Paper Menu >>
Journal Menu >>
 Advances in Bioscience and Biotechnology, 2011, 2, 89-96 ABB doi:10.4236/abb.2011.22014 Published Online April 2011 (http://www.SciRP.org/journal/abb/). Published Online April 2011 in SciRes. http://www.scirp.org/journal/ABB Pectinases yeast production using grape skin as carbon source María Arévalo-V illena1*, Mercedes Fernández2, Jesús López2, Ana Briones1 1Tecnología de Alimentos, Facultad de Ciencia Químicas, Universidad de Castilla la Mancha, Ciudad Real, Spain; 2Escuela de Ingeniería Técnica Industrial, Universidad de Castilla la Mancha, Ciudad Real, Spain. Email: Maria.Arevalo@uclm.es Received 17 January 2011; revised 9 March 2011; accepted 11 March 2011. ABSTRACT Pectinases are used in Enology for some different utilities. Enzymatic preparations from moulds are a mixed of different enzymes with strong and unspe- cific activities. Some Saccharomyces cerevisiae pro- duce pectinases which can be used instead of com- mercial preparations. The objectives of this work were to study the enzyme secretion by one Saccharo- myces cerevisiae (CECT 11783) for growing on grape skin (industry oenological by-product) as carbon source. Preliminary experiments showed that the strain produced pectinases for growing on grape skin without any other carbon source. Statistical treat- ment (factorial design 25) was applied to evaluate the influences of related factors (agitation, temperature, presence of peptone and detergent in the medium and time of growth) Variables with the most significant interactions for pectinase production were agitation and nitrogen source concentration. Response surface methodology showed that a first order model was not adequate for results. Nevertheless, the built of a sec- ond order model offered a polynomial equation which surface predicted a maximum of activity (52.68 enzymatic units) for specific values of the studied variables (147.8 rpm of agitation and 15.9 g of pep- tone/L culture medium). Keywords: Pectinase Enzyme from Yeast; Enology; Grape Skin; Statistical Treatment; Response Surface Methodology 1. INTRODUCTION Pectinolytic enzymes are found mainly in moulds and bacteria, but they also occur in some yeasts [1-3]. Given the role played by yeasts, especially of the genus Sac- charomyces, in fermented products, further research into their pectinolytic enzymes would be useful for two pur- poses: one, so that yeast can be used to synthesize then purify the enzymes for addition to fruit juices as clarifi- cation and extraction enhancers; and two, in the case of fermented products, so that the enzyme can be produced by the yeast as part of the process rather than having to be added to the medium. Most commercial pectinase preparations used in the food industry are derived from Aspergillus niger, a GRAS microorganism producing large quantities of these enzymes. However, this mould secretes other en- zymes which may trigger collateral reactions, such as th e release of volatile phenols less desirable for the produc- tion of wine or fruit juices, for instance arabinofuranosi- dase, which can cause turbidity [4]. Pectinases are used in winemaking to enhance must extraction by degrading structural polysaccharides which interfere with