 Neuroscience & Medicine, 2013, 4, 290-298 Published Online December 2013 (http://www.scirp.org/journal/nm) http://dx.doi.org/10.4236/nm.2013.44043 Open Access NM Affinity Crosslinking of Y1036 to Nerve Growth Factor Identifies Pharmacological Targeting Domain for Small Molecule Neurotrophin Antagonists Joseph K. Eibl1, Zouleika Abdallah1, Allison E. Kennedy2, John A. Scott1, Gregory M. Ross1,2 1Northern Ontario School of Medicine, Sudbury, Canada; 2Biology Department, Laurentian University, Sudbury, Canada. Email: gross@nosm.ca Received October 24th, 2013; revised November 20th, 2013; accepted December 10th, 2013 Copyright © 2013 Joseph K. Eibl et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. ABSTRACT Classically, small molecule antagonists have targeted membrane bound receptors and intracellular enzyme targets. While this drug discovery strategy is extremely successful, the number of new chemical entities in the pharmaceutical pipeline is diminishing and complementary strategies are in need. A particularly attractive therapeutic strategy is to neutralize soluble signalling proteins using small molecules. Small molecule-based technologies have the potential to sufficiently alter the molecular topology of a given ligand and inhibit ligand/receptor interactions—effectively neutral- izing the ligand’s signalling capacity. Recent technical advances in the field of structural biology have enabled the elu- cidation of ligand/receptor complexes at atomic resolution enabling a detailed appreciation of the molecular interactions governing ligand-mediated receptor activation. Exploiting molecular modeling techniques to study these signalling complexes allows for a paradigm shift from “receptorcentric” to “ligandcentric” screening strategies. Nerve growth fac- tor (NGF) is a prototypical protein signalling ligand, which binds two receptors, TrkA and p75NTR. We first explore the molecular landscape governing the ligand/receptor interactions of NGF/TrkA and NGF/p75 structures. Next, we use the recently reported NGF neutralizing small-molecule, Y1036, as an affinity probe to determine residues in proximity to the pharmacological targeting domain of NGF and perform theoretical docking experiments to predict the residues which comprise distinct pharmacological targeting domains on the surface of NGF. Exploiting such strategies may fa- cilitate “ligandcentric” drug discovery and could further the development of a trophic-factor-selective compound such as a BDNF-selective antagonist. Keywords: NGF; BDNF; Inhibitor; Antagonist; Pain 1. Introduction Historically, small molecule-based therapeutics have generally focused on compounds directed towards mem- brane bound receptors, channels and intracellular enzyme targets. However, the physiological role of several of these targets is governed by soluble protein ligands such as cytokines or growth factors. Traditional “receptorcen- tric” screening strategies yielded clinically effective therapeutics for most of the past century [1]. Obvious examples in the field of pain research include targets such as cyclooxygenase (Cox1/2), canabinoid and opioid receptors, and a variety of ion-channel inhibitors [2]. However, in the last decade, the success of these strate- gies has not kept pace with the demand for new chemical entities and, complementary approaches may help to re- vitalize lead identification in many drug discovery pro- grams. In an attempt to counteract declining productivity, re- search and development in the pharmaceutical industry are moving towards unprecedented targets and novel technologies [3]. By definition, unprecedented targets are proteins or enzymes for which a pharmacological (or biological) therapy has yet to be approved in a clinical setting [3]. In the field of neuropharmacology, a prime example of an unprecedented target is nerve growth fac- tor (NGF). NGF is a soluble ~26 kDa homodimeric protein which is known to be critical for the development and mainte- nance of the central and peripheral nervous system [4]. NGF is a member of the neurotrophin family of proteins and mediates its physiological role by binding the com-  Affinity Crosslinking of Y1036 to Nerve Growth Factor Identifies Pharmacological Targeting Domain for Small Molecule Neurotrophin Antagonists 291 mon low-affinity receptor p75NTR [5] and the selective high-affinity receptor TrkA [6]. Interestingly, the dys- regulation of NGF has also been implicated in several disease states of the nervous system [4,7]. Thus, identi- fying mechanisms to inhibit pathological signalling may have significant therapeutic potential in the field of neu- ropharmacology. The therapeutic efficacy of targeting the ligand in a ligand/receptor system has been demonstrated by the neutralizing effect of monoclonal antibodies in the clinic [8]. However, antibody-mediated therapies have several technical, practical, and economical limitations that re- strict their widespread application in many clinical set- tings. A similar strategy using small-molecules to neu- tralize soluble peptide ligands in a pathological setting is just starting to be explored. NGF neutralizing small-molecules (which bind NGF rather than the TrkA or p75NTR receptors) has been de- scribed by our group and others. For example, ALE- 0540 [9], Ro 08-2750 [10], PD90780 [11] and Y1036 [12] are known to effectively neutralize NGF activity. Inter- estingly, Y1036 is reported to also bind the related neu- rotrophin brain-derived neurotrophic factors. Better un- derstanding the high resolution of structural biology gov- erning the mode-of-action of these molecules may facili- tate the identification and optimization of such com- pounds in drug discovery programs directed at unprece- dented targets. In this study, we describe the identification of a puta- tive pharmacological targeting domain on the structure of NGF. Beginning with high-resolution structural biology data, we dissect the molecular interactions governing the NGF/p75NTR and NGF/TrkA interface. Next, we use the NGF-binding antagonist Y1036 as an affinity probe to identify the putative pharmacological targeting domain. From these data, we perform theoretical docking experi- ments which demonstrate that the binding of Y1036 can substantially alter the molecular topology of NGF. The results of this study provide valuable information to- wards the identification of a pharmacological targeting domain on the surface of a soluble signalling ligand and may prove usefulness in rationally modifying small molecules directed towards the neutralization of NGF. 2. Methods 2.1. Molecular Modeling All molecular modeling was performed in the Sybyl 8.0 environment (Tripos; St. Louis, MO). The 3D coordi- nates of the protein backbone were kept fixed at their X- ray geometry, and a water environment was not included in the model. Ribbon structures of NGF [RCSB ID-1BET [13]], NGF/TrkA [RCSB ID-2IFG [6]] and NGF/p75NTR [RCSB ID-1SG1[5]] were generated using the Molcad rendering suite of Sybyl 8.0. External surfaces were cal- culated using the Fast Connoly approximation of the MolCad suite. Surface to charge ratios were approxi- mated using the MMFF94 molecular mechanical force field [14]. 2.2. NGF Crosslinking and Digest NGF was purchased from Cedarlane Laboratories (Bur- lington, ON, Canada). NGF (1 mg/mL) and Y1036 (50 μM) were incubated for 1 hour in 25 mM phosphate buffer (pH 7.4) at room temperature in a volume of 100 μl. Covalent crosslinking of NGF to Y1036 was per- formed via 1-ethyl-3-(3-dimethyl amino-propyl) carbo- diimide (EDC) and N-hydroxysuccinimide (NHS)-me- diated reaction (Pierce; Rockford, IL). 5 mM EDC and 2 mM NHS were added to the solution and incubated at 25˚C temperature for 30 minutes. Crosslinked NGF was reduced in 200 mM DTT in 100 mM ammonium bicar- bonate buffer (pH = 8.0) for 1hr at room temperature. The reaction mixture was then acetylated with iodoace- tamide (1 M) via 1hr incubation at room temperature in the dark. Excess iodoacetamide was neutralized using dithiotheritol (DTT) for 1hr at room temperature. A pro- tein digest was then performed by incubating 1 μg of se- quencing grade trypsin (Madison, WI, USA) with 50 μg of NGF or crosslinked NGF-Y1036 at 37˚C for 18 hrs. 2.3. Mass Spectroscopy Peptide analysis was performed at the Biological Mass Spectrometry Laboratory at the University of Western Ontario (London, ON, Canada). Peptides were separated on a CapLC high-performance liquid chromatography system using a Nano-Acquity C18 column and analyzed using Q-TOF Micro mass spectrometer (Waters, Missis- sauga ON, Canada). NGF-peptides were identified via the MASCOT analysis (Matrix Science; Boston MA). Analysis was then used to identify peptides which ex- actly matched the m/z of a given NGF