Paper Menu >>
Journal Menu >>
 Materials Sciences and Applications, 2011, 220-225 doi:10.4236/msa.2011.24028 Published Online April 2011 (http://www.scirp.org/journal/msa) Copyright © 2011 SciRes. MSA Corrosion Inhibition of Copper in 3% NaCl Solution by Derivative of Aminotriazole Karima Rhattas1, Mohammed Benmessaoud1,2, Mostafa Doubi1, Najjat Hajjaji1, Abdellah Srhiri1 1Laboratory of Electrochemistry, Materials and Environment (LEME), Faculty of Sciences, Ibn Tofail University, Morocco; 2University of Mohammed V Agdal, Energy Systems Laboratory, Materials and Environment, High School of Technology, Salé Medina, Morocco E-mail: benmessaoud_mma@yahoo.fr Received November 6th, 2010; revised January 26th, 2011; accepted February 21st, 2011. ABSTRACT The inhibiting effect of N-decyl-3-amino-1,2,4-triazole (TN10) against Copper corrosion in aerated 3% NaCl, has been developed. Potentiodynamic measurements and electrochemical impedance spectroscopy have been applied to deter- mine the corrosion rate. Scanning electron microscopy (SEM) studied surface morphology has been also used to char- acterize electrode surface. The obtained results indicate that TN10 acts as a good mixed-type inhibitor retarding both anodic and cathodic reactions. An increase of TN10 concentration leads to a decrease of corrosion rate and inhibition efficiency increase. Keywords: Copper, Triazole, 3% NaCl Solution, Corrosion, Inhibition 1. Introduction Due to its high electrical and thermal conductivity and good mechanical workability, copper is a material widely used in pipelines for domestic and industrial water utili- ties, including sea water, heat conductors, and heat ex- changers [1]. In spite of the relatively noble potential of copper, its corrosion takes place at a significant rate in sea water and chloride environments [2-9]. It is generally accepted that the anodic dissolution of copper in chloride environments is influenced by the chloride ions concen- tration. At chloride concentrations lower than 1 M, the dissolution of copper occurs through the formation of CuCl, which is not protective enough and is converted to the soluble CuCl2 by reacting with excess chloride [10]. Heterocyclic compounds, especially nitrogen-based ones, are effective inhibitors, being coordinate with Cu(0), Cu(I) or Cu(II) via their nitrogen atoms through lone pair electrons to form polymeric complexes with copper [11-17]. These form an adsorbed protective film on the copper surface, providing inhibition of corrosion by acting as a barrier to aggressive ions such as chloride [18-20]. Polar functional groups such as nitrogen, sulfur, and oxygen and conjugated double bonds are considered reaction centers in establishing the adsorption process [21,22]. In others previous work, the influence of organic compounds on the corrosion of copper in neutral solution have been studied [23-28]. The present investigations discusses the results ob- tained in studying the corrosion behavior of pure copper in aerated 3% NaCl solution in absence and in presence of an aminotriazole derivative like N-decyl-3-amino-1, 2,4-triazole noted (TN10). The electrochemical methods and surface characterization have been used to charac- terize the inhibiting effect. 