the extraction process [5], thus increasing the release of colour and aroma compounds in musts both before and during fermentation. At the same time, the addition of pectinases improves maceration, clarifi- cation and filtration during the winemaking process [6-8]. Pectinolytic enzymes derived from Saccharomyces cerevisiae would provide a useful alternative to mould- derived pectinases, since a genuine product can only be obtaine d f rom yeasts . Certain strains of S. cerevisiae have been found to break down polygalacturonic acid, which could be im- portant for the fermentation of plant-derived substrates [1,9]. It has been demonstrated that when the enzyme ex- tract from Saccharomyces bayanus is added to fresh must, the effects on turbidity are the same as when a commercial enzyme preparation is added [10]. A study reported that when PG+ strains of S. cere- visiae were used in winemaking, in some cases the fil- tration time was reduced by half without any appreciable changes in viscosity [11]. Moreover, a transformed strain with good winemaking qualities has more recently been engineered using the PGU1 gene from another strain, transcriptionally bonded to the PGK1 gene promoter, in order to enhance its expression during growth [12].  M. Arévalo-V illena et al. / Advances in Bioscience and Biotechnology 2 (2011) 89-96 Copyright © 2011 SciRes. ABB 90 In some countries, current legislation prohibits the u se of genetically-modified organisms, though not of GMO- derived enzymes, in winemaking. The first step towards achieving this goal is to develop appropriate enzyme- production technology. Spanish grape-skin production, as a by-product of the winemaking process, is estimated at around 750 000 ton- nes per year. At present, it is used mostly as animal feed. Polygalacturonase activity in grape musts has been shown to increase markedly one day after the addition of yeast, whereas no enzyme activity was detected through- out fermentation in must made from juice al one [13]. The incorporation of grape skin in the formulation of culture media for use in industrial enzyme production would bring both economic and environmental benefits for winemaking areas, by enabling commercial exploita- tion of this by-product. The composition of grape skin may well enhance yeast growth as well as inducing pecti- nase synthesis. The aim of this research was to optimize the culture medium using grape skin as substrate for the growth and synthesis of pectinases derived from a genetically- modified yeast strain. Statistical optimization was preferred because it en- abled evaluation of interactions between parameters and involved a specific experimental design [14,15]. 2. MATERIAL AND METHODS A genetically-modified Saccharomyces cerevisiae strain (CECT 11783) (12) was used, containing the gene PGU1 from a spontaneous winemaking yeast, which conferred the ability to hydrolyze polygalacturonic acid and there- fore pectins. For all assays, cells were precultured in YPD broth to enable inoculation of a final population of 107 cells/mL onto each tested growth medium. 