it is possible to conclude that the degradation did not occur homogene- ously in the sample and according to Barbanti and col- laborators [13], the mechanism of in vitro degradation of bioabsorbable polymers have been evaluated in recent years and proves to be a heterogeneous process on the extent of the material. Through the surface morphology of the surface of blend PHBV/PCL (50/50) samples illustrated in Figures 12(a)-(c), it is observed that there was not a relevan t dif- ference in the surface between the samples degraded for 90 days (Figure 12(b)) and the sample de- graded for 180 days (Figure 12(c)). This behavior was similar to that observed in pure PCL polymer samples, illustrated in Figures 13(a)-(c). This behavior of the PCL polymer was also observed by Barbanti and collaborators [13], where the authors observed that the PCL surface in saline environment was virtually unchanged, but after 15 weeks of degradation in alkaline environment, NaOH pH13, the PCL samples indicated several areas of erosion. 4. Conclusions Through the study of in vitro degradation of PCL and PHBV polymers, it is concluded that both polymers are more stable to hydrolytic degradation when compared with other bioresorbable polymers. It was noticed that the material already starts to degrade after 30 days, losing molar mass, but only after 180 days of degradation the materials studied indicated significant changes in their (a) (b) Figure 6. FTIR spectra of PHBV 0 days, PHBV 90 days, PHBV 180 days. (a) in the region between 4000 and 500 cm–1 (b) in the region between 3400 and 3000 cm–1. 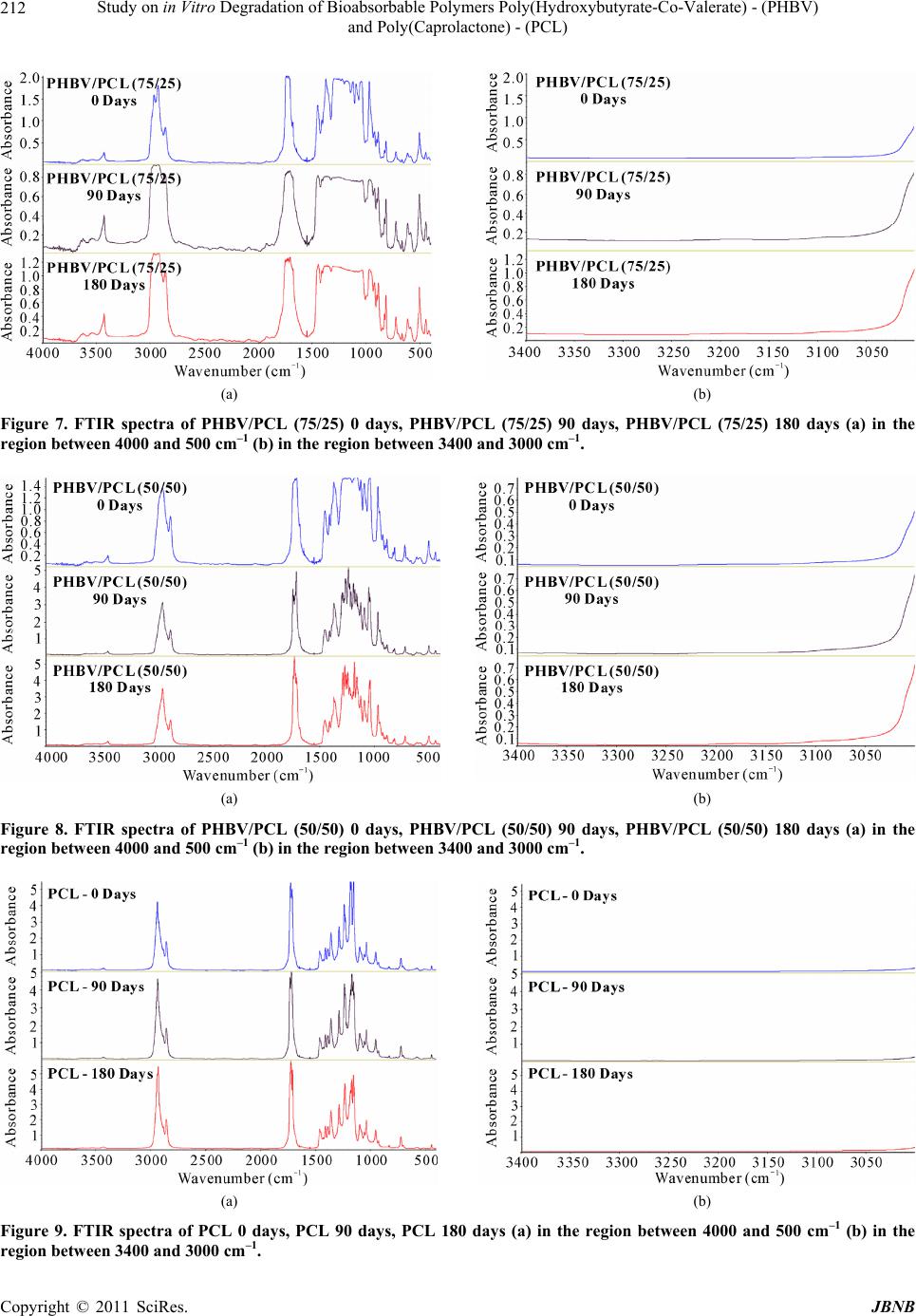 Study on in Vitro Degradation of Bioabsorbable Polymers Poly(Hydroxybutyrate-Co-Valerate) - (PHBV) and Poly(Caprolactone) - (PCL) Copyright © 2011 SciRes. JBNB 212 (a) (b) Figure 7. FTIR spectra of PHBV/PCL (75/25) 0 days, PHBV/PCL (75/25) 90 days, PHBV/PCL (75/25) 180 days (a) in the region between 4000 and 500 cm–1 (b) in the region between 3400 and 3000 cm–1. (a) (b) Figure 8. FTIR spectra of PHBV/PCL (50/50) 0 days, PHBV/PCL (50/50) 90 days, PHBV/PCL (50/50) 180 days (a) in the region between 4000 and 500 cm–1 (b) in the region between 3400 and 3000 cm–1. (a) (b) Figure 9. FTIR spectra of PCL 0 days, PCL 90 days, PCL 180 days (a) in the region between 4000 and 500 cm–1 (b) in the region between 3400 and 3000 cm–1.  