 American Journal of Analytical Chemistry, 2013, 4, 689-695 Published Online December 2013 (http://www.scirp.org/journal/ajac) http://dx.doi.org/10.4236/ajac.2013.412083 Open Access AJAC Bioremediation of Lead(II) from Polluted Wastewaters Employing Sulphuric Acid Tr eated Maize Tassel Biomass Mambo Moyo, Linda Chikazaza Department of Chemical Technology, Midlands State University, Gweru, Zimbabwe Email: moyom@msu.ac.zw Received September 30, 2013; revised October 29, 2013; accepted November 15, 2013 Copyright © 2013 Mambo Moyo, Linda Chikazaza. This is an open access article distributed under the Creative Commons Attribu- tion License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. ABSTRACT The ability to modify a waste by-product precursor, maize tassel biomass using sulfuric acid as the activating agent with specific focus on Lead(II) ion from water has been proposed. The treating of maize tassel using sulphuric acid is be- lieved to enhance sorption capacity of Lead(II) ions. For this, batch adsorption mode was adopted for which the effects of initial pH, adsorbent dosage, contact time and initial concentration were investigated. Consequently, it was found that the adsorbent capacity depends on pH; since it increases up to 4.5 and then decreases. The highest percentage of Lead(II) ion removal was achieved in the adsorbent dosage of 1.2 g and at an initial concentration of 10 mg/L metal ion. In an attempt to determine the capacity and rate of Lead(II) removal, isotherm and kinetic data were modeled using appropri- ate equations. To this end, the adsorption data fitted best into the Langmuir model with an R2 (0.9997) while kinetically the Lead(II) adsorption followed the pseudo-second-order model. Furthermore, as a way to address issues related to sustainability, maize tassel is recommended since the process is considered to be a dual solution for environmental cleaning. From one side, it represents a better way to dispose the maize tassel which has no use after fertilization and on the other hand it is an economic source of carbonaceous materials. Keywords: Maize Tassel; Adsorption; Removal; Wastewater Treatment, Lead(II) Ion 1. Introduction The presence of heavy metals in the environment poses a serious and complex environmental and public problem due to their non-degradability [1]. Among the heavy metals, Lead (Pb) is very toxic because it is carcinogenic in nature [2]. Lead is the most significant toxin of the trace metal ions, and human exposure is through inges- tion of food and water, and inhalation. Lead affects mainly the peripheral nervous system and hematopoietic, renal, gastrointestinal, cardiovascular and reproductive systems. According to World Health Organization [3], the permis- sible level for Pb in drinking water is 0.05 mg/L. Conse- quently, the occurrence of very low concentration levels of lead in drinking water is considered as highly toxic and requires more efficient removal, extraction, and treat- ment methodologies. The removal of metals from water has been previously achieved by various methods such as ion exchange, pre- cipitation, oxidation, reduction and membrane filtration [4,5]. Currently, adsorption technology using cheap, eas- ily available agricultural plants, algal biomass and cyano- bacteria has increased in recent years. However, the use of raw sorbents in adsorption may cause problems since most plants contain a green pigment known as chloro- phyll (sparingly soluble in water) and some organic mat- ter may be leached out, consequently affecting the taste and colour of the treated waters [6,7]. In spite of the ver- satility of commercial activated carbon as an adsorbent in wastewater