2.1. Enzyme Method for Determining Pectinolytic Activity Pectinolytic activity was evaluated by quantifying the amount of galacturonic acid released from apple pectin (Fluka) using the DNSA (dinitrosalicylic acid) reaction. The method was optimized by adjusting reagent concen- trations and incubation times. A commercial pectinase was used as positive control, and a commercial Sac- charomyces strain (UCLM S325) not possessing pecti- nolytic activity served as negative control. Results were plotted on a galacturonic-acid calibration curve covering the appropriate range of concentrations. 2.2. Preliminary Tests. Relationship between Yeast Growth and Pectinase Production 2.2.1. First Experiment A number of prior experiments were performed to con- firm the ability of yeast to grow and to synthesize poly- galacturonase in the presence of grape skin. The yeast was grown on the following media: 7 g/L grape skin 7 g/L grape skin + 5 g/L gl ucose 7 g/L grape ski n + 10 g/ L pep t o ne 7 g/L grape skin + 5 g/L gl ucose + 10 g/L pepto ne A set of 100-mL flasks containing 20 mL of each me- dium were inoculated and incubated at 28ºC in a ther- mostatically-controlled water bath shaker (150 rpm). Polygalacturonase activity was measured at 24, 46 and 96 hours of yeast growth. All assays were performed in triplicate, and results were expressed in enzyme units (i.e. the amount of en- zyme required to liberate 10 ug of galacturonic acid from apple pectin in the conditions outlined above). 2.2.2. Second Experiment In view of the results obtained, a second experiment was performed to ascertain whether grape skin and/or glu- cose concentrations significantly influenced enzyme synthesis. For this purpose, peptone concentration and growth time (determinant variables) were fixed at 10 g/L and 24 h, respectively. Yeast was grown in a r efriger ated orbital shaker which was used until the end of th e work. The new media formulations were as follows: 0 g/L grape skin + 5 g/L gl ucose + 10 g/L peptone 7 g/L grape skin + 5 g/L gl ucose + 10 g/L peptone 21 g/L grape skin + 5 g/L glucose + 10 g/L peptone 7 g/L grape ski n + 10 g/ L pep t o ne 21 g/L grape skin + 10 g/L peptone 2.3. Optimization of the Culture Medium for Pectinase Production. Statistical Analysis In the light of the preliminary study results, the grape skin concentration was set at 21 g/L and glucose was omitted from the culture broth; a study was therefore made of other variables potentially influencing enzyme synthesis. A two-stage statistical analysis was performed: the first stage identified significant factors, while in the second stage response surface methodology (RSM) was used to maximize enzyme activity. 2.3.1. Fi r st Stage The conditioning variables studied were: agitation (shaking speed), temperature, presence of detergent (Tween 80), cell harvest time and presence of a nitrogen source (peptone); these variables were selected in view of their marked influence on enzyme synthesis by yeasts (16). Each variable was studied at two levels, so that the combination of five variables (factorial design 25 with two replications) gave a total of 64 runs. The experimental design used is shown in Table 1.  