2. Experimental Conditions A classic electrochemical cell with three-electrode con- figuration was used in this study: a platinum grid as a counter electrode, a rotating disk of pure copper (99.9%) as working electrode, and Ag/AgCl in 3 M KCl (SSE) as a reference one. The working electrode was made of cylinder rod of pure copper (Goodfellow) of 12.5 mm in diameter. A cylinder rod of about 1 cm height was fixed to a stain- less-steel shaft, and then the lateral part was covered with a cataphoretic epoxyamine base paint (PPG; WT724 + P962). First, the paint was deposited at a constant voltage of 180 V during 4 min, and then cured at 180˚C for 30 min. After that, the electrode was embedded into an ep- oxy resin (Buhler; Epoxycure), and worked out to the cylinder shape, the outer diameter was 21 mm. Only the cross-section of the alloy rod embedded in the epoxy resin was used to form a rotating disk electrode. The cataphoretic coating allowed avoiding any infiltration of electrolyte between the metal and epoxy resin interface. 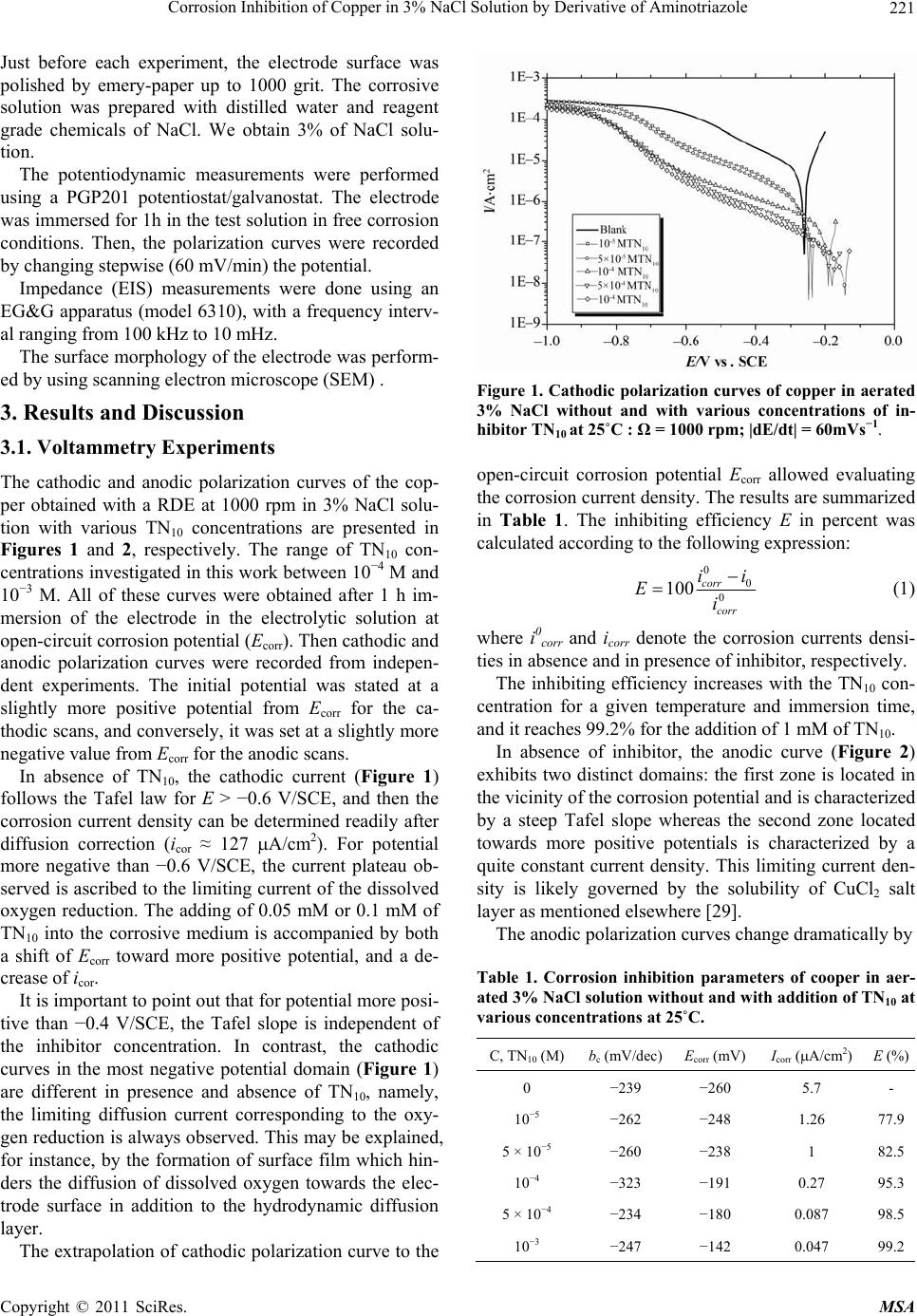 Corrosion Inhibition of Copper in 3% NaCl Solution by Derivative of Aminotriazole Copyright © 2011 SciRes. MSA 221 Just before each experiment, the electrode surface was polished by emery-paper up to 1000 grit. The corrosive solution was prepared with distilled water and reagent grade chemicals of NaCl. We obtain 3% of NaCl solu- tion. The potentiodynamic measurements were performed using a PGP201 potentiostat/galvanostat. The electrode was immersed for 1h in the test solution in free corrosion conditions. Then, the polarization curves were recorded by changing stepwise (60 mV/min) the potential. Impedance (EIS) measurements were done using an EG&G apparatus (model 6310), with a frequency interv- al ranging from 100 kHz to 10 mHz. The surface morphology of the electrode was perform- ed by using scanning electron microscope (SEM) . 3. Results and Discussion 3.1. Voltammetry Experiments The cathodic and anodic polarization curves of the cop- per obtained with a RDE at 1000 rpm in 3% NaCl solu- tion with various TN10 concentrations are presented in Figures 1 and 2, respectively. The range of TN10 con- centrations investigated in this work between 10−4 M and 10−3 M. All of these curves were obtained after 1 h im- mersion of the electrode in the electrolytic solution at open-circuit corrosion potential (Ecorr). Then cathodic and anodic polarization curves were recorded from indepen- dent experiments. The initial potential was stated at a slightly more positive potential from Ecorr for the ca- thodic scans, and conversely, it