Journal of Water Resource and Protection
Vol.5 No.11(2013), Article ID:40068,10 pages DOI:10.4236/jwarp.2013.511108
Coliforms and Helminth Eggs Removals by Coagulation-Flocculation Treatment Based on Natural Polymers
1Bioprocesess Department, UPIBI-Instituto Politécnico Nacional, Barrio la Laguna Ticomán, Mexico
2Engineering Institute UNAM, Environmental Engineering Department, Coyoacán, Mexico
Email: *LTorresBustillos@gmail.com
Copyright © 2013 Evelyn Zamudio-Pérez et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received April 16, 2013; revised May 18, 2013; accepted June 16, 2013
Keywords: Coagulation; Fecal Coliforms; Flocculation; Helminth Eggs; Total Coliforms; Wastewaters
ABSTRACT
Wastewater from a municipal wastewater treatment plant (Mexico) was treated by a coagulation-flocculation using natural gums. The residual water was characterized in terms of organic load, and biological contaminants, i.e. total Coliforms (TC), fecal Coliforms (FC) and helminth eggs (HE). The sample has values of 2.8 × 107 MPN/100 ml, 8.48 × 106 MPN/100 ml and 470 HE/L is regarded to TC, FC and HH, respectively. The HE identified in the wastewaters were Diphyllobothrium latum, Ascaris lumbricoides, Hymenolepis nana and Toxocara canis. From several gums, Hydroxypropyl triammonium chloride guar gum (HPTAC-guar) was selected as the most adequate coagulant-flocculant for the class of municipal wastewater (MWW). A diminution of 82% for TC, 94% for FC and 99% for HE was obtained. Besides, the reduction of chemical oxygen demand (COD) and turbidity removals were 46% and 39% respectively. The natural gum mixed with Ca(OH)2, obtained removals of 52% for TC, 100% for HE, 47% for COD and 30% for turbidity.
1. Introduction
Recent studies of two more important Mexico City wastewater flows demonstrated that MWW is having a medium-weak charge with strong bacteriological characteristics, high alkalinity and mineral content [1]. Bacteriological factors and global epidemiological studies on the use of wastewater for agricultural purposes, particularly in Latin American countries, have shown a great public health risk by gastrointestinal diseases such as diarrhea, caused by the presence of viruses, bacteria, protozoa and helminth eggs [2].
Pathogens in water infect approximately 250 million people each year, resulting in 10 ± 20 million deaths. The microbial pathogen concentrations are different depending of many factors such as the month in the year, the specific zone, etc. Thus, determining the number of different microbial pathogens in a sample of water or wastewater is essential to prevent or control a feasible epidemic [3]. Not all countries have HE in wastewater or sludges (Table 1 gives an insight on this fact in some countries). Based on epidemiological studies, the world health organization (WHO) recommends a criterion of ≤ 1 HE/ L in wastewater used for irrigation without using drip irrigation crops eaten without cooking [4].
Many scientific studies have been focused on the removal of bacteria (total and fecal coliforms) and HE as biological indicators of water microbiological quality [5]. Most of these works can be divided into two clear classifications. Those works are oriented to water or wastewater disinfection with acids [6], alkalis , radiations [7], ozone [2], etc. The second group contains reports of the removal efficiencies for physicochemical or biological systems which were designed for solids, carbonaceous or nitrogenated material removals, with a no controlled degree of elimination efficiency for bacteria and/or pathogenic parasites.
Physical methods have been employed for removing (not inactivation) many pathogens in waters and wastewaters. Among others filtration, sedimentation, coagulation-flocculation and the use of membranes can be men
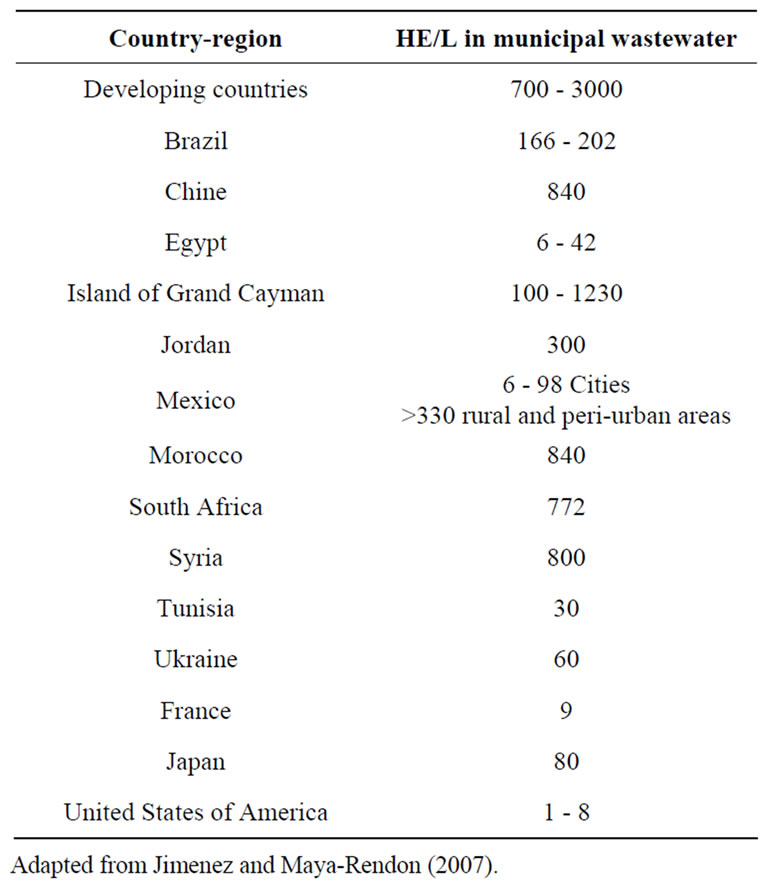
Table 1. Helminth egg content in waste waters and sludges at different countries.