M. Arévalo-V illena et al. / Advances in Bioscience and Biotechnology 2 (2011) 89-96 Copyright © 2011 SciRes. ABB 91 Table 1. Experimental design for identification of significant factors in enzyme production. (Agitation, temperature, presence of detergent (Tween 80), cell harvest time and presence of a nitrogen source (peptone)). Experiment A Experiment B Experiment C Experiment D Tween 80 Time (h) Peptone A1 B1 C1 D1 – 12 – A2 B2 C2 D2 + 12 – A3 B5 C5 D5 + 48 – A4 B6 C6 D6 + 12 + A5 B3 C3 D3 – 48 – A6 B4 C4 D4 – 12 + A7 B8 C8 D8 + 48 + A8 B7 C7 D7 – 48 + - Experiment A: Agitation 50 rpm, temperatu re 18ºC - Experiment B: Agitation 150 rpm, temperature 18ºC - Experiment C: Agitation 5 0 rp m, temperature 28ºC - Experiment D: Agitation 150 rpm, temperature 28ºC The four possible combinations of shaking speed (50 and 150 rpm) and temperature (18 and 28ºC) were fixed; 8 experiments were performed in duplicate for each com- bination to quantify the amount of enzyme produced, expressed as enzyme units. The replicated 25 factorial model was constructed us- ing the SPSS statistical software package. The univariate GLM procedure was used to examine the magnitude and direction of fac t o r effe ct s. The design model was based on the equation: ()( ) ijklmhij kl mij ik illm ijkklm jklm ijklmh y ijkl ijklm e where i, j, k, l, m took the values 1 and 2, and h varied between 1 and the number of replicates (in this case, 2). In the first instance, a full factorial model was con- structed using the five main factors, ten second-order interactions and ten third-order interactions. Factor ef- fects and significant interactions were then estimated by the UNIANOVA procedure using pairwise comparisons and profile plots. 2.3.2. Second Sta ge The results from the first stage indicated that neither detergent (Tween 80) nor cell harvest time were signifi- cant variables, and that the optimum temperature was 28ºC. For technical and economic reasons, therefore, the following variables were fixed: temperature (28ºC), grape skin concentration (21 g/L) and cell harvest time (24 h); thus only shaking speed and peptone concentra- tion were studied at the seco nd stage. First, a linear approach to optimal cond itions was car- ried out using first-order strategies and a 22 factorial design with three replications of the centre-point (values of the central conditions of each assay): agitation = 150 rpm, peptone concentration = 10 g/L. Model suitability was assessed by analysing fit and curvature, and esti- mating experimental error. Shaking speed was set at 150 rpm, and peptone concentrations were tested at 2 g/L intervals up to 20 g/L, giving a total of 7 assays. Since results were not determinant (data not shown), the test was repeated with a new centre-point (shaking speed = 150 rpm, peptone = 14 g/L), which involved performing a further 7 experiments. 3. RESULTS AND DISCUSSION 3.1. Enzyme Method for Determining Pectinolytic Activity The enzyme reaction providing the best results was a mixture of 500 µL of supernatant (enzyme) with 500 µL of 0.25% apple pectin, incubated for 30 minutes at 37ºC. Once the reaction was complete, 500 µL of the mixture was reacted with 500 µL of DNSA, and incubated for 10 minutes at 100ºC. The cooled reaction mixture was diluted with 1.2 mL of water, and data were plotted on a galacturonic acid calibration curve ranging from 0.1 to 1 mg/mL. 3.2. Preliminary