 Journal of Geoscience and Environment Protection, 2014, 2, 42-47 Published Online April 2014 in SciRes. http://www.scirp.org/journal/gep http://dx.doi.org/10.4236/gep.2014.22007 How to cite this paper: He, F. et al. (2014). Simultaneous Removal of Perchlorate and Nitrate Using Biodegradable Poly- mers Bioreactor Concept. Journal of Geoscience and Environment Protection, 2, 42-47. http://dx.doi.org/10.4236/gep.2014.22007 Simultaneous Removal of Perchlorate and Nitrate Using Biodegradable Polymers Bioreactor Concept Fang He1,2*, Haihong Zhou1, Xiuju Wang1, Liguo Wang1 1School of Resources and Environment, University of Jinan, Jinan, China 2Shandong Provincial Engineering Technology Research Center for Ecological Carbon Sink and Capture Utilization, Jina n, China Email: *hefanghhd@163.com Received Dec emb er 2013 Abstract Simultaneous perchlorate and nitrate removal from contaminated groundwater in one reactor has been realized with different methods in the past. The usage of biodegradable polymers as biofilm carriers and carbon source is new. Polymer in this paper was designed out of the copolymer of starch and polyvinyl alcohol. Under polluted water with 2 mg/L of perchlorate and 20 mg/L of NO3-N, it was possible to produce completely denitrification only for 5 h and below the detection limit to perchlorate within 9 h. Results indicating a significant impact of liquor pH on the biode- gradation of but slight effect on nitrate reduction. Packed-bed reactor filled with polymer granules could remove 2 mg/L perchlorate and 25 mg/L NO3-N completely with influent flow rate of 1.17 mL/min. Morphological observation indicated the developed biofilm coverage on the outer surfaces of the carriers was dense and primarily composed of bacillus and coccus. The microbes in biofilm decomposed polymer, the chink and filament structure on the carrier surface developed, through metabolism and provided carbon source for them by releasing small organic molecules. Keywords Perchlorate; Nitrate; Biodegradation; Biodegradable Polymers; Biofilm 1. Introduction Perchlorate ( ) in surface and groundwater has become an ever-increasing water quality concern (Sriniva- san et al., 2009; Baidasa et al., 2011). Nitrate is a well-known water pollutant harmful to human health (Bau- chard et al., 1992). Specially, nitrate is often a co-contaminant with due to fertilizer application or explo- sives. As methods for effective remediation of perchlorate and nitrate are sought, the most promising techniques appear to involve use of bacteria that respire and degrade perchlorate and nitrate (Coates et al., 2004; Coates et al., 2000; Wallace et al., 1998; Herman et al., 1999). It has been found that many perchlorate respiring bacteria *Corresponding author.  