 Surgical Science, 2013, 4, 494-499 Published Online November 2013 (http://www.scirp.org/journal/ss) http://dx.doi.org/10.4236/ss.2013.411096 Open Access SS Accuracy of Fine Needle Aspiration Cytology of Solitary Thyroid Nodules in Tertiary versus Community Hospital H. M. El Hennawy1, H. O. Abu Zaid2, Imaad Bin Mujeeb3, Eihab A. El Kahlout4, El S. M. Bedair5 1Trauma Surge ry Department, Hama d General Hospital, Hamad Medical Corporation, Doha, Qatar 2Medicine Department, Al Khor Hospital, Hamad Medical Corporation, Doha, Qatar 3Pathology Department, Hamad General Hospital, Hamad Medi cal Corporation, Doha, Qatar 4Surgery Department, Al Khor Hospital, Ham ad Med ical Corporation, Doha, Qatar 5Radiology Department, Al Khor Hospital, Hamad Medical Corporation, Doha, Qatar Email: hennawyhany@hotmail.com Received October 11, 2013; revised November 2, 2013; accepted November 10, 2013 Copyright © 2013 H. M. El Hennawy et al. This is an open access article distributed under the Creative Commons Attribution Li- cense, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. ABSTRACT Background: Fine needle aspiration cytology (FNAC) is considered as the gold standard diagnostic test for the diagno- sis of solitary thyroidnodules (STN). Aim: To compare the accuracy of FNAC in diagnosis of thyroid nodules in Al Khor community hospital versus that of tertiary hospital (Hamad General Hospital) in Qatar. Methods: Retrospective, descriptive and comparative study of 320 patients of any age group and both sexes who have thyroid nodules and are subjected to surgical treatment [160 patients from Al Khor community hospital (group A) and 160 patients from Hamad General Hospital (group B)] during the period from May 2005 to Dec 2012. Each group was subdivided into free hand FNAC (done by physicians) an d ultrasonography guided FN AC. All samples were prepared in the same h istopathology laboratory and interpreted by the same histopathology team. Results: Free hand FNAC in group A and group B achieved a sensitivity of (10% vs. 50 %), specificity of 93% in both groups, positive predictive valu e of (25% vs. 17%), negative predictive value of (82% vs. 98%), and a total accuracy of (82% vs. 94%) respectively, while US guided FNAC in group A and group B achieved a sensitiv ity of (10% vs. 86%), specificity of (98% vs. 94%), positive pred ic- tive value of (50% vs. 60%), negative predictive value of (86% vs. 98%), and a total accuracy of (85% vs. 97%) respec- tively. Conclusion: The overall accuracy of FNAC of STN in tertiary hospital was better than community hospital. More training is required for community hospital staff. Keywords: Needle Aspiration; Solitary Thyroid Nodules; Accuracy 1. Introduction Thyroid nodules are common clinical findings and have a reported prevalence of 4% - 7% of adult population. However, fewer than 5% of adult thyroid nodules are malignant, and the vast majority is non-neoplastic lesions [1]. Solitary thyroid nodule (STN) is defined clin ically as the localized thyroid enlarg ement with apparently normal rest of the gland. The main goal of evaluating these nod- ules is to identify nodules with malignant potential [2]. FNAC is safe, simple and quick with a low complication rate so considered as the gold standard initial investiga- tion in the diagnosis of thyroid swellings [3-5]. With the increase use of imaging over the last decades, the number of incidentally discovered thyroid nodules is rising [6]. Either palpation (free hand) or ultrasonography (US) may be used for guidance of FNA, but US has several advan- tages over palpation [7,8]. Limitations of FNAC include false negative and false positive results and a proportion of FNA results that are