Tests. Relationship between Yeast Growth and Pectinase Production 3.2.1. First Experiment Extracellular pectinase activity at various harvest times using different growth media is shown in Figure 1. Ac- tivity was influenced by medium composition and the presence of a nitrogen source (peptone) stimulating pectinase synthesis (growth media C and D). One-way analysis of variance (ANOVA, 95% CI) re- vealed that enzyme production differed significantly in all tested media at 46 h. Maximum enzyme production was observed with the grape skin + glucose + peptone  M. Arévalo-V illena et al. / Advances in Bioscience and Biotechnology 2 (2011) 89-96 Copyright © 2011 SciRes. ABB 92 AAA BB B CC D D D C 0 20 40 60 80 20 30 4050 60 70 8090100 Tim e (h) Enzym e units Figure 1. Evolution of pectinolitic activity in some culture media at different harvest time. A. 7 g/L skin; B. 7 g/L skin + 5 g/L glucose; C. 7 g/L skin + 10 g/L peptone; D. 7 g/L skin + 5 g/L glucose + 10 g/L peptone. combination (medium D) at 46 hours; the maximum value of 78.25 ± 0.2 enzyme units was significantly higher than that obtained with all the other combin ations. This higlights the importance of peptone for yeast growth and thus for enzyme synthesis; indeed, the sec- ond-highest value (60.3 ± 1.4) was obtained with pep- tone-containing medium C. The influence of cell harvest time on enzyme produc- tion in each medium varied as a function of medium composition: variations in harvest time had no signifi- cant effect on production in the medium containing only grape skin, but prompted significant differences in the other three media. For the grape skin + glucose + pep- tone combination (medium D), enzyme production peaked at 46 h. When glucose was removed (medium C), production peaked earlier (24 h); however, the difference between the two harv est times, though significant, was not marked. Finally, in the medium containing glucose but not peptone (medium B), enzyme synthesis was sig- nificantly inhibited in the early stages; although some activity was detected later, values never approached those obtained using a nitrogen source. 3.2.2. Second Experiment Yeast was grown in media I, II, III, IV and V (detailed under Material and Methods) to determine the influence of glucose and/or grape skin on pectinolytic activity. No significant differences (95% CI) in enzyme production were noted, values of around 40 enzyme units being re- corded in all cases (Tabl e 2). The positive effect of glu- cose in medium II was matched by the increased grape-skin concentration in medium V. In the absence of statistically-significant differences, the decision to use a peptone-containing medium in which the carbon source (glucose) was replaced by an increased grape skin concentration (21 g/L) was prompt- ed by the fact that this medium proved cheaper and also made profitabl e use of a wi nemaking by-pro duct . 