 Open Journal of Forestry 2014. Vol.4, No.1, 14-23 Published Online January 2014 in SciRes (http://www.scirp.org/journal/ojf) http://dx.doi.org/10.4236/ojf.2014.41003 OPEN ACCESS The Effectiveness of Arbuscular Mycorrhizal Inoculation and Bio-Compost Addition for Enhancing Reforestation with Argania spinosa in Morocco Said El Mrabet1,3*, Lahcen Ouahmane2, Abdelhamid El Mousadik3, Fouad Msanda3, Younes Abbas4 1Regional Department of Forestry and Combating Desertification, Agadir, Morocco 2Polydisciplinary Faculty Safi, Laboratory of Ecology and Environment, Faculty of Sciences Semlalia, University Cadi Ayyad, Marrakesh, Morocco 3Laboratory of Biotechnology and Valuation of Natural Resources—Faculty of Sciences, Agadir, Morocco 4Root Symbiosis Laboratory, Sylviculture Department, Forest Research Centre, Rabat, Morocco Email: *saidov2007@gmail.com Received September 6th, 2013; revised October 4th, 2013; accepted November 2nd, 2013 Copyright © 2014 Said El Mrabet et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. In accordance of the Creative Commons Attribution License all Copyrights © 2014 are reserved for SCIRP and the owner of the intellectual property Said El Mrabet et al. All Copyright © 2014 are guarded by law and by SCIRP as a guardian. A field experiment was carried out in arid area to assess the influence of mycorrhizal inoculation with a native complex and bio-compost addition on establishment of Argania spinosa. The experimental area was located in the Admine forest at Agadir (Southwestern Morocco). The results showed a positive effect of arbuscular mycorrhizal fungi (AMF) on the growth of Argania spinosa seedlings in the nursery. Six months after planting, the mycorrhizal complex revealed an increase in the growth of Argan seedlings (51%) compared to non mycorrhizal plants. In the field conditions, after one year of transplantation, this benefit was maintained. Results showed that the height of Argan seedlings treated with AMF was double that of the control group. An additional positive effect of inoculation with AMF on plant biomass was observed and it was closely related to colonization by these microorganisms. There was an estimated 169% increase in biomass compared to contro l plants. The use of bio-compost alone or in combination with AMF improved the production of shoot biomass of Argan plants (84% and 108% respectively compared to control plants). In addition, AMF improved the survival rate and the contents of nitrogen (N) and phosphorus (P) in the tissues of A. spinosa plants. A significant positive correlation between dry biomass and nutrient content in plant tissue was detected. The content of (P) in the leaves and roots of inoculated plants was higher than those in non-inoculated and pla nted seedlings in amended soils. Th is result reaffirms the prime necessity of mycorrhiza in arid conditions. Thus the introduction of mycorrhizal fungi in forest nurseries is a key tool to improve the quality of seedlings produced and their resistance in reforestation sites. Keywords: Argania spinosa; Arbuscular Mycorrhizal Fungi; Bio-Compost; Regeneration Introduction The Argan tree (Argania spinosa L. Skeels) is one of the most remarkable species of Moroccan forest landscape. Argan ecosystems, which cover 871,000 ha (IFN, 1996), were origi- nally the most special agroforestry system in Morocco. Argan forests have been exploited for edible oil, firewood, timber, as a forage for goats and sheep, and as a shade tree for cereal crops, thereby, supporting the economy of the indigenous population (Alados, 2008). Indeed, the sustainability of the Argan ecosys- tem in particular in the plain is affected by the grazing pressure that trees are encountering intensive agriculture and the arid climate. Currently this ecosystem continues to be destroyed with all its components of biodiversity. There are only some Argan trees scattered in areas that are polluted by pesticides (Benabid, 2000). The ecosystem has regressed in terms of area and especially density, mainly due to clearing of Argan trees and removal of floristic cortege. Between the years of 1969- 1986, the Argan plain lost almost 9900 hectares, an average of 550 ha/year (EL Yousfi, 1988). Due to overexploitation of these forest resources, there has been an increase in the deteri- oration of the various components of the ecosystem, especially plant communities. However, it is recognized that such conse- quences necessarily lead to the degradation of the physico- chemical and biological properties of soils (Skujins & Allen, 1986; Albaladejo et al., 1998; Requena et al., 2001; Azcon- Aguilar et al., 2002). This degradation is manifested by a re- duction in the diversity and/or terrestrial microbial activity (Kenny & Smith, 1995). Consequently, the reduction or loss of this potential can influence the nutritional status of plants and limit the success of native species plantations (Sylvia, 1990; *Corresponding a uthor.  S. EL MRABET ET AL. OPEN ACCESS Roldan et al., 1997; Van der Heijden et al., 1998). However, research has proven that inoculation of plants by mycosymbi- otes not only facilitates installation of plants (Herrera et al., 1993; Smith & Read, 2008; Alguacil, 2011) but also improves the physico-chemical and biological properties of the soil, the- reby improving soil quality (Carrillo-Garccia et al., 1999; Rillig and Mummey, 2006; Schmid et al., 2008). This quality can be improved and the productivity of degraded soils can also be improved by adding organic soil amendments (Roldan et al., 1994; Garcia et al., 1998; Zendejas et al., 2011). The beneficial effects of these amendments include reducing soil bulk density, improving the capacity of water retention and infiltration rate, and aggregating stability and development of biochemical ac- tivities (Zebarth et al., 1999; Caravaca et al., 2002; Fuentes et al., 2010). Regeneration programs of Argan tree in the Argan plain for- est have been initiated since 2000, but have not yielded the expected results. The reasons for this include: 1) the use of reforestation techniques is not adapted to difficult soil and cli- matic conditions prevailing in the arid and semi-arid south-west of Morocco, 2) the Argan seedlings produced are often non mycorrhized, and 3) there is no assessment of land mycorrhizal potential before the launch of any reforestation program. Re- forestation strategy based on the use of mycorrhizal plant bio- technology as a tool has never been tested in reconstruction of the Argan ecosystem. This strategy essentially adapted to semi-arid and arid areas (Nelson & Safir, 1982) should be based on an assessment of the chemical and biological soil fertility, in particular, the estimation of mycorrhizal capacity of soils. Therefore, the use of local mycorrhizal potential deriving from shrubs could be considered as a preferential inoculation strategy to ensure the success of reforestation with native spe- cies in arid and semi-arid degraded areas (Requena et al., 2001; Caravaca et al., 2002; Duponnois et al., 2011). Similarly, it has been shown that in