treatment with its high surface area, micro- porous characteristics, and high adsorption capacity; its high cost and loss during the regeneration restrict its ap- plications in developing countries [8,9]. Given these dis- advantages, efforts have now been geared towards intro- ducing low cost precursors that can serve as alternative sources for water cleaning after modification using dif- ferent activation agents. In the recent years, modified sorbents derived from locally available materials such as Ceiba pentandra hulls [8], Euphorbia rigida [10], ha- zelnut husks [11], wheat bran [12], coirpith [13], and coconut shell [14] have received increasing attention for the removal and recovery of heavy metals from water  M. MOYO, L. CHIKAZAZA 690 and wastewater systems. Maize tassel is an inexhaustible, non-edible, mesopor- ous and renewable polymeric material resource, which is discarded as waste by most farmers after fertilization [15]. Hence, the present study aims to assess the applica- bility of acid treated maize tassel for the adsorptive re- moval of Lead(II) from aqueous solution and to investi- gate the effect of operating parameters on the adsorption process. The parameters studied include contact time, initial Lead(II) concentration, adsorbent dosage and ini- tial solution pH. 2. Materials and Methods 2.1. Modification of Maize Tassel The maize tassel was collected from Morris farm in Northlea, Gweru, Zimbabwe. Maize tassel was plucked off the woody parts of the maize plant, thoroughly washed with water and sun dried for 5 days. The dry biomass was milled and then fractionated using 100 - 300 µm analytical sieves. Then, the modification was prepared according to reported procedure [16-18]. Briefly, the frac- tionated maize tassel powder (200 g) was weighed in a clean dry beaker of capacity 1 L containing (200 mL, 97% H2SO4 for 24 h) followed by refluxing in a fume hood for 4 h. After cooling, the reaction mixture was filtered, and the filtrate was washed repeatedly with ul- tra-pure water and soaked in 1% NaHCO3 solution to remove any remaining acid. The sample was then washed with distilled water until the pH of the acid treated maize tassel was between 6 - 7, dried in an oven at 120˚C over- night and kept in a glass bottle until use. 2.2. Characterization of Prepared Acid Treated Maize Tassel The bulk density, loss of mass on ignition, simple spe- cific surface area, pH and moisture content of the acid treated maize tassel were determined following reported procedures [19,20]. Briefly, for bulk density, acid treated maize tassel was transferred to a 10 mL measuring cyl- inder of about 1.0 cm diameter. Sufficient quantity of powder was added to occupy a volume of 10 mL under the condition, and it was subsequently weighed. The bulk density was expressed as grams per litre (the weight of the acid treated maize tassel filling a graduated cylinder on gentle tapping, divided by the volume of the cylinder 10 mL). The loss of mass on ignition was done by weighing 15 g of the acid treated maize tassel and put inside furnace at constant temperature of 600˚C for 2 h. After roasting, the sample became charred and was re- moved from the furnace then put in a desiccator for cooling. The residual product was then weighed, and the difference in mass represented the mass of organic mate- rial present in the sample. For moisture content, 10 g of acid treated maize tassel was weighed into a crucible and heated in the oven for 5 h at constant temperature of 105˚C. The sample was then removed and put in a desic- cator in so that moisture from atmosphere could not be absorbed. The sample was reweighed. This procedure was repeated several times until a constant weight was ob- tained. The difference in the mass constitutes the amount of moisture content of the adsorbent 23 21 %moisture 100ww ww (1) where w w1 is weight of crucible, w2 is initial weight of crucible with sample, w3 is final weight of crucible with sample. 