peptide + Y1036 – H2O. 2.4. Theoretical Docking Molecular modeling and in silico docking of Y1036 to NGF [RCSB ID-1BET (Wehrman et al., 2007)] was carried out using the software program Sybyl 8.0 (Tripos, St. Louis, MO). The structure of NGF was prepared for docking using the Biopolymer suite of Sybyl 8.0. Co- structures were deleted and hydrogens were added, the appropriate formal charges were applied to the N- and C-termini and the structure was optimized using the MMFF94 molecular mechanical force field [14]. Flexible docking of Y1036 was performed using the Surflex-Doc suite [15] incorporated into Sybyl 8.0. The docking Open Access NM 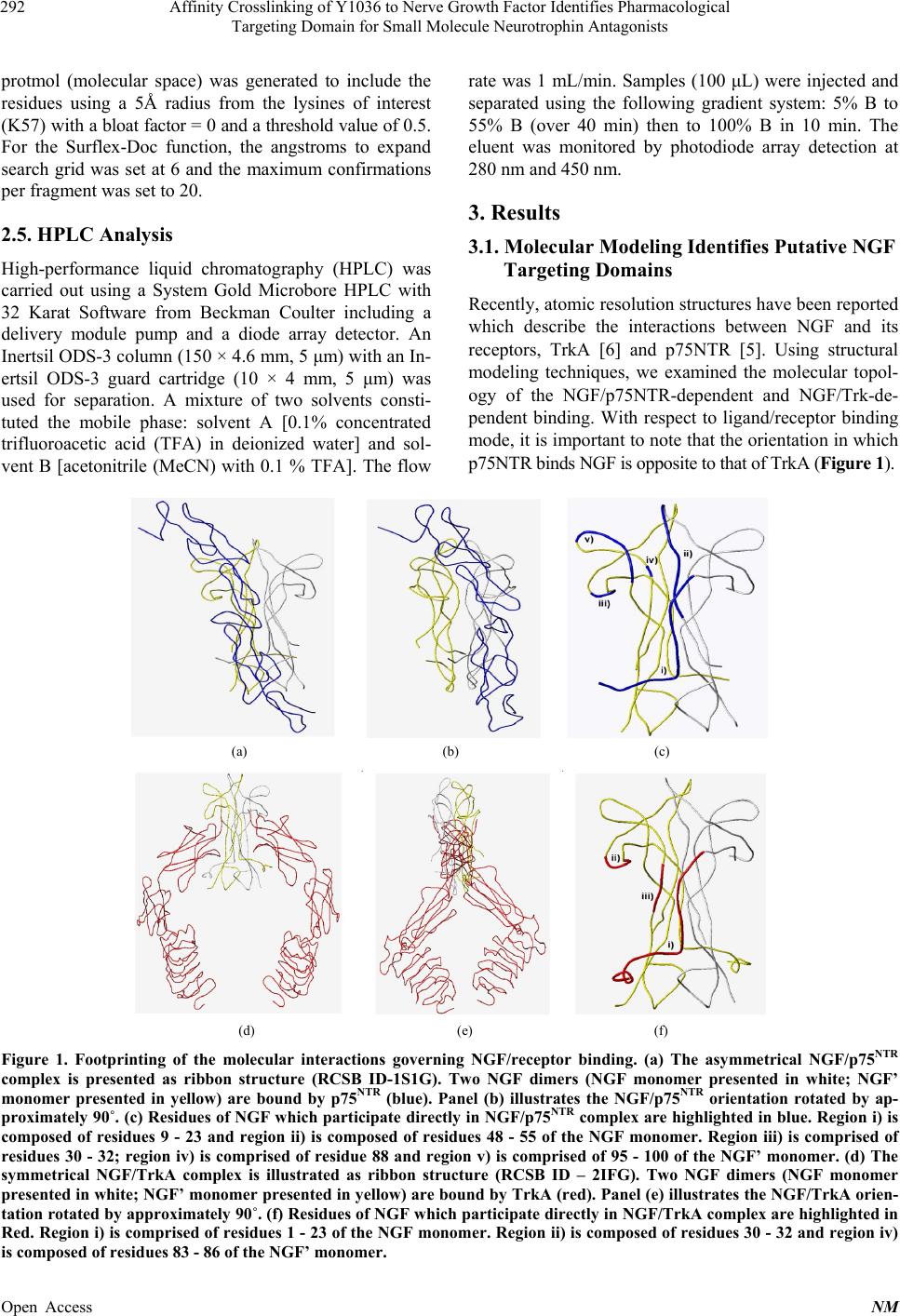 Affinity Crosslinking of Y1036 to Nerve Growth Factor Identifies Pharmacological Targeting Domain for Small Molecule Neurotrophin Antagonists Open Access NM 292 rate was 1 mL/min. Samples (100 μL) were injected and separated using the following gradient system: 5% B to 55% B (over 40 min) then to 100% B in 10 min. The eluent was monitored by photodiode array detection at 280 nm and 450 nm. protmol (molecular space) was generated to include the residues using a 5Å radius from the lysines of interest (K57) with a bloat factor = 0 and a threshold value of 0.5. For the Surflex-Doc function, the angstroms to expand search grid was set at 6 and the maximum confirmations per fragment was set to 20. 3. Results 2.5. HPLC Analysis 3.1. Molecular Modeling Identifies Putative NGF Targeting Domains High-performance liquid chromatography (HPLC) was carried out using a System Gold Microbore HPLC with 32 Karat Software from Beckman Coulter including a delivery module pump and a diode array detector. An Inertsil ODS-3 column (150 × 4.6 mm, 5 μm) with an In- ertsil ODS-3 guard cartridge (10 × 4 mm, 5 μm) was used for separation. A mixture of two solvents consti- tuted the mobile phase: solvent A [0.1% concentrated trifluoroacetic acid (TFA) in deionized water] and sol- vent B [acetonitrile (MeCN) with 0.1 % TFA]. The flow Recently, atomic resolution structures have been reported which describe the interactions between NGF and its receptors, TrkA [6] and p75NTR [5]. Using structural modeling techniques, we examined the molecular topol- ogy of the NGF/p75NTR-dependent and NGF/Trk-de- pendent binding. With respect to ligand/receptor binding mode, it is important to note that the orientation in which p75NTR binds NGF is opposite to that of TrkA (Figure 1). (a) (b) (c) (d) (e) (f) Figure 1. Footprinting of the molecular interactions governing NGF/receptor binding. (a) The asymmetrical NGF/p75NTR complex is presented as ribbon structure (RCSB ID-1S1G). Two NGF dimers (NGF monomer presented in white; NGF’ monomer presented in yellow) are bound by p75NTR (blue). Panel (b) illustrates the NGF/p75NTR orientation rotated by ap- proximately 90˚. (c) Residues of NGF w hich participate directly in NGF/p75NTR complex are highlighted in blue. Region i) is composed of residues 9 - 23 and region ii) is composed of residues 48 - 55 of the NGF monomer. Region iii) is comprised of residues 30 - 32; region iv) is comprised of residue 88 and region v) is comprised of 95 - 100 of the NGF’ monomer. (d) The symmetrical NGF/TrkA complex is illustrated as ribbon structure (RCSB ID – 2IFG). Two NGF dimers (NGF monomer presented in white; NGF’ monomer presented in yellow) are bo und by TrkA (red). Panel (e) illustr ates the NGF/TrkA orien- tation rotated by approximately 90˚. (f) Residues of NGF which participate directly in NGF/TrkA complex are highlighted in Red. Region i) is comprised of residues 1 - 23 of the NGF monomer. Region ii) is composed of residues 30 - 32 and region iv) is composed of residues 83 - 86 of the NGF’ monomer.  Affinity Crosslinking of Y1036 to Nerve Growth Factor Identifies Pharmacological Targeting Domain for Small Molecule Neurotrophin Antagonists 293 Interestingly, select regions participating in p75NTR binding of NGF are shared with TrkA binding while other regions are primarily distinct from those involved in TrkA interactions. For instance, the N-terminal resi- dues of NGF participate in binding with both receptors, but the loop I/IV region is of critical importance to p75NTR interactions. The NGF/TrkA complex reported by Wehrmann et al. [6] involves the residues of loop IV, but it is unclear if loop I participates in binding. NGF/TrkA interactions also occur at the C-terminal domain of NGF which are outside the contact region of p75NTR. Figure 1 illustrates the NGF ligand/receptor footprint with respect to p75NTR and TrkA. The molecular topology of NGF’s ligand/receptor complex suggests that an inhibiting small molecule would likely bind in proximity to these regions of interaction. 