these areas, mycorrhizal fungi help plants growth and cope with nutrient deficiency, drought, soil distur- bance and other environmental stresses (Barea et al., 2007; Martínez-García & Pugnaire, 2009; Martínez-García, 2010). Some studies have only investigated the effects of mycorrhi- za on seedlings of Argania spinosa in controlled conditions. They showed that mycorrhizal inoculation with an artificial inoculum containing Glomus intraradices has improved growth and nutrition of seedlings (Nouaim, 1994; Bousselmame et al., 2002; Echairi et al., 2008). Whereas in this study, we attempt to evaluate the effect of mycorrhization while using a native in- oculum, first on Argan seedlings at the nursery stage and se- condly, it is important to check if the pre-inoculation of seedl- ings could insure a sustainable advantage after transplantation on natural environment. In the present study, we used Argania spinosa, which is well adapted to water stress conditions and belongs to the natural succession of arid ecosystems in south western of Morocco. The aims of this study were to evaluate the benefits of the use of mycorrhizal symbionts to produce vigorous Argan plants that are able to thrive in threatened land, and to evaluate the effectiveness of the mycorrhizal inoculation and the addition of bio-compost through their impact on the physico-chemical characteristics of rhizosphere soil, on growth, nutrition and survival of Argania spinosa seedlings planted in field condi- tions under arid Mediterranean bioclimate. Indeed, two inde- pendent experiments were carried out in this study and were individually addressed in the Methods section, as well as in the Results a nd the Discussion: 1) Evaluation of the effect of native mycorrhizal complex on growth and nutrition of the Argan seedlings under greenhouse conditions and 2) Evaluation of mycorrhiza and the addition of bio-compost on the growth and nutrition of the Argan seedlings in field conditions. Material and Methods Study Site The experimental area was located in the Argan forest “Ad- mine” at Agadir in southwestern of Morocco (coordinates 9˚36'22''W and 33˚55'39''N). The bio-climate can be described as Mediterranean arid with an average annual rainfall of 243 mm and the elevation above sea level is 63 m. The analytical characteristics of the soil in the experimental plot, determined by standard methods (Page et al., 1982) are shown in Table 1. The plot chosen, occupies an area of 32 a, is devoid of Argan trees to avoid their influence on young seedlings. This ecosys- tem had known since the 70s a considerable development of vegetable and fruit crops which consuming very large amounts of water drawn by pumping groundwater. However, it is recog- nized that many communities of AMF are sensitive to distur- bance associated with agriculture and fertilizer use (Helgason et al., 1998). Material Origin of Argan Seeds Argan trees can reach 10 to 12 m high and are characterized by a rugged trunk with rough and cracked bark. This endemic species in Morocco and irreplaceable in its range is the only species capable of stopping severe desertification phenomena observed in recent years in the region (Msanda, 2004) and therefore it is frequently used in reforestation programs in semi-arid and arid disturbed lands. The seeds used in this study were collected under the canopy of Argan trees in the “Admine ” forest. Table 1 . Physico-chemical and biological characteristics of the soil in Ad- mine forest. Texture Fine silty-cl a y-sand pH (H2O) 8.35 Total carbon (%) 0.98 MO (%) 1.69 Total Nit ro g en ( % ) 0.15 C/N 6.53 Available P (mg/Kg) 9 Extractable K (mg/kg) 497 Magnesia (MgO) (mg/kg) 748 Copper (mg/kg) 1.98 Zinc (mg/kg) 1.06 Iron (mg/kg) 6.69 Total limestone % 5.3 (MPN∙g−1 dry soil) 0.14 Note: MPN: Most Probable Number.  S. EL MRABET ET AL. OPEN ACCESS Characteristics of bio-Compost The bio-compost used is organic vegetable compost disin- fected by thermotherapy. The chemical characteristics of the bio-compost are shown in Table 2. Experiment 1: Evaluation of the Effect of Native Mycorrhi zal Comp l e x on Growth and Nutrition of the Argan Seedlings under Greenhouse Conditi ons Production of Inoculum and Inoculation of the Argan Seedlings The corn (Zea mays L.) was used as endophytic plant for the production of endomycorrhizal inoculum. Corn seeds were disinfected in a solution of 30% hydrogen peroxide for 30 min and rinsed several times with sterile distilled water and then planted for three months in plastic pots containing soil from a preserved Argan forest mountain “Mesguina” at Agadir in southwestern Morocco (coordinates 9˚47'14''W and 34˚5'4''N). The soil was taken near the roots (rhizosphere) of Argan trees and shrubs associated with the tree. Therefore the inoculum consisted of a mixture of rhizosphere containing spores, hyphae, and fragments of infected corn roots. This endomycorrhizal fungi complex, dominated by the Glomus genus is also consti- tuted by Scutellospora and Acaulospora genus. The inoculum was brought to the substrate consisting of peat disinfected in containers of 500 ml. The sowing operation of Argan pre-ger- minated seeds was carried out in October 2011. Half seedlings (84U) were planted in a mixture of substrate consisting of peat and inoculum which is 5% of the substrate. The second half of seedlings were planted in the same amount of mixture disin- fected using the autoclave. Seedlings were grown in a green- house where the temperature was 25˚C to 35˚C with daily wa- tering. After four months, a total of sixty four each inoculated and non-inoculated seedlings were planted in the experimental plot “Admine”. Experiment in greenhouse was continued on twenty plants each for two months. After the completion of the observation period in the greenhouse (six months), five plants were randomly collected to assess the dry biomass, the rate of mycorrhizal and nutrition of Argan seedlings. Relative Mycorrhizal Dependency Index (RMDI) Relative Mycorrhizal Dependency Index is calculated from the a verage values of shoot and root dry weight of mycorrhizal Table 2. Chemical characteristics of bio-compost used in the experiment. Parameters Bio-compost pH (H2O) 6 Total ca r bon (%) 35 MO (%) 60 Total Nitrogène (%) 3 C/N 11.6 P (%) 2 K (%) 2 Copper (mg/kg) 150 Zinc (mg/ kg) 100 iron (mg/kg) 2000 plants (DWM) and non-mycorrhizal (DWNM) as described by Plenchette et al. (1983): ( ) *100.RMDIDWM DWNM DWM = − Experiment 2: Evaluation of Mycorrhiza and the Addition of Bio-Compost on the Growth and Nutrition of the Argan Seedlings in Field Conditions Experimental Design and Layout The experimental plot sufficiently homogeneous was pre- pared in august 2011 (Opening 128 holes cubic shape 60 * 60 * 60 cm in sixteen lines). The experimental was a randomized complete block design with two factors of classification: in- oculation or not mycorrhizal complex and the addition or not of bio-compost. During the experiment, four treatments and eight plants per treatment with four repetitions were held on the field in the form of blocks. Each block