Paper Menu >>
Journal Menu >>
 Materials Sciences and Applications, 2011, 2, 131-140 doi:10.4236/msa.2011.23017 Published Online March 2011 (http://www.SciRP.org/journal/msa) Copyright © 2011 SciRes. MSA 131 Design of 2,4-Dichlorophenoxyacetic Acid Imprinted Polymer with High Specificity and Selectivity Kizhakekuthiathottil Mathew Annamma1,2, Beena Mathew1 1School of Chemical Sciences, Mahatma Gandhi University, Kottayam, India; 2Department of Chemistry, Devamatha College, Kot- tayam, India. Email: beenamathewscs@hotmail.com Received July 9th, 2010; revised December 7th, 2010; accepted January 28th, 2011. ABSTRACT A widely used herbicide 2,4-dichlorophenoxyacetic acid (2,4-D) was imprinted on poly (4-vinylpyridine) (4-VP) using (40%) ethyleneglycol dimethacrylate (EGDMA) as crosslinking agent. The classical imprinting technolo gy ma kes use o f a high degree of crosslinking which does not allow the template molecules to move freely. So the binding sites, lo cated in the central area of the three dimensional polym er matrix are hard to be accessed and the template molecules canno t be extracted totally. But here we propose a low crosslinked system with high specificity and selectivity. The imprinted and non-imprinted polymers were characterized by various spectroscopic techniques. The extent of binding was fol- lowed by batch equilibration method and compared with the respective non-imprinted polymer. Conditions for maxi- mum specific rebinding were set by altering certain factors like template/monomer ratio, concentration o f template so- lution, rebinding medium, mass of polymer and time of incubation. The selectivity of the imprinted polymer was inves- tigated by comparing the binding with structural analogues of 2,4-D like, phenoxyacetic acid (POA), 4-chlorophen- oxyacetic acid (4-CPOA) and 2,4,5-trichlorophenoxy acetic acid (2,4,5-T). The imprinted polymer exhibited high affinity towards the template molecule and was selectively rebound to the specific sites. The binding towards the structural analogues depends on the number of chlorine in the benzene ring. Keywords: Molecular Imprinting, 2,4-Dichlorophenoxyacetic Acid, Specificity, Selectivity, Sepa ration Factor 1. Introduction The agricultural efficiency of the world depends on the effective control of a variety of diseases and pests, espe- cially weeds. Herbicides are the chemicals that are widely used in agricultural field for controlling the growth of herbs, weeds and bushes. Thus the herbicides and their hydrolysis products are the most abundant pollutants found in the environment and in agricultural products. Concern about the health hazards connected with pesti- cide use has focused on 2,4-D and 2,4,5-T as suspected cancer-causing agents. Molecular imprinting is a tech- nique for preparing synthetic polymers possessing recog- nition sites complementary to a template molecule [1-3]. MIPs can be used for the removal of pesticides, herbi- cides, endocrine disrupting compounds and heavy metals from waste and drinking water [4]. The high selectivity of MIPs for organic compounds, along with other useful properties opens up wide opportunities for the use of these materials in analytical chemistry [5-7]. Attempts were made previously to imprint herbicides in polymers and to use them in various applications [8-10]. But the traditional synthesis results in a highly crosslinked rigid structure, which does not allow the template molecules to move freely. So the binding sites, located in the central area of the three dimensional polymer matrix are hard to be accessed and the template molecules cannot be ex- tracted totally. Hereafter, many imprinting strategies have been introduced to solve the problems faced by the tradi- tional MIPs [11-15]. The reported techniques for herbi- cide detection using MIPs require high crosslinking, so- phisticated instruments, skilled technician and longer analysis time. The most common methods of herbicide detection are gas chromatography with mass spectrome- try detection (GC-MS) and HPLC. These methods re- quire one or more preconcentration steps for trace level  Design of 2,4-Dichlorophenoxyacetic Acid Imprinted Polymer with High Specificity and Selectivity Copyright © 2011 SciRes. MSA 132 detection and quantification of herbicides due to insuffi- cient sensitivity of these methods. In the present work high specific and selective recognition is given by low crosslinked polymers. Further the UV-visible spectro- photometric method is technically simple and can per- form easily. The present paper deals with the synthesis of low crosslinked (40%) 2,4-dichlorophenoxyacetic acid (2,4-D) imprinted 4-vinylpyridine (4-VP) polymer, and the specificity and selectivity studies. The imprinted polymers were prepared by non-covalent method of im- printing polymerization in the presence of polar solvents like methanol and water. Non-imprinted polymers were also prepared without the template. Factors affecting specificity and selectivity were also discussed. The re- binding ability and selectivity performance of the poly- mer was followed by UV-vis. spectrophotometric method. The nature of binding sites was followed by Scatchard analysis. 