3.2. Proximity Cross-Linking Identifies Pharmacological Targeting Domains Y1036 is a small molecule which neutralizes the signal- ling activity of NGF and inhibits NGF interaction with the p75NTR and TrkA receptors [12]. The minimized en- ergy structure of Y1036 is presented in Figure 2. The free carboxyl group of Y1036 has the potential to form H-bonds with the ε-amine of a given lysine residue on the surface of NGF. Importantly, this pharmacological property may allow Y1036 to be used as chemical probe to identify a functional pharmacological targeting do- main(s) on the surface of NGF. Thus, we performed a zero-distance EDC/NHS crosslinking proximity assay. Recombinant NGF was incubated in the presence of ex- cess Y1036 at room temperature for 1 hr. Pre-activation of Y1036 or NGF with EDC/NHS did not affect the effi- ciency of the crosslinking reaction. Crosslinking was then performed via an EDC/NHS reaction. Native NGF and crosslinked NGF-Y1036 then underwent tryptic di- gest. Using time-of-flight mass spectrometry, we were able to resolve the full sequence coverage of NGF via Mascot analysis of tryptic peptides as summarized in Figure 3(a). Similar analysis of NGF crosslinked to Y1036 identified a principal peak corresponding in mass to the Y1036- modified peptide QYFFETK(-Y1036) (Figures 3(b) and (c)). As EDC/NHS crosslinking occurs between the car- boxyl group of Y1036 and primary amines of lysine resi- dues these data suggests that Y1036 crosslinked with residues K57 (Figure 3(c)). Figure 4 illustrates the spa- tial orientation of the QYFFETK(-Y1036) on the struc- ture of NGF. Theoretical docking experiments support the involve- ment of K57 in pharmacological targeting domains. To evaluate the potential contribution of K57 to the binding mode of Y1036, we created an ab initio docking Figure 2. Structure of Y1036. (a) The structure of Y1036, 3- [(5E)-4-oxo-5-[[5-(4-sulfamoylphenyl)-2-furyl]methylene]-2- thioxo-thiazolidin-3-yl]propanoic acid. (b) The minimized energy conformation of Y1036. site biased to the residues within a 5Å radius of the K57 crosslinking site. We then performed flexible docking experiments to obtain a theoretical binding mode for Y1036 in the proximity to the dimer interface (K57 site). A favorable docking mode for Y1036 was obtained with a consensus score of 4. In the case of the pharmacological targeting domain near the hydrophobic interface, the docking results pre- dicted the formation of three stabilizing H-bonds: the first H-bond is formed between the ε-amine of Lys57 and the carboxyl group of Y1036; another H-bond is shared by the aminosulfonyl group of Y1036 and the α-carbonyl group of Trp21; and a third H-bond with the double bonded oxygen of the sulfonamyl group of Y1036 and the α-amide of Gly23 (Figure 5(a)). Accordingly, the docking mode also demonstrates a favorable pose with little conformational strain consistent with the minimized energy conformation of Y1036. Importantly, the results of the docking experiments suggest the formation of an H-bond between the carboxyl group of Y1036 and the ε-amine of the lysine (K57). Thus, these theoretical docking experiments are consis- tent with the mass spectra data obtained the Y1036 proximity crosslinking assay. These results narrow the possible pharmacological targeting domains of NGF to the hydrophobic interface with involvement of K57. In order to rule out non-specific crosslinking/binding activity, we attempted to crosslink a non-active analogue of Y1036 (Figure 6(a)). Y410 (Figure 6(b) shares struc- tural homology to Y1036, but is not an active NGF an- Open Access NM 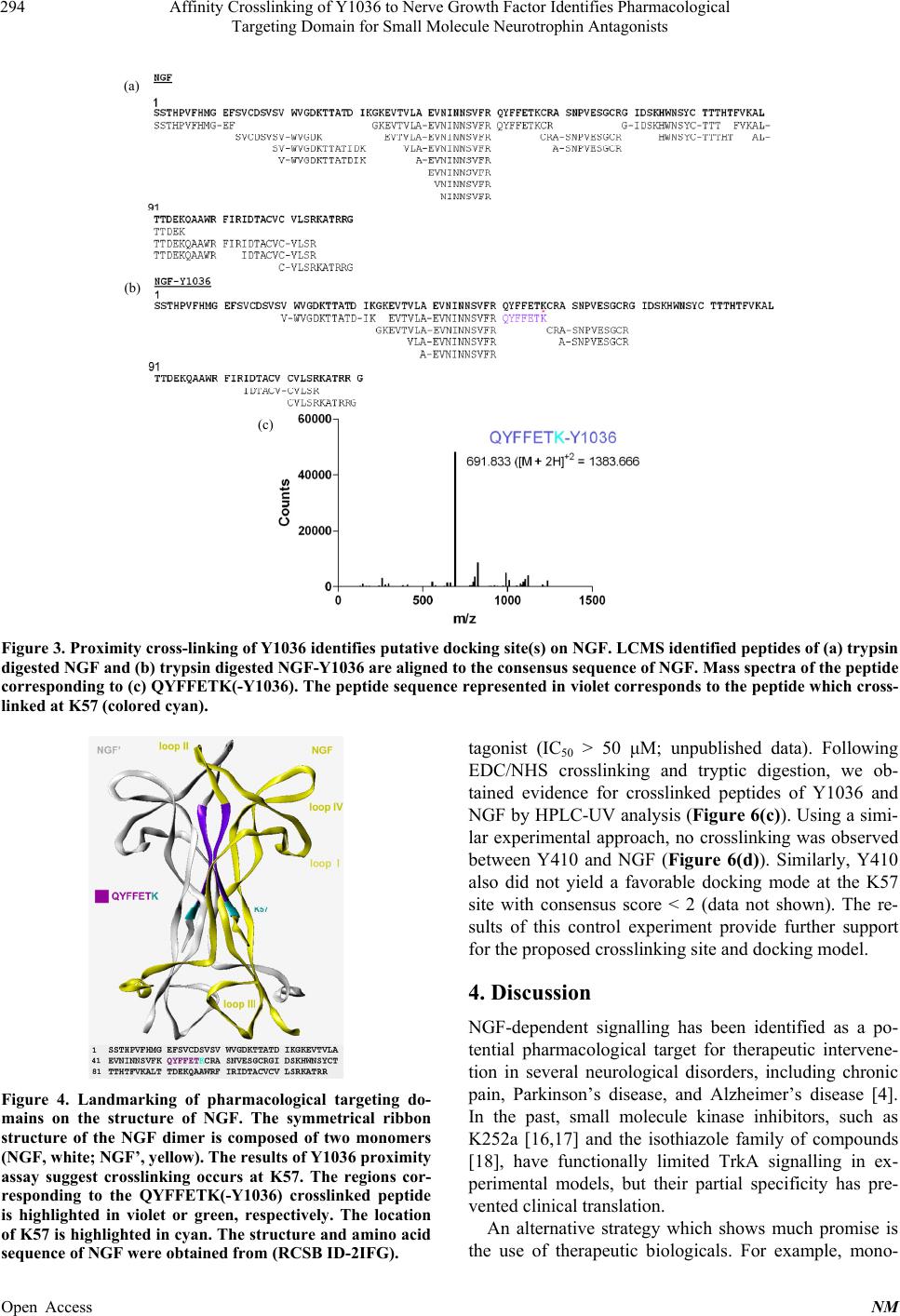 Affinity Crosslinking of Y1036 to Nerve Growth Factor Identifies Pharmacological Targeting Domain for Small Molecule Neurotrophin Antagonists 294 (a) (b) (c) Figure 3. Proximity cross-linking of Y1036 identifies putative docking site(s) on NGF. LCMS identified peptides of (a) trypsin digested NGF and (b) trypsin digested NGF-Y1036 are aligned to the consensus sequence of NGF. Mass spectra of the