F. He et al. are also capable of reducing nitrate, but it has not been yet clarified whether the reduction of perchlorate and ni- trate is catalyzed by a single reductase (Xu et al., 2003, 2004; Choi et al., 2008). It is known that organic carbon is needed as the electron donor in the process of reduction of perchlorate and nitrate (Shrout et al., 2006; Ghosh et al., 2011; Zhou et al., 2009). However, electron donors are extremely in- sufficient in perchlorate- and nitrate-contaminated groundwater. Therefore, an exogenous electron donor must often be added in significant quantities (at significant cost). The challenge of in situ perchlorate and nitrate re- mediation is to provide an effective electron donor source for bacteria that respires perchlorate and nitrate. This work was to evaluate the feasibility of starch/ polyvinyl alcohol (PVA) polymer as the carbon source and the only physical support for microorganisms for synchronous removing nitrate and perchlorate. In addition, the changes of biofilm morphology and microbiology community on the carrier were also investigated before and after removing nitrate and perchlorate cultivation. The results may give us the insight into the synergistic inte- raction, dynamics in degradation activity of the microbial community on the biodegradable polymer. 2. Materials and Methods 2.1. Materials The synthetic groundwater composition was consisted of deionized water supplemented with 10 mg/L KH2PO4 (as P), 50 mg/L NH4Cl (as N), 20 mg/L NaNO3 (as N), 2 mg/L NaClO4 (as ), 10 mL trace metal solution. All chemicals used were of ACS grade. Working standards were prepared daily from the stock solution. The biodegradable particles were produced by an extrusion process and were a copolymer of starch and PVA mate- rials. The product was insoluble in water and would not lose its strength in the water for a long time. The main characteristics of the carriers are given in Table 1. 2.2. Instruments To each reactor, 30 g polymer carrier with perchlorate and nitrate biofilm and 100 ml of synthetic groundwater were added and then incubated. Batch experiments were performed using glass reactors placed on water-bath shakers with adjustable water temperatures. Flasks were sealed by rubber plugs to maintain anoxic condition. The packed bed reactor used in this study was an upflow column. A schematic representation of the experimen- tal setup was shown in Figure 1. Polymer granules were used as support media for biofilm growth and packed the column up to a height of 30 cm. Synthetic groundwater mixed with mature activated sludge was pumped at a flow rate of 1.80 mL/min and flew into the bottom of column, which was recirculated for about 7 days until microbial films on the granules formed and became gradually thicker. And then starch/PVA granules packed bed reactor began the operation. 