2.3. Preparation of Synthetic Solution A stock solution of Lead(II) ion (1000 mg/L) was pre- pared by dissolving Pb(NO3)2 (Merck, South Africa) in ultra-pure water (resistivity ˃ 18 MΩ·cm−1). The pro- gressive dilution procedure of the stock solution was employed in the preparation of working solutions. The pH of the working solutions was adjusted to the required value with 0.1 M·NaOH or 0.1 M·HCl. All the chemicals used were of analytical reagent grade. 2.4. Batch Adsorption Studies A weighed amount of acid treated maize tassel was in- troduced into stoppered reagent bottles containing vari- ous concentrations with 100 mL aqueous solutions of Lead(II) ions. The suspensions were shaken at room tem- perature (25˚C ± 1˚C) using a mechanical shaker for a prescribed time at 160 rpm. The solutions were filtered through Whatman 42 filter paper and the residual con- centration of metal ion was determined by AAS method. The effects of concentration (10 - 50 mg/L), contact time (5 - 300 min), solution pH (2 - 12) and adsorption dose (0.1 - 2.5 g) were studied. The percentage of removed Lead(II) ions (R%) in solution was calculated by using Equation (2): 100 %ie i CC RC (2) The amount of metal adsorbed by acid treated maize tassel was calculated from the difference between metal quantity added to the biomass and metal content of the supernatant using Equation (3): ie e CCV q (3) where qe is the metal uptake (mg metal adsorbed per g adsorbent), Ci and Ce are the initial and equilibrium metal concentration in solution (mg/L), V is the volume of the solution (mL) and M is the weight of acid treated maize Open Access AJAC 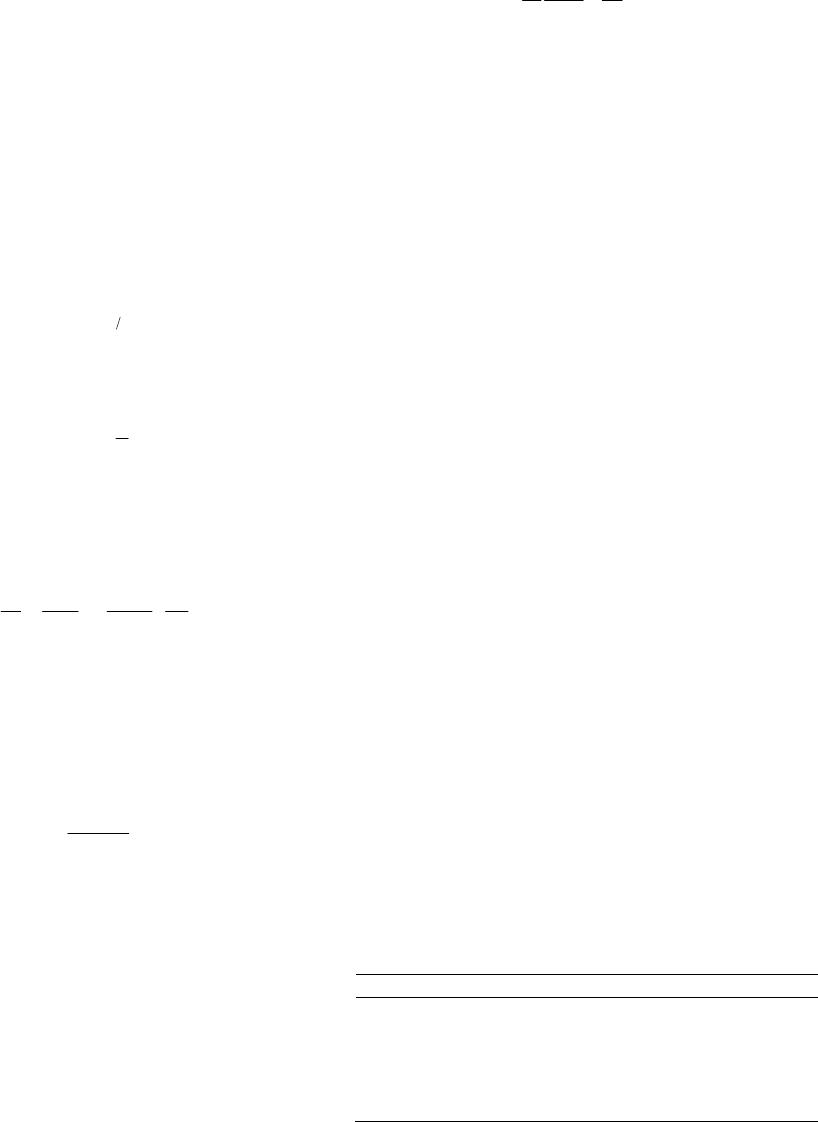 M. MOYO, L. CHIKAZAZA 691 tassel (g). 2.5. Desorption Studies For the desorption studies, contact was made between 1.2 g of acid treated maize tassel and a 100 mL Lead(II) so- lution. After Lead(II) ion sorption, the acid treated maize tassel was filtered, washed three times with ultra-pure water to remove residual Lead(II) ions on the surface, and kept in contact with different concentrations (0.05 - 0.3 mol/L) of 100 mL·HCl solution. The mixtures were shaken in a rotary shaker for 60 min. The filtrates were analyzed to determine the concentration of Lead(II) ions after desorption using AAS. 2.6. Adsorption Isotherms In the present study, the equilibrium data for the ad- sorbed Lead(II) ions onto acid treated maize tassel was expressed using Langmuir and Freundlich isotherm. The Freundlich isotherm [21] is given by Equation (4): 1n eFe qKC (4) The parameters can be linearized by taking logarithms to find the parameters KF and n: 1 ln lnln eF qK C n e (5) A plot of ln qe versus ln Ce gives a straight line and KF and n can be calculated from the intercept and slope, re- spectively. The linear form of the Langmuir isotherm model [22] can be represented by Equation (6): max max 11 11 ee qqbq C (6) A plot of 1/qe versus 1/Ce was found to be a straight line with 1/qmax as intercept and 1/qmax b as slope and hence qmax and b can be calculated. In addition, a dimen- sionless constant called separation factor, RL can be used to express an essential feature of Langmuir isotherm [23]: 1 1 Lm RaC (7) where, Cm is the initial concentration of Lead(II). The value of RL indicates the type of the isotherm to be either unfavorable when RL ˃ 1, linear if RL = 1, favorable if 0 ˂ RL ˂ 1 or RL = 0. 