was set at a slightly more negative value from Ecorr for the anodic scans. In absence of TN10, the cathodic current (Figure 1) follows the Tafel law for E > −0.6 V/SCE, and then the corrosion current density can be determined readily after diffusion correction (icor ≈ 127 A/cm2). For potential more negative than −0.6 V/SCE, the current plateau ob- served is ascribed to the limiting current of the dissolved oxygen reduction. The adding of 0.05 mM or 0.1 mM of TN10 into the corrosive medium is accompanied by both a shift of Ecorr toward more positive potential, and a de- crease of icor. It is important to point out that for potential more posi- tive than −0.4 V/SCE, the Tafel slope is independent of the inhibitor concentration. In contrast, the cathodic curves in the most negative potential domain (Figure 1) are different in presence and absence of TN10, namely, the limiting diffusion current corresponding to the oxy- gen reduction is always observed. This may be explained, for instance, by the formation of surface film which hin- ders the diffusion of dissolved oxygen towards the elec- trode surface in addition to the hydrodynamic diffusion layer. The extrapolation of cathodic polarization curve to the Figure 1. Cathodic polarization curves of copper in aerated 3% NaCl without and with various concentrations of in- hibitor TN10 at 25˚C : Ω = 1000 rpm; |dE/dt| = 60mVs−1. open-circuit corrosion potential Ecorr allowed evaluating the corrosion current density. The results are summarized in Table 1. The inhibiting efficiency E in percent was calculated according to the following expression: 0 0 0 100 corr corr ii Ei (1) where i0corr and icorr denote the corrosion currents densi- ties in absence and in presence of inhibitor, respectively. The inhibiting efficiency increases with the TN10 con- centration for a given temperature and immersion time, and it reaches 99.2% for the addition of 1 mM of TN10. In absence of inhibitor, the anodic curve (Figure 2) exhibits two distinct domains: the first zone is located in the vicinity of the corrosion potential and is characterized by a steep Tafel slope whereas the second zone located towards more positive potentials is characterized by a quite constant current density. This limiting current den- sity is likely governed by the solubility of CuCl2 salt layer as mentioned elsewhere [29]. The anodic polarization curves change dramatically by Table 1. Corrosion inhibition parameters of cooper in aer- ated 3% NaCl solution without and with addition of TN10 at various concentrations at 25˚C. C, TN10 (M)bc (mV/dec)Ecorr (mV) Icorr (A/cm2)E (%) 0 −239 −260 5.7 - 10−5 −262 −248 1.26 77.9 5 × 10−5 −260 −238 1 82.5 10−4 −323 −191 0.27 95.3 5 × 10−4 −234 −180 0.087 98.5 10−3 −247 −142 0.047 99.2 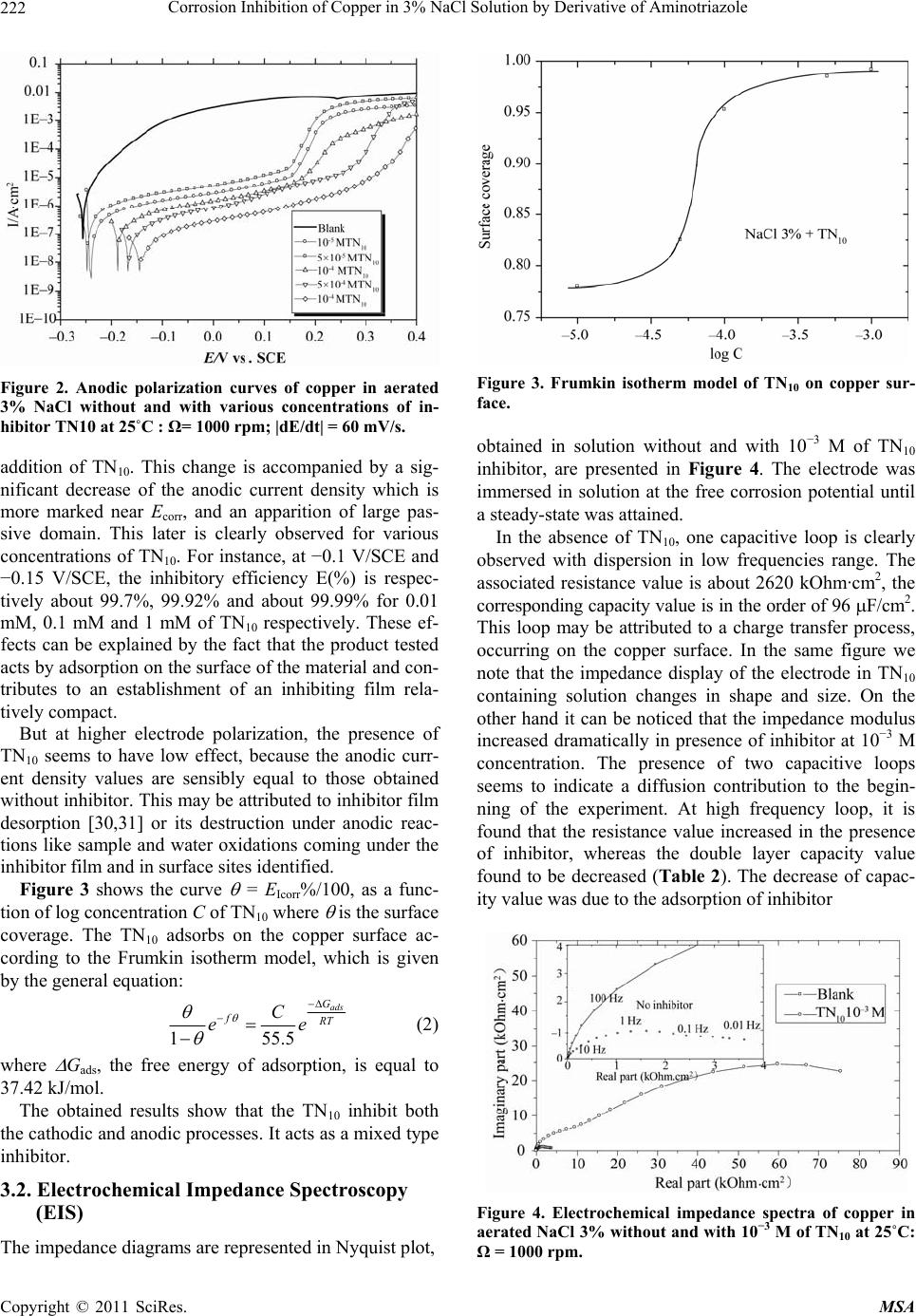 Corrosion Inhibition of Copper in 3% NaCl Solution by Derivative of Aminotriazole Copyright © 2011 SciRes. MSA 222 Figure 