2. Experimental 2.1. Materials and Methods 4-chlorophenoxyacetic acid (4-CPOA), 2,4-dichlorophen- oxyacetic acid (2,4-D), 2,4,5-trichlorophenoxyacetic acid (2,4,5-T), and 4-vinylpyridine (4-VP) were obtained from Sigma-Aldrich (USA). Ethyleneglycol dimethacrylate (EGDMA) was from Merck (Germany). Phenoxyacetic acid (POA) and 2,2’-azobisisobutyronitrile (AIBN) were purchased from SRL (Mumbai). Methanol, acetonitrile and chloroform were obtained from Merck (India). 4- vinylpyridine was distilled under reduced pressure prior to use. Doubly distilled water was used throughout. The imprinted polymers were characterized by FT-IR (8400 S, Shimadzu), UV-vis. (Shimadzu 2450), and 1H (BRUKER AMX-400) and 13C CP-MAS-NMR (BRUKER DSX-300) spectrometer. 2.2. Synthesis of EGDMA-Crosslinked 2,4-D Imprinted and Non-Imprinted Polymers in Methanol/Water EGDMA-crosslinked 2,4-D imprinted poly (4-vinylpyri- dine) with 1:1, 1:2 and 1:4 ratios of 2,4-D and 4-vinyl- pyridine were prepared (Table 1). The template 2,4-D (1 mmol), functional monomer (4-VP), (4 mmol), required amount of the crosslinking agent ethyleneglycol dime- thacrylate and the initiator AIBN (0.32 mmol) were weighed into glass tubes and dissolved in10 ml solvent (methanol/water, 4:1, v/v). The resulting mixture was purged with nitrogen for 15 min. The tubes were then sealed and inserted in a water bath at 60˚C for 4 h, fol- lowed by 2 h at 70˚C (Figure 1). The resultant hard bulk polymers were ground in a mechanical mortar and wet- sieved in acetone through a 25 m sieve. Non-imprinted Figure 1. Synthesis of 2,4-D imprinted and non-imprinted polymers. polymers were also prepared using the same recipe but without the addition of the template. 2.3. Extraction of Imprinted Template: General Procedure The fine polymer particles were carefully washed by in- cubation in methanol/acetic acid (7:3, v/v, 2x), acetoni- trile/acetic acid (9:1, v/v, 2x), acetonitrile (1x), methanol (2x) for 2 h and Soxhlet extracted with methanol until no template could be detected under UV (max = 284 nm) in the wash solution. The particles were then suspended in acetone and allowed to settle for 4 h. The solvent was removed by centrifugation and the particles were dried to constant weight in vacuum. 2.4. Swelling Studies Fixed amount of the polymer particles were packed into sintered crucibles, which were filled with different sol- vents. After 24 h of equilibration, the excess solvent was removed from the polymer by applying reduced pressure for 1 min. and the weight of the swollen particles (Ww) was measured. Subsequently the particles were freeze- dried for 24 h and were weighed again to obtain the dry weight (Wd). The swelling ratio (SR) of the polymer in each solvent was calculated from the following equation: SR = (Ww − Wd)/Wd. 2.5. Rebinding of Template: General Procedure The template-desorbed polymers were treated with solu- tions of the desorbed template and the extent of rebinding was followed by UV measurements at 284 nm. The poly- mer particles were put into sample tubes, and the template solutions of known concentrations were introduced and equilibrated for a period of time. After this incubation, the  Design of 2,4-Dichlorophenoxyacetic Acid Imprinted Polymer with High Specificity and Selectivity Copyright © 2011 SciRes. MSA 133 Table 1. Synthesis of 40% EGDMA-crosslinked MIPs and NIPs with varying 2,4-D/4 VP ratio. Yield (%) Ratio of 2,4-D:4-VP 2,4-D (1 mmol) 4-VP EGDMA MIP NIP 1:1 0.221 g 0.11 ml (1 mmol) 0.14 ml (0.75 mmol) 35 45 1:2 0.221 g 0.22 ml (2 mmol) 0.28 ml (1.5 mmol) 65 68 1:4 0.221 g 0.44 ml (4 mmol) 0.56 ml (3 mmol) 72 75 polymer particles were filtered off, and the remaining concentrations of the template were determined spectro- photometrically. The amount of template bound to the polymer Sb (mM/g polymer) was calculated by the equa- tion, Sb = (Co – Ct)/W. Where Co and Ct are the 2,4-D molar concentration in the solution at initial and after interval time ‘t’, respectively. ‘W’ is the weight of the dry polymer used. 2.6. Optimization of the Conditions of 2,4-D Rebinding The optimum conditions for maximum 2,4-D rebinding were set by changing certain factors like templatemonomer ratio, rebinding solvent, time, concentration of 2,4-D solution and mass of polymer. 