not obviously benign or malig- nant and fall into the indeterminate or suspicious group [9]. Moreover, the pitfalls of FNAC are related to speci- men adequacy, sampling techniques, the skill of the ph y- sician performing the aspiration, the experience of the pathologist interpreting the aspirate and the overlapping cytological features between some benign and malignant thyroid lesions [10]. No previous researchers studied if the level of hospital (community or tertiary) affects the accuracy of FNAC. This study aimed to compare the accuracy of FNAC in STN in a tertiary versus commu- nity hospitals and to determine the possible reasons of differences in accuracy if present.  H. M. EL HENNAWY ET AL. 495 2. Methods This is a retrospective comparative descriptive study of 320 Consecutive diagnosed cases of STN treated surgi- cally from two hospitals; 160 patients from Al Khor community Hospital (group A) and 160 patients from Hamad General tertiary Hospital (group B) during the period from May 2005 to Dec 2012. Informed consents were taken from all patients. Institution research com- mittee approved the study. Detailed history, physical examination, thyroid function tests, neck ultrasonography, FNAC, surgical intervention and post-operative histopa- thology were reviewed. All cytological and postoperative histopathology specimens were prepared in the same cyto/histopathology laboratory and interpreted by same cyto/histopathology team. Each group was subdivided into free hand FNAC (done by surg eons in the outpatient clinic) and ultrasonography guided FNAC. Free hand FNAC were performed using aspiration technique when a 23-gauge needle was connected to a 10-ml syringe mounted on a syringe holder. Multiple needle passes were made within the lesion 3 - 4 times at varying angles and depths and with constant negative pressure (never emerging outside the skin). Before final withdrawal, the negative pressure was released prior to the needle emerging from the skin. The cytological material was transferred on to glass slides. For US-guided FNAC Technique, after the lesion is localized using a high- resolution (7.5 - 15 MHz) linear-array transducer, the patient is instructed not to swallow or speak during the insertion of the needle. A freehand biopsy technique is used, and the syringe attached to the needle is placed just above the transducer. During the procedure, all needle movements should be continuously visualized in real time. It is recommended that aspiration be performed at least twice. Before aspiration, scanning is performed in the transverse plane for lesion localization, followed by color Doppler mapping to depict any large blood vessels in and around. The collected material is placed on four glass slides, smeared, and fixed in 95% ethyl alcohol for about 30 minutes. All the slides were stained with Pa- panicolaou stain. We categorized our results into insuffi- cient for diagnosis, benign, atypical follicular lesion of undetermined significance, follicular neoplasm, suspi- cious for malignancy, and malignant sampling according to the recent Bethesda classification [11]. The smear was considered adequate if there were at least five groups of well-visualized follicular cells, each group containing ten or more cells [12]. All patients subsequently had a thy- roid resection and a definitive diagnosis was reached FNAC results were then compared with the definitive histological diagnosis which was considered the gold standard. No blinding was done, in Al Khor community hospital group (A) we included all STN FNAC speci- mens during the study period while in tertiary hospital group (B) we included only the first consecutive 160 FNAC specimen done during the