peptide corresponding to (c) QYFFETK(-Y1036). The peptide sequence represented in violet corresponds to the peptide which cross- linked at K57 (colored cyan). Figure 4. Landmarking of pharmacological targeting do- mains on the structure of NGF. The symmetrical ribbon structure of the NGF dimer is composed of two monomers (NGF, white; NGF’, yellow). The results of Y1036 proximity assay suggest crosslinking occurs at K57. The regions cor- responding to the QYFFETK(-Y1036) crosslinked peptide is highlighted in violet or green, respectively. The location of K57 is highlighted in cyan. The structure and amino acid sequence of NGF were obtained from (RCSB ID-2IFG). tagonist (IC50 > 50 μM; unpublished data). Following EDC/NHS crosslinking and tryptic digestion, we ob- tained evidence for crosslinked peptides of Y1036 and NGF by HPLC-UV analysis (Figure 6(c)). Using a simi- lar experimental approach, no crosslinking was observed between Y410 and NGF (Figure 6(d)). Similarly, Y410 also did not yield a favorable docking mode at the K57 site with consensus score < 2 (data not shown). The re- sults of this control experiment provide further support for the proposed crosslinking site and docking model. 4. Discussion NGF-dependent signalling has been identified as a po- tential pharmacological target for therapeutic intervene- tion in several neurological disorders, including chronic pain, Parkinson’s disease, and Alzheimer’s disease [4]. In the past, small molecule kinase inhibitors, such as K252a [16,17] and the isothiazole family of compounds [18], have functionally limited TrkA signalling in ex- perimental models, but their partial specificity has pre- vented clinical translation. An alternative strategy which shows much promise is the use of therapeutic biologicals. For example, mono- Open Access NM 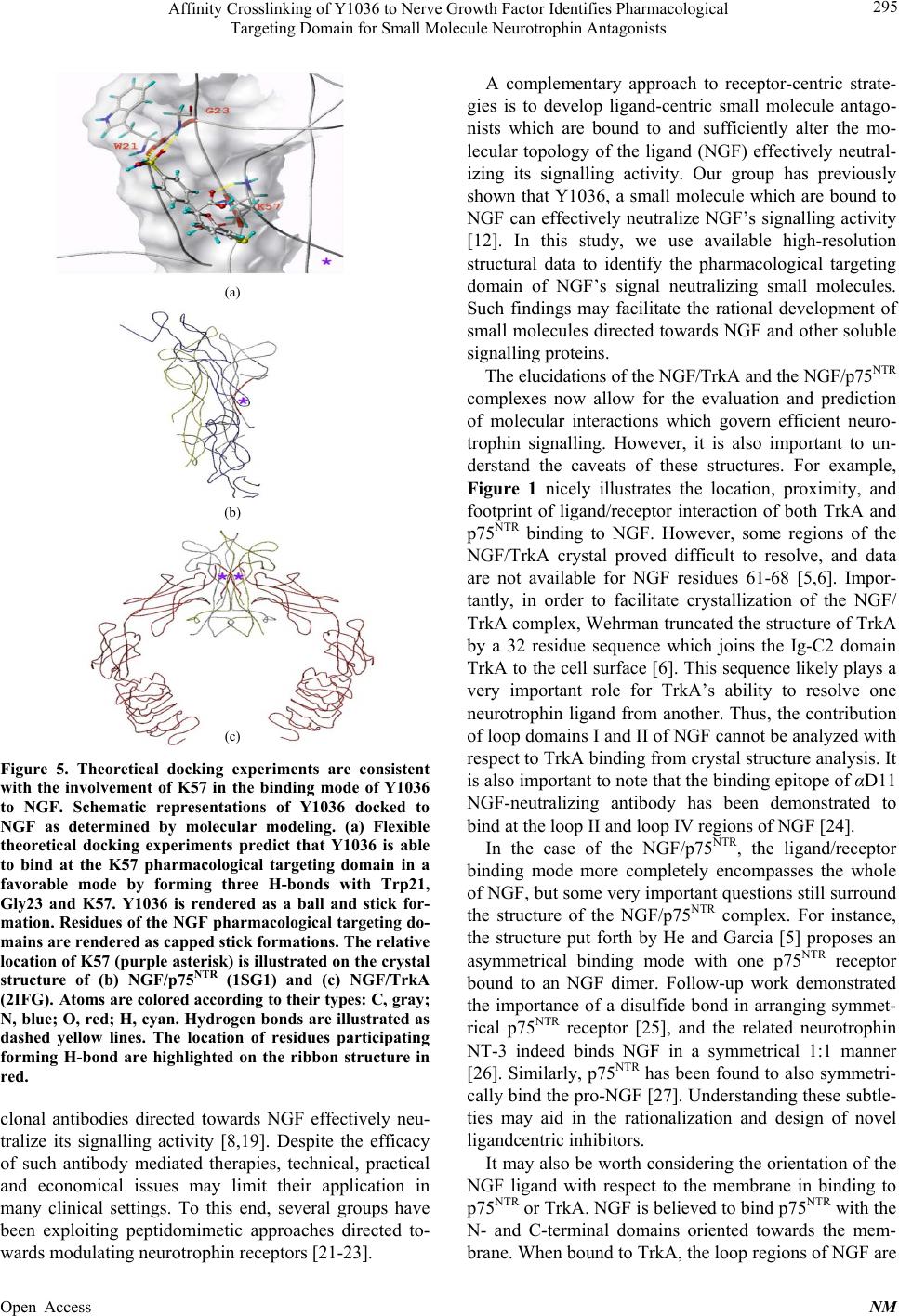 Affinity Crosslinking of Y1036 to Nerve Growth Factor Identifies Pharmacological Targeting Domain for Small Molecule Neurotrophin Antagonists 295 (a) (b) (c) Figure 5. Theoretical docking experiments are consistent with the involvement of K57 in the binding mode of Y1036 to NGF. Schematic representations of Y1036 docked to NGF as determined by molecular modeling. (a) Flexible theoretical docking experiments predict that Y1036 is able to bind at the K57 pharmacological targeting domain in a favorable mode by forming three H-bonds with Trp21, Gly23 and K57. Y1036 is rendered as a ball and stick for- mation. Residues of the NGF pharmacological targeting do- mains are rendered as capped stick formations. The relative location of K57 (purple asterisk) is illustrated on the crystal structure of (b) NGF/p75NTR (1SG1) and (c) NGF/TrkA (2I FG). Ato ms are colored according to their types: C, gray; N, blue; O, red; H, cyan. Hydroge n bonds are illustrated as dashed yellow lines. The location of residues participating forming H-bond are highlighted on the ribbon structure in red. clonal antibodies directed towards NGF effectively neu- tralize its signalling activity [8,19]. Despite the efficacy of such antibody mediated therapies, technical, practical and economical issues may limit their application in many clinical settings. To this end, several groups have been exploiting peptidomimetic approaches directed to- wards modulating neurotrophin receptors [21-23]. A complementary approach to receptor-centric strate- gies is to develop ligand-centric small molecule antago- nists which are bound to and sufficiently alter the mo- lecular topology of the ligand (NGF) effectively neutral- izing its signalling activity. Our group has previously shown that Y1036, a small molecule which are bound to NGF can effectively neutralize NGF’s signalling activity [12]. In this study, we use available high-resolution structural data to identify the pharmacological targeting domain of NGF’s signal neutralizing small molecules. Such findings may facilitate the rational development of small molecules directed towards NGF and other soluble signalling proteins. The elucidations of the NGF/TrkA and the NGF/p75NTR complexes now allow for the evaluation and prediction of molecular interactions which govern efficient neuro- trophin signalling. However, it is also important to un- derstand the caveats of these structures. For example, Figure 1 nicely illustrates the location, proximity, and footprint of ligand/receptor interaction