contains 32 plants (eight × four treatment plants). Half of the holes were amended by 3 kg/hole of bio-compost (Table 2) at a depth of 0 - 20 cm a month before planting. Thus, mycorrhizal plants and controls were planted in January 2012 in Admine site. Regular watering was assured in shearling 10L/plant/month until august 2012. In September 2012, the irrigation operations were suspended after registration of significant rainfall slices. Sampling Pr oc edures One year after planting, four soil samples from each treat- ment were collected (16 soil samples in total). Each sample consisted of five sub-samples (200 cm3 soil cores) randomly collected at 0 - 20 cm in the rhizosphere of five individual plants. The samples were taken in early February 2013 (before the dry season) when the highest microbial activity would be expected (Lax et al., 1997). Furthermore, four plants of each treatment (one per block) were harvested for analysis. Plant Analysis and Plant Growth Twelve months after planting, samples (leaf and root) were oven dried at 68˚C for 72 h, then crushed. The total nitrogen (N) was determined by the Kjeldahl method (Page et al., 1982). Available P was determined by colorimetry according to Mur- phy and Riley (1962). Extractable K was determined by flame photometry (Schollemberger and Simon, 1954).The percentage of root length colonized by AM fungi was calculated by the gridline intersect method (Giovannetti and Mosse, 1980) after staining with trypan blue (Phillips & Hayman, 1970). Regular monitoring of Argan plants was also carried out during the period of observation. Thus, basal stem diameter, plant height (main and secondary branches) and survival rates were recorded after 4, 7, 9 and 12 months from the date of planting. Soil Physical-Chemical Analysis Soil pH was measured in a 1:5 (w:v) aqueous solution. The total organic carbon (C) was determined by the method of Yeomans and Bremner, 1989. Total nitrogen (N), available P (with sodium bicarbonate (Olsen et al., 1954)), and extractable K (with ammonium acetate) were determined following the methods described above for plant tissues. Statistical Analysis The effects of bio-compost, mycorrhizal inoculation and their 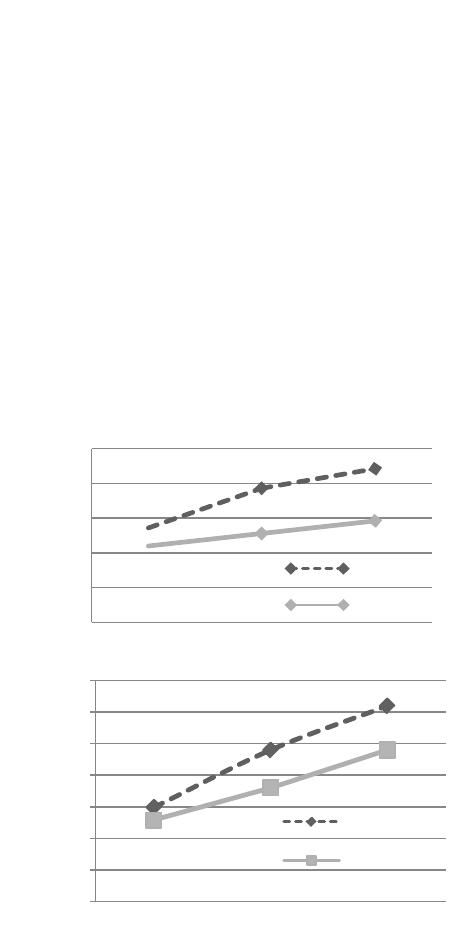 S. EL MRABET ET AL. OPEN ACCESS interactions on measured variables were tested by a two-way analysis of variance and comparisons among means were made using least significant difference (LSD) calculated at P < 0.05. The analysis of correlation between the measured parameters was performed using Pearson’s rank correlation coefficients. All data were processed using SPSS Version 18 software. Results Experiment 1: Effect of Mycorrhiza on Growth and Nutrition of the Argan Seedlings under Greenhouse Conditions Roots Mycorrhizal Colonization, Growth and Nutrition of Argan Plant s under Greenhou se Conditions A positive effect of the AM fungi on Argan seedlings was shown (Figure 1). Six months after planting, mycorrhizal in- oculation improved growth of Argan seedlings by 51%. A sim- ilar trend was observed with basal diameter (on average 29%) The influence on the biomass production was important. The average dry root weight was 66% higher compared to control plants (Table 3). With regard to dry shoot weight, the average was 60% higher than non-inoculated plants. The mycorrhiza has also a significant effect on the N and P content of foliar tissues of Argan plants. The concentrations of N and P are re- spectively 185% and 118% higher in mycorrhizal plants com- pared with controls. Colonization rate of Argan seedling roots had shown that at least 54.9% of roots are occupied by the mycorrhizal fungi (Table 3). Relative Mycorrhizal Dependency Index (RMDI ) Relative Mycorrhiza l Dependency Index, cal culated on the ba - sis of the a verage dry weight of shoots and roots of mycorrhizal and non-mycorrhizal plants is 39% after six months of growth. Figure 1. Plant height and basal diameter of Argan seedlings inoculated and not after 4, 5 and 6 months under greenhouse conditions. Experiment 2: Effect of Mycorrhiza and the Addition of Bio-Compost on the Growth and Nutrition of the Argan Seedlings after 12 Months Transplantation in Field Conditions Physico-Chemical Parameters of Rhizosphere Soil of Argan Plants One year after planting, the addition of composted residue and mycorrhizal inoculation significantly decreased soil pH (Table 4). The combination of these two methods of reforesta- tion (CRM) produced significantly higher values of total nitro- gen (N) compared to control soil. The addition of bio-compost increased significantly the levels of total organic carbon and extractable K, while the total limestone content was signifi- cantly lower compared to non-amended soils (M and C). Fur- thermore, the content of available phosphorus in the soil was increased by the addition of bio-compost. Thus, the content of available phosphorus in soils amended with bio-compost (CR and CRM) was about four times higher than in non-amended soil (C and M). The analysis also showed that mycorrhizal in- oculation (M) has slightly higher values in total organic C, N, P and K compared to the values recorded in the rhizosphere soil of the control plants (C). However, there was no statistically significant difference between treatments (LSD test). Both me- thods of reforestation (M, CR and CRM) have a C/N ratio ranged between 8 and 10, which means the presence of a good biological activity, while the rhizosphere of control plants present a ratio which is about 6.9, indicating that there is a low biological activity in the control soil (Table 4). Roots Colonization, Biomass Production and Mineral Nutrition of Argan Plants Twelve months after planting, the mycorrhizal colonization of non-inoculated seedlings planted in amended and non- amended soil (CR and C) have increased by an average of 26.25% and 20% as a result of natural infection (Table 5). However, there were no significant differences between the values of these two treatments. Mycorrhizal plants (M) in non-amended soils showed the highest percentages of root co- lonization (77.5%) followed by inoculated plants planted in amended soil CRM (55%). Thus, application of the amendment had a negative effect on the mycorrhizal colonization of inocu- lated plants. Furthermore, inoculation had the greatest effect on the growth of Argan plants producing about 169% shoot dry matter more than in the control plants at the end of the observa- tion period. Therefore, stimulating the production of biomass observed in inoculated plants (M) can be linked to the ability of the fungi to increase the absorption of nutrients including NPK from soil relatively low in nutrients compared to amended soils (Tables 4 and 5). Amended plants (CR and CRM) have shown also that shoot dry biomass was significantly higher compared to those of control plants (at least 84%). Results had shown that the two methods of reforestation (mycorrhizal inoculation or addition of bio-compost) contributed separately (M or CR) and simultaneously (CRM) to improve NPK contents in foliar and