3.3. Optimization of the Culture Medium for Pectinase Production. Statistical Analysis 3.3.1. Fi r st Stage Enzyme activity for the 64 runs detailed under Material and Methods is shown in Table 3. Enzyme production was significantly affected by all tested variables except presence of detergent. In experiment A (agitation 50 rpm, temperature 18ºC), activity was n eg ligible or no nexisten t due to minimal yeast growth, attributable to stress condi- tions. With these results, statistical analysis was repeated, Table 2. Production of pectinolitic enzyme in media with different skin and glucose concentration. Culture medium Enzyme units I. 0 g/L skin + 5 g/L glucose + 10 g/L peptone 43.3 ± 3.1 II. 7 g/L skin + 5 g/L glucose + 10 g/L peptone 48.5 ± 2.2 III. 21 g/L skin + 5 g/L glucose + 10 g/L peptone 39.8 ± 7.9 IV. 7 g/L skin + 10 g/L peptone 42.4 ± 4.5 V. 21 g/L skin + 10 g/L peptone 47.5 ± 1.5 Table 3. Pectinolitic activity obtained by Table 1 design. Experiment A (50 rpm/18ºC) Tw een 80 Time (h) Peptone Enzyme units 1 – 12 – 3.2 ± 0.6 2 + 12 – 3.6 ± 2 .6 3 + 48 – 4 + 12 + 5 – 48 – 6 – 12 + 7 + 48 + 8 – 48 + Nd* 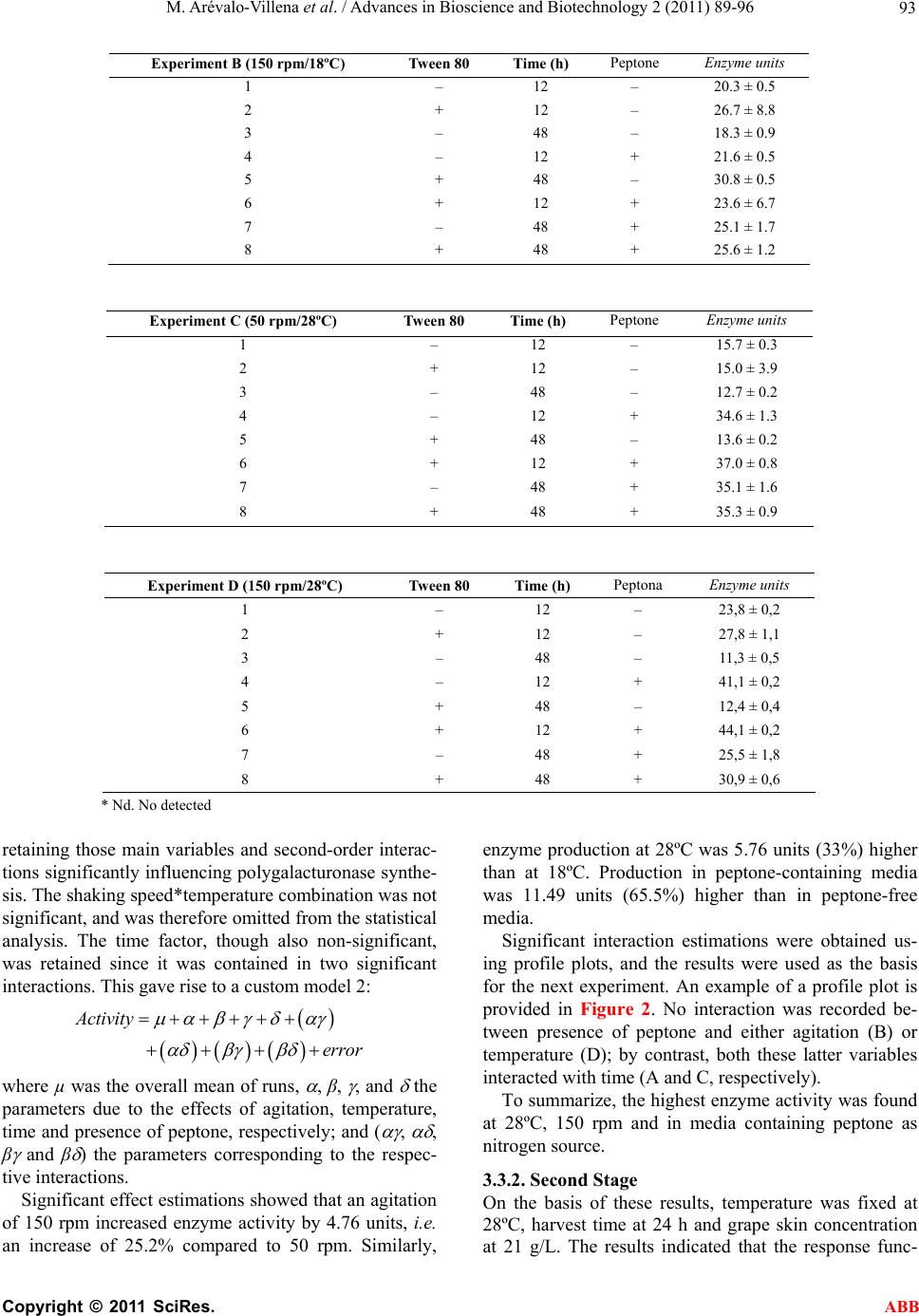 M. Arévalo-V illena et al. / Advances in Bioscience and Biotechnology 2 (2011) 89-96 Copyright © 2011 SciRes. ABB 93 Experiment B (150 rpm/18ºC) Tween 80 Time (h) Peptone Enzyme units 1 – 12 – 20.3 ± 0.5 2 + 12 – 26.7 ± 8.8 3 – 48 – 18.3 ± 0.9 4 – 12 + 21.6 ± 0.5 5 + 48 – 30.8 ± 0.5 6 + 12 + 23.6 ± 6.7 7 – 48 + 25.1 ± 1.7 8 + 48 + 25.6 ± 1.2 Experiment C (50 rpm/28ºC) Tween 80 Time (h) Peptone Enzyme units 1 – 12 – 15.7 ± 0.3 2 + 12 – 15.0 ± 3.9 3 – 48 – 12.7 ± 0.2 4 – 12 + 34.6 ± 1.3 5 + 48 – 13.6 ± 0.2 6 + 12 + 37.0 ± 0.8 7 – 48 + 35.1 ± 1.6 8 + 48 + 35.3 ± 0.9 Experiment D (150 rpm/28ºC) Tween 80 Time (h) Peptona Enzyme units 1 – 12 – 23,8 ± 0,2 2 + 12 – 27,8 ± 1,1 3 – 48 – 11,3 ± 0,5 4 – 12 + 41,1 ± 0,2 5 + 48 – 12,4 ± 0,4 6 + 12 + 44,1 ± 0,2 7 – 48 + 25,5 ± 1,8 8 + 48 + 30,9 ± 0,6 * Nd. No detected retaining those main variables and second-order interac- tions significantly influencing polygalacturonase synthe- sis. The shaking speed*temperature combination was not significant, and was therefore omitted from the statistical analysis. The time factor, though also non-significant, was retained since it was contained in two significant interactions. This gave rise to a custom model 2: Activity error where µ was the overall mean of runs, , β, , and the parameters due to the effects of agitation, temperature, time and presence of peptone, respectively; and ( , , β and β ) the parameters corresponding to the respec- tive interactions. Significant effect estimations showed that an agitation of 150 rpm increased enzyme activity by 4.76 units, i.e. an increase of 25.2% compared to 50 rpm. Similarly, enzyme production at 28ºC was 5.76 units (33%) higher than at 18ºC. Production in peptone-containing media was 11.49 units (65.5%) higher than in peptone-free media. Significant interaction estimations were obtained us- ing profile plots, and the results were used as the basis for the next experiment. An example of a profile plot is provided in Figure 2. No interaction was recorded be- tween presence of peptone and either agitation (B) or temperature (D); by contrast, both these latter variables interacted with time (A and C, respectively). To summarize, the highest enzyme activity was found at 28ºC, 150 rpm and in media containing peptone as nitrogen source. 