2.7. Factors Affecting Rebinding 2.7.1. Concentration of 2,4-D Solution The influence of concentration of template solution on rebinding was evaluated by batch binding experiments. From the difference in concentration of template solution before and after incubation, the amount of template bound by the polymer was determined. 2.7.2. Rebinding Solvent The template solutions were prepared in different solvents and the rebinding in each solvent was determined spec- trophotometrically. 2.7.3. Time of Rebinding The time required for the saturation of binding sites was determined by measuring the absorbance at regular in- tervals of time till saturation was attained at 30˚C. 2.7.4. Mass of Polymer Varying amounts of polymers were introduced into equal volume of template solution for a fixed time and rebinding was followed. 2.8. Selectivity Studies: General Procedure The selectivity of imprinted polymers were evaluated by incubating template desorbed imprinted polymer in solu- tions of structural analogues for definite time and the rebinding was followed spectrophotometrically. Separa- tion and selectivity factors were also determined. 3. Results and Discussion 3.1. Synthesis of EGDMA-Crosslinked 2,4-D Imprinted and Non-Imprinted Polymers with Varying Composition of Functional Monomers and Template EGDMA-crosslinked 2,4-D imprinted and non-imprinted polymers with varying template-monomer ratio were synthesized by free radical polymerization (Figure 1). 3.2. Characterization of 2,4-D Imprinted and Non-Imprinted Polymers 3.2.1. FT-IR The incorporation of EGDMA in both imprinted and non-imprinted polymers was supported by the IR spectra. The bands at 1396 and 1458 cm−1 correspond to C–H bend of CH3 and CH2 respectively. The C=O stretch of the EGDMA crosslinking was observed at 1647 cm−1. The IR spectrum of 4-vinylpyridine showed an intense band at 1596 cm−1 due to the C=N stretching. The spec- tral characteristics of 4-vinylpyridine changed upon addi- tion of 2,4-D. The 2,4-D/4-vinylpyridine complex re- vealed a new band at 1546 cm−1 within the C=N mode. The peaks at 1596 and 1546 cm−1 were assigned to mono- meric 4-vinylpyridine and H-bonded 4-vinylpyridine respectively. The C=O and O–H stretching of the car- boxyl group of 2,4-D at 1735 and 3263 cm−1 were also shifted and broadened on complexation with 4-vinylpy- ridine. These changes strongly suggest the formation of intermolecular H-bonding between the carboxylic group of 2,4-D and the nitrogen of 4-vinylpyridine. 3.2.2. 1H NMR The pre-polymerisation complex formation during the preparation of molecular imprinted polymers can be in- vestigated by 1H NMR. Due to the low solubility of 2,4-D in chloroform, the 1H NMR titration studies of the 2,4-D/4-VP system were performed in CD3OD. The study was conducted by adding increasing amounts of 4-VP into 2,4-D solution and the spectra were recorded after each addition. Because of the rapid exchange be- tween labile proton of the template and solvent deute-  Design of 2,4-Dichlorophenoxyacetic Acid Imprinted Polymer with High Specificity and Selectivity Copyright © 2011 SciRes. MSA 134 rium atom, the signal due to acidic proton of the carbox- ylic acid is not seen in the spectrum. Therefore the change in chemical shifts of the aromatic and CH2 pro- tons of 2,4-D as well as aromatic protons of 4-VP were investigated (Figure 2). As a result of ion-pair formation between basic nitrogen and acidic proton, the chemical environment of H2 and H6 (8.8 ppm), H3 and H5 (7.6 ppm) of 4-VP changes and the peaks corresponding to them were shifted upfield. An upfield shifting of the peaks corresponding to the aromatic protons of 2,4-D (H3 - 7.7, H5 - 7.5, H6 - 7.2 ppm) was also observed and attributed to the participation in - stacking interactions. The signals due to CH2 protons of 2,4-D (4.9 ppm) was broadened along with upfield shifting and gradually the peaks disappeared due to complex formation with 4-VP. The results confirmed the existence of a 1:3 (2,4-D/4-VP) complex prior to polymerisation. 3.2.3. 13C CP-MAS-NMR The incorporation of crosslinking agent and functional monomer 4-VP in the polymer backbone of imprinted polymer was confirmed by 13C NMR spectrum (Figure 3). The ester carbonyl of the EGDMA crosslinking was characterised by the strong peak at 178.8 ppm. The small peak at 65.0 and the one at 16.7 ppm correspond to the OCH2 and CH3 carbons of EGDMA. The intense peak at 2,4-D 4-VP Figure 2. 