root tissues of Argan plants. Therefore, leaf tissues of plants in amended soil (CR) have significantly higher N, P and K content than the control plants (C). The combination of the two me- thods of reforestation (CRM) also produced plants with a leaves and roots content of NP significantly higher than the control plants (C). The analysis had shown that the P content in leaf  S. EL MRABET ET AL. OPEN ACCESS Table 3. Growth, nutrition and roo ts mycorrhizal colonization o f inoculated and non-inoculated Argan plants after 6 months under greenhouse conditions (n = 5). Height (cm) Basal Diameter (mm) Aerial dry weight (g) 1 Root d ry weight (g) 2 tot al dry wei ght (g) 3 = 1 + 2 Total N % P mg /plant Roots Mycorrhizal colonization (%) Mycorrhizal Plants 24.1a (±4.47) 3.5a (±0.42) 0.93a (±0.21) 0.74a (±0.18) 1.67a (±0.38) 4a (±0.86) 24a (±1.58) 54.9 (±13.39) controls 16.2b (±1.52) 2.83b (0.13) 0.56b (±0.13) 0.46b (±0.14) 1.02b (±0.14) 1.4b (±0.15) 11b (±1.59) 0 Note: Values sharing the sa me lette r within a column are not sig nificantly different at 5% by the LSD test. Table 4. Changes in soil chemical properties in response to mycorrhizal inoculation (M) and the addition of bio-compost (CR) (n = 4) pH (H2O) Total organic C % total N % P Available (mg/kg) K Extractable (mg/kg) C/N Total l imestone (%) M 8.51a (±0.43) 1.51ab (±0.53) 0.18ab (±0.15) 27.25a (±2.5) 847a (±54.3) 8.30ab (±2.26) 12.42a (±2.85) CRM 8.52a (±0.25) 1.92a (±0.5) 0.19a (±0.13) 143b (±42.4) 850a (±112.6) 10.26a (±1.9) 6.25b (±2.29) CR 8.55a (±0.31) 1.80a (±0.22) 0.18ab (±0.04) 119.2b (±18.2) 1218b (±195) 10.06a (±1.33) 6.7b (±2.25) C 8.62b (±0.14) 1.16b (±0.93) 0.17b (±0.05) 25.75a (±2.9) 775a (±30.7) 6.91b (±0.73) 13.77a (±0.99) Note: Values sharing the same letter within a column are not significantly different at 5% (LSD test). (C: control soil, without mycorrhizal ino culation and without com- posted re sidue addition a nd CRM: c omposted residue addition + mycorrhizal inoculation). Table 5. Roots colonization, biomass prod uction and mineral nutrition of Argan plants after 1 2 months transplan tation after mycorrhizal inoculatio n (M) and composted re s idue (CR) addit ion (n = 4). Shoot aerial dry weight (g) (%) Foliaire tis sue s Root tissues P mg/kg K mg /kg Total N % P mg/kg K mg/kg Total N % M 28.95a (±1.17) 77.5a (±8.6) 1.64a (±0.17) 10.80ab (±1.70) 3.32a (±0.12) 0.92a (±0.07) 7.54a (±0.14) 0.886a (±0.017) CRM 22.45b (±4.08) 55b (±17.3) 1.04b (±0.68) 10.51ab (±2.23) 3.11a (±0.23) 0.67b (±0.04) 7.62a (±0.05) 0.889a (±0.009) CR 19.84b (±4.77) 26.25c (±4.7) 1.01b (±0.76) 13.74a (±1.94) 3.09a (±0.11) 0.6bc (±0.05) 3.4b (±1.05) 0.901a (±0.024) C 10.76c (±3.29) 20c (±7) 0.74c (±0.51) 7.80b (±4.29) 2.71b (±0.16) 0.55c (±0.03) 4.06b (±0.09) 0.545b (±0.013) Note: Values sharing the same letter within a column are not significantly different at 5% (LSD test). (C: control soil, without mycorrhizal inoculation and without bio- compost r esidue additio n and CRM: compost residue addition + mycorr hizal inoculation). and root tissues of inoculated plants (M) is significantly higher than that of plants in the other treatments (CR, CRM and C). The inoculated plants (M) present a difference of the P content in leaf and root tissues, respectively, 121% and 67% compared to the values recorded in control plants (Table 5). The growth and survival of Argan plants At the moment of planting, the inoculated plants showed higher growth in height (13.5 cm) than non-inoculated plants (10.9 cm). This difference was significant at the 5% level (LSD). Twelve months after planting, the addition of bio- compost and mycorrhizal inoculation improved both the growth compared to the control soil (C). Height was improved respec- tively by 79% and 158% (Figure 2). However, the combined mycorrhizal× bio-compost treatment (CRM) had only a slight additive effect but not significant with respect to the addition of bio-compost (CR). This effect improved the growth of Argan plants approximately 87% compared to control plants, while the comparison between the two methods of planting on the growth of Argan plants showed that the effect of inoculation is indeed net. Consequently, the mycorrhizal plants (M) are much greater than non-mycorrhizal plants planted in amended soil (CR). The difference was significant between the two treatments on the total height of the main axes and secondary axes and it main- tains over time to record the seventh month after the date of planting 58%, 39% in the ninth month and 43% in the 12th month. A similar trend was observed on the growth of basal diameter (Figure 2). 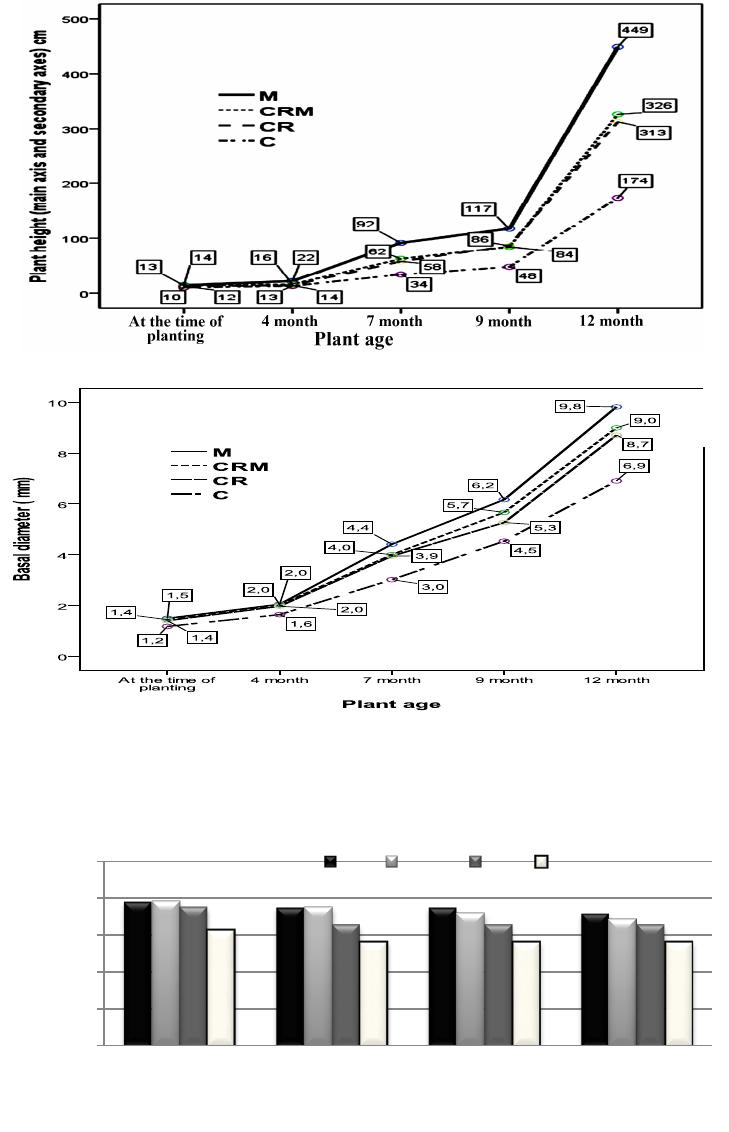 S. EL MRABET ET AL. OPEN ACCESS Mycorrhizal symbiosis also helps Argan seedlings to survive under extreme climatic conditions in this arid area. Indeed, as Figure 3 shows, twelve months after the plantation the percen- tage of survival inoculated plants (71.8% for M and 68.7% for Figure 2. Effect of composted residue addition (CR) and mycorrhizal inoculation (M) on growth (Height and basal di- ameter) of Argania spinosa under field conditions at the time of planting and after 4, 7, 9 and 12 months from the date of planting (n = 4). (C: control soil, withou t mycorrhizal inoculation an d without composted residue a d- dition and CRM: composted residue addition + mycorrhizal inoculation). Figure 3. Survival plants of Argania spinosa in response to mycorrhizal inoculation (M) and the addition of bio-compost (CR) after 4, 7, 9 and 12 months from the date of planting in the experimental plot. (C: contro l soil, without mycorrhizal inoculation and without composted resid ue addition and CRM: composted resid ue addition + mycorrhizal inoculation).  S. EL MRABET ET AL. OPEN ACCESS CRM) was much higher than within non-inoculated plants (65.6% for CR and 56.2% for C), although there was no statis- tically significant difference between the first three treatments (M, CR and CRM). However, the values of the survival rate of the control plants (C) are significantly lower than those of other treatments (M, CR and CRM). Discussion Effectiveness of Mycorrhiza under Greenhouse Conditions This study showed that mycorrhizal inoculation with a native complex has a positive and significant effect on height, basal diameter, and biomass and NP contents of leaf tissues of Argan plants which were planted six months under greenhouse. This study confirms the results obtained in nursery stage by several authors (Nouaim, 1994; Bousselmame et al., 2002; Echairi et al., 2008). The study also shows that the Argan tree is depen- dent on mycorrhiza for its development and mineral nutrition. The RMDI calculated (39%), after six months, is lower than those found by Nouaim (1994) and Echairi et al. (2008) using a non-native inoculum containing Glomus intraradices to per- form the mycorrhiza of Argan plants produced respectively by in vitro culture and from seeds. Nouaim (1994) found 78% of RMDI after six months of growth. However, Echairi et al. (2008) found 48% after nine months of aging under controlled conditions. The last authors observed