same period. Follicular neoplasm Specimens were excluded from our results. We studied the details of inconclusive specimen. We excluded patients with inconclusive and follicular adenoma/neoplasm results. Total number of Group A specimens became 120 vs. 134 in group B. Free hand FNAC was 55 in group A vs. 64 in group B. US guided FNAC was 65 in group A vs. 70 in group B. We com- pared the results of free hand FNAC and US guided FNAC in both hospitals separately. Data were analyzed statistically using SPSS software to assess the sensitiv ity, specificity and accuracy of the result. Diagnostic accu- racy represents combination of sensitivity and specificity. 3. Results The FNAC results in community hospital group A vs. tertiary hospital group B included; benign diagnosis in [114 patients (71.2%) vs. 123 (76.8%)], malignant in [6 patients (3.7%) vs. 11 (6.8%)], follicular neoplasm in 9 patients (5.6%) vs. 8 (5%)] and inconclusive specimens in 31 patients (19.4%) vs. 18 (11.2%)] (Table 1). Inconclusive specimens in free hand FNAC were 29% and 15% in group A and B respectively, while in US FNAC were 10% and 6.25% in group A and B respec- tively. Free hand FNAC repeated twice in group A and group B was (15% vs. 10%) while US FNAC repeated twice in group A and group B was (6% vs. 5%). 90% of the repeated FNAC revealed satisfactory results. The remaining 10% (5 patients) underwent total thyroidec- tomy; 3 of them were papillary carcinoma. We excluded patients with inconclusive and follicular adenoma/neo- plasm results. Total number of Group A specimens be- came 120 vs. 134 in group B. Free hand FNAC was 55 in group A vs. 64 in group B. US guided FNAC was 65 in group A vs. 70 in group B. FNAC results were compared to that of postoperativ e histopathology. Free hand FNAC in both groups (Table 2) and US guided FNAC in both groups (Table 3). Free hand FNAC in group A and group B achieved a sensitivity of (10% vs. 50%), specificity of 93% in both groups, positive predictive value of (25% vs. 17%), negative predictive value of (82% vs. 98%), and a total accuracy of (82% vs. 94%) respectively (Ta b le 4). While in US guided FNAC in group A and group B achieved a sensitivity of (10% vs. 86%), specificity of (98% vs. 94%), positive predictive value of (50% vs. 60%), nega- tive predictive value of (86% vs. 98%), and a total accu- racy of (85% vs. 97%) respectively (Table 5). 4. Discussion In current study, we can notice a significant differen ce in accuracy of FNAC of STN in tertiary versus community hospital. While the sensitivity of free hand FNAC in the Open Access SS 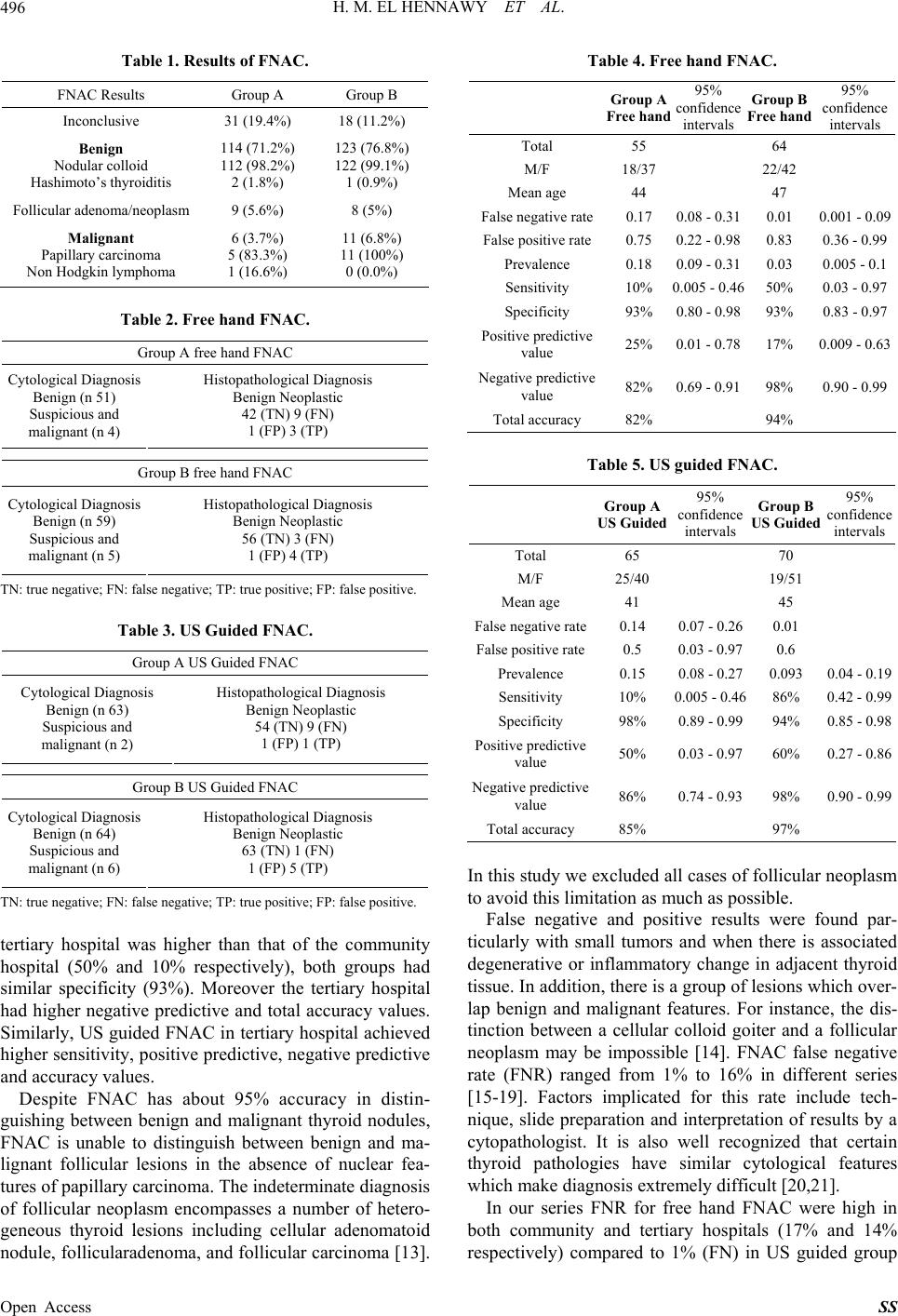 H. M. EL HENNAWY ET AL. 496 Table 1. Results of FNAC. FNAC Results Group A Group B Inconclusive 31 (19.4%) 18 (11.2%) Benign Nodular colloid Hashimoto’s thyroiditis 114 (71.2%) 112 (98.2%) 2 (1.8%) 123 (76.8%) 122 (99.1%) 1 (0.9%) Follicular adenoma/neoplasm 9 (5.6%) 8 (5%) Malignant Papillary carcinoma Non Hodgkin lymphoma 6 (3.7%) 5 (83.3%) 1 (16.6%) 11 (6.8%) 11 (100%) 0 (0.0%) Table 2. Free hand FNAC. Group A free hand FNAC Cytological Diagnosis Benign (n 51) Suspicious and malignant (n 4) Histopathological Diagnosis Benign Neoplastic 42 (TN) 9 (FN) 1 (FP) 3 (TP) Group B free hand FNAC Cytological Diagnosis Benign (n 59) Suspicious and malignant (n 5) Histopathological Diagnosis Benign Neoplastic 56 (TN) 3 (FN) 1 (FP) 4 (TP) TN: true negative; FN: false ne gative; TP: true positive; FP: false positive. Table 3. US Guided FNAC. Group A US Guided FNAC Cytological Diagnosis Benign (n 63) Suspicious and malignant (n 2) Histopathological Diagnosis Benign Neoplastic 54 (TN) 9 (FN) 1 (FP) 1 (TP) Group B US Guided FNAC Cytological Diagnosis Benign (n 64) Suspicious