1H NMR spectra of 2,4-D with increasing concen- tration of 4-VP. Figure 3. 13C NMR spectrum of EGDMA-crosslinked im- printed polymer. 42.8 ppm corresponds to CH2 carbon of the polymer backbone. 3.3. Determination of Swelling Ratio (SR) In order to determine the swelling ratios of 2,4-D im- printed and non-imprinted polymers, the polymers were immersed in four different solvents such as acetonitrile, methanol, methanol/water and water. The dispersed poly- meric particles were recovered by vacuum filtration and the swollen weight (Ww) of the particles was measured. Subsequently, the particles were dried and weighed again to obtain the dry weight (Wd). The swelling ratio [16] of the polymer was calculated as given below and summa- rised in the experimental part. The polymer exhibited higher swelling in methanol- water, the porogen. Here the swelling ratios of non-im- printed polymers are higher than that of the imprinted polymers. This is due to the reluctance of the crosslinked polymer matrix to expand from the designed geometry around the template molecule. Whereas there is no such complementary cavity and framework in the non-im- printed polymer and so it could undergo extensive swell- ing. The swelling ratio of both imprinted and non-im- printed polymer in water is very high compared to other solvents. This anomalous behaviour in water is due to the possibility of extensive H-bonding with the nitrogen in 4-vinylpyridine. 3.4. 2,4-D Rebinding Studies The methodology of molecular imprinting induces the formation of recognition sites with predetermined selec- tivity into a synthetic matrix. After removal of the tem-  Design of 2,4-Dichlorophenoxyacetic Acid Imprinted Polymer with High Specificity and Selectivity Copyright © 2011 SciRes. MSA 135 Table 2. Swelling ratios of 2,4-D imprinted and non-im- printed polymers. Swelling Ratio Solvent MIP NIP CH3CN 0.15 0.28 CH3OH 1.72 0.97 CH3OH/H2O 2.25 4.63 H2O 7.09 4.30 plate, complementary binding sites are revealed and a molecular memory is introduced into the polymer, capa- ble of rebinding the template with high specificity and selectivity [3,4,17]. According to this principle of mo- lecular imprinting, the 2,4-D imprinted polymers must specifically rebind the print molecule than the non-im- printed polymers. The imprinted and non-imprinted poly- mers were investigated for their rebinding capacity by incubating a fixed amount of each polymer in template solution of known concentration and equilibrated for a period of time ‘t’. The concentration of template solution before and after incubation was determined spectropho- tometrically. The amount of template bound to the poly- mer [S]b (mM/g) was calculated as discussed earlier. Specificity in 2, 4-D Rebinding by Imprinted Polymers The 2,4-D imprinted polymers showed high specificity towards the template than the non-imprinted polymer. The investigation was done by comparing the specific binding of imprinted polymer (MIP) with a non-im- printed polymer (NIP) of same crosslinking (40%). Equal amounts of MIP and NIP were introduced into 0.9 mM solution of 2, 4-D for a definite time and the amount of 2,4-D bound was determined. From the difference in binding by the MIP and NIP, it is evident that the im- printed polymer has a significant affinity towards 2,4-D than the non-imprinted polymer. Further, the specificity shown by the imprinted polymer supports the memory of the polymer towards the print molecule (Figure 4). 3.5. Template-Monomer Ratio on 2,4-D Rebinding It has been found that the molar relationship between the template and functional monomer influences the number and quality of MIP recognition sites. Hence 2,4-D im- printed and non-imprinted polymers were prepared in 1:2 and 1:4 template-monomer ratio. To investigate the in- fluence of this ratio on rebinding, imprinted and non- imprinted polymers were incubated in equal volume of 2,4-D solutions of definite concentration for a fixed time. After filtration the 2,4-D binding was followed spectro- photometrically. The results are summarised in Figure 5. The 1:4 system offered high specific binding com- pared to the 1:2 system. In order to accomplish highly efficient imprinting, the template-monomer adduct must exist in large excess with respect to free template and functional monomer. Otherwise non-selective polymeri- sation would occur concurrently and diminish the effi- ciency of molecular imprinting. Thus the 1:4 system is more specific than the 1:2 system. Hence for further studies the imprinted and non-imprinted polymers in the 1:4 template-monomer ratios were used. 3.6. Optimisation of the Conditions of 2,4-D Rebinding There is the need to optimise the conditions for maximum template rebinding by the imprinted polymers. The total Figure 4. Specificity in 2,4-D rebinding by 40% EGDMA- crosslinked MIP and NIP. Figure 5. Effect of template-monomer ratio on 2,4-D re- binding by MIP and NIP.  