of both TrkA and p75NTR binding to NGF. However, some regions of the NGF/TrkA crystal proved difficult to resolve, and data are not available for NGF residues 61-68 [5,6]. Impor- tantly, in order to facilitate crystallization of the NGF/ TrkA complex, Wehrman truncated the structure of TrkA by a 32 residue sequence which joins the Ig-C2 domain TrkA to the cell surface [6]. This sequence likely plays a very important role for TrkA’s ability to resolve one neurotrophin ligand from another. Thus, the contribution of loop domains I and II of NGF cannot be analyzed with respect to TrkA binding from crystal structure analysis. It is also important to note that the binding epitope of αD11 NGF-neutralizing antibody has been demonstrated to bind at the loop II and loop IV regions of NGF [24]. In the case of the NGF/p75NTR, the ligand/receptor binding mode more completely encompasses the whole of NGF, but some very important questions still surround the structure of the NGF/p75NTR complex. For instance, the structure put forth by He and Garcia [5] proposes an asymmetrical binding mode with one p75NTR receptor bound to an NGF dimer. Follow-up work demonstrated the importance of a disulfide bond in arranging symmet- rical p75NTR receptor [25], and the related neurotrophin NT-3 indeed binds NGF in a symmetrical 1:1 manner [26]. Similarly, p75NTR has been found to also symmetri- cally bind the pro-NGF [27]. Understanding these subtle- ties may aid in the rationalization and design of novel ligandcentric inhibitors. It may also be worth considering the orientation of the NGF ligand with respect to the membrane in binding to p75NTR or TrkA. NGF is believed to bind p75NTR with the N- and C-terminal domains oriented towards the mem- brane. When bound to TrkA, the loop regions of NGF are Open Access NM 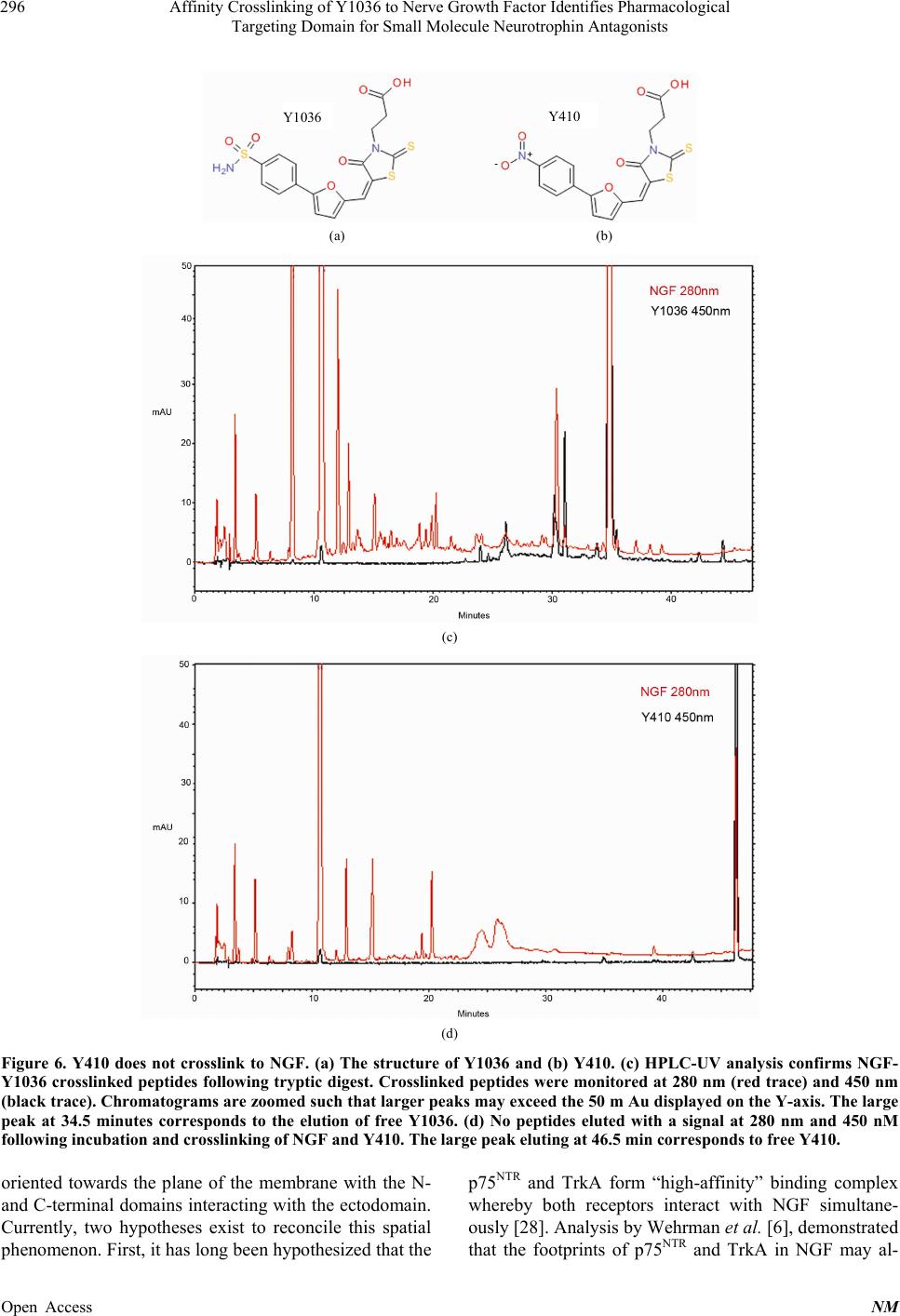 Affinity Crosslinking of Y1036 to Nerve Growth Factor Identifies Pharmacological Targeting Domain for Small Molecule Neurotrophin Antagonists Open Access NM 296 Y1036 Y410 (a) (b) (c) (d) Figure 6. Y410 does not crosslink to NGF. (a) The structure of Y1036 and (b) Y410. (c) HPLC-UV analysis confirms NGF- Y1036 crosslinked peptides following tryptic digest. Crosslinked peptides were monitored at 280 nm (red trace) and 450 nm (black trace). Chromatograms are zoomed such that larger peaks may exceed the 50 m Au displayed on the Y-axis. The large peak at 34.5 minutes corresponds to the elution of free Y1036. (d) No peptides eluted with a signal at 280 nm and 450 nM following incubation and crosslinking of NGF and Y410. The large peak eluting at 46.5 min corresponds to free Y410. oriented towards the plane of the membrane with the N- and C-terminal domains interacting with the ectodomain. Currently, two hypotheses exist to reconcile this spatial phenomenon. First, it has long been hypothesized that the p75NTR and TrkA form “high-affinity” binding complex whereby both receptors interact with NGF simultane- ously [28]. Analysis by Wehrman et al. [6], demonstrated that the footprints of p75NTR and TrkA in NGF may al-  Affinity Crosslinking of Y1036 to Nerve Growth Factor Identifies Pharmacological Targeting Domain for Small Molecule Neurotrophin Antagonists 297 low for simultaneous binding. Cross-linking experiments have demonstrated that a NGF/p75NTR/TrkA complex forms [29], but it’s not clear if this is a stable or transient complex. Alternatively, a “hand-over” mechanism has also been proposed whereby p75NTR first binds NGF and a hand-to-hand exchange from p75NTR to TrkA occurs [28]. It is thought that this mechanism may account for the binding of NGF in opposite orientation. For the pur- poses of rational drug design, it is important to consider the dynamics of such receptor interplay. With respect to the pharmacological targeting domains put forward in this study, the potential mechanism at the hydrophobic interface (K57 site) is not obvious from crystallography data. The molecular topology altered by Y1036 binding at the K57 site is in proximity, but outside of the static interaction surfaces of NGF/p75NTR or NGF/TrkA. The static nature of crystallography provides very useful in- sights, but further real-time high-resolution techniques will undoubtedly refine our current understanding of these events. 