2.3. Analytical Methods The concentration of was determined using an ion chromatograph (Dionex, ICs2000) equipped with a suppressed conductivity detector, an AS20 column, an AG20 guard column. The analysis of was made using a mobile phase of 35 mM of NaOH (flow rate 1 mL/min). For the determination of , the mobile phase (flow rate 1 mL/min) was a 5 mM solution of NaOH. Table 1. Main characteristics of the starch/PBS polymer carriers. Parameter Characteristics and value Material Starch 60%: PVA 40% composite Color Light yellow Diameter 3.0 mm Height 3.0 mm Densit y 1 .2 2kg/m3 Draw intensity ≥15 MPa Specific surface area 1735 m2/m3 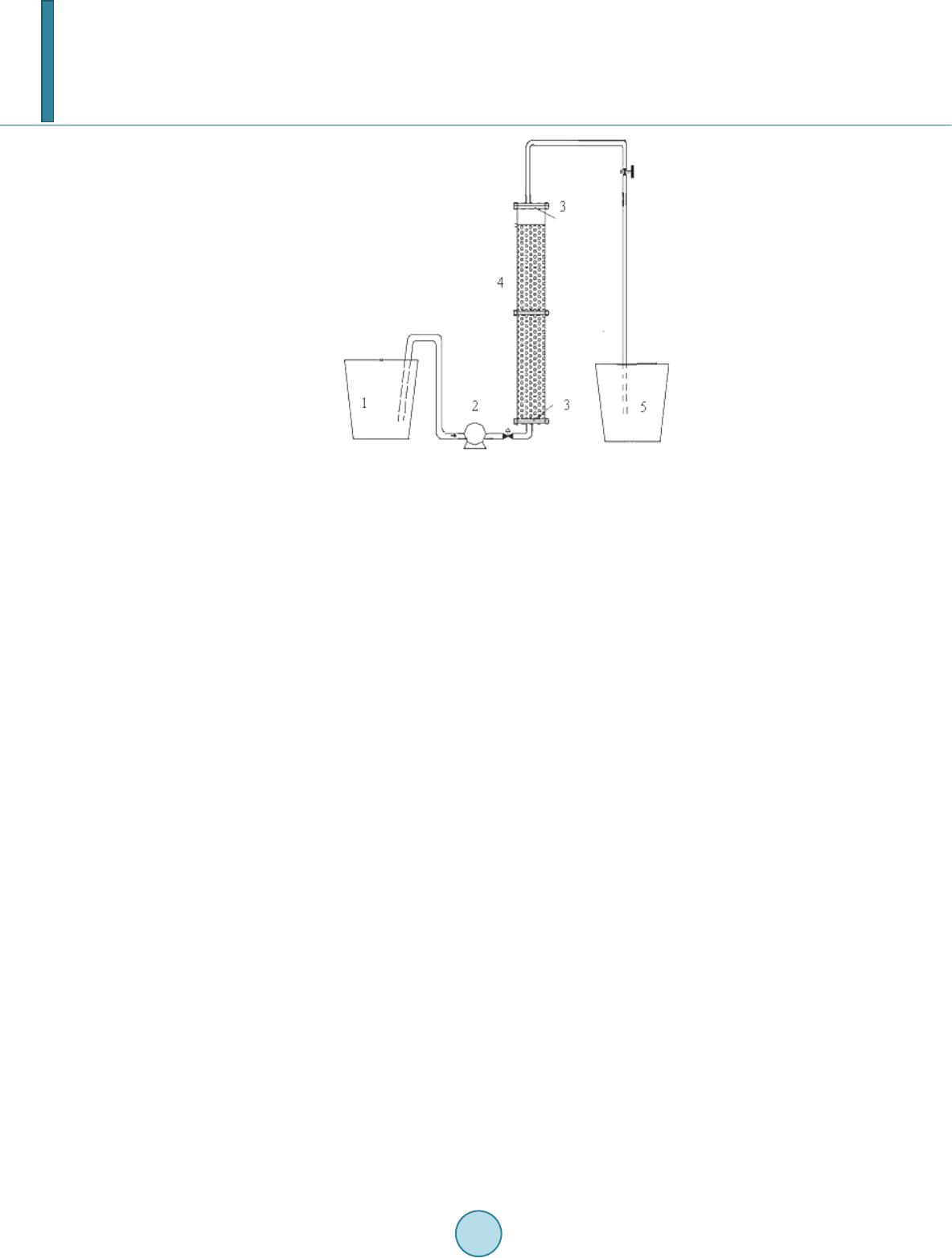 F. He et al. Figure 1. Process scheme of pac ked-bed reactor (1) in- fluent reservoir (2) pump (3) glass wool (4) column (5) effluent tank. 3. Results and Discussion 3.1. Simultaneous Removal Performance of and in the Polymer Bioreactor The microcosm degradation curves of and NO3 -N spiked into bioreactor are illustrated in Fig ure 2. The Starc h/PVA biofilm reactor was treated at the initial ClO4- concentration of 2 ppm and NO3-N concentra- tion of 20 ppm, corresponding to the nitrate concentration at a typical perchlorate-contaminated site. Indicating rapid decrease at the initial period, NO3-N dropped to 0.11 mg/L after 4 h from the spiked concentration of 20 mg/L and was totally disappeared after running for 5 h. Compared to NO3-N, the degradation of took place in a manner much slower. After running for 5 h, the residual in the reactor fell only to about 500 μg/L, a much higher l e ve l. Complete disappearance of the spiked was noticed in association with the data point after running for 9 h. 3.2. Effect of Influent pH To examine the effect of the pH of the water sample on the reduction of perchlorate and nitrate in the bioreactor, a series of experiments were performed by changing the pH of the water samples from 5 to 10.0, as shown in Figure 3. The results indicated that the optimal pH of perchlorate and nitrate was pH 6.7. The influent pH had signif i- cant influence on perchlorate reduction but has sli gh t effect on nitrate reduction, which may be attributed to tha t denitrification biofilm on polymer surface can tolerate a certain pH shock and perchlorate reduction is affected by many factors. Owing to the density structure of the biofilm on starch/PVA polymer surface, it protected the internal degradation bacteria and could not lead to significant removal decrease. 3.3. Performance of the Packed Bed Bioreactor Afte r the end of start-up period (for 40 days), the flow rate was changed to 1.17 mL/min and the influent per- chlorate and nitrate was removed completely (Figure 4). During the following period, the perchlorate and nitrate removal varied in the range of 84% - 100% and 97% - 100%, respectively, while the nitrite concentration re- mained between 0.004 and 0.015 mgNO2-N/L. Except one sample on the 70th day effluent nitrite was as high as 0.26 mgNO2-N/ L. After the effluent perchlorate and nitrate changed steadily, the effluent DOC level (ca.10 mg/L) became rela- tive stable. The above results demonstrated clearly that packed bed reactor filled with starch/PVA polymer gra- nules could effectively and synchronously remove perchlorate and nitrate from drinking water. 