Design of 2,4-Dichlorophenoxyacetic Acid Imprinted Polymer with High Specificity and Selectivity Copyright © 2011 SciRes. MSA 136 rebinding to a MIP is the sum of specific binding to the imprinted binding sites and non-specific binding to the polymer. The non-specific binding to the polymer is measured as the binding, under identical conditions to a non-imprinted polymer. If this non-specific binding to the polymer is dominant, specific binding to the imprinted sites will be obscured. Thus the ratio of the specific to non-specific binding should be optimised to observe the binding at the imprinted sites. 3.6.1. Effect of Concentration of 2,4-D Solution To evaluate the variation of rebinding with concentration of 2,4-D solution, batch methods were carried out using stock solutions of 2,4-D with concentrations ranging from 0.3 to 1.5 mM. Definite amount of MIPs were in- cubated in these solutions for a fixed interval of time and the amount of 2,4-D bound at the saturation point [S]b, was calculated as described earlier. It was plotted against initial concentration Co and is given in Figure 6. The extent of 2,4-D binding increased with concentra- tion up to 0.9 mM and then remained constant. Thus the binding attained a saturation point at 0.9 mM. The indi- vidual cavities in the polymer vary in their affinity and selectivity towards the template [18]. The binding of the template molecules to high affinity sites concentrated inside the polymeric domains led to the increase in spe- cific binding, which later resulted in shape change of the polymer network. Binding sites on the surface of the polymer bound the template without causing much reor- ganisation of the polymer structure. 3.6.2. Determination of Binding Parameters and Guest Binding Constants In the molecular imprinting technique, Scatchard model was often used to evaluate the binding characteristics of the imprinted polymers [19,20]. The binding data ob- tained were treated with Scatchard equation [S]b/[S]f = (Smax − [S]b)/KD, where KD is an equilibrium dissociation constant of the binding site, Smax an apparent maximum number of binding sites and [S]b is the amount of 2,4-D bound to MIP. [S]f is the fraction of 2,4-D left in solution. The plot of [S]b/[S]f against [S]b is shown in Figure 7. The Scatchard plot is not linear and composed of two straight lines indicating that the binding sites in the polymer matrix are heterogeneous in respect to the affin- ity for 2,4-D. Thus two classes of binding sites are mainly produced in the studied template concentration. From the slope and intercept of the straight lines obtained, the values of KD and Smax are determined, where KD1 (50 M/L) and KD2 (201 M/L) are the dissociation constants, and Smax1 (12.5 mM/g) and Smax2 (19 mM/g) are the ap- parent maximum number of binding sites at high and low affinity sites. Since the dissociation constant at high af- finity site (KD1) is lower than that at low affinity site, Figure 6. Binding isotherm for the binding of 2,4-D by MIP. Figure 7. Scatchard plot to determine the nature of binding sites. guest binding at high affinity site is stronger. 3.6.3. Effect of Rebinding Medium Molecular imprinted polymers utilise a solvent in po- lymerisation to provide porosity within the network poly- mer. Binding properties of molecular imprinted polymers are influenced by the type of solvent, or porogen, used in the polymer synthesis and the solvent used in the par- ticular application of the MIP [21]. A number of reports in the literature indicated that the best recognition of im- printed polymer occurs when the rebinding medium and the porogen used are the same [22,23]. The ability of the rebinding medium to recreate the binding site dimensions formed during the polymerisation determines the binding performance of the imprinted polymer in that medium. To determine the effect of binding medium on specific binding properties, imprinted and non-imprinted polymers were equilibrated with solutions of 2,4-D in acetonitrile, methanol, methanol/water, and water. The EGDMA cross- linked polymer offered maximum specificity in metha- nol/water, the porogen. The high binding observed in water is due to a partitioning effect of the lipophilic 2,4-D into the hydrophobic polymer matrix. The results are given  Design of 2,4-Dichlorophenoxyacetic Acid Imprinted Polymer with High Specificity and Selectivity Copyright © 2011 SciRes. MSA 137 in Figure 8. 