5. Conclusion The results of our molecular modeling suggest that the binding of Y1036 to NGF is sufficient to alter the mo- lecular topology such that the resultant change in sur- face charge density may alter the receptor: neurotrophin interface and prevent neurotrophin mediated signalling. However, there are limitations to the interpretation of our data. In order to have definitive proof of binding me- chanism of Y1036, co-crystallization experiments will be required. The docking model presented in this study is consistent with our experimental observations. Under- standing the structural and molecular features of the residues in proximity to the NGF crosslinking site now gives researchers the ability to modify chemical scaffolds in a rational manner. Taking advantage of similar high resolution structural biology, it may be possible to apply this strategy to soluble protein ligands more generally. Adopting such emerging high resolution strategies may ultimately enhance drug discovery activities and aid in revitalizing the collective pharmaceutical pipeline. REFERENCES [1] B. Munos, “Lessons from 60 Years of Pharmaceutical In- novation,” Nature Reviews Drug Discovery, Vol. 8, No. 12, 2009, pp. 959-968. http://dx.doi.org/10.1038/nrd2961 [2] P. Imming, C. Sinning and A. Meyer, “Drugs, Their Tar- gets and the Nature and Number of Drug Targets,” Nature Reviews Drug Discovery, Vol. 5, No. 10, 2006, pp. 821- 834. http://dx.doi.org/10.1038/nrd2132 [3] S. Paul, “Steven Paul,” Nature Reviews Drug Discovery, Vol. 8, No. 1, 2009, p. 14. http://dx.doi.org/10.1038/nrd2800 [4] M. V. Chao, R. Rajagopal and F. S. Lee, “Neurotrophin Signalling in Health and Disease,” Clinical Science (Lon- don), Vol. 110, 2006, pp. 167-173. http://dx.doi.org/10.1042/CS20050163 [5] X. L. He and K. C. Garcia, “Structure of Nerve Growth Factor Complexed with the Shared Neurotrophin Recep- tor p75,” Science, Vo. 304, No. 5672, 2004, pp. 870-875. http://dx.doi.org/10.1126/science.1095190 [6] T. Wehrman, X. He, B. Raab, A. Dukipatti, H. Blau and K. C. Garcia, “Structural and Mechanistic Insights into Nerve Growth Factor Interactions with the TrkA and p75 Receptors,” Neuron, Vol. 53, No. 1, 2007, pp. 25-38. http://dx.doi.org/10.1016/j.neuron.2006.09.034 [7] D. W. Sah, M. H. Ossipo and F. Porreca, “Neurotrophic Factors as Novel Therapeutics for Neuropathic Pain,” Na- ture Reviews Drug Discovery, Vol. 2, No. 6, 2003, pp. 460- 472. http://dx.doi.org/10.1038/nrd1107 [8] A. Dray, “Neuropathic Pain: Emerging Treatments,” Bri- tish Journal of Anaesthesia, Vol. 101, No. 1, 2008, pp. 48-58. http://dx.doi.org/10.1093/bja/aen107 [9] J. B. Owolabi, G. Rizkalla, A. Tehim, G. M. Ross, R. J. Riopelle, R. Kamboj, M. Ossipov, D. Bian, S. Wegert, F. Porreca and D. K. Lee, “Characterization of Antiallodynic Actions of ALE-0540, a Novel Nerve Growth Factor Re- ceptor Antagonist, in the Rat,” Journal of Pharmacology and Experimental Therapeutics, Vol. 289, 1999, pp. 1271-1276. [10] O. Niederhauser, M. Mangold, R. Schubenel, E. A. Kusznir, D. Schmidt and C. Hertel, “NGF Ligand Alters NGF Sig- naling via p75(NTR) and trkA,” Journal of Neuroscience Research, Vol. 61, No. 3, 2000, pp. 263-272. http://dx.doi.org/10.1002/1097-4547(20000801)61:3<263 ::AID-JNR4>3.0.CO;2-M [11] K. Spiegel, D. Agrafiotis, B. Caprathe, R. E. Davis, M. R. Dickerson, J. H. Fergus, T. W. Hepburn, J. S. Marks, M. Van Dorf, D. M. Wieland, et al., “PD 90780, a Non Pep- tide Inhibitor of Nerve Growth Factor’s Binding to the P75 NGF Receptor,” Biochemical and Biophysical Re- search Communications, Vol. 217, No. 2, 1995, pp. 488- 494. http://dx.doi.org/10.1006/bbrc.1995.2802 [12] J. K. Eibl, S. A. Chapelsky and G. M. Ross, “Multipotent Neurotrophin Antagonist Targets Brain-Derived Neuro- trophic Factor and Nerve Growth Factor,” Journal of Pharmacology and Experimental Therapeutics, Vol. 332, No. 2, 2010, pp. 446-454. http://dx.doi.org/10.1124/jpet.109.159079 [13] N. Q. McDonald, R. Lapatto, J. Murray-Rust, J. Gunning, A Wlodawer and T. L. Blundell, “New Protein Fold Re- vealed by a 2.3-A Resolution Crystal Structure of Nerve Growth Factor,” Nature, Vol. 354, No. 6352, 1991, pp. 411-414. http://dx.doi.org/10.1038/354411a0 [14] T. A. Halgren, “Maximally Diagonal Force Constants and Dependent Angle Bending Coordinates II. Implecations for the Design of Emperical Forcefields,” Journal of the American Chemical Society, Vol. 112, No. 12, 1990, pp. 4710-4723. http://dx.doi.org/10.1021/ja00168a015 [15] A. N. Jain, “Scoring