and malignant (n 6) Histopathological Diagnosis Benign Neoplastic 63 (TN) 1 (FN) 1 (FP) 5 (TP) TN: true negative; FN: false ne gative; TP: true positive; FP: false positive. tertiary hospital was higher than that of the community hospital (50% and 10% respectively), both groups had similar specificity (93%). Moreover the tertiary hospital had higher negative predictive and total accuracy values. Similarly, US guided FNAC in tertiary hospital achieved higher sensitivity, po sitive predictive, negative pred ictive and accuracy values. Despite FNAC has about 95% accuracy in distin- guishing between benign and malignant thyroid nodules, FNAC is unable to distinguish between benign and ma- lignant follicular lesions in the absence of nuclear fea- tures of papillary carcinoma. The indeterminate diagno sis of follicular neoplasm encompasses a number of hetero- geneous thyroid lesions including cellular adenomatoid nodule, follicularaden oma, and follicular carcinoma [13]. Table 4. Free hand FNAC. Group A Free hand 95% confidence intervals Group B Free hand 95% confidence intervals Total 55 64 M/F 18/37 22/42 Mean age 44 47 False negative rate0.17 0.08 - 0.31 0.01 0.001 - 0.09 False positive rate0.75 0.22 - 0.98 0.83 0.36 - 0.99 Prevalence 0.18 0.09 - 0.31 0.03 0.005 - 0.1 Sensitivity 10% 0.005 - 0.46 50% 0.03 - 0.97 Specificity 93% 0.80 - 0.98 93% 0.83 - 0.97 Positive predictive value 25% 0.01 - 0.78 17% 0.009 - 0.63 Negative predictive value 82% 0.69 - 0.91 98% 0.90 - 0.99 Total accuracy 82% 94% Table 5. US guided FNAC. Group A US Guided 95% confidence intervals Group B US Guided 95% confidence intervals Total 65 70 M/F 25/40 19/51 Mean age 41 45 False negative rate0.14 0.07 - 0.26 0.01 False positive rate0.5 0.03 - 0.97 0.6 Prevalence 0.15 0.08 - 0.27 0.093 0.04 - 0.19 Sensitivity 10% 0.005 - 0.46 86% 0.42 - 0.99 Specificity 98% 0.8 9 - 0.99 94% 0.85 - 0.98 Positive predictive value 50% 0.03 - 0.97 60% 0.27 - 0.86 Negative predictive value 86% 0.74 - 0.93 98% 0.90 - 0.99 Total accuracy 85% 97% In this study we excluded all cases of follicular neoplasm to avoid this limitation as much as possible. False negative and positive results were found par- ticularly with small tumors and when there is associated degenerative or inflammatory change in adjacent thyroid tissue. In addition, th ere is a group of lesions which over- lap benign and malignant features. For instance, the dis- tinction between a cellular colloid goiter and a follicular neoplasm may be impossible [14]. FNAC false negative rate (FNR) ranged from 1% to 16% in different series [15-19]. Factors implicated for this rate include tech- nique, slide preparation and interpretation of results by a cytopathologist. It is also well recognized that certain thyroid pathologies have similar cytological features which make diagnosis extremely difficult [20,21]. In our series FNR for free hand FNAC were high in both community and tertiary hospitals (17% and 14% respectively) compared to 1% (FN) in US guided group Open Access SS  H. M. EL HENNAWY ET AL. 497 in both hospitals. FNAC false positive rate (FPR) indicates that a patient with malignant FNAC result was found on histological examination to have benign lesion. In our series the FPR for free hand FNAC were significantly low in tertiary hospital group for both free hand and US guided FNAC (5% and 6% respectively) compared to higher values in community hospital group for both free hand and US guided FNAC (75% and 83% respectively) which agrees with other series that ranged from 0% to 8% [21]. FNAC has inherent limitations related to inadequate sampling, reportedly from 10% to 31% [22-24]. Speci- men procurement may be affected by the level of opera- tor