3.6.4. Effect of Time To optimise the time taken for maximum binding of 2,4-D by MIP and NIP, definite amounts of the polymer was equilibrated with 2,4-D solution of known concentration and the binding was followed spectrophotometrically at definite intervals of time (Figure 9). The imprinted polymers took more time for saturation of binding sites compared to the non-imprinted polymers. The imprinted polymers possessing shape complementary binding sites took more time to attain saturation, since the template molecules have to penetrate through the highly cross- linked polymer network to access the imprinted cavities for rebinding. In non-imprinted system, there is no such specific arrangement of the binding sites, and non-spe- cific interactions occur only at the surface of the polymer, the binding of template is rather fast and random. The MIP attained saturation within 4-6 h. So it is not neces- sary to equilibrate the polymers for 24 h for getting the imprinted sites saturated. Figure 8. Effect of solvent on 2,4-D rebinding by EGDMA- crosslinked MIP and NIP. Figure 9. Time dependence on 2,4-D rebinding by MIP and NIP. 3.6.5. Amount of Polymer Increasing the amount of polymer is expected to provide more binding sites for a substrate and as a result the amount of the substrate bound is also expected to increase. The effect of the mass of polymer on rebinding was ob- served by incubating varying amounts of MIP and NIP in fixed volume of 0.9 mM 2,4-D solution. As the amount of polymer is increased, the amount of bound substrate also increased (Figure 10). When higher amount of polymer was used the substrate bound to the higher affinity binding sites in preference to the low affinity binding sites. The increase in the number of binding sites with increase in mass of the polymer leads to the increase in specificity. But the substrate bound per gram decreased. This is due to the possible changes in binding parameters with the dif- ferent amounts of the polymers used in the batch rebind- ing studies. The non-imprinted polymers bound signify- cantly less template than the imprinted polymers. 3.7. Selectivity of 2,4-D Imprinted Polymers 3.7.1. Template Selectivity The selectivity of MIP is based on the configuration of the binding sites and the orientation of the functional groups in the sites. Three structural analogues of 2,4-D, viz. phenoxyacetic acid (POA), 4-chlorophenoxyacetic acid (4-CPOA) and 2,4,5-trichlorophenoxyacetic acid (2,4,5-T) were used to evaluate the selectivity of the imprinted polymer. The molecular structures of these compounds are given in Figure 11. The molecules related to 2,4-D in the acidic part and different in the aromatic part were recognised by the im- printed polymers, but to a lesser extent than the template (Figure 12). Since the molecules exactly matching the template molecule in the carboxylic substructure can be recognised by the imprinted polymer, it is clear that this interaction is strongly governed by the ion pair formation. It is also observed that molecules resembling 2,4-D in the Figure 10. Effect of the mass of polymer on 2,4-D rebind- ing by MIP and NIP. 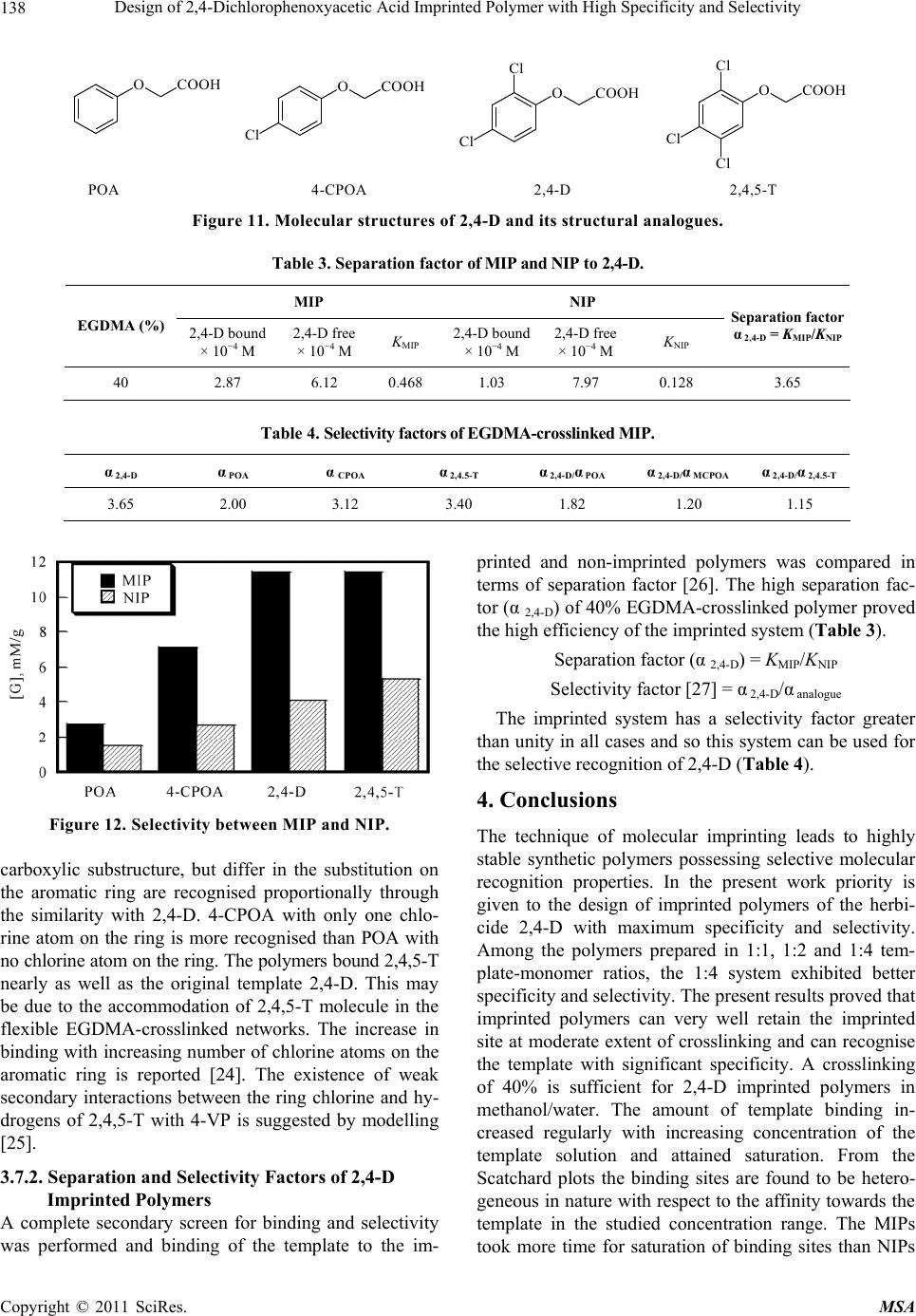 Design of 2,4-Dichlorophenoxyacetic Acid Imprinted Polymer with High Specificity and Selectivity Copyright © 2011 SciRes. MSA 138 OCOOH Cl Cl Cl OCOOH Cl Cl OCOOH OCOOH Cl POA 4-CPOA 2,4-D 2,4,5-T Figure 11. Molecular structures of 2,4-D and its structural analogues. Table 3. Separation factor of MIP and NIP to 2,4-D. MIP NIP EGDMA (%) 2,4-D bound × 10−4 M 2,4-D free × 10−4 M KMIP 2,4-D bound × 10−4 M 2,4-D free × 10−4 MKNIP Separation factor α 2,4-D = KMIP/KNIP 40 2.87 6.12 0.468 1.03 7.97 0.128 3.65 Table 4. Selectivity factors of EGDMA-crosslinked MIP. α 2,4-D α POA α CPOA α 2,4.5-T α 2,4-D/α POA α 2,4-D/α MCPOA α 2,4-D/α 2,4.5-T 3.65 2.00 3.12 3.40 1.82 1.20 1.15 Figure 12. Selectivity between MIP and NIP. carboxylic substructure, but differ in the substitution on the aromatic ring are recognised proportionally through the similarity with 2,4-D. 4-CPOA with only one chlo- rine atom on the ring is more recognised than POA with no chlorine atom on the ring. The polymers bound 2,4,5-T nearly as well as the original template 2,4-D. This may be due to the accommodation of 2,4,5-T molecule in the flexible EGDMA-crosslinked networks. The increase in binding with increasing number of chlorine atoms on the aromatic ring is reported [24]. The existence of weak secondary interactions between the ring chlorine and hy- drogens of 2,4,5-T with 4-VP is suggested by modelling [25]. 