3.3.2. Second Sta ge On the basis of these results, temperature was fixed at 28ºC, harvest time at 24 h and grape skin concentration at 21 g/L. The results indicated that the response func- 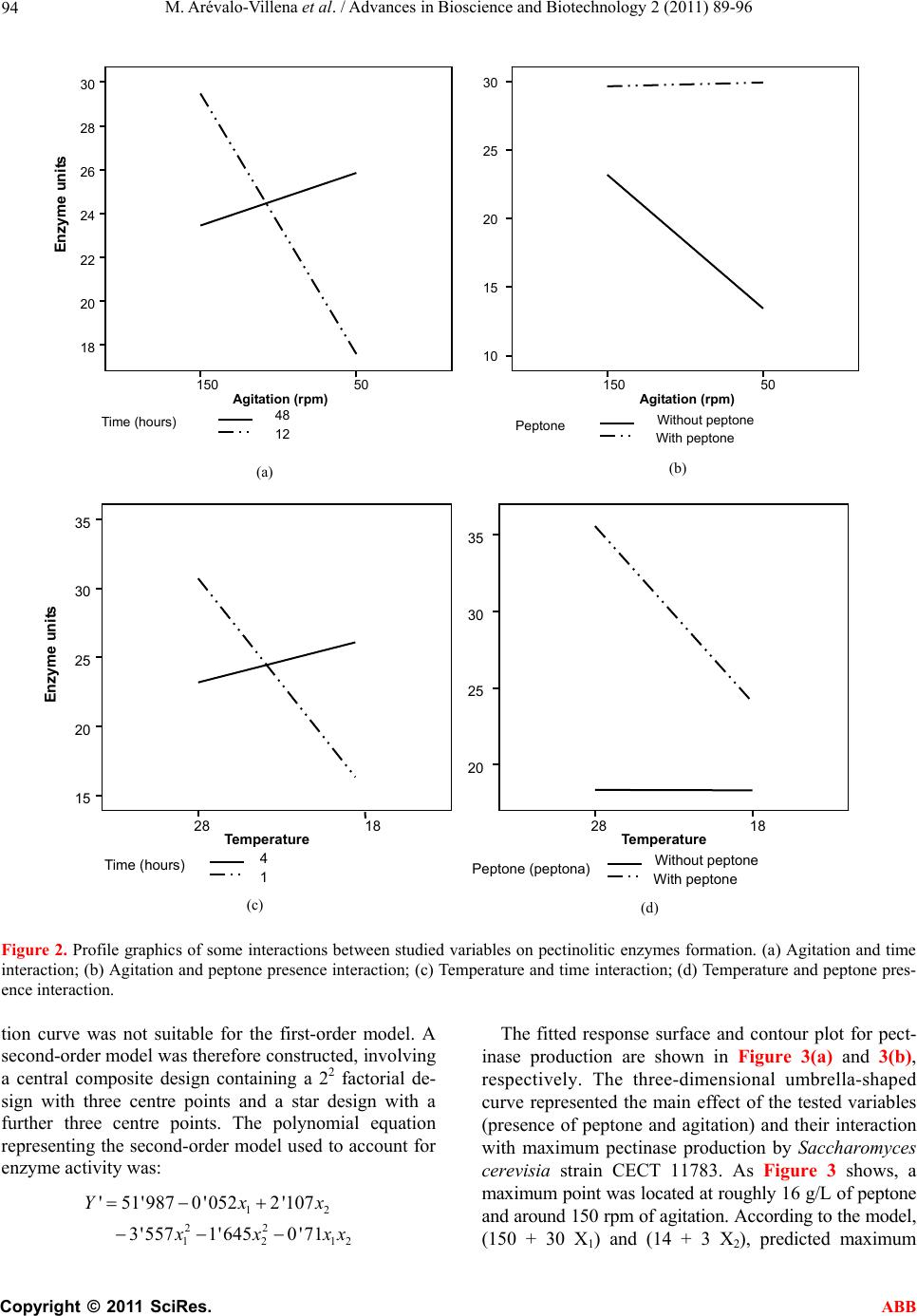 M. Arévalo-V illena et al. / Advances in Bioscience and Biotechnology 2 (2011) 89-96 Copyright © 2011 SciRes. ABB 94 Figure 2. Profile graphics of some interactions between studied variables on pect inolitic enzymes formation. (a) Agitation a nd time interaction; (b) Agitation and peptone presence interaction; (c) Temperature an d time interacti on; (d) Temperature and peptone pres- ence interaction. tion curve was not suitable for the first-order model. A second-order model was therefore constructed, involving a central composite design containing a 22 factorial de- sign with three centre points and a star design with a further three centre points. The polynomial equation representing the second-order model used to account for enzyme activity was: 12 22 1212 '51'987 0'0522'107 3'557 1'6450'71 Yxx x xxx The fitted response surface and contour plot for pect- inase production are shown in Figure 3(a) and 3(b), respectively. The three-dimensional umbrella-shaped curve represented the main effect of the tested variables (presence of peptone and agitation) and their interaction with maximum pectinase production by Saccharomyces cerevisia strain CECT 11783. As Figure 3 shows, a maximum point was located at roughly 16 g/L of peptone and around 150 rpm of agitation. According to the model, (150 + 30 X1) and (14 + 3 X2), predicted maximum Agitation (rpm) 50150 Enz y me units 30 28 26 24 22 20 18 12 48 Time (hours) Agitation (rpm) 50 150 With peptone Without peptone Peptone 30 25 20 15 10 (b) (a) Temperature 1 28 Enzyme units 35 30 25 20 15 1 4 Time (hours) Temperature 18 28 35 30 25 20 With peptone Without peptone Peptone (peptona) 18 (c) (d) 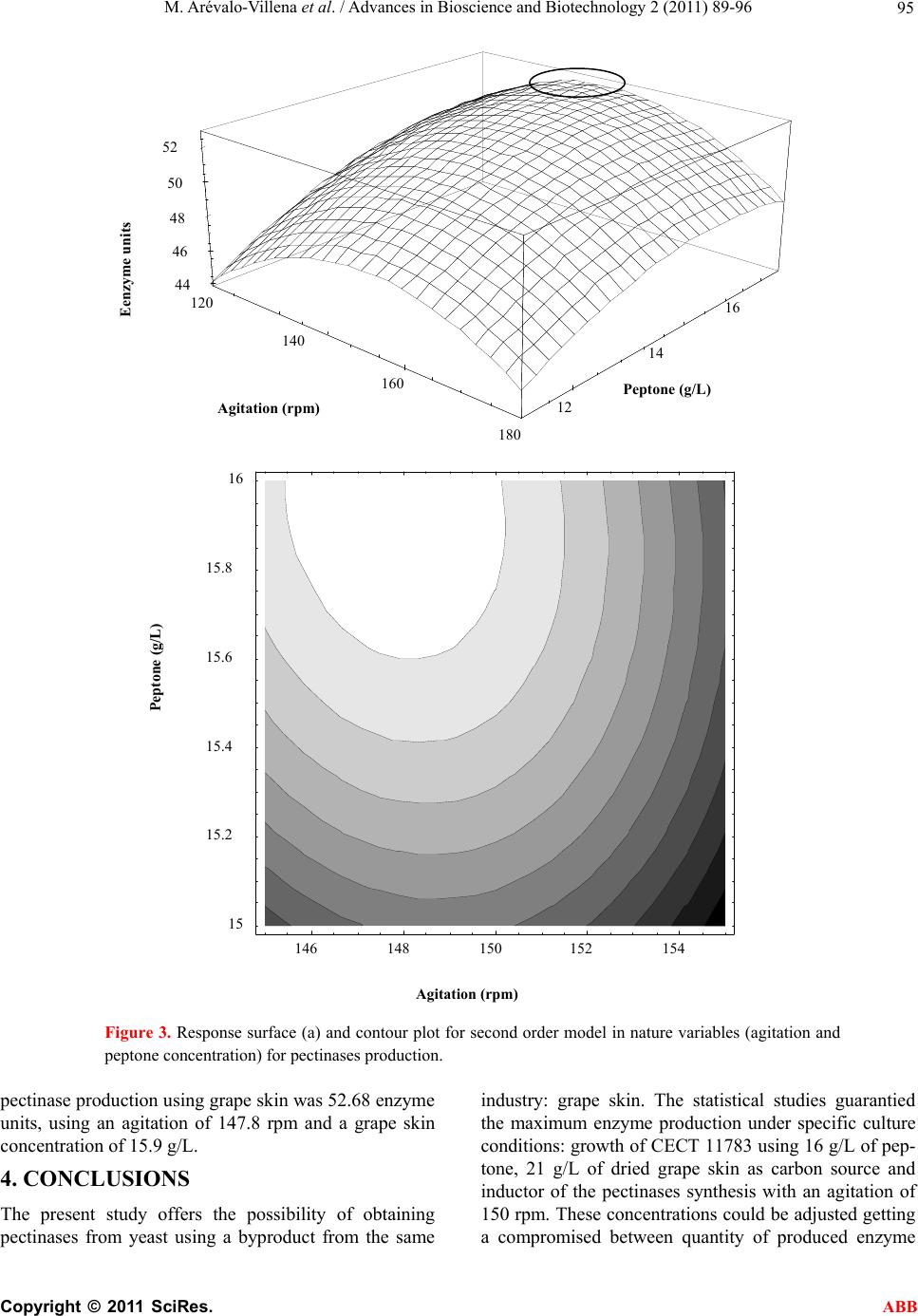 M. Arévalo-V illena et al. / Advances in Bioscience and Biotechnology 2 (2011) 89-96 Copyright © 2011 SciRes. ABB 95 Figure 3. Response surface (a) and contour plot for second order model in nature variables (agitation and peptone concentration) for pectinases production. pectinase produ ction u sing grap e sk in was 52.68 enzyme units, using an agitation of 147.8 rpm and a grape skin concentration of 15.9 g/L. 4. CONCLUSIONS The present study offers the possibility of obtaining pectinases from yeast using a byproduct from the same industry: grape skin. The statistical studies guarantied the maximum enzyme production under specific culture conditions: growth of CECT 11783 using 16 g/L of pep- tone, 21 g/L of dried grape skin as carbon source and inductor of the pectinases synthesis with an agitation of 150 rpm. These concentrations could be adjusted getting a compromised between quantity of produced enzyme Eenzyme units Agitation (rpm) 12 14 16 Peptone (g/L) 44 46 48 50 52 120 140 160 180 Agitation (rpm) Peptone (g/L) 146 148 150 152 154 15 15.2 15.4 15.6 15.8 16  M. Arévalo-V illena et al. / Advances in Bioscience and Biotechnology 2 (2011) 89-96 Copyright © 2011 SciRes. ABB 96 and economic factors. 