2.7. Adsorption Kinetics The pseudo-second order model has been widely used for adsorption for the following reasons: it does not have the problem of assigning an effective adsorption capacity, the adsorption constant capacity, rate constant and initial adsorption rate can all be determined from the equation without knowing any parameter beforehand [24]. Hence in the present study, adsorption of Lead(II) ions on acid treated maize tassel has been described by pseudo-sec- ond-order model [18]. The general form of the pseudo- second-order kinetic model: 2 2 11 te e tt qq kq (8) where qe and qt (mg/g) are the amounts of the Lead(II) ions sorbed at equilibrium and at time t (min), respec- tively and k2 (mg/g·min) is the second order rate constant. The slope of the plot (t/qt) versus t gives the value of qe and from the intercept k2 can be calculated 3. Results and Discussion 3.1. Physicochemical Characteristics of Acid Treated Maize Tassel The physico-chemical characteristics like pH, moisture content, bulk density, surface area, and loss of mass on ignition were determined and given in Table 1. 3.2. Effects of Dosage The effect of biomass dosage on the biosorption of Pb (II) ions was studied using biomass dosage in the range 0.1 - 2.5 g (Figure 1). As shown in Figure 1, the percentage removal increases sharply from 58.5% to 92.2%, but be- yond this, the percentage removal did not increase sig- nificantly and reached a maximum at 92.3%. This phe- nomenon can be due to the greater availability of activity sites or surface area making easier penetration of the Lead(II) ions to the adsorption sites and increasing this number had no effect after equilibrium is reached. These results are in agreement to other results reported in lit- erature [2,25]. 3.3. Effect of pH The effect of initial pH of a solution is a major factor used to determine the adsorption property of an adsorb- ent from wastewater. The possible reasons are its effects on the chemistry of the ions and the activity of functional groups (carboxylate, phosphate and amino groups) on the cell walls [12]. As shown in Figure 2, there is an in- Table 1. Characteristics of the acid treated maize tassel developed from maize tassel. Parameter Value pH 6.9 Moisture (%) 0.3 Bulk density (g·mL−1) 0.52 Surface area (m2/g) 250 Particle size range 100 - 300 µm % Loss of mass on ignition 0.7 Open Access AJAC 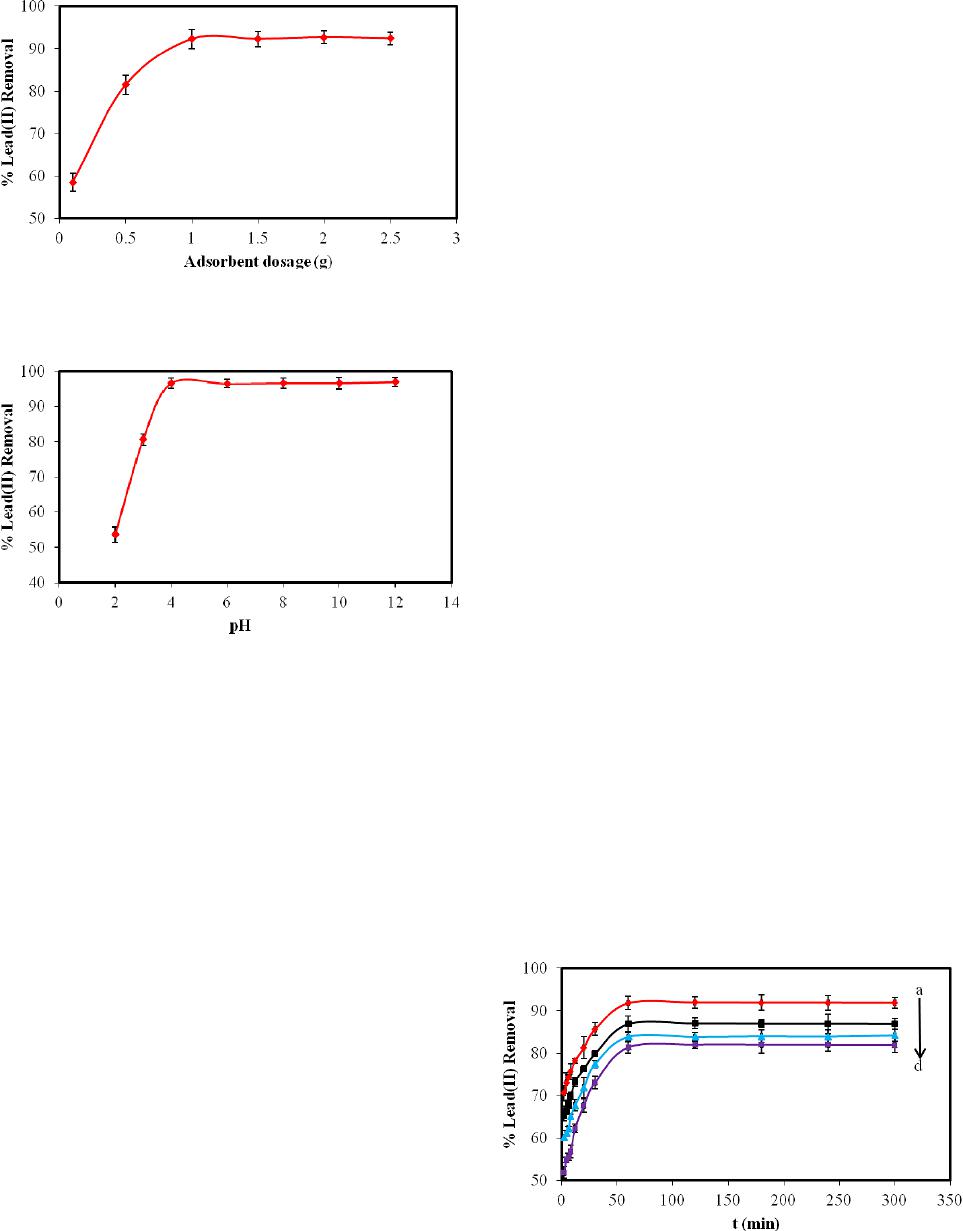 M. MOYO, L. CHIKAZAZA 692 Figure 1. Dosage versus percentage removal of Lead(II) ion (contact time: 60 min; pH 5.4; initial concentration 10 mg/L; Error bar = ±S.D. and n = 3). Figure 2. pH against percentage removal of Lead(II) ion (contact time: 60 min; dosage 1.2 g; initial concentration 10 mg/L; Error bar = ±S.D. and n = 3). crease in the quantity of adsorbed Lead(II) ions onto acid treated maize tassel by increasing pH of the medium up to a maximum value of pH 5.4. At low pH values, the adsorbent is positively charged since the pH is lower than the isoelectric point or point of zero charge (PZC) [26]. Hence the removal yield of Lead(II) ions is very low due to the electrostatic repulsion forces between positively charged H3O+ and Pb2+ ions. Thereafter the adsorption percentage decreased in alkaline medium perhaps due to the formation of Pb(OH)2 and soluble hydroxyl com- plexes such as PbOH+, aqueous Pb(OH)2 and Pb(OH)3− and adsorbent was deteriorated with the accumulation of metal ions making true adsorption studies impossible [25,27]. The results obtained in this study showed that the acid treated maize tassel is more effective in remov- ing Lead(II) ions from an aqueous system with more than 94% removal as compared to raw maize tassel 75% [28]; physically prepared AC 78% [29]. Therefore, pH 5.4 was selected to be the optimum pH for all further studies. 