an increase in height, shoot and root biomass respectively 40%, 93% and 41% of mycorrhizal plants compared to control plants. Effectiveness of Mycorrhizal Inoculation Several authors have emphasized the role of AMF in the ab- sorption of water, absorption of nutrients and stimulation of the growth of many plant species (Roldan et al., 1996b; Smith & Read, 1997; Jeffries et al., 2003; Ouahmane et al., 2007). In this study, inoculation of Argania spinosa seedlings has signifi- cantly stimulated the production of biomass during the first year of planting, which is the most critical period for reforesta- tion, especially in the Mediterranean semi-arid and arid areas (Meddad-Hamza et al., 2010; Ouahmane et al., 2012). Fur- thermore, the effect of inoculation on plant biomass is posi- tively correlated to the level of colonization by AMF (Table 6). At the end of the period of growth, mycorrhiza increased shoot biomass of Argan plants to a greater extent, about 169% com- pared to control plants. Previous studies in Mediterranean areas have shown similar results. Indeed, Caravaca et al. (2002), showed that the biomass of inoculated seedlings of Olea euro- pea subsp. sylvestris and Pistacia lentiscus raised after a year of planting, respectively to 630% and 300% compared to non- inoculated plants. Caravaca et al. (2003b) also showed that the production of root plants biomass of Dorycnium pentaphyllum inoculated by Glomus intraradices increased by 116% com- pared to non-inoculated plants. The total content of plant nu- trients can be considered as a representative indicator of the effectiveness of mycorrhiza, because it takes into account the well balanced effects of nutrient uptake and biomass production (Jeffries et al., 2003). Indeed, the highest levels of P and N in the leaf tissues were observed in the inoculated plants, which could explain why the growth of Argania spinosa seedlings was the highest in this treatment. Thus, we noticed that there is a positive and significant correlation between shoot dry biomass Table 6. Pearson rank correlation between foliar and root levels of NPK, shoot aerial dry weight and root colonization % (n = 4)a. Root colonisation% FP 0.789** 0.804** FK 0.265ns 0.037ns FN 0.806** 0.758** RP 0.696** 0.827** RK 0.626** 0.818** RN 0.753** 0.547* Root coloni s ation% 0.878** 1 Note: aCorrelation coefficient (significance level); *, **: Respectively significant at P < 0.05 and P < 0.01; ns: not significant. FP: Foliar P content, FK: foliar K content; FN: foliar N content, RP: P content in root, RK: K content in root; RN: N content in root. and nutrient N and P in plant tissue (Table 6). It is also impor- tant to note that mycorrhizal inoculation was more effective than the addition of composted residue on the leaf and root P content of A. spinosa plants, even if the available P in the rhi- zosphere soil of plants treated with composted waste is four times higher than in the rhizosphere soil of inoculated plants. These results once again reaffirm the capital role of AMF in P uptake (Harrison, 1999; Bago et al., 2002; Ohtomo & Saito, 2005; Helgason & Fitter, 2009; Smith et al., 2004, 2009). Si- milarly, increasing the nitrogen content in the tissues of my- corrhizal plants may be due to the ability of AMF to improve the decomposition of organic matter and nitrogen capture (Hodge et al., 2001), and increase the absorption of P which strongly favors the biological N2 fixation (Azcon & Barea, 1992). The study also showed that inoculation with the AMF is able to improve the survival rate of transplanted Argan seedlings in the Mediterranean degraded environments. This result is in agreement with the results obtained through the mycorrhizal symbiosis by several authors in similar conditions. They showed significant improvement in the survival of many forest species after transplantation in unfavorable environments (Boutekrabt et al., 1999; Requena et al., 2001; Ouahmane et al., 2007, Abbas et al., 2013). Mycorrhiza is an essential component that facilitates the success of programs of plant regeneration on degraded soils and before initiating these programs, so it is necessary to study the existing vegetation and their partners, particularly mycorrhizal propagules (Jasper, 1994) that control the biogeochemical cycles of major elements of soil (Kennedy & Smith, 1995; Re- quena et al., 2001; Palenzuela et al., 2002; Jeffries et al., 2003; Ouahmane, 2007). Thus, these programs must include the re- construction of the mycorrhizal population (Barea et al., 1990) which can be done through: 1) assessment of mycorrhizal status of soils including isolation, identification and characterization of local AM fungi targeted land and 2) the production of a se- lected inoculum from these AMF which are able to improve the quality of plants to thrive in arid conditions via improving the assimilation of nutrients especially P and N (Toro et al., 1997), mitigation of water stress (Augé, 2001; Herrera et al., 1993; Roldan et al., 1996b; Barea et al., 2008; Honrubia, 2009) and  S. EL MRABET ET AL. OPEN ACCESS improving the quality of soil (Jeffries & Barea, 2001) and fi- nally the development of resistance against diseases (Pozo et al., 1999; Dalpe, 2005; Tahat et al., 2012). Effectiveness of Soil Amendments The result of this study has shown the effectiveness of the addition of bio-compost to improve the growth and nutritional status (NPK) of young Argan plants. This result is based on the improvement of soil fertility. In this regard, the addition of bio-compost increased levels of total organic C, total N, availa- ble P, extractable K and C/N ratio of the soil, with the largest increase being observed for available P. This is in agreement with the conclusion of Roldán et al. (1996a); Caravaca et al. (2002); and Zendejas et al. (2011), which found that the posi- tive effect of composted residue on chemical parameters is primarily due to phosphorus. We also noticed that the soil amendment has led to a decrease in pH soil. The application of organic amendments to the soil is an effective method to im- prove the physico-chemical and microbiological properties of degraded soils, which in turn promote the creation of stable vegetation (Roldán et al., 1994). Thus, at the end of the growth period, the addition of bio-compost increased the biomass of the Argan seedlings to about 84% compared to control plants. Effectiveness of the Combination of Soil Amendment and Mycorrhizal Inoculation The field experiment also showed that the combination of soil amendment and mycorrhizal inoculation caused the largest increase in the total nitrogen content in the rhizosphere of Ar- gania spinosa. It should also be noted that this combined treat- ment significantly stimulates the production of biomass in arid conditions. Thus, it has increased the growth of A. spinosa seedlings but to a lesser extent as the only treatment of mycorr- hizal inoculation. There was almost a slightly additive effect but not significant, compared to adding only bio-compost. This result is consistent with the widely accepted