5. ACKNOWLEDGEMENTS This study was funded by INIA pr o j ec t (RM 2007 00004-00-00). REFERENCES [1] Fernández-González, M., Ubeda, J.F., Vasudevan, T.G., Otero and Briones, R.A. (2004) Evaluation of polygalac- turonase activity in Saccharomyces cerevisiae wine strains. FEMS Microbiology Letters, 237, 261-266. [2] Jayani, R. Singh, Saxena, S. and Gupta, R. (2005) Mi- crobial pectinolitic enzymes: A review. Proccess Bio- chemistry, 40, 2931-2944. doi:10.1016/j.procbio.2005.03.026 [3] Oliveira, K.F., Malavolta, L., Souza, C.S., Vicente, E.J. and Laluce, C. (2006) Pectinoytic activity secreted by yeasts isolated from fermented citrus molasses. Journal of Applied Microbiology, 100, 633-640. do i:10 .1111/j. 1365 -2672.2006.02823.x [4] Whitaker, J.R. (1990) Microbial pectinolitic enzymes. In: Fogarty, W.M. and Kelly, C.T., Eds., Microbial Enzymes and Biotechnology. 2nd Edition, Elsevier Science Ltd., London, 133-176. [5] Roldán, A., Palacios, V., Peñatez, X., Benitez, T. and Pérez, L. (2006) Use of Trichoderma enzymatic extracts on vinification of Palomino fino grapes in the Sherry re- gion. Journal of Food Engineering, 75, 375-382. doi:10.1016/j.jfoodeng.2005.03.065 [6] Villetaz, J.C. (1990) Les colloides colmatant et la filtration des vins. Reveu Francaise d’oenologie, 122, 59-63 [7] Villetaz, J.C. (1996) Utilisitain les enzymes en oenologie pour l’extraction de la couleur et por l’extration et la revelation des aromes. Bulletin de l'OIV, 787, 843-869. [8] Servili, M., Begliomini, A.L. and Montedor, G. (1992) Utilization of a yeast pectinase in olive oil extraction and red wine making processes. Journal of Science and Food Agriculture, 58, 253-260. doi:10.1002/jsfa.2740580214 [9] McKay, A.M. (1990) Degradation of polygalacturonic acid by Saccharomyces cerevisiae. Letter in Applied Mi- crobiology, 11, 41-44. [10] Gainvors, A., Frézier, V., Lemarasquier, H., Lequart, C., Aigle, M. and Belarbi, A. (1994) Detection of polygalac- turonase, pectin-lyase and pectin-esterase activities in a Saccharomyces cerevisiae strain. Yeast, 10, 1311-1319. doi:10.1002/yea.320101008 [11] Blanco, P., Sieiro, C., Díaz, A., Reboredo, M. and Villa, T.G. (1997) Grape juice biodegradation by polygalactu- ronases from Saccharomyces cerevisiae. International Biodeterioration and Biodegradation, 40, 115-118. doi:10.1016/S0964-8305(97)00055-3 [12] Fernández-González, M., Úbeda, J.F., Cordero-Otero, R.R., Thanvanthri Gururajan, V. and Briones, A.I. (2005) Engineering of an oenological Saccharomyces cerevisiae strain with pectinolytic activity and its effect on wine. International Journal of Food Microbiology, 102, 173- 183. doi:10.1016/j.ijfoodmicro.2004.12.012 [13] Takayanagi, T., Uchibori, T. and Yokotsuka, K. (2001) Characteristics of yeast polygalacturonases induced dur- ing fermentation on grape skins. American Journal of Enology and Viticulture, 52, 41-44. [14] Fernández Valdivia, D.G., Espinola Lozano, F. and Moya Vilar, M. (2008) Influencia de diferentes coadyuvantes tecnológicos en la calidad y rendimiento del aceite de oliva virgen utilizando la metodología de superficies de respuesta. Grasas y Aceites, 59, 39-44. [15] Vasconcelos, A.F., Barbosa, A., Dekker, R., Scarminio, I. and Rezende, M.I. (2000) Optimization of lacase produc- tion by Botriosphaeria sp., in the presence of veratryl alcohol by the response-surface method. Process in Bio- chemistry, 35, 1131-1138. doi:10.1016/S0032-9592(00)00149-7 |