2. Anodic polarization curves of copper in aerated 3% NaCl without and with various concentrations of in- hibitor TN10 at 25˚C : Ω= 1000 rpm; |dE/dt| = 60 mV/s. addition of TN10. This change is accompanied by a sig- nificant decrease of the anodic current density which is more marked near Ecorr, and an apparition of large pas- sive domain. This later is clearly observed for various concentrations of TN10. For instance, at −0.1 V/SCE and −0.15 V/SCE, the inhibitory efficiency E(%) is respec- tively about 99.7%, 99.92% and about 99.99% for 0.01 mM, 0.1 mM and 1 mM of TN10 respectively. These ef- fects can be explained by the fact that the product tested acts by adsorption on the surface of the material and con- tributes to an establishment of an inhibiting film rela- tively compact. But at higher electrode polarization, the presence of TN10 seems to have low effect, because the anodic curr- ent density values are sensibly equal to those obtained without inhibitor. This may be attributed to inhibitor film desorption [30,31] or its destruction under anodic reac- tions like sample and water oxidations coming under the inhibitor film and in surface sites identified. Figure 3 shows the curve = EIcorr%/100, as a func- tion of log concentration C of TN10 where is the surface coverage. The TN10 adsorbs on the copper surface ac- cording to the Frumkin isotherm model, which is given by the general equation: 1 55.5 ads G fRT C ee (2) where Gads, the free energy of adsorption, is equal to 37.42 kJ/mol. The obtained results show that the TN10 inhibit both the cathodic and anodic processes. It acts as a mixed type inhibitor. 3.2. Electrochemical Impedance Spectroscopy (EIS) The impedance diagrams are represented in Nyquist plot, Figure 3. Frumkin isotherm model of TN10 on copper sur- face. obtained in solution without and with 10−3 M of TN10 inhibitor, are presented in Figure 4. The electrode was immersed in solution at the free corrosion potential until a steady-state was attained. In the absence of TN10, one capacitive loop is clearly observed with dispersion in low frequencies range. The associated resistance value is about 2620 kOhm·cm2, the corresponding capacity value is in the order of 96 F/cm2. This loop may be attributed to a charge transfer process, occurring on the copper surface. In the same figure we note that the impedance display of the electrode in TN10 containing solution changes in shape and size. On the other hand it can be noticed that the impedance modulus increased dramatically in presence of inhibitor at 10−3 M concentration. The presence of two capacitive loops seems to indicate a diffusion contribution to the begin- ning of the experiment. At high frequency loop, it is found that the resistance value increased in the presence of inhibitor, whereas the double layer capacity value found to be decreased (Table 2). The decrease of capac- ity value was due to the adsorption of inhibitor Figure 4. Electrochemical impedance spectra of copper in aerated NaCl 3% without and with 10−3 M of TN10 at 25˚C: Ω = 1000 rpm. 