3.4. Effect of Agitation Time and Initial Concentration The effect of agitation time is one of the important fac- tors when designing batch sorption systems for eco- nomical wastewater treatment plant application [13]. As shown in Figure 3, the relationship between agitation time and Lead(II) ion sorption onto acid treated maize tassel at different initial Lead(II) concentrations is deter- mined by plotting the percentage biosorption of Lead(II) ion against agitation time for the interaction time inter- vals of between 2 to 300 min. It was observed that 52%, 60%, 65% and 70% of Lead(II) ions was removed in the first 2 min for the dif- ferent concentrations 10, 20, 30 and 50 mg/L respec- tively and the process was rapid up to 60 min reaching 91.8%. Beyond 60 min, the percentage of biosorption is almost constant indicating the attainment of equilibrium conditions. The presence of adequate external surface area on the acid treated maize tassel may have boosted the rate of adsorption to be fast in the initial stages which was followed by a slower internal diffusion process, which maybe the rate determining step. From the observed ad- sorption trend of Lead(II), the binding may be through van der Waals forces of attraction present on the surface of the acid treated maize tassel. Therefore, all other ex- periments were conducted at an agitation time of 60 min. The effect of initial concentration on the percentage removal is also shown in Figure 3. The concentration range from 10 to 50 mg/L for the metal ion has been studied. The removal of Lead(II) ions by acid treated maize tassel was found to decrease with increase in ini- tial Lead(II) concentration. The observed behavior can be attributed to the increase in the amount of Lead(II) ions to the unchanging number of available active sites on the acid treated maize tassel. Hence, more metal ions were left in solution. Thus it can be said that removal of Lead(II) ion is highly concentration dependent. 3.5. Interference Studies The selectivity of the acid treated maize tassel on the adsorption efficiency of Lead(II) were studied in the Figure 3. Agitation time against percentage removal Lead (II) ion (concentrations: a to d: 10 to 50 mg/L, adsorbent dosage: 1.2 g; pH: 5.4, Error bar = ± S.D. and n = 3). Open Access AJAC  M. MOYO, L. CHIKAZAZA 693 presence of anions such as F−, Cl−, 3, NO 2 4 SO and and cations such as Fe2+, Mg2+, Ca2+, Na+ and K+. There was no significant reduction in the adsorption of Lead(II) when the concentration of the above ions in- creased up to 50 mg/L in aqueous solution. It can be concluded that with proper treatment of the industrial wastewater, adsorption of Lead(II) ions on the surface of the adsorbent up to 94% in the presence of the studied anions and cations up to 50 mg/L is possible. 3 4 PO 3.6. Isotherm and Kinetics Study Figures 4(a) and (b) show the Langmuir isotherm and the Freundlich adsorption isotherm respectively. The Freundlich constants n and KF, Langmuir constants b and qmax and the correlation coefficient R2 are given in Table 2. The calculated value of Freundlich constant n is within the range (0.1 < n < 1), reported in literature [30] show- ing that adsorption is favorable. However, the linearized equation did not give a good correlation for the removal of Lead(II) onto acid treated maize tassel, indicating that Lead(II) adsorption by acid treated maize tassel fits bet- ter to the Langmuir model than to the Freundlich model. The calculated RL was 0.62 indicating that the adsorption of the Lead(II) was a favorable process. The plot of t/qt versus t is shown in Figure 4(c). The values of k2 and qe were 0.135 g/mg min and 2.02 mg/g respectively. The R2 value was 0.998, indicating a chemisorptions process. 