3.7.2. Separation and Selectivity Factors of 2,4-D Imprinted Polymers A complete secondary screen for binding and selectivity was performed and binding of the template to the im- printed and non-imprinted polymers was compared in terms of separation factor [26]. The high separation fac- tor (α 2,4-D) of 40% EGDMA-crosslinked polymer proved the high efficiency of the imprinted system (Table 3). Separation factor (α 2,4-D) = KMIP/KNIP Selectivity factor [27] = α 2,4-D/α analogue The imprinted system has a selectivity factor greater than unity in all cases and so this system can be used for the selective recognition of 2,4-D (Table 4). 4. Conclusions The technique of molecular imprinting leads to highly stable synthetic polymers possessing selective molecular recognition properties. In the present work priority is given to the design of imprinted polymers of the herbi- cide 2,4-D with maximum specificity and selectivity. Among the polymers prepared in 1:1, 1:2 and 1:4 tem- plate-monomer ratios, the 1:4 system exhibited better specificity and selectivity. The present results proved that imprinted polymers can very well retain the imprinted site at moderate extent of crosslinking and can recognise the template with significant specificity. A crosslinking of 40% is sufficient for 2,4-D imprinted polymers in methanol/water. The amount of template binding in- creased regularly with increasing concentration of the template solution and attained saturation. From the Scatchard plots the binding sites are found to be hetero- geneous in nature with respect to the affinity towards the template in the studied concentration range. The MIPs took more time for saturation of binding sites than NIPs  Design of 2,4-Dichlorophenoxyacetic Acid Imprinted Polymer with High Specificity and Selectivity Copyright © 2011 SciRes. MSA 139 and overnight equilibration is not necessary for getting the sites saturated. Selectivity studies with structural analogues revealed that in addition to complementarity in size and shape, optimal spatial fit also had a role in bind- ing interactions. The present work in molecular imprint- ing provided an alternative method against the conven- tional analytical methods. In the present work high spe- cific and selective recognition is given by low cross- linked polymers. Further the UV-visible spectrophoto- metric method is technically simple and can perform easily. The attempts made in this work led to the suc- cessful design of 2,4-D imprinted polymers with high specificity and selectivity. 5. Acknowledgements Annamma is grateful to the University Grants Commis- sion (UGC) for the award of Teacher Fellowship. REFERENCES [1] K. Ensing and de B. Theo, “Tailor-Made Materials for Tailor-Made Applications: Application of Molecular Im- prints in Chemical Analysis,” TrAC Trends in Analytical Chemistry, Vol. 18, No. 3, 1999, pp. 138-145. [2] D. Kriz, O. Ramstrom and K. Mosbach, “Molecular Im- printing-New Possibilities for Sensor Technology,” Ana- lytical Chemistry News and Features, Vol. 69, 1997, pp. 345-349. [3] F. G. Tamayo, J. L. Casillas and A. Martin-Esteban, “Cleanup of Phenylurea Herbicides in Plant Sample Ex- tracts Using Molecularly Imprinted Polymers,” Analytical and Bioanalytical Chemistry, Vol. 381, No. 6, 2005, pp. 1234-1240. doi:10.1007/s00216-005-3071-1 [4] S. A. Piletsky, S. Alcock and A. P. F. Turner, “Molecular Imprinting: At the Edge of Third Millennium,” Trends in Biotechnology, Vol. 19, No. l, 2001, pp. 9-12. doi:10.1016/S0167-7799(00)01523-7 [5] L. I. Anderson, “Molecular Imprinting: Developments and Applications in the Analytical Chemistry Field,” Journal of Chromatography B: Biomedical Sciences and Applications, Vol. 745, No. l, 2000, pp. 3-13. doi:10.1016/S0378-4347(00)00135-3 [6] B. Sellergren, “Molecularly Imprinted Polymers: Man-Made Mimics of Antibodies and their Application in Analytical Chemistry,” Elsevier, 2001. [7] K. Haupt, “Imprinted Polymers—Tailor-Made Mimics of Antibodies and Receptors,” ChemInform, Vol. 34. No. 15, 2003, pp. 171-178. doi:10.1002/chin.200315284 [8] T. D. Panasyuk, M. Vladimir, U. Mathias and S. Otto, “Impedometric Herbicide Chemosensors Based on Mole- cularly Imprinted Polymers,” Analytica Chimica Acta, Vol. 435, No. 1, 2001, pp. 157-162. doi:10.1016/S0003-2670(00)01280-0 [9] M. T. Muldoon and L. H. Stanker, “Molecularly Im- printed Solid Phase Extraction of Atrazine from Beef Liver Extracts,” Analytical Chemistry, Vol. 69, No. 5, 1997, pp. 803-808. doi:10.1021/ac9604649 [10] K. P. L. Mitch, F. C. Cheuk and H. W. L. Michael, “A Sol-Gel Derived Molecular