idea that mycorr- hiza has few advantages over plants in amended soils (Yanai et al., 1995; Caravaca et al., 2004). However, our results are in disagreement with the work of Caravaca et al. (2002, 2003a). They showed an additive and positive effect of composted waste and mycorrhizal inoculation on the growth of Retama sphaerocarpa and Olea europaea subsp. sylvestris plants in Mediterranean semi-arid condi tions. T he additi on of bio-compost, inoculation with AM fungi and the combination of these two treatments had no significant effect on the survival of Argan plants during the first year of planting. The survival rate of three treatments ranges from 66% to 72%. These results are quite similar to those obtained by Alguacil et al. (2008). In conclusion, the addition of bio-compost was effective to improve the physical, chemical and biological quality of the rhizosphere soil of seedlings. Yet, the mycorrhizal inoculation was the most effective treatment for stimulating the growth of Argania spinosa plants on abandoned farmlands and subject to Mediterranean arid climatic conditions. This treatment also has a significant positive effect on height, basal diameter, biomass, N and P contents of the plant tissues. Acknowledgements This research was partially supported by the Deutsche Ge- sellschaft für Internationale Zusammenarbeit (GIZ)/(Adaptation to climate change project). REFERENCES Abbas, Y., Bakkali, Y. S., Prin, Y., Arahou, M., Abourouh, M., & Duponnois, R. (2013). Growth and nutrition of Tetraclinis articulata (Vahl) Mast.cultivated in different rhizosphere soils collected from Tetraclinis stand. Biotechnology, Agronomy, Society and Environ- ment, 17, 3-11. Alados, C. L., & El Aich, A. (2008). Stress assess ment of Argan (Ar- gania spinosa (L.) Skeels) in response to land uses across an aridity gradient: Translational asymmetry and branch fractal di mension. Journal of Arid E nvironment, 72, 338-349. http://dx.doi.org/10.1016/j.jaridenv.2007.06.015 Albaladejo, J., Martinez-Mena, M., Roldan, A., & Castillo, V. (1998). Soil degradation and desertification induced by vegetation removal in a semiarid environment. Soil Use Manage, 14, 1-5. http://dx.doi.org/10.1111/j.1475-2743.1998.tb00602.x Alguacil, M. M., Caravaca, F., Azcónb, R., & Roldán, A. (2008). Changes in biological activity of a degraded Mediterranean soil after using microbially-treated dry olive cake as a biosolid amendment and arbuscular mycorrhizal fungi. European Journal of Soil Biology, 44, 347-354. http://dx.doi.org/10.1016/j.ejsobi.2008.02.001 Alguacil, M. M., Torrecillas, E., Kohler, J., & Roldan, A. (2011). Amo- lecular approach to ascertain the success of “in situ” AM fungi in- oculation in the revegetation of a semiarid, degraded land. Science of the Total Environment, 409, 2874-2880. http://dx.doi.org/10.1016/j.scitotenv.2011.04.029 Augé, R. M. (2001). Water relations, drought and vesicular-arbuscular mycorrhizal symbiosis. Mycorrhiza, 11, 3-42. http://dx.doi.org/10.1007/s005720100097 Azcon, R., & Barea, J. M. (1992). Nodulation, N2 fixation (5N) and N nutrition relationships in mycorrhizal or phosphate-amended alfalfa plants. Symbiosis, 12, 33-41. Azcón-Aguilar, C., Palenzuela, E. J., Roldan, A., Bautista, S., Vallejo, R., & Barea, J. M. (2002). Analysis of the mycorrhizal potential in the rhizosphere of representative plant species from desertification- threatened Mediterranean shrublands. Applied Soil Ecology, 21, 1-9. Bago, B., Pfeffer, P. E., Zipfel, W., Lammers, P., & Shachar-Hill, Y. (2002). Tracking metabolism and imaging transport in arbuscular mycorrhizal f ungi. Metabolism and transport in AM fungi. Plant and Soil, 244, 189-197. http://dx.doi.org/10.1023/A:1020212328955 Barea, J. M., Salamanca, C. P., Herrera, M. A., & Roldán-Fajardo, B. E. (1990). La simbiosis microbio planta en el establecimiento de unacubiertavegetal sobre suelosdegradados. In: J. Albaladejo, M. Stocking, & E. Díaz (Eds.), Soil degradation and rehabilitation in Mediterranean environmental conditions (pp. 139-156). Murcia: Consejo Superior de Investigaciones Científicas. Barea, J. M., Palenzuela, J., Cornejo, P., Sánchez, I., Navarro, C., Quiñones, P. B., Azcón, R., Ferrol, N., & Azcón-Aguilar, C. (2007). Significado, diversidad e impacto de los hongos de las micor- rizasarbusculares en ambientesmediterráneos. In: J. M. Barea-Azcón, M. Moleón, R. Travesí, E. Ballesteros, J. M. Luzón, & J. M. Tierno (Eds.), Biodiversidad y conservación de fauna y flora en ambientes Mediterráneos (pp. 155-185). Granada: Sociedad Granatense de Historia Natural. Barea, J. M., Ferrol, N., Azcón-Aguilar, C., & Azcón, R. (2008). My- corrhizal symbioses. Series. In: P. J. White, & J. P. Hammond (Eds.), The ecophysiology of plant-phosphorus interactions. Plant cophy- siology, Vol. 7 (pp. 143-163). Dordrecht: Springer. Benabid, A. (2000). Flore et écosystèmes du Maroc. Paris: Ed Ibis Press. Bousselmane, F., Kenny, L., & Achouri, M. (2002). Effet des mycorhizes à vésicules et arbuscules sur la croissance et la nutrition de l’arganier (Argania spinosa L.). Revue Marocaine des Sciences Agronomiques et Vétérinaires, 22, 193-198. Boutekrabt, A., Chevalier, G., Pargney, J. C., & Dexheimer, J. (1999). Mycorhization par Tuber melanosporumVitt de vitroplants de Que- rcus robur L et Quercus pubescensWilld. Agronomie, 2, 127-132.  S. EL MRABET ET AL. OPEN ACCESS Brundrett, M. C., Piche, Y., & Peterson, R. L. (1985). A developmental study of the early stages in vesicular-arbuscular mycorrhizal forma- tion. Canadian Journal of Botany, 63, 184-194. http://dx.doi.org/10.1139/b85-021 Carrillo-García, A., León de la Luz, J. L., Bashan, Y., & Bethlenfalvay, G. J. (1999). Nurse plants, mycorrhizae, and plant establishment in a disturbed area of the Sonoran desert. Restoration Ecology, 7, 321- 335. http://dx.doi.org/10.1046/j.1526-100X.1999.72027.x Caravaca, F., Barea, J. M., Figueroa, D., & Roldań, A. (2002). Assess- ing the effectiveness of mycorrhizal inoculation and soil compost addition for reafforestation with Olea europaea subsp. sylvestris through changes in soil biological and physical parameters. Applied Soil Ecology, 20, 107-118. http://dx.doi.org/10.1016/S0929-1393(02)00015-X Caravaca, F., Alguacil, M. M., Figuer oa, D. , Bar ea, J. M., & Roldán, A. (2003a). Re-establishment of Retama sphaerocarpa as a target spe- cies for reclamation of soil physical and biological properties in a semiarid Mediterranean land . Forest Ecology and Management, 182, 49-58. http://dx.doi.org/10.1016/S0378-1127(03)00067-7 Caravaca, F., Barea, J. M., Palenzuela, J., Figueroa, D., Alguacil, M. M., & Roldán, A. (2003b). Establishment of shrub species in a degraded semiarid site after inoculation with native or allochthonous arbuscu- lar mycorrhizal fungi. Applied Soil Ecology, 22, 103-111. http://dx.doi.org/10.1016/S0929-1393(02)00136-1 Caravaca, F., Alguacil, M. M., Azcón, R., Díıaz, G., & Roldán, A. (2004). Comparing the effectiveness of mycorrhizal inoculation and amendment with sugar beet, rock phosphate and Aspergillus niger to enhance field performance of the leguminous shrub