3.7. Treatment of Industrial Wastewater The suitability of the sulphuric acid treated maize tassel for the removal of Lead(II) was tested with the leachates from an electroplating plant. The leachate sample pH was maintained between 5.4 - 5.9 and the determined compo- sition of leachate from an electroplating plant is tabulated in Table 3. The treatment of Lead(II) in leachates was signifi- cantly good (p < 0.05). Almost 93.9% removal from wastewater was possible with 1.2 g of the adsorbent. Thus, the results corroborate well with what is obtained from the batch adsorption mode conducted for Lead(II) removal in synthetic wastewater samples. Furthermore, preliminary treatment of the leachates is essential before application of acid treated maize tassel as the adsorbent. 3.8. Desorption Studies For wastewater treatment, the successful application of desorption reduces the dependence on thermal activation, incineration, and land disposal, which directly or indi- rectly increases environmental pollution. In this study, effect of HCl concentration on the desorption of Lead(II) ion is shown in Figure 5. It can be observed that desorp- tion rate increases with the increase in HCl concentration but attained a constant at 0.2 M·HCl. (a) (b) (c) Figure 4 (a) Langmuir isotherm; (b) Freundlich isotherm for Lead(II) ion removal; (c). Pseudo-second-order kinetic fit for Lead(II) adsorption. Table 2. Langmuir and Freundlich constants for Lead(II) adsorption using acid treated maize tassel. Adsorption isotherm Parameter Value Langmuir qmax (mg/g) 37.31 b (L/mg) 0.062 R2 0.9997 Freundlich KF 0.077 n 0.482 R2 0.9515 4. Conclusion The present study showed that acid treated maize tassel can be used for the effective removal of Lead(II) ions Open Access AJAC 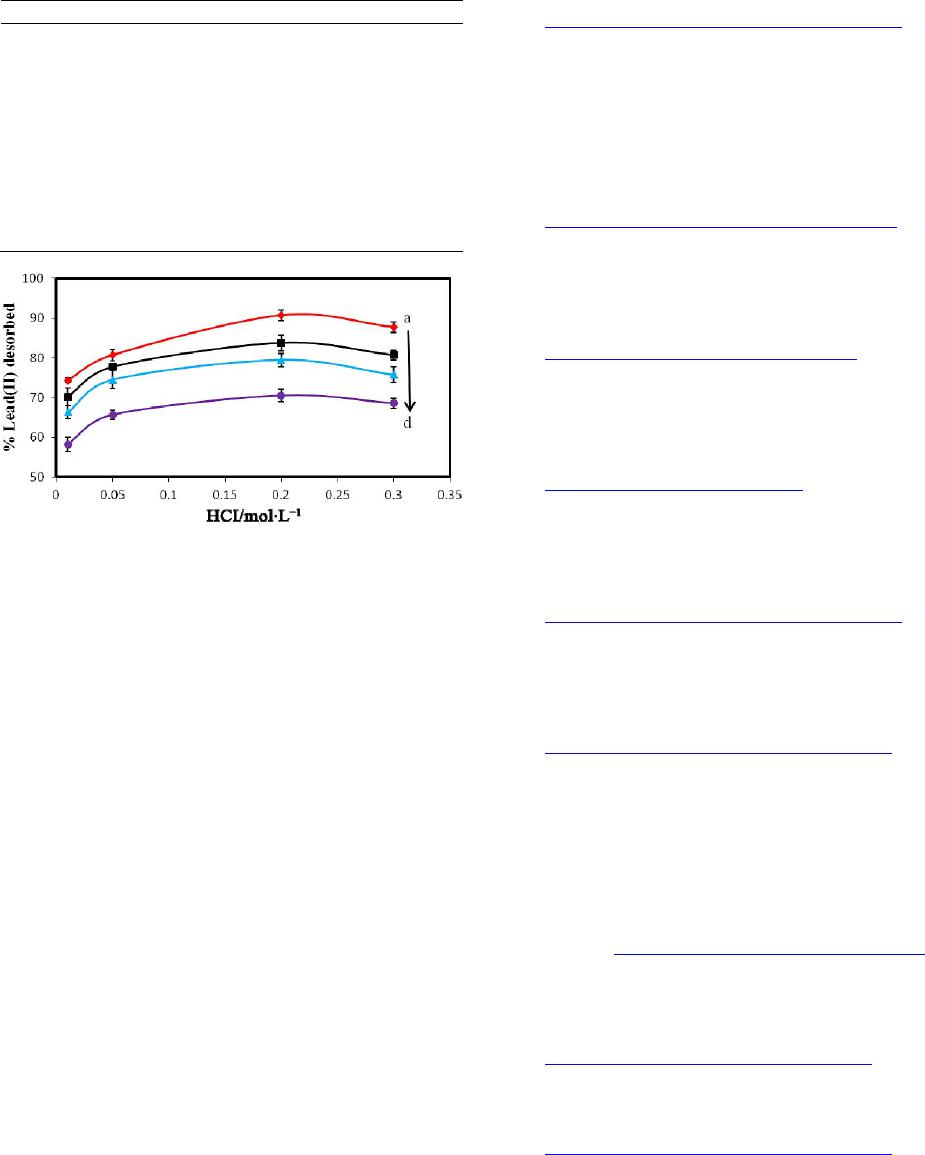 M. MOYO, L. CHIKAZAZA 694 Table 3. Composition of leachates sample from an electro- plating plant. Parameter Value pH 3.9 Total dissolved salts (mg/L) 5056.44 Turbidity (NTU) 1.22 Electrochemical conductivity (μmhos/cm) 3044.28 COD (mg/L) 30.56 Chloride (mg/L) 320.58 Sulphate (mg/L) 1005.56 Calcium (mg/L) 95.35 Lead (mg/L) 98.28 Zinc (mg/L) 18.25 Figure 5. Effect of different HCl concentrations on desorp- tion of Lead(II) ion (concentrations: a to d: 10 to 50 mg/L Error bar = ±S.D. and n = 3). from wastewater. Lead(II) adsorption was found to be pH dependent and maximum removal was observed at pH 5.4. An increase in the acid treated maize tassel dosage leads to an increase in Lead(II) ions removal due to a corresponding increase in the number of active sites. The Langmuir adsorption isotherm was demonstrated to pro- vide the best correlation (R2 = 0.9997) for the adsorption of Lead(II) ions onto acid treated maize tassel confirming monolayer coverage. Adsorption obeyed a pseudo-sec- ond-order model. It can be concluded that the acid treated maize tassel from maize tassel adds to the global discus- sion of its cost-effective utilization in wastewater treat- ment. 5. Acknowledgements The authors are grateful for the Department of Chemical Technology, Midlands State University, Gweru, Zimbab we for providing facilities. REFERENCES [1] P. S. Kumar, “Adsorption of Lead(II) Ions from Simu- lated Wastewater Using Natural Waste: A Kinetic, Ther- modynamic and Equilibrium Study,” Environmental Pro- gress & Sustainable Energy, 2013, pp. 1-10. [2] A. B. Dekhil, Y. Hannachi, A. Ghorbel and T. Boubaker, “Removal of Lead and Cadmium Ions from Aqueous So- lutions Using the Macroalga Caulerpa racemosa,” Chem- istry and Ecology, Vol. 