experience, accuracy of localization of the lesion and the needle, method of guidance (palpation or US), num- ber of aspirations, needle gauge, sampling technique, capability for immediate on-site cytologic analysis, and many other factors. Unsatisfactory specimen may be due to cystic fluid, bloody smears or poor technique in ob- taining the sample and/or preparing the slides. The Pa- panicolaou Society of Cytopathology task force on Stan- dards of Practice recommends that aspirators who per- sistently produce a high rate of unsatisfactory aspirates (>15%) should be identified and given r emedial training. To develop and maintain the necessary level of staff ex- pertise in an institution, the number of staff members who perform aspiration biopsies and the interpreting cy- topathologists should be kept small. Each staff member who performs aspiration biopsies must complete at least 1 - 5 such procedures per month [25]. Criteria of speci- men adequacy are: 1) aminimum of five or six groups of well-preserved cells, with each group containing ap- proximately 10 - 15 cells; 2) six clusters of benign cells on at least two slides prepared from separate FNA biopsy samples; 3) 10 clusters of follicular cells, with each clus- ter containing at least 20 cells. Adherence to rigid criteria leads to higher nondiagno stic rates and lower false-nega- tive rates; and high nondiagnostic rates exacerbate pa- tient anxieties and lead to the performance of unneces- sary repeat aspiration and unnecessary surgical excision, thereby reducing the overall efficiency and cost-effec- tiveness of the FNA biopsy procedure [26]. In our study, Inconclusive specimens were higher in community hos- pital for both free hand FNAC (29% vs. 15%) and US FNAC (10% vs. 6.25%). A large nodule is easier to sample than a smaller one, and the diagnostic yield from FNA of large nodules may be higher. Degirmenci et al. reported that the highest specimen adequacy rate was observed among nodules smaller than 1 cm (76.4%) and the lowest rate was ob- served among nodules larger than 3 cm (56.9%). They inferred that the lower rate in larger nodules probably resulted from increased vascularity and the larger size of blood vessels, with resultant blood staining of the mate- rial acquired at fine needle biopsy [18]. Another probable cause of the inadequacy of specimens from larger nod- ules in the study by Degirmenci et al. is that large nod- ules more often are cystic and contain necrotic areas. Selecting finer needles (24 - 25 G) for sonography- guided FNB of thyroid nodules and using the capil- lary-action technique decreased the rate of inadequate material in cytological examination [18]. One of the limitations of this study was that we did not include the STN size and site. Moreover we did not mention in can- cer cases if the lesions originated from the same STN or originated from other foci. FNAC has high sensitivity and specificity for predict- ing thyroid malignancies averaging 83% and 92% re- spectively [27,28]. Inpatients with thyroid nodules 4 cm or larger, the FNAC results are highly inaccurate, mis- classifying half of all patients with reportedly benign lesions. Furthermore, those patients with a non diagnostic FNAC result display a high risk of differentiated thyroid carcinoma. Therefore, Scott N. Pinchot et al. recom- mended that diagnostic lobectomy be strongly considered in patients with thyro id nodules 4 cm or larger regard less of FNAB cytologic test results [29]. A possible way to reduce such error is to do aspirations from different parts which could reveal the typical nuclear features of papil- lary carcinoma [30]. The determinant factor for such a wide range of sensitivity, specificity