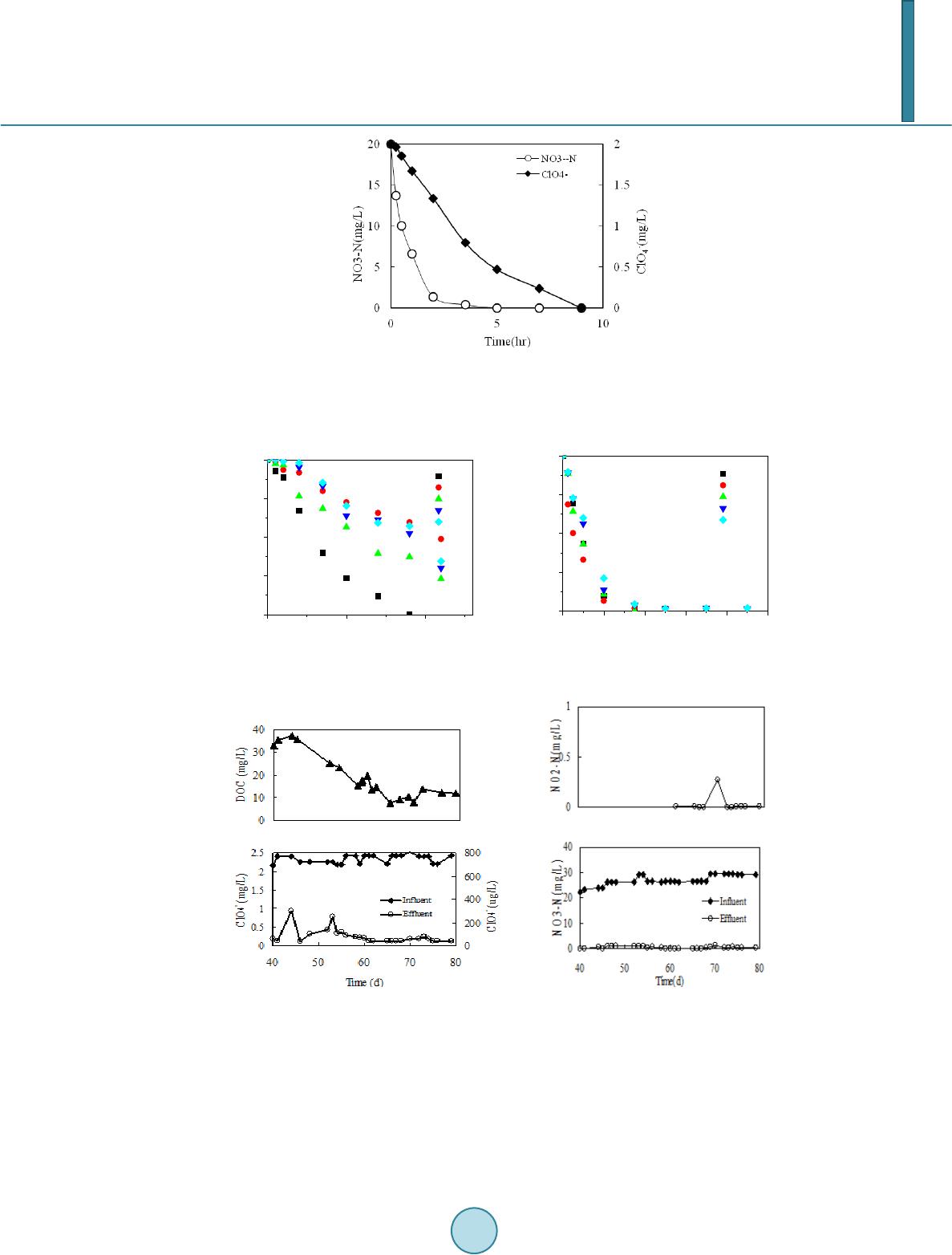 F. He et al. Figure 2. Concentration profiles of and NO3-N operated by spiking 2 mg/L of and 20 mg/L of NO3-N. Figure 3. Effect of pH on nitrate and perchlorate degradation. (a) (b) Figure 4. (a) Concentration profiles of DOC and perchlorate in effluent in polymer bi oreact or; (b) Concentration profiles of nitrite and nitrate in effluent in polymer bi oreacto r. 3.4. Biofilm Development and Changes of PBS St ru c t u re Figure 5(a) showed biofilm coverage on the outer surfaces of the carriers was dense, which had a good layered structure and primarily composed of bacillus and coccus. Due to the subsequent degradation of the carrier material the diameter decreased by inches over the cultivation 05 10 0. 0 0. 5 1. 0 1. 5 2. 0 ClO 4 - ( mg /L) Time(hr) 6.7 5.55 8 9 10 0 2 4 6 810 0 5 10 15 20 5.55 6.7 8 9 10 NO3-N(mg/ L ) Time(hr)  F. He et al. (a) (b) (c) Figure 5. The biofilm morphology on starch/PVA polymer surface (a). The surface morphology of raw (b) and used (c) ma- terial of starch/PV A polymer after cultivation. time. Especially during the experimental phase the chink and filament structure on the carrier surface developed, the surface of the raw material was oppositely smooth and had no chink and filament. The deepness of the pores reached several hundred micrometers and, thus, provided space for anoxic and anaerobic microbial activity. In- side the pores of the carrier large amounts of bacteria could be detected. The results indicated that after biofilm formed, the microorganisms in biofilm decomposed PBS through me- tabolism and provided carbon source for themselves by releasing small organic molecules, which causing the changes of PBS surface morphology. 4. Conclusion The results presented clearly show that the biodegradable polymers process allows for a simultaneous removal of perchlorate and nitrate from contaminated drinking water to below their recommended limits, without sec- ondary c o ntami nat ion. The results showed that complete denitrifi cation only for 5 h and below the detection limit of perchlorate within 9 h occured at a typical perchlorate-contaminated simulate reactor. Morphological observation indicated the developed biofilm coverage on the outer surfaces of the carriers was dense and primar- ily composed of bacillus and coccus. The microbes in biofilm decomposed polymer, the chink and filament structure on the carrier surface developed, through metabolism and provided carbon source for them by releas- ing small organic molecules. Acknowledgements This work was financially supported by the National Natural Science Foundation of China (No. 21107031), the Doctor Foundation of Shandong Province, China (No. BS2010HZ003, BS2010HZ004), and the Project of Jinan Science and Technology Board (No. 201202261). References Baidasa, S., Ga o , B. Y., & Meng, X. G. (2011). Perchlorate Removal by