Noncovalent Protein-Ligand Interac- Open Access NM  Affinity Crosslinking of Y1036 to Nerve Growth Factor Identifies Pharmacological Targeting Domain for Small Molecule Neurotrophin Antagonists Open Access NM 298 tions: A Continuous Differentiable Function Tuned to Compute Binding Affinities,” Journal of Computer-Aided Molecular Design, Vol. 10, No. 5, 1996, pp. 427-440. http://dx.doi.org/10.1007/BF00124474 [16] M. M. Berg, D. W. Sternberg, L. F. Parada and M. V. Chao, “K-252a Inhibits Nerve Growth Factor-Induced trk Proto-Oncogene Tyrosine Phosphorylation and Kinase Activity,” The Journal of Biological Chemistry, Vol. 267, 1992, pp. 13-16. [17] B. Knusel and F. Hefti, “K-252 Compounds: Modulators of Neurotrophin Signal Transduction,” Journal of Neuro- chemistry, Vol. 59, No. 6, 1992, pp. 1987-1996. http://dx.doi.org/10.1111/j.1471-4159.1992.tb10085.x [18] B. Lippa, J. Morris, M. Corbett, T. A. Kwan, M. C. Noe, S. L. Snow, T. G. Gant, M. Mangiaracina, H. A. Coffey, B. Foster, E. A. Knauth and M. D. Wessel, “Discovery of Novel Isothiazole Inhibitors of the TrkA Kinase: Struc- ture-Activity Relationship, Computer Modeling, Optimi- zation, and Identification of Highly Potent Antagonists,” Bioorganic & Medicinal Chemistry Letters, Vol. 16, No. 13, 2006, pp. 3444-3448. http://dx.doi.org/10.1016/j.bmcl.2006.04.003 [19] J. J. Watson, S. J. Allen and D. Dawbarn, “Targeting Nerve Growth Factor in Pain: What Is the Therapeutic Poten- tial?” BioDrugs, Vol. 22, No. 6, 2008, pp. 349-359. http://dx.doi.org/10.2165/0063030-200822060-00002 [20] S. M. Massa, T. Yang, Y. Xie, J. Shi, M. Bilgen, J. N. Joyce, D. Nehama, J. Rajadas and F. M. Longo, “Small Molecule BDNF Mimetics Activate TrkB Signaling and Prevent Neuronal Degeneration in Rodents,” Journal of Clinical Investigation, Vol. 120, No. 5, pp. 1774-1785. http://dx.doi.org/10.1172/JCI41356 [21] F. M. Longo and S. M. Massa, “Small Molecule Modula- tion of p75 Neurotrophin Receptor Functions,” CNS & Neurological Disorders—Drug Targets, Vol. 7, No. 7, 2008, pp. 63-70. http://dx.doi.org/10.2174/187152708783885093 [22] F. Brahimi, A. Malakhov, H. B. Lee, M. Pattarawarapan, L. Ivanisevic, K. Burgess and H. U. Saragovi, “A Pepti- domimetic of NT-3 Acts as a TrkC Antagonist,” Peptides, Vol. 30, No. 10, 2009, pp. 1833-1839. http://dx.doi.org/10.1016/j.peptides.2009.07.015 [23] D. Chen, F. Brahimi, Y. Angell, Y. C. Li, J. Moscowicz, H. U. Saragovi and K. Burgess, “Bivalent Peptidomimetic Ligands of TrkC Are Biased Agonists and Selectively In- duce Neuritogenesis or Potentiate Neurotrophin-3 Tro- phic Signals,” ACS Chemical Biology, Vol. 4, No. 9, 2009, pp. 769-781. http://dx.doi.org/10.1021/cb9001415 [24] S. Covaceuszach, A. Cassetta, P. V. Konarev, S. Gonfloni, R. Rudolph, D. I. Svergun, D. Lamba and A. Cattaneo, “Dissecting NGF Interactions with TrkA and p75 Recep- tors by Structural and Functional Studies of an Anti-NGF Neutralizing Antibody,” Journal of Molecular Biology, Vol. 381, No. 4, 2008, pp. 881-896. http://dx.doi.org/10.1016/j.jmb.2008.06.008 [25] M. Vilar, I. Charalampopoulos, R. S. Kenchappa, A. Simi, E. Karaca, A. Reversi, S. Choi, M. Bothwell, I. Mingarro, W. J. Friedman, G. Schiavo, P. I. Bastiaens, P. J. Verveer, B. D. Carter and C. F. Ibanez, “Activation of the p75 Neu- rotrophin Receptor through Conformational Rearrange- ment of Disulphide-Linked Receptor Dimers,” Neuron, Vol. 62, No. 1, 2009, pp. 72-83. http://dx.doi.org/10.1016/j.neuron.2009.02.020 [26] Y. Gong, P. Cao, H. J. Yu and T. Jiang, “Crystal Struc- ture of the Neurotrophin-3 and p75NTR Symmetrical Complex,” Nature, Vol. 454, 2008, pp. 789-793. [27] D. Feng, T. Kim, E. Ozkan, M. Light, R. Torkin, K. K. Teng, B. L. Hempstead and K. C. Garcia, “Molecular and Structural Insight into proNGF Engagement of p75NTR and Sortilin,” Journal of Molecular Biology, Vol. 396, No. 4, 2010, pp. 967-984. http://dx.doi.org/10.1016/j.jmb.2009.12.030 [28] P. A. Barker, “High Affinity Not in the Vicinity?” Neu- ron, Vol. 53, No. 1, 2007, pp. 1-4. http://dx.doi.org/10.1016/j.neuron.2006.12.018 [29] G. M. Ross, I. L. Shamovsky, G. Lawrance, M. Solc, S. M. Dostaler, D. F. Weaver and R. J. Riopelle, “Reciprocal Modulation of TrkA and p75NTR Affinity States Is Me- diated by Direct Receptor Interactions,” European Jour- nal of Neuroscience, Vol. 10, No. 3, 1998, pp. 890-898. http://dx.doi.org/10.1046/j.1460-9568.1998.00094.x
|