and accuracy may be how the cytopathologists classify “suspicious” as well as false positive and negative samples. Some authors include follicular lesion in malignant/neoplastic group, whereas others exclude them from the calculations. In our study sensitivity rate was low compared to others as three out of four (75%) “indeterminate” FNAC results were later found to have malignancy on histological ex- amination. This was desp ite adequ ate number of cells (by definition) and suggests that the FNAC interpretation is operator dependent. Our positive predictive values and negative predictive values for both techniques in both hospitals were comparable with published data [31,32]. The accuracy of US-guided FNA was significantly higher than that of palpation-guided FNA (68% vs. 48%), particularly for tumors smaller than 2 cm and those that were cystic or in deep locations. In our study the total accuracy of free hand FNAC was higher in tertiary hos- pital (94% vs. 82%) similarly, total accuracy of US guided FNAC was higher in tertiary hospital (97% vs. 85%). US-guided FNA is the best available diagnostic pro- cedure to evaluate thyroid nodules to submit to surgery [33]. Kim DW et al. showed a good adequacy and effi- cacy of US-FNAC for thyroid nodules despite one-sam- pling [34]. Specimen adequacy is not dependent on the vascularity and echogenicity of the sampled thyroid nod- ule but on components such as cystic change, calcifica- Open Access SS  H. M. EL HENNAWY ET AL. 498 tion, and fibrosis. Similarity, in our study US guided FNAC had better accuracy and less inconclusive results than free hand FNAC. One of the limitations of this study group is that it comprised FNAC samples taken by different operators, hence with varying sk ill levels and experience. Moreover, patients’ selection process not similar in both hosp itals as it is consecutive 160 patients in the community hospital (all patients done in this hospital) and we selected ran- domly 160 patients from the about 700 patients done in the tertiary hospital. It is anticipated that improvement in diagnosis will develop from specific cytopathological training in thy- roid FNAC, consideration of the cytopathologist per- forming the aspiration biopsy, and the development of immunohistochemical and molecular techniques applied to the cytologic smears. 5. Conclusion The overall accuracy of both free hand and US guided FNAC in tertiary hospital was better than community hospital. We recommend better training of community hospital staff and encourage more liberal use of US guided FNAC. REFERENCES [1] S. A. Roman, “Endocrine Tumors. Evaluation of Thyroid Nodule,” Current Opinion in Oncology, Vol. 15, No. 1, 2003, pp. 66-70. http://dx.doi.org/10.1097/00001622-200301000-00010 [2] H. K. Zygnunt, “The Thyroid Gland and Thyroglossal Tract: Short Practice of Surgery,” 24th Edition, Arnold, London, 2004, pp. 784-786. [3] P. Caruso and E. L. Muzzaferri, “Fine Needle Aspiration Biopsy in the Management of Thyroid Nodules,” Endo- crinology, Vol. 1, 1991, pp. 194-202. http://dx.doi.org/10.1097/00019616-199106000-00009 [4] U. Handa, S. Garg, H. Mohan and N. Nagarkar, “Role of Fine Needle Aspiration Cytology in the Diagnosis and Management of Thyroid Lesions: A Study on 434 Pa- tients,” Journal of Cytology, Vol. 25, No. 1, 2008, pp. 13- 17. [5] M. Amrikachi, I. Ramzy, S. Rubenfeld and T. M. Whee l e r, “Accuracy of Fine Needle Aspiration of Thyroid: A Re- view of 6226 Cases and Correlation with Surgical and Clinical Outcome,” Archives of Pathology & Laboratory Medicine, Vol. 125, 2001, pp. 484-488. [6] N. Coorough, K. Hudak, D. Buehler, S. Selvaggi, R. Sip- pel and H. Chen, “Fine Needle Aspiration of the