Quaternary Amine Modifi ed Reed. Journal of Ha- zardous Materials, 189, 54-61. http://dx.doi.org/10.1016/j.jhazmat.2011.01.124 Bauchard, D. C., Williams, M. K., & Surampalli, R. Y. (1992). Nitrate Contamination of Groundwater: Sources and Poten- tial Health Effects. Journal of the American Water Works Association, 84, 85-90 . Choi, H., & JoAnn, S. (2008). Inhibition of Perchlorate Reduction by Nitrate in a Fixed Biofilm Reactor. Journal of Hazardous Materials, 159, 440 -445. http://dx.doi.org/10.1016/j.jhazmat.2008.02.038 Coates, J. D., & Achenbach , L. A. (2004). Microbial Perchlorate Reduction: Rocket-Fuelled Metabolism. Nature Reviews Microbiology, 2, 569-580. http://dx.doi.org/10.1038/nrmicro926 Coates, J. D., & Anderson, R. T. (2000 ). Emerging Techniques for Anaerobic Bioremediation of Contaminated Environ- ments. Trends in Biotechnology, 18, 408-412. http://dx.doi.org/10.1016/S0167-7799(00)01478-5 Ghosh, A., Pakshirajan, K., Ghosh, P. K., & Sahoo, N. K. (2011). Perchlorate Degradation Using an Indigenous Microbial  F. He et al. Consortium Predominantly Burkholderia sp. Journal of Hazardous Materials, 187, 133 -139. http://dx.doi.org/10.1016/j.jhazmat.2010.12.130 Herman, D. C., & Frankenberger, W. T. (1999). Bacterial Reduction of Perchlorate and Nitrate in Water. Journal of Envi- ronmental Quality, 28, 1018-10 24 . http://dx.doi.org/10.2134/jeq1999.00472425002800030036x Shrout, J. D., Struckhoff, G. C., Parkin, G. F., & Schnoor, J. L. (2006). Stimulation and Molecular Characteri zatio n of Bac- terial Perchlorate Degradation by Plant-Produced Electron Donors. Environmental Science & Technology, 40, 310-317 . http://dx.doi.org/10.1021/es051130g Srin ivasan, R., & Sorial, G. A. (2009). Treatment of Perchlorate in Drinking Water: A Critical Review. Separation and Puri- fication Technology, 69, 7-21. http://dx.doi.org/10.1016/j.seppur.2009.06.025 Wallace, W., Beshear, S., Williams, D., Hospadar, S., & Owe ns, M. (1998 ). Perchlorate Reduction by Amixed Culture in an Upflow Anaerobic Fixed Bed Reactor. Journal of Industrial Microbiology and Biotechnology, 20, 126-131. http://dx.doi.org/10.1038/sj.jim.2900494 Xu, J., Song, Y., Min, B., Steinberg, L., & Logan, B. E. (2003). Microb ial Degradation of Perchlorate Principles and Ap- plications. Environmental Engineering Science, 20, 405-422 . http://dx.doi.org/10.1089/109287503768335904 Xu, J., Timble, J. J., Steinberg, L., & Logan, B. E. (2004). Chlorate and Nitrate Reduction Pathways Are Separately Induced in the Per chlorat e-Respiring Bacterium Dechlorosoma sp. KJ and the Chlorate-Respiring Bacterium Pseudomonas sp. PDA. Water Research, 38 , 673 -680 . http://dx.doi.org/10.1016/j.watres.2003.10.017 Zhou , H. H., Zhao, X., & Wan g , J. L. (2009). Nitrate Removal from Groundwater Using Biodegradable Polymers as Carbon Source and Biofilm Support. International Journal of Environment and Pollution, 38, 339 -348. http://dx.doi.org/10.1504/IJEP.2009.027234
|