Dorycnium pen- taphyllum L. Applied Soil Ecology, 25, 169-180. http://dx.doi.org/10.1016/j.apsoil.2003.08.002 Dalpé, Y. (2005). Les mycorhizes: Un outil de protection des plantes mais non une panacée. Phytoprotection, 86, 53-59. http://id.erudit.org/iderudit/011715ar Duponnois, R., Ouahmane, L., Kane, A., Thioulouse, J., Hafidi, M., Boumezzough, A., Prin, Y., Baudoin, E., Galiana, A., & Dreyfus, B. (2011). Nurse shrubs increased the early growth of Cupressus seedl- ings by enhancing belowground mutualism and soil microbial activ- ity. Soil Biology & Biochemistry, 43, 2160-2168. Echairi, A., Nouaim, R., & Chaussod, R. (2008). Intérêt de la my- corhization contrôlée pour la production de plants d’arganier (Ar- gania spinosa) en conditions de pépinière. Sécheresse, 19, 277-281. EL Yousfi, M. (1988). Dégradation du couvert forestier dans le Sud Marocain: Cas de l’arganeraie d’Admine. Rabat: Mémoire de 3 ème cycle IAV Hassan II, 117p. Fisher, R. A., & Yates, F. (1970). Statistical tables for biological agri- culture and medical research (6th ed.). Davien: Hafner Publishing Company. Fuentes, D., Valdecantos, A., Llovet, J., Cortina, J., & Vallejo, V. R., (2010). Fine-tuning of sewage sludge application to pro mote the es- tablishment of Pinus halepensis seedlings. Ecological Engineering, 36, 1213-1221. Garcia, C., Hernández, T., Albaladejo, J., Castillo, V., & Roldán, A. (1998). Revegetation in semiarid zones: Influence of terracing and organic refuse on microbial activity. Soil Science Socie ty of America Journal, 62, 670-676. http://dx.doi.org/10.2136/sssaj1998.03615995006200030018x Garcia, C., León de la Luz, J.-L., Bashan, Y., & Bethlenfalvay, G. J. (1999). Nurse plants, mycorrhizae, and plant establishment in a dis- turbed area of the Sonoran desert. Restoration E cology, 7, 321-335. http://dx.doi.org/10.1046/j.1526-100X.1999.72027.x Giovannetti, M., & Mosse, B. (1980). An evaluation of techniques for measuring vesicular-arbuscular mycorrhizal infection in roots. New Phytologist, 84, 489-499. http://dx.doi.org/10.1111/j.1469-8137.1980.tb04556.x Harrison, M. J. (1999). Molecular and cellular aspects of the arbuscular mycorrhizal symbiosis. Annual Review of Plant Physiology and Plant Molecular Biology, 50, 361-389. http://dx.doi.org/10.1146/annurev.arplant.50.1.361 Helgason, T., Daniell, T. J., Husband, R., Fitter, A. H., & Young, J. P. Y. (1998). Ploughing up the wood-wide web? Nature, 394, 431. http://dx.doi.org/10.1038/28764 Helgason, T., & Fitter, A. H. (2009). Natural selection and evolution in the arbuscular mycorrhizal fungi (Phylum Glomeromycota). Journal of Experimental Botany, 60, 2465-2480. Herrera, M. A., Salamanca, C. P., & Barea, J. M. (1993). Inoculation of woody legumes with selected arbuscular mycorrhizal fungi and rhi- zobia to recover desertified Mediterranean ecosystems. Applied and Environmental Microbiology, 59, 129-133. Hodge, A., Campbell, C. D., & Fitter, A. H. (2001). An arbuscular mycorrhizal fungus accelerates decomposition and acquires nitrogen directly from organic material. Nature, 413, 297-299. http://dx.doi.org/10.1038/35095041 Honrubia, M. (2009). The Mycorrhizae: A plant-fungus relation that has existed f or more than 400 million years. Anales del Jardín Botánico de Madrid, 66, 133-144. IFN (1996). Synthèse de l’Inventaire forestier national marocain. Rabat: Direction de Développement Forestier. Jasper, D. A. (1994). Management of mycorrhiza in revegetation. In A. D. Robson, L. K. Abbot, & N. Malajczuk (Eds.), Management of mycorrhizas in agriculture, horticulture and forestry (pp. 211-219). Dordrecht: Kluwer Academic Press. Jeffries, P., & Barea, J. M. (2001). Arbuscular mycorrhiza—A key component of sustainable plant-soil ecosystems. In: B. Hock (Ed.), The Mycota. IX Fungal Associations (pp. 95-113). Berlin: Springer- Verlag. http://dx.doi.org/10.1007/978-3-662-07334-6_6 Jeffries, P., Gianinazzi, S., Perotto, S., Turnau, K., & Barea, J. M. (2003). The contribution of arbuscularmycorrhizal fungi in sustaina- ble maintenance of plant health and soil fertility. Biolog y and Fertil- ity of Soils, 37, 1-16. Kennedy, A. C., & Smith, K. L. (1995). Soil microbial diversity and the sustainability of agricultural soils. Plant and Soil, 170, 75-86. http://dx.doi.org/10.1007/BF02183056 Lax, A., Roldán, A., Caravaca, F., & García-Orenes, F. (1997). Rela- tionships between aggregate i mprovement, microbiological activity and organo-mineral complex formation in soils from semiarid areas. In: S. G. Pandalai (Ed.), Recent resea rch developments in so il biolo- gy and biochemistry (pp. 77-92). Trivandrum: Research Signpost. López-Bermúdez, F., & Albaladejo, J. (1990). Factores ambientales de la degradación delsuelo en el áreamediterránea. In: J. Albadejo, M. A. Stocking, & E. Díaz (Eds.), Soil degradation and rehabilitation in Mediterranean environmental conditions (pp. 15-45). Murcia: Con- sejo Superior de Investigaciones Científicas. Martínez-García, L. B., & Pugnaire, F. I. (2009). Interacciones entre las comunidades de hongosformadores de micorrizasarbusculares y de plantas. Algunosejemplos en los ecosistemassemiáridos. Ecosistemas, 18, 44-54. Martínez-García, L. B. (2010). Micorrizas arbusculares en ecosistem- assemiáridos. Respuesta a factoresde estrésambiental. Tesis Doc- torales, Almería: Universidad de Almería. Meddad-Hamza, A., Beddiar, A., Gollotte, A., Lemoine, M. C., Kus- zala, C., & Gianinazzi, S. (2010). Arbuscular mycorrhizal fungi im- prove the growth of olive trees and their resistance to transplantation stress. African Journal of B iotechnology, 9, 1159-1167. Msanda, F. (2004). Végétation de l’Anti Atlass occidental et de sa retombée saharienne (Maroc). Essai de Synthèse, Thèse de Doctorat ès Sciences, Agadir: Université Ibn Zohr. Murphy, J., & Riley, J. P. (1962). A modified single solution method for determination of phosphate in natural waters. Analytica Chimica Acta, 27, 31-36. http://dx.doi.org/10.1016/S0003-2670(00)88444-5 Nelson, C. E., & Safir, G. R. (1982). Increased drought tolerance of mycorrhizal onion plants caused by improved phosphorus nutrition. Planta, 154, 407-413. http://dx.doi.org/10.1007/BF01267807 Nouaïm, R. (1994). Écologie microbienne des sols d’arganeraies. Activités microbiologiques des sols et rôle des endomycorhizes dans la croissance et la nutrition de l’Arganier. Thèse d’État, Agadir: Université Ibn Zohr. Olsen, S. R., Cole, C. V., Watanabe, F. S., & Dean, L. A. (1954). Esti- mation of available phosphorus in soils by extraction with sodium bicarbonate. Circular, Vol 939 (p. 19). Washington, DC: US De- partment of Agriculture.  