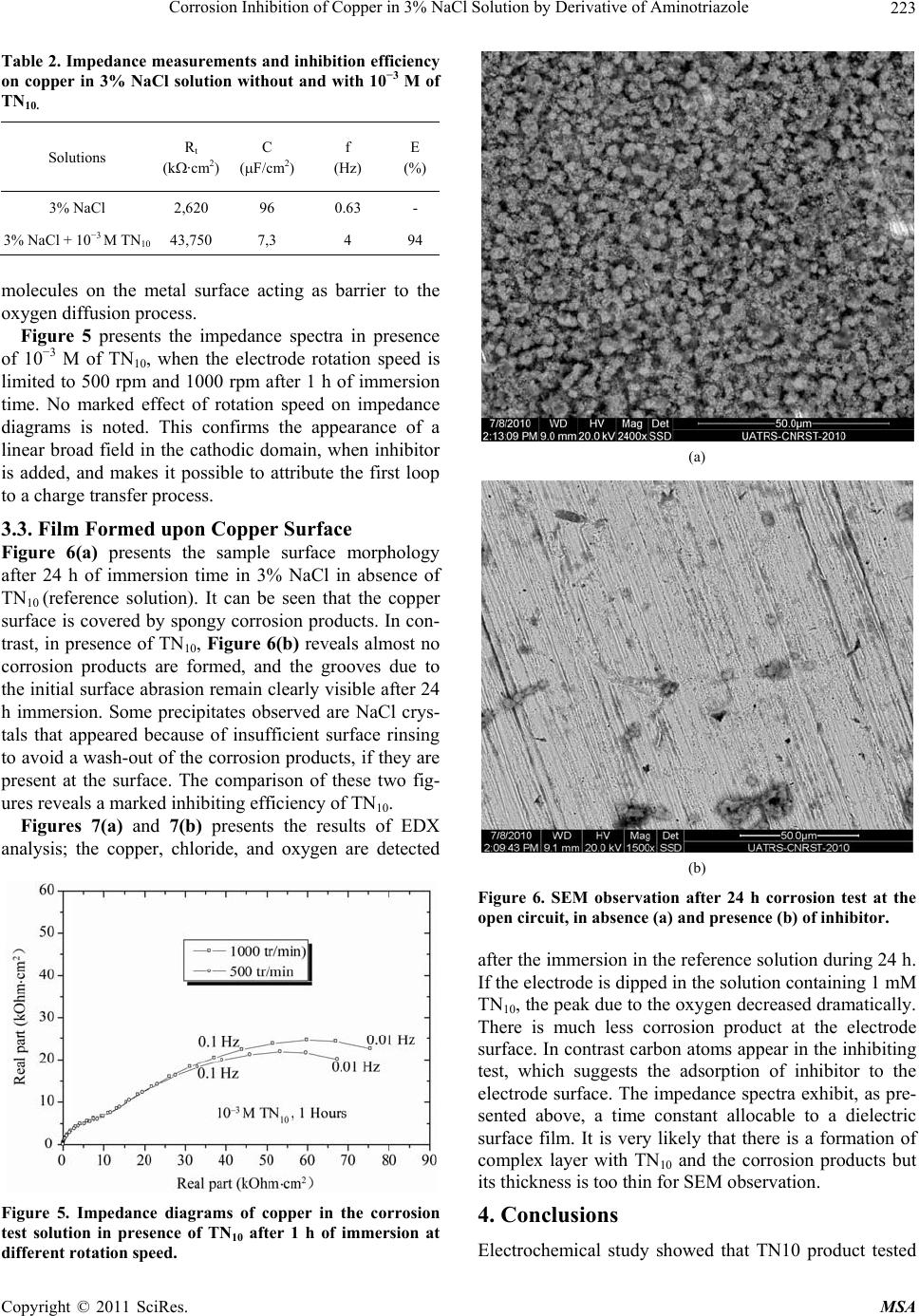 Corrosion Inhibition of Copper in 3% NaCl Solution by Derivative of Aminotriazole Copyright © 2011 SciRes. MSA 223 Table 2. Impedance measurements and inhibition efficiency on copper in 3% NaCl solution without and with 10−3 M of TN10. Solutions Rt (k·cm2) C (F/cm2) f (Hz) E (%) 3% NaCl 2,620 96 0.63 - 3% NaCl + 10−3 M TN10 43,750 7,3 4 94 molecules on the metal surface acting as barrier to the oxygen diffusion process. Figure 5 presents the impedance spectra in presence of 10−3 M of TN10, when the electrode rotation speed is limited to 500 rpm and 1000 rpm after 1 h of immersion time. No marked effect of rotation speed on impedance diagrams is noted. This confirms the appearance of a linear broad field in the cathodic domain, when inhibitor is added, and makes it possible to attribute the first loop to a charge transfer process. 3.3. Film Formed upon Copper Surface Figure 6(a) presents the sample surface morphology after 24 h of immersion time in 3% NaCl in absence of TN10 (reference solution). It can be seen that the copper surface is covered by spongy corrosion products. In con- trast, in presence of TN10, Figure 6(b) reveals almost no corrosion products are formed, and the grooves due to the initial surface abrasion remain clearly visible after 24 h immersion. Some precipitates observed are NaCl crys- tals that appeared because of insufficient surface rinsing to avoid a wash-out of the corrosion products, if they are present at the surface. The comparison of these two fig- ures reveals a marked inhibiting efficiency of TN10. Figures 7(a) and 7(b) presents the results of EDX analysis; the copper, chloride, and oxygen are detected Figure 5. Impedance diagrams of copper in the corrosion test solution in presence of TN10 after 1 h of immersion at different rotation speed. (a) (b) Figure 6. SEM observation after 24 h corrosion test at the open circuit, in absence (a) and presence (b) of inhibitor. after the immersion in the reference solution during 24 h. If the electrode is dipped in the solution containing 1 mM TN10, the peak due to the oxygen decreased dramatically. There is much less corrosion product at the electrode surface. In contrast carbon atoms appear in the inhibiting test, which suggests the adsorption of inhibitor to the electrode surface. The impedance spectra exhibit, as pre- sented above, a time constant allocable to a dielectric surface film. It is very likely that there is a formation of complex layer with TN10 and the corrosion products but its thickness is too thin for SEM observation. 