Imprinted Luminiscent PET Sensing Materials for 2,4-Dichlorophenoxyacetic Acid,” Journal of Materials Chemistry Articles, Vol. 11, 2001, pp. 2985-2991. [11] E. Yimaz, K. Haupt and K. Mosbach, “The Use of Immo- bilized Templates—A New Approach in Molecular Im- printing,” Angewandte Chemie International Edition, Vol. 39, No. 12, 2000, pp. 2115-2118. doi:10.1002/1521-3773(20000616)39:12<2115::AID-AN IE2115>3.0.CO;2-V [12] J. F. Wang, A. G. Cormack, D. C. Sherrington and E. Khoshdel, “Monodisperse, Molecularly Imprinted Poly- mer Microspheres Prepared by Precipitation Polymeriza- tion for Affinity Separation Applications,” Angewandte Chemie International Edition, Vol. 42, No. 43, 2003, pp. 5336-5338. doi:10.1002/anie.200352298 [13] S. C. Zimmerman, M. S. Wendland, N. A. Rakow, I. Zharov and K. S. Suslick, “Synthetic Hosts by Monomo- lecular Imprinting inside Dendrimers,” Nature, Vol. 418, 2002, pp. 399-403. doi:10.1038/nature00877 [14] C. H. Lu, W. H. Zhou, B. Han, H. H. Yang, X. Chen and X. R. Wang, “Surface Imprinted Core-Shell Nanoparticles for Sorbent Assays,” Analytical Chemistry, 79, No. 14, 2007, pp. 5457- 5461. doi:10.1021/ac070282m [15] C. D. Ki and J. Y. Chang, “Preparation of a Molecularly Imprinted Polymeric Nanocapsule with Potential Use in Delivery Applications,” Macromolecules, Vol. 39, No. 9, 2006, pp. 3415-3419. doi:10.1021/ma052480q [16] S. Lu, G. Cheng and X. Pang, “Study on Preparation of Protein-Imprinted Soft-Wet Gel Composite Microspheres with Magnetic Susceptibility and Their Characteristics 1. Preparation and Particle Morphology,” Journal of Applied Polymer Science, Vol. 100, No. 1, 2006, pp. 684-694. doi:10.1002/app.23411 [17] G. Wulff, “Molecular Imprinting in Cross-Linked Materi- als with the Aid of Molecular Templates—A Way to- wards Artificial Antibodies,” Angewandte Chemie Inter- national Edition, Vol. 34, No. 17, 1995, pp. 1812-1832. doi:10.1002/anie.199518121 [18] D. Kriz and K. Mosbach, “Competitive Amperometric Mor- phine Sensor Based on an Agarose Immobilized Molecularly Imprinted Polymer,” Analytica Chimica Acta, Vol. 300, No. 1-3, 1995, pp. 71-75. doi:10.1016/0003-2670(94)00368-V [19] M. Kempe and K. Mosback, “Binding Studies on Sub- strate and Enantioselective Molecularly Imprinted Poly- mers,” Analytical Letters, Vol. 24, No. 7, 1991, pp. 1137-1145. [20] C. Yu and K. Mosbach, “Influence of Mobile Phase Composition and Cross-Linking Density on the Enan- tiomeric Recognition Properties of Molecularly Imprinted Polymers,” Journal of Chromatography A, Vol. 888, No. 1-2, 2000, pp. 63-72. doi:10.1016/S0021-9673(00)00556-2  Design of 2,4-Dichlorophenoxyacetic Acid Imprinted Polymer with High Specificity and Selectivity Copyright © 2011 SciRes. MSA 140 [21] D. J. O’Shannessy, G. Ekberg, L. I. Anderson and K. Mosbach, “Recent Advances in the Preparation and Use of Molecularly Imprinted Polymers for Enantiomeric Resolution of Aminoacid Derivatives,” Journal of Chro- matography A, Vol. 470, No. 2, 1989, pp. 391-399. doi:10.1016/S0021-9673(01)83567-6 [22] Z. Quing-Zhi, K. Haupt, K. Dietmar and N. Reinhard, “Molecularly Imprinted Polymers for Met Sulfuron-Methyl and Its Binding Characteristics for Sulfonylurea Herbi- cides,” Analytica Chimica Acta, Vol. 468, 2002, pp. 217-227. doi:10.1016/S0003-2670(01)01437-4 [23] R. J. Umpleby, G. T. Rushton, R. N. Shah, A. M. Rampey, J. C. Bradshaw, J. K. Berch and K. D. Shimizu, “Recogni- tion Directed Site-Selective Chemical Modification of Molecularly Imprinted Polymers,” Macromolecules, Vol. 34, No. 24, 2001, pp. 8446-8452. doi:10.1021/ma010903s [24] S. Lachlan, I. H. Clovia, A. McCluskey and M. C. Bowyer, “Synthesis and Evaluation of a Molecularly Imprinted Polymer Selective to 2,4,6-Trichlorophenol,” Australian Journal of Chemistry, Vol. 57, 2004, pp. 759-764. doi:10.1071/CH04004 [25] C. Baggiani, C. Giovannoli, L. Anfossi and C. Tozzi, “Molecularly Imprinted Solid-Phase Extraction Sorbent for the Clean-up of Chlorinated Phenoxyacids from Aqueous Samples,” Journal of Chromatography A, Vol. 938, No. 1-2, 2001, pp. 35-44. doi:10.1016/S0021-9673(01)01126-8 [26] F. Lanza and B. Sellergren, “Method for Synthesis and Screening of Large Groups of Molecularly Imprinted Polymers,” Analytical Chemistry, Vol. 71, 1999, pp. 2092-2096. doi:10.1021/ac981446p [27] H. S. Andersson, J. G. Karlsson, S. A. Piletsky, A. Koch-Schmidt, K. Mosbach and I. A. Nicholls, “Study of the Nature of Recognition in Molecularly Imprinted Polymers, II [1]: Influence of Monomer-Template Ratio and Sample Load on Retention and Selectivity,” Journal of Chromatography A, Vol. 848, 1999, pp. 39-49. doi:10.1016/S0021-9673(99)00483-5 |