S. EL MRABET ET AL. OPEN ACCESS Ohtomo, R., & Saito, M. (2005). Polyphosphate dynamics in mycorr- hizal roots during colonization of an arbuscular mycorrhizal fungus. New Phytologist, 167, 571-578. http://dx.doi.org/10.1111/j.1469-8137.2005.01425.x Ouahmane, L. (2007). Rôles des plantes associées (Lavandula et Thymus) dans la régénération du cypres de l’atlas—Effets sur la diversité rhizosphérique. Thèse de Doctorat National, Marrakech: Université cadi Ayyad. Ouahmane, L., Hafidi, M., Thioulouse, J., Ducousso, M., Kisa, M., Prin, Y., Galiana, A., Boumezzough, A., & Duponnois, R. (2007). Im- provement of Cupressus atlantica Gaussen growth by inoculation with native arbuscular mycorrhizal fungi. Journal of Applied Micro- biology, 103, 683-690. Ouahmane, L., Ndoye, I., Morino, A., Ferradous, A., Sfairi, Y., Al Faddy, M. N., & Abourouh, M. (2012). Inoculation of Ceratonia si- liqua L. with native arbuscular mycorrhizal fungi mixture improves seedling establishment under g reenhouse con ditions. African Journal of Biotechnology, 11, 16422-16426. Page, A. L., Miller, R. H., & Keeny, O. R. (1982). Methods of soil analysis. Madison: American Society of Agronomy. Palenzuela, J., Azcoń-Aguilar, C., Figueroa, D., Caravaca, F., Roldań, A., & Barea, J. M. (2002). Effects of mycorrhizal inoculation of shrubs from Mediterranean ecosystems and composted residue ap- plication on transplant performance and mycorrhizal developments in a desertified soil. Biology and Fertility of Soils, 26, 170-175. Phillips, J. M., & Hayman, D. S. (1970). Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular my- corrhizal fungi for rapid assessment of infection. Tra nsactions of the British Mycological Society, 55, 158-161. http://dx.doi.org/10.1016/S0007-1536(70)80110-3 Plenchette, C., Fortin, J. A., & Furlan, V. (1983). Growth responses of several plant species to mycorrhizae in a soil of moderate P fertility. I. Mycorrhizal dependency under field conditions. Plant and Soil, 70, 199-209. http://dx.doi.org/10.1007/BF02374780 Pozo, M. J., Azcón-Aguilar, C., Dumas-Gaudot, E., & Barea, J. M. (1999). β-1,3-glucanase activities in tomato roots inoculated with arbuscular mycorrhizal fungi and/or Phytophthora parasitica: Time course analysis and possible involvement in bioprotection. Plant Science, 141, 149-157. http://dx.doi.org/10.1016/S0168-9452(98)00243-X Requena, N., Perez-Solis, E., Azcon-Aguilar, C., Jeffries, P., & Barea J. M. (2001). Management of indigenous plant-microbe symbioses aids restoration of desertified ecosystems. Applied and Environmental Microbiology, 67, 495-498. http://dx.doi.org/10.1128/AEM.67.2.495-498.2001 Rillig, M., & Mummey, D. L. (2006). Mycorrhizas and soil structure. New Phytologist, 171, 41-53. http://dx.doi.org/10.1111/j.1469-8137.2006.01750.x Roldán, A., García-Orenes, F., & Lax, A. (1994). An incubation expe- riment to determine factors involving aggregation changes in an arid soil receiving urban refuse. S oil Biolog y and Biochemis try, 26, 1699- 1707. http://dx.doi.org/10.1016/0038-0717(94)90323-9 Roldán, A., Albaladejo, J., & Thornes, J. B. (1996a). Aggregate stabili- ty changes in a semiarid soil after treatment with different organic amendments. Arid Soil Research and Rehabilitation, 10, 139-148. http://dx.doi.org/10.1080/15324989609381428 Roldán, A., Querejeta, J. I., Albaladejo, J., & Castillo, V. (1996b). Growth response of Pinus halepensis to inoculation with Pisolithus arhizus in a terraced rangeland amended with urban refuse. Plant and Soil, 179, 35-43. http://dx.doi.org/10.1007/BF00011640 Roldán, A., García, C., & Albaladejo, J. (1997). AM fungal abundance and activity in a chronosequence of abandoned fields in a semiarid Mediterranean site. Arid Soil Research and Rehabilitation, 11, 211- 220. http://dx.doi.org/10.1080/15324989709381474 Schmid, T., Meyer, J., & Oehl, F. (2008). Integration of mycorrhizal inoculum in high alpine revegetation. Mycorrhiza works. In: F. Feldmann, Y. Kapulnik, & J. Baar (Eds.), Proceedings of the Inter- national Symposium “Mycorrhiza for Plant Vitality” and the Joint Meeting of Working Groups 1 - 4 of COST Action 870 (pp. 278-288). Braunschweig: Deutsche Phytomedizinische Gesellschaft. Schollemberger, C. J., & Simon, R. H. (1954). Determination of ex- change capacity and exchangeable bases in soils. Soil Science, 59, 13-24. http://dx.doi.org/10.1097/00010694-194501000-00004 Sieverding, E. (1991). Vesicular-arbuscular mycorrhiza management in tropical agrosystems. Eschborn: GTZ. Skujins, J., & Allen, M. F. (1986). Use of mycorrhizae for land rehabil- itation. MIRCEN Journal of Applied Microbiology and Biotechnolo- gy, 2, 161-176. http://dx.doi.org/10.1007/BF00937191 Smith, S. E., & Read, D. J. (1997). Mycorrhizal symbiosis (2nd ed.). Academic Press, London. Smith, S. E., Smith, F. A., & Jakobsen, I. (2004). Functional diversity in arbuscular mycorrhizal (AM) symbioses: The contribution of the mycorrhizal P uptake pathway is not correlated with mycorrhizal responses in growth or total P uptake. New Phytologist, 162, 511-524. http://dx.doi.org/10.1111/j.1469-8137.2004.01039.x Smith, S. E., & Read, D. J., (2008). Mycorrhizal symbiosis (3rd ed.). London: Academic Press. Smith, F. A., Grace, E. J., & Smith, S. E. (2009). More than a carbon economy: nutrient trade and ecological sustainability in facultative arbuscular mycorrhizal symbioses. New Phytologist, 182, 347-358. http://dx.doi.org/10.1111/j.1469-8137.2008.02753.x Sylvia, D. M. (1990). Inoculation of native woody p lants with ves icular- arbuscular mycorrhizal fungi for phosphate mine land reclamation. Agriculture, Ecosystems & Environment, 31, 253-261. http://dx.doi.org/10.1016/0167-8809(90)90224-2 Tahat, M. M., Kamaruzaman, S., & Radziah, O. (2012). The potential of endomycorrhizal fungi in controlling tomato bacterial wilt Ralsto- nia solanacearum under glasshouse conditions. African Journal of Biotechnology, 11, 13085-13094. Toro, M., Azcón, R., & Barea, J. M. (1997). Improvement of arbuscular mycorrhiza development by inoculaltion of soil phosphate solubiliz- ing rhizobacteria to improve rock phosphate bioavailability (32P) and nutrient cycling. Applied and Environmental Microbiology, 63, 4408-4412. Van der Hejden, M. G. A., Klironomos, J. N., Ursic, M., Moutoglis, P., Streitwolf-Engel, R., Boller, T., Wiemken, A., & Sanders, I. R. (1998). Mycorrhizal fungal diversity determines plant biodiversity, ecosystem variability and productivity. Nature, 396, 69-72. http://dx.doi.org/10.1038/23932 Yanai, R. D., Fahey, T. J., & Miller, S. L. (1995). Efficiency of nutrient acquisition by fine roots and mycorrhizae. In: W. K. Smith, & T. M. Hinckley (Eds.), Resource physiology of conifers: Acquisition, allo- cation and utilization (pp. 75-103). London: Academic Press. http://dx.doi.org/10.1016/B978-0-08-092591-2.50008-X Yeomans, J. C., & Bremner, J. M. (1989). A rapid and precise method for routine determination of organic carbon in soil. Communications in Soil Science and Plant Analysis, 19, 1467-1476. http://dx.doi.org/10.1080/00103628809368027 Zebarth, L. B. J., Neilsen, G. H., Hogue, E., & Neilsen, D. (1999). Influence of organic waste amendments on selected soil physical and chemical properties. Canadian Journal of Soil Science, 79, 501-504. http://dx.doi.org/10.4141/S98-074 Zendejas, L. H. S., Solís, O. M., López, W. W., Vera, R. A., & Gon- zález, P. J. M. (2011). Effects of compost made with sludge and or- ganic residues on bean (Phaseolus vulgaris L.) crop and arbuscular mycorrhizal fungi density. African Jou rnal of Agricultur al Research, 6, 1580-1585.
|