4. Conclusions Electrochemical study showed that TN10 product tested 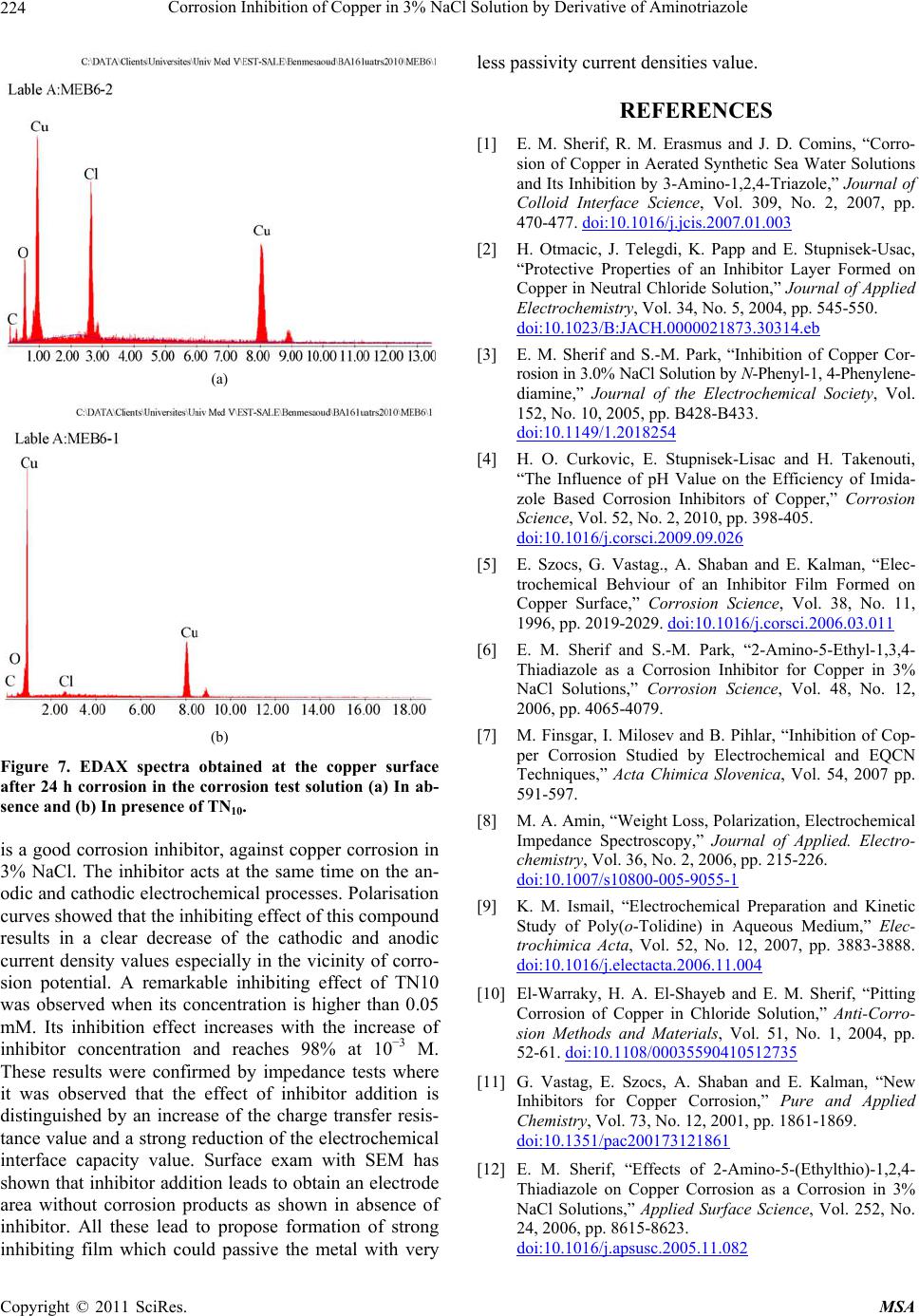 Corrosion Inhibition of Copper in 3% NaCl Solution by Derivative of Aminotriazole Copyright © 2011 SciRes. MSA 224 (a) (b) Figure 7. EDAX spectra obtained at the copper surface after 24 h corrosion in the corrosion test solution (a) In ab- sence and (b) In presence of TN10. is a good corrosion inhibitor, against copper corrosion in 3% NaCl. The inhibitor acts at the same time on the an- odic and cathodic electrochemical processes. Polarisation curves showed that the inhibiting effect of this compound results in a clear decrease of the cathodic and anodic current density values especially in the vicinity of corro- sion potential. A remarkable inhibiting effect of TN10 was observed when its concentration is higher than 0.05 mM. Its inhibition effect increases with the increase of inhibitor concentration and reaches 98% at 10−3 M. These results were confirmed by impedance tests where it was observed that the effect of inhibitor addition is distinguished by an increase of the charge transfer resis- tance value and a strong reduction of the electrochemical interface capacity value. Surface exam with SEM has shown that inhibitor addition leads to obtain an electrode area without corrosion products as shown in absence of inhibitor. All these lead to propose formation of strong inhibiting film which could passive the metal with very less passivity current densities value. REFERENCES [1] E. M. Sherif, R. M. Erasmus and J. D. Comins, “Corro- sion of Copper in Aerated Synthetic Sea Water Solutions and Its Inhibition by 3-Amino-1,2,4-Triazole,” Journal of Colloid Interface Science, Vol. 309, No. 2, 2007, pp. 470-477. doi:10.1016/j.jcis.2007.01.003 [2] H. Otmacic, J. Telegdi, K. Papp and E. Stupnisek-Usac, “Protective Properties of an Inhibitor Layer Formed on Copper in Neutral Chloride Solution,” Journal of Applied Electrochemistry, Vol. 34, No. 5, 2004, pp. 545-550. doi:10.1023/B:JACH.0000021873.30314.eb [3] E. M. Sherif and S.-M. Park, “Inhibition of Copper Cor- rosion in 3.0% NaCl Solution by N-Phenyl-1, 4-Phenylene- diamine,” Journal of the Electrochemical Society, Vol. 152, No. 10, 2005, pp. B428-B433. doi:10.1149/1.2018254 [4] H. O. Curkovic, E. Stupnisek-Lisac and H. Takenouti, “The Influence of pH Value on the Efficiency of Imida- zole Based Corrosion Inhibitors of Copper,” Corrosion Science, Vol. 52, No. 2, 2010, pp. 398-405. doi:10.1016/j.corsci.2009.09.026 [5] E. Szocs, G. Vastag., A. Shaban and E. Kalman, “Elec- trochemical Behviour of an Inhibitor Film Formed on Copper Surface,” Corrosion Science, Vol. 38, No. 11, 1996, pp. 2019-2029. doi:10.1016/j.corsci.2006.03.011 [6] E. M. Sherif and S.