Current Distortion Evaluation in Traction 4Q Constant Switching Frequency Converters Copyright © 2011 SciRes. JBNB 213 (a) (b) (c) Figure 10. Micrograph (a) PHBV 0 days (b) PHBV 90 days (c) PHBV 180 days. (a) (b) (c) Figure 11. Micrograph (a) PHBV/PCL (75/25) 0 days (b) PHBV/PCL (75/25) 90 days (c) PHBV/PCL (75/25) 180 days.  Study on in Vitro Degradation of Bioabsorbable Polymers Poly(Hydroxybutyrate-Co-Valerate) - (PHBV) and Poly(Caprolactone) - (PCL) Copyright © 2011 SciRes. JBNB 214 (a) (b) (c) Figure 12. Micrograph (a) PHBV/PCL (50/50) 0 days; (b) PHBV/PCL (50/50) 90 days; (c) PHBV/PCL (50/50) 180 days. (a) (b) (c) Figure 13. Micrograph (a) PCL 0 days (b) PCL 90 days (c) PCL 180 days.  Study on in Vitro Degradation of Bioabsorbable Polymers Poly(Hydroxybutyrate-Co-Valerate) - (PHBV) and Poly(Caprolactone) - (PCL) Copyright © 2011 SciRes. JBNB 215 mechanical properties, suggesting their use in osteosyn- thesis devices that require a longer permanence in the organism. Through the micrographs, it can be concluded that the degradation does not occur homogeneously throughout the specimen. 5. Acknowledgments The authors appreciate the contribution of PHB Industrial S/A for providing the poly mers used. REFERENCES [1] H. A. Yuehuei, K. W. Shane and R. J. Friedman, “Pre-clinical in vivo Evaluation of Orthopedic Bioab- sorbable Devices,” Biomaterials, Vol. 21, No. 24, 2000, pp. 2635-2652.doi:10.1016/S0142-9612(00)00132-0 [2] M. Elke, M. Rolf-Joachim and D. Wolf-Dieter, “Studies on the Enzymatic Hydrolysis of Polyesters I. Low Mo- lecular Mass Model Esters and Aliphatic Polyesters,” Polymer Degradation and Stability, Vol. 80, No. 3, 2003, pp. 485-501.doi:10.1016/S0141-3910(03)00032-6 [3] M. H. Huang, S. Li, D. W. Hutmacher, J. T. Schantz, C. A. Vacanti, C. Braud and M. Vert, “Degradation and Cell Culture Studies on Block Copolymers Prepared by Ring Opening Polymerization of Epsilon-Caprolactone in the Presence of Poly(Ethylene Glycol),” Journal of Biomedi- cal Materials Research. Vol. 69A, No. 3, 2004, pp. 417- 427.doi:10.1002/jbm.a.30008 [4] R. M. Luciano, C. A. C. Zavaglia, E. A. R. Duek and M. C. Alberto-Rincon, “Synthesis and Characterization of Poly(L-lactic acid) Membranes: Studies in Vivo and in Vivo,” Journal of Materials Science: Materials in Me- dicine, Vol. 14, No. 1, 2003, pp. 87-94. doi:10.1023/A:1021509722296 [5] P. Laine, R. Kontio, C. Lindqvist and R. Suuronen, “Are there Any Complications