Thyroid: A Contemporary Experience of 3981 Cases,” Journal of Surgical Research, Vol. 170, No. 1, 2011, pp. 48-51. http://dx.doi.org/10.1016/j.jss.2011.02.048 [7] P. W. Wiest , M. F. Hartshorne and P. D. Inskip, “Thyroid Palpation versus High-Resolution Thyroid Ultrasonogra- phy in the Detection of Nodules,” Journal of Ultrasound in Medicine, Vol. 17, No. 8, 1998, pp. 487-496. [8] K. M. Clary, J. L. Condell and Y. Liu, “Interobserver Variability in the Fine Needle Aspiration Biopsy Diagno- sis of Follicular Lesions of the Thyroid Gland,” Acta Cy- tologica, Vol. 49, No. 4, 2005, pp. 378-382. http://dx.doi.org/10.1159/000326169 [9] H. Galera-Davidson, “Diagnostic Problems in Thyroid Fine Needle Aspirations,” Diagnostic Cytopathology, Vol. 17, No. 6, 1997, pp. 422-428. http://dx.doi.org/10.1002/(SICI)1097-0339(199712)17:6< 422::AID-DC9>3.0.CO;2-A [10] Z. W. Baloch, M. J. Sack, G. H. Yu, V. A. Livolsi and P. K. Gupta, “Fine Needle Aspiration of Thyroid an Institu- tional Experience,” Thyroid, Vol. 8, No. 7, 1998, pp. 565- 569. http://dx.doi.org/10.1089/thy.1998.8.565 [11] E. S. Cibas and E. Z. Ali, “The Bethesda System for Re- porting Thyroid Cytopathology,” Thyroid, Vol. 19, No. 11, 2009, pp. 1159-1165. http://dx.doi.org/10.1089/thy.2009.0274 [12] E. Saggiorato, P. R. De and M. Volante, “Characteriza- tion of Thyroid ‘Follicularneoplasms’ in Fine-Needle As- piration Cytological Specimens Using a Panel of Immu- nohistochemical Markers: A Proposal for Clinical Appli- cation,” Endocrine-Related Cancer, Vol. 12, No. 2, 2005, pp. 305-317. http://dx.doi.org/10.1677/erc.1.00944 [13] E. A. Sinna and N. Ezzat, “Diagnostic Accuracy of fine Needle Aspiration Cytology in Thyroid Lesions,” Journal of the Egyptian National Cancer Institute, Vol. 24, No. 2, 2012, pp. 63-70. http://dx.doi.org/10.1016/j.jnci.2012.01.001 [14] H. A. Nggada, A. B. Musa, B. M. Gali and M. I. Khalil, “Fine Needle Aspiration Cytology of Thyroid Nodule(s): A Nigerian Tertiary Hospital Experience,” Internet Jour- nal of Pathology, 2006. www.Ispub.com/ostia/index.Php/ijpa [15] D. C. Howlett, B. Harper, M. Quante, A. Berresford, M. Morley, J. Grant, et al., “Diagnostic Adequacy and Ac- curacy of Fine Needle Aspiration Cytology in Neck Lump Assessment: Results from a Regional Cancer Network Over a One Year Period,” Journal of Laryngology & Otology, Vol. 121, No. 6, 2007, pp. 571-579. [16] K. Jarvi, R. Mokka and M. Vorne, “Problems of Cancer Diagnosis in Thyroid Nodules,” Acta Chirurgica Scandi- navica, Vol. 154, 1988, pp. 93-96. [17] P. Perros, Ed., “British Thyroid Association, Royal Col- lege of Physicians. Guidelines for the Management of Thy - roid Cancer,” Report of the Thyroid Cancer Guidelines Update Group, 2nd Edition, Royal College of Physicians, London, 2007. [18] B. Degirmenci, A. Haktanir, R. Albayrak, M. Acar, D. A. Sahin, O. Sahin, A. Yucel and G. Caliskan, “Sonographi- cally Guided Fine-Needle Biopsy of Thyroid Nodules: The Effects of Nodule Characteristics, Sampling Tech- nique, and Needle Size on the Adequacy of Cytological Material,” Clinical Radiology, Vol. 62, No. 8, 2007, pp. 798-803. http://dx.doi.org/10.1016/j.crad.2007.01.024 [19] M. R. Castro and H. Gharib, “Thyroid Fine-Needl e Aspi- ration Biopsy: Progress, Practice, and Pitfalls,” Endocrine Open Access SS  H. M. EL HENNAWY ET AL. Open Access SS 499 Practice, Vol. 9, No. 2, 2003, pp. 128-136. http://dx.doi.org/10.4158/EP.9.2.128 [20] M. Gupta, S. Gupta and V. B. Gupta, “Correlation of Fine Needle Aspiration Cytology with Histopathology in the Diagnosis of Solitary Thyroid Nodule,” Journal of Thy- roid Research, 2010, Article ID: 379051. http://dx.doi.org/10.4061/2010/379051 [21] B. Mundasad, I. Mcallister, J. Carson and