-M. Park, “2-Amino-5-Ethyl-1,3,4- Thiadiazole as a Corrosion Inhibitor for Copper in 3% NaCl Solutions,” Corrosion Science, Vol. 48, No. 12, 2006, pp. 4065-4079. [7] M. Finsgar, I. Milosev and B. Pihlar, “Inhibition of Cop- per Corrosion Studied by Electrochemical and EQCN Techniques,” Acta Chimica Slovenica, Vol. 54, 2007 pp. 591-597. [8] M. A. Amin, “Weight Loss, Polarization, Electrochemical Impedance Spectroscopy,” Journal of Applied. Electro- chemistry, Vol. 36, No. 2, 2006, pp. 215-226. doi:10.1007/s10800-005-9055-1 [9] K. M. Ismail, “Electrochemical Preparation and Kinetic Study of Poly(o-Tolidine) in Aqueous Medium,” Elec- trochimica Acta, Vol. 52, No. 12, 2007, pp. 3883-3888. doi:10.1016/j.electacta.2006.11.004 [10] El-Warraky, H. A. El-Shayeb and E. M. Sherif, “Pitting Corrosion of Copper in Chloride Solution,” Anti-Corro- sion Methods and Materials, Vol. 51, No. 1, 2004, pp. 52-61. doi:10.1108/00035590410512735 [11] G. Vastag, E. Szocs, A. Shaban and E. Kalman, “New Inhibitors for Copper Corrosion,” Pure and Applied Chemistry, Vol. 73, No. 12, 2001, pp. 1861-1869. doi:10.1351/pac200173121861 [12] E. M. Sherif, “Effects of 2-Amino-5-(Ethylthio)-1,2,4- Thiadiazole on Copper Corrosion as a Corrosion in 3% NaCl Solutions,” Applied Surface Science, Vol. 252, No. 24, 2006, pp. 8615-8623. doi:10.1016/j.apsusc.2005.11.082  Corrosion Inhibition of Copper in 3% NaCl Solution by Derivative of Aminotriazole Copyright © 2011 SciRes. MSA 225 [13] C. T. Wang, S. H. Chen and S. Y. Zhao, “Inhibition Ef- fect of Ac-Treated Mixed Self-Assembled Film of Phenylthiourea and 1-Dodecanethiol on Cooper Corro- sion, Journal of the Electrochemical Society, Vol. 151, No. 1, 2004, pp. B11-B15. doi:10.1149/1.1630595 [14] L. Nunèz, E. Reguera, F. Corvo, F. Gonzalez and C. Vazquez, “Corrosion of Copper in Seawater and Its Aerosols in a Tropical Island,” Corrosion Science, Vol. 47, No. 2, 2005, pp. 461-484. doi:10.1016/j.corsci.2004.05.015 [15] E. M. Sherif, R. M. Erasmus and J. D. Comins, “Effects of 3-Amino-1,2,4-Triazole on the Inhibition of Copper Corrosion in Acidic Chloride Solutions,” Journal of Col- loid Interface Science, Vol. 311, No. 1, 2007, pp. 144- 151. doi:10.1016/j.jcis.2007.02.064 [16] F. Zucchi, G. Trabanelli and C. Monticelli, “The Inhibi- tion of Copper Corrosion in 0.1 M NaCl under Heat Ex- change Conditions,” Corrosion Science, Vol. 38, No. 1, 1996, pp. 147-154. doi:10.1016/0010-938X(96)00121-7 [17] V. Lakshminarayanan, K. Kannan and S. R. Rajagopalan, “Cyclic Voltammetric Behavior of Certain Copper-Azole Systems Using Carbon Paste Electrodes,” Journal of Electroanalytical Chemistry, Vol. 364, No. 1-2, 1994, pp. 79-86. doi:10.1016/0022-0728(93)02924-7 [18] M. Scendo and M. Hepel, “Inhibiting Properties of Ben- zimidazole Films for Cu(II)/Cu(I) Reduction in Chloride Media Studied by RDE and EQCN Techniques,” Journal of Electroanalytical Chemistry, Vol. 613, No. 1, 2008, pp. 35-50. doi:10.1016/j.jelechem.2007.10.014 [19] E. M. M Sutter, F. Ammeloot, M. J. Pouet, C. Fiaud and R. Couffignal, “Heterocyclic Compounds Used as Corro- sion Inhibitors: Correlation between 13C and 1H NMR Spectroscopy and Inhibition Efficiency,” Corrosion Sci- ence, Vol. 41, No. 1, 1999, pp. 105-111. doi:10.1016/S0010-938X(98)00099-7 [20] F. Zucchi, G. Trabanelli and M. Fonsati, “Tetrazole De- rivatives as Corrosion Inhibitors for Copper in Chloride Solutions,” Corrosion Science, Vol. 38, No. 11, 1996, pp. 2019-2029. doi:10.1016/S0010-938X(96)00094-7 [21] E. M. Sherif and S.