with Bioabsorbable Fixation Devices? A 10 Year Review in Orthognathic Surgery,” International Journal of Oral and Maxillofacial Surgery, Vol. 33, No. 3, 2004, pp. 240-244. doi:10.1006/ijom.2003.0510 [6] M. Vanin, “Obtaining, Characterization and Study of Ad- sorption on Protein on Biorreabsorbable Blend (b- Ydroxybutyrate) (PHB)/Poly(L-Lactic Acid) ( PLLA),” Ph. D Thesis in Chemical Engineering, State University of Campinas, Campinas, 2003, p. 212. [7] J. C. Middleton and A. J. Tipton, “Synthetic Biodegrad- able Polymers as Orthopaedic Devices,” Biomaterials, Vol. 21, No. 23, 2000, pp. 2335-2346. doi:10.1016/S0142-9612(00)00101-0 [8] M. C. Wake, et al., “Effects of Biodegradable Polymer Particles on Rat Marrow-Derrived Stromal Osteoblasts in Vitro,” Biomaterials, Vol. 19, No. 14, 1998, pp. 1255- 1268.doi:10.1016/S0142-9612(98)00022-2 [9] ASTM D-638-02, “Standard Test Method for Tensile Properties of Plastics,” Annual Book of ASTM Standards, 2002. [10] ASTM F 1635-04a, “Standard Test Method for in Vitro Degradation Testing of Hydrolytically Degradable Poly- mer Resins and Fabricated Forms for Surgical Implants,” Annual Book of ASTM Standards, Philadelphia, 2004. [11] G. M. Vinha s, Y. M. B. Almeida, M. A. G. A. Li ma and L. A. Santos, “Estudo das Propriedades e Biodegrada- bilidade de Blendas de Poliéster/Amido Submetidas ao Ataque Microbiano,” Química Nova, Vol. 30, No. 7, 2007, pp. 1584-1588. doi:10.1590/S0100-40422007000700016 [12] E. L. Hedberg, C. K. Shih, J. J. Lemoine, M. D. Timmer, M. A. K. Liebschner, J. A. Jansen and A. G. Mikos, “In Vitro Degradation of Porous Poly(propylene fumarate)/ poly(DL-Lactic-Co-Glycolic Acid) Composite Scaffolds,” Biomaterials, Vol. 26, No. 16, 2005, pp. 3215-3225. doi:10.1016/j.biomaterials.2004.09.012 [13] S. H. Barbanti, C. A. Zavaglia and E. A. R. Duek, “De- gradação Acelerada de Suportes de Poli(-ca- prolactona) e Poli(D,L-ácido láctico-co-ácido glicólico) em Meio Alcalino,” Polímeros: Ciência e Tecnologia, Vol. 16, No. 2, 2006, pp. 141-148. [14] E. A. R. Duek, C. A. C. Zavaglia and W. D. Belangero, “In Vitro Study of Poly(Lactic Acid) Pin Degradation,” Polymer, Vol. 40, No. 23, 1999, pp. 6465-6473. doi:10.1016/S0032-3861(98)00846-5 [15] L. B. Canto and L. A. Pessan, “Resistência à tração, flexão e compressão” In: S. V. Canevarolo Ed., Técnicas de Caracterização de Polímeros, 2nd Edition, Artliber, São Paulo, 2007. |