27, No. 3, 2011, pp. 221-234. http://dx.doi.org/10.1080/02757540.2010.529077 [3] World Health Organization, “Guidelines for Drinking Water Quality,” Vol. 1-2, Geneva, 1984. [4] J. M. Lezcano, F. González, A. Ballester, M. L. Blázquez, J. A. Muñoz and C. García-Balboa, “Sorption and De- sorption of Cd, Cu and Pb Using Biomass from an Eu- trophized Habitat in Monometallic and Bimetallic Sys- tems,” Journal of environmental management, Vol. 92, No. 10, 2011, pp. 2666-2674. http://dx.doi.org/10.1016/j.jenvman.2011.06.004 [5] A. B. Marín, J. F. Ortuño, M. I. Aguilar, V. F. Meseguer, J. Saez and M. Lloréns, “Use of Chemical Modification to Determine the Binding of Cd(II), Zn(II) and Cr(III) Ions by Orange Waste,” Biochemical Engineering Journal, Vol. 53, No. 1, 2010, pp. 2-6. http://dx.doi.org/10.1016/j.bej.2008.12.010 [6] J. P. Chen and L. Yang, “Chemical Modification of Sar- gassum sp. for Prevention of Organic Leaching and En- hancement of Uptake during Metal Biosorption,” Indus- trial & Engineering Chemistry Research, Vol. 44, No. 26, 2005, pp. 9931-9942. http://dx.doi.org/10.1021/ie050678t [7] I. W. Mwangi, J. C. Ngila, and J. O. Okonkwo, “A Com- parative Study of Modified and Unmodified Maize Tas- sels for Removal of Selected Trace Metals in Contami- nated Water,” Toxicological & Environmental Chemistry, Vol. 94, No. 1, 2012, pp. 20-39. http://dx.doi.org/10.1080/02772248.2011.638636 [8] M. Madhava Rao, A. Ramesh, G. Purna Chandra Rao and K. Seshaiah, “Removal of Copper and Cadmium from the Aqueous Solutions by Activated Carbon Derived from Ceiba pentandra Hulls,” Journal of Hazardous Materials, Vol. 129, No. 1, 2006, pp. 123-129. http://dx.doi.org/10.1016/j.jhazmat.2005.08.018 [9] S. T. Ong, S. P. Yip, P. S. Keng, S. L. Lee and Y. T. Hung, “Papaya (Carica papaya) Seed as a Low-Cost Sor- bent for Zinc Removal,” African Journal of Agricultural Research, Vol. 7, No. 5, 2012, pp. 810-819. [10] Ö. Gerçel and H. F. Gerçel, “Adsorption of Lead(II) Ions from Aqueous Solutions by Activated Carbon Prepared from Biomass Plant Material of Euphorbia rigida,” Chemical Engineering Journal, Vol. 132, No. 1, 2007, pp. 289-297. http://dx.doi.org/10.1016/j.cej.2007.01.010 [11] M. Imamoglu and O. Tekir, “Removal of Copper(II) and Lead(II) Ions from Aqueous Solutions by Adsorption on Activated Carbon from a New Precursor Hazelnut Husks,” Desalination, Vol. 228, No. 1, 2008, pp. 108-113. http://dx.doi.org/10.1016/j.desal.2007.08.011 [12] A. Özer, “Removal of Pb(II) Ions from Aqueous Solu- tions by Sulphuric Acid-Treated Wheat Bran,” Journal of Hazardous Materials, Vol. 141, No. 3, 2007, pp. 753-761. http://dx.doi.org/10.1016/j.jhazmat.2006.07.040 [13] K. Kadirvelu and C. Namasivayam, “Activated Carbon from Coconut Coirpith as Metal Adsorbent: Adsorption of Cd(II) from Aqueous Solution,” Advances in Environ- mental Research, Vol. 7, No. 2, 2003, pp. 471-478. Open Access AJAC  M. MOYO, L. CHIKAZAZA Open Access AJAC 695 http://dx.doi.org/10.1016/S1093-0191(02)00018-7 [14] O. S. Amuda, A. Giwa and I. A. Bello, “Removal of Heavy Metal from Industrial Wastewater Using Modified Activated Coconut Shell Carbon,” Biochemical Engineer- ing Journal, Vol. 36, No. 2, 2007, pp. 174-181. http://dx.doi.org/10.1016/j.bej.2007.02.013 [15] M. Moyo, J. O. Okonkwo and N. M. Agyei, “A Novel Hydrogen Peroxide Biosensor Based on Adsorption of Horseradish Peroxidase onto a Nanobiomaterial Compo- site Modified Glassy Carbon Electrode,” Electroanalysis, Vol. 25, 2013, pp. 1946-1954. http://dx.doi.org/10.1002/elan.201300165 [16] A. El Nemr, A. El Sikaily, A. Khaled and O. Abdelwahab, “Removal of Toxic Chromium(VI) from Aqueous Solu- tion by Activated Carbon Using Casuarina