-M. Park, “Effect of 2-Amino-5- Ethylthio-1,3,4-Thiadiazole on Copper Corrosion Aerated Acidic Pickling Solutions,” Electrochimica Acta, Vol. 51, No. 28, 2006, pp. 6556-6562. doi:10.1016/j.electacta.2006.04.047 [22] K. F. Khaled, “Guanidine Derivative as a New Corrosion Inhibitor for Copper in 3% NaCl Solution,” Materials Chemistry and Physics, Vol. 112, No. 1, 2008, pp. 104-111. doi:10.1016/j.matchemp hy s.2008. 05.052 [23] El-Sayed M. Sherif, R. M. Eramus and J. D. Comins “In- hibition of Corrosion Processes on Copper in Aerated So- dium Chloride Solution by 5-(Aminophenyl)-Tetrazole,” Journal of Applied Electrochemistry, Vol. 39, No. 1, 2009, pp. 83-91. doi:10.1007/s10800-008-9641-0 [24] El-Sayed M. Sherif, A. M. El Shamy, M. M. Ramla and A. O. H. El Nazhawy, “5-(Phenyl)4H-1,2,4-Triazole-3-Thiol as a Corrosion Inhibitor for Copper in 3.5% NaCl Solu- tions,” Materials Chemistry and Physics, Vol. 102, No. 2-3, 2007, pp. 231-239. [25] M. Yadav and D. Sharma, “Dithiobiruts as Corrosion Inhibitors for Copper in 3.5% NaCl Solution,” Portuga- liae Electrochimica Acta, Vol. 28, No. 1, 2010, pp. 51-62. doi:10.4152/pea.201001051 [26] El-Sayed M. Sherif and A. A. Almajid, “Surface Protec- tion of Copper in Aerated 3.5% Sodium Chloride Solu- tions by 3-Amino-5-Mercapto-1,2,4-Triazole as a Copper Corrosion Inhibitor,” Journal Applied Electrochem, Vol. 40, No. 8, 2010, pp. 1555-1562. doi:10.1007/s10800-010-0140-8 [27] H. O. Curkovic, E. Stupnisek-Lisac and H. Takenouti “Electrochemical Quartz Crystal Microbalance and Elec- trochemical Impedance Spectroscopy Study of Copper Corrosion Inhibition by Imidazoles,” Corrosion Science, Vol. 51, No. 10, October 2009, pp. 2342-2348. doi:10.1016/j.corsci.2009.06.018 [28] E. D’Elia, O. E. Barcia, O. R. Mattos, N. Pébère and B. Tribollet, “High-Rate Copper Dissolution in Hydrochloric Acid Solution,” Journal of the Electrochemical Society, Vol. 143, No. 3, 1996, pp. 961-967. doi:10.1149/1.1836565 [29] Z. A. Chikh, “Contribution à L'étude de L'inhibition de la Corrosion d'un Alliage Cu-Zn en Milieu NaCl 3% par Couplage de Méthodes Électrochimiques et Spectro- scopiques,” Doctoral Dissertation, Ibn Tofail University, Kenitra, 2001. [30] Z. A. Chikh, B. Assouli, A. B. Bachir, N. Hajjaji, A. Frignani, F. Zucchi and A. Srhiri, “A New Corrosion In- hibitor for Cu60-Zn Brass in 3% NaCl Solution,” Pro- ceedings of the 9th European Symposium on Corrosion Inhibitors, Ferrara, Vol. 2, 2000, pp. 687-698. [31] A. Srhiri, B. Trachli, N. Hajjaji, M. Keddam, H. Ta- kenouti, A. Frignani and F. Zucchi, “Corrosion Inhibition of Copper in Chloride Solution by Triazole Derivatives,” Proceedings of the 9th European Symposium on Corro- sion Inhibitors, Ferrara, Vol. 2, 2000, pp. 627-637. |