P. Pyper, “Ac- curacy of Fine Needle Aspiration Cytology in Diagnosis of Thyroid Swellings,” The Internet Journal of Endocri- nology, Vol. 2, No. 2, 2006. [22] M. Radetic, Z. Kralj and I. Padovan, “Reliability of Aspi- ration Biopsy in Thyroid Nodes: Study of 2190 Operated Patients,” Tumori, Vol. 70, No. 3, 1984, pp. 271-276. [23] D. C. Howlett, B. Harper, M. Quante, A. Berresford, M. Morley and J. Grant, “Diagnostic Adequacy and Accu- racy of Fine Needle Aspiration Cytology in Neck Lump Assessment: Results fro m a Regional Cancer Network over a One Year Period,” Journal of Laryngology and Otology, Vol. 121, No. 6, 2007, pp. 571-579. http://dx.doi.org/10.1017/S0022215106004944 [24] S. A. Kantasueb, K. O. Sukpan and P. O. Mahanupab, “The Study of Thyroid Lesions and the Correlation be- tween Histological and Cytological Findings,” Chiang Mai Medical Journal, Vol. 49, No. 3, 2010, pp. 105-110. [25] A. Bartolazzi, A. Gasbarri and M. Papotti, “Application of an Immunodiagnostic Method for Improving Preopera- tive Diagnosis of Nodular Thyroid Lesions,” Lancet, Vol. 357, No. 9269, 2001, pp. 1644-1650. http://dx.doi.org/10.1016/S0140-6736(00)04817-0 [26] C. Cappelli, I. Pirola, D. Cumetti, L. Micheletti, A. Tironi, E. Gandossi, E. Martino, L. Cherubini, B. Agosti, M. Castellano, C. Mattanza and E. A. Rosei, “Is the Antero- posterior and Transverse Diameter Ratio of Nonpalpable Thyroid Nodules a Sonographic Criteria for Recommend- ing Fine-Needle Aspiration Cytology?” Clinical Endo- crinology, Vol. 63, No. 6, 2005, pp. 689-693. [27] M. Bouvet, J. I. Feldman and G. N. Gill, “Surgical Man- agement of the Thyroid Nodule: Patient Selection Based on the Results of Fine-Needle Aspiration Cytology,” La- ryngoscope, Vol. 102, No. 12, 1992, pp. 1353-1356. http://dx.doi.org/10.1288/00005537-199212000-00008 [28] J. Cap, A. Ryska, P. Rehorkova, E. Hovorkova, Z. Kere- kes and D. Pohnetalova, “Sensitivity and Specificity of the Fine Needle Aspiration Biopsy of the Thyroid: Clini- cal Point of View,” Clinical Endocrinology, Vol. 51, No. 4, 1999, pp. 509-515. [29] S. N. Pinchot, H. Al-Wagih, S. Schaefer, R. Sippel and H. Chen, “Accuracy of Fine-Needle Aspiration Biopsy for Predicting Neoplasm or Carcinoma in Thyroid Nodules 4 cm or Larger,” Archives of Surgery, Vol. 144, No. 7, 2009, pp. 649-655. http://dx.doi.org/10.1001/archsurg.2009.116 [30] X. J. Cai, N. Valiyaparambat h, P. Nixon, A. Waghorn, T. Giles and T. Helliwell, “Ultrasound-Guided Fine Needle Aspiration Cytology in the Diagnosis and Management of Thyroid Nodules,” Cytopathology, Vol. 17, No. 5, 2006, pp. 251-256. http://dx.doi.org/10.1111/j.1365-2303.2006.00397.x [31] T. Yokozawa, S. Fukata and K. Kuma, “Thyroid Cancer Detected by Ultrasound-Guided Fine-Needle Aspiration Biopsy,” World Journal of Surgery, Vol. 20, No. 7, 1996, pp. 848-853, Discussion 853. http://dx.doi.org/10.1007/s002689900129 [32] L. F. Morris, N. Ragavendra and M. W. Yeh, “Evidence- Based Assessment of the Role of Ultrasonography in the Management of Benign Thyroid Nodules,” World Journal of Surgery, Vol. 32, No. 7, 2008, pp. 1253-1263. http://dx.doi.org/10.1007/s00268-008-9494-z [33] B. T. Torres, G. M. Castro, L. I. Cucalón, B. C. Santaren, J. De Diego and R. E. Larreina, “The Accuracy of Fine Needle Aspiration Cytology in the Thryroid Nodule S tu dy, in a Tertiary Hospital,” Endocrine Abstract s, Vol. 22, 2010, p. 834. [34] D. W. Kim, H. J. Choo, J. S. Park, E. J. Lee, S. H. Kim, S. J. Jung and J. H. Ryu, “Ultrasonography-Guided Fine- Needle Aspiration Cytology for Thyroid Nodules: An Em - phasis on One-Sampling and Biopsy Techniques,” Diag- nostic Cytopathology, Vol. 40, Suppl. 1, 2012, pp. E48- E54. http://dx.doi.org/10.1002/dc.21669
|