equisetifolia,” Chemistry and Ecology, Vol. 23, No. 2, 2007, pp. 119- 129. http://dx.doi.org/10.1080/02757540701197754 [17] A. Esmaeili, S. Ghasemi and J. Sohrabipour, “Biosorption of Copper from Wastewater by Activated Carbon Prepa- ration from Alga Sargassum sp,” Natural Product Re- search, Vol. 24, No. 4, 2010, pp. 341-348. http://dx.doi.org/10.1080/14786410903064915 [18] P. S. Kumar, S. Ramalingam, R. V. Abhinaya, K. V. Thi- ruvengadaravi, P. Baskaralingam and S. Sivanesan, “Lead (II) Adsorption onto Sulphuric Acid Treated Cashew Nut Shell,” Separation Science and Technology, Vol. 46, No. 15, 2011, pp. 2436-2449. http://dx.doi.org/10.1080/01496395.2011.590174 [19] K. Nath, S. Panchani, M. S. Bhakhar and S. Chatrola, “Preparation of Activated Carbon from Dried Pods of Prosopis cineraria with Zinc Chloride Activation for the Removal of Phenol,” Environmental Science and Pollu- tion Research, 2013, pp. 4030-4045. http://dx.doi.org/10.1007/s11356-012-1325-y [20] A. O. Dada, J. O. Ojediran, A. P. Olalekan, “Sorption of Pb2+ from Aqueous Solution unto Modified Rice Husk: Isotherms Studies,” Advances in Physical Chemistry, Vol. 2013, 2013, pp. 1-6. http://dx.doi.org/10.1155/2013/842425 [21] H. M. F. Freundlich, “Over the Adsorption in Solution,” Journal of Physical Chemistry, Vol. 57, 1906, pp. 384- 470. [22] I. Langmuir, “The Constitution and Fundamental Proper- ties of Solids and Liquids. II. Liquids. 1.,” Journal of the American Chemical Society, Vol. 39, No. 9, 1917, pp. 1848-1906. http://dx.doi.org/10.1021/ja02254a006 [23] K. R. Hall, L. C. Eagleton, A. Acrivos and T. Vermeulen, “Pore- and Solid-Diffusion Kinetics in Fixed-Bed Ad- sorption under Constant-Pattern Conditions,” Industrial & Engineering Chemistry Fundamentals, Vol. 5, No. 2, 1966, pp. 212-223. http://dx.doi.org/10.1021/i160018a011 [24] W. Li, L. Zhang, J. Peng, N. Li, S. Zhang and S. Guo, “Tobacco Stems as a Low Cost Adsorbent for the Re- moval of Pb(II) from Wastewater: Equilibrium and Ki- netic Studies,” Industrial Crops and Products, Vol. 28, No. 3, 2008, pp. 294-302. http://dx.doi.org/10.1016/j.indcrop.2008.03.007 [25] J. Srinivasa Rao, C. Kesava Rao and G. Prabhakar, “Op- timization of Biosorption Performance of Casuarina Leaf Powder for the Removal of Lead Using Central Compos- ite Design,” Journal of Environmental & Analytical Toxi- cology, Vol. 3, No. 2, 2013, p. 166. [26] I. W. Mwangi and J. C. Ngila, “Removal of Heavy Metals from Contaminated Water Using Ethylenediamine-Mo- dified Green Seaweed Caulerpa serrulata,” Physics and Chemistry of the Earth, Parts A/B/C,” Vol. 50-52, 2012, pp. 111-120. http://dx.doi.org/10.1016/j.pce.2012.08.015 [27] S. Doyurum and A. Celik, “Pb(II) and Cd(II) Removal from Aqueous Solutions by Olive Cake,” Journal of Haz- ardous Materials, Vol. 138, No. 1, 2006, pp. 22-28. http://dx.doi.org/10.1016/j.jhazmat.2006.03.071 [28] C. M. Zvinowanda, J. O. Okonkwo, M. M. Sekhula, N. M. Agyei and R. Sadiku, “Application of Maize Tassel for the Removal of Pb, Se, Sr, U and V from Borehole Water Contaminated with Mine Wastewater in the Presence of Alkaline Metals,” Journal of Hazardous Materials, Vol. 164, No. 2, 2009, pp. 884-891. http://dx.doi.org/10.1016/j.jhazmat.2008.08.110 [29] O. F. Olorundare, R. W. M. Krause, J. O. Okonkwo and B. B. Mamba, “Potential Application of Activated Carbon from Maize Tassel for the Removal of Heavy Metals in Water,” Physics and Chemistry of the Earth, Parts A/B/C, Vol. 50-52, 2012, pp. 104-110. [30] C. Raji and T. S. Anirudhan, “Batch Cr(VI) Removal by Polyacrylamide-Grafted Sawdust: Kinetics and Thermo- dynamics,” Water Research, Vol. 32, No. 12, 1998, pp. 3772-3780. http://dx.doi.org/10.1016/S0043-1354(98)00150-X
|