tioned [5]. Jimenez et al. [8], reported the use of sand and synthetic medium filtration of advanced primary treatment effluent from Mexico City for the elimination of HE. The sand and fuzzy filters removed the 70% and 91.3% of HE (initially 22.7 HE/L), respectively, but no reduction of FC was observed (76.5% and 88.4% of the TSS were removed). Jimenez and Maya-Rendon [5] established that membrane bioreactors (99.99%), dualmedia filtration (99%), coagulation-flocculation as tertiary treatment (99%) and granular sand filtration (99%) are the methods with higher efficiencies for HE removal. The removal of simulated helminth eggs by micro screening (98.95+/−0.83% of samples containing between 74.37 and 77.13 eggs/L) was studied [9]. The simulated wastewaters contained 20 mm latex spheres with size distribution and densities very similar to those observed for HE. Sengupta et al. [10], studied the natural sedimentation of HE. These authors found that the measured settling velocities for these eggs were in average 0.0612, 0.1487 and 0.1262 m/h. Nouri et al. [11], reported the removal of Ascaris spp. and Fasciola hepatica eggs from drinking water using the horizontal (HRF) and downflow (DRF) roughing filters. For Fasciola hepatica, HRF and DRF filters were capable of eliminating up to 91.3% and 82.1% of the initial amount of eggs at the lower filtration rate (0.5 m/h). In the case of Ascaris spp., the filters removed up to 60.7% and 57.3%, again, at the lower filtration rate (0.5 m/h).
Biological methods for HE and FC elimination are less utilized in wastewater treatments. Cabirol et al. [12], reported the HE and FC removal by anaerobic thermophilic sludge digestion. They achieved coliforms removals of 1000 MPN/gTS and HE diminished to 0.28 HE/gTS. Von Sperling et al. [13], reported a good COD removal with efficiencies ranging from 69% to 84% for the UASB reactor, from 43% to 56% for the activated sludge systems only and from 85% to 93% for the overall system. La Motta et al. [14], describe the use of a combined UASB/aerobic solid contact system for the treatment of municipal wastewater; the UASB reactor has a total COD removal efficiency of 34%, and the result indicates 36% to TSS. Guo et al. [15], studied the effect to the bioaugmentation in biological tanks at low temperatures, the removal efficiency was 84% for COD with an optimal temperature between 13˚C and 23˚C. Reinoso et al. [16], reported HE, TC and fecal streptococci removals founded in facultative ponds (92.46%, 84.82% and 89.61%), surface flow wetlands (>99.99%, 36.07% and 61.97%) and subsurface flow wetlands (0%, 69.27% and 54.67%). Biological aerated filter (BAF) is an effective bioreactor that provides a small footprint process at various stages of wastewater treatment. Liu et al. [17], reported a COD removal of 85.1% when the hydraulic retention time was longer to 4 h, and 65.7 h to 2 h, when oyster shell was applied as the media to the filter.
Finally, chemical methods have shown higher removal efficiencies. However, their unitary cost of FC and HE removal is the highest compared to all other wastewater treatments. Rojas-Valencia [18] reported that ozone destroyed a wide variety of microorganisms (Escherichia coli was removed 4 logs), including biological indicators (TC, FC with a removal of 2 - 3 logs, Streptococcus, Salmonella, etc.) as well as algae, bacteria, fungi, virus and HE (81% and 94%). Lin et al. [19], reported the use of UV irradiation and the UV-TiO2 such effective to inactivate microorganisms in wastewater disinfection, reported a TC removal of 96.78% (UV) and 93.32% (UV-TiO2). Bhatti et al. [20], integrated chemical treatment using waste hydrogen peroxide and UV light about 93% COD, 90% BOD and 83% turbidity reduction and complete disinfection coliform bacteria occurred when 40% waste H2O2/UV was used. García et al. [21], studied the effect in the HE inactivation with homogeneous photocatalysis. The Ascaris eggs were 79% inactivated with He/H2O2 and 36% with Co2+/PMS with an irradiation to 800 - 900 W/m2.
Mijaylova et al. [1], reported for the Gran Canal and the deep drainage flows, average total coliforms values of 1.5 × 108 and 3.4 × 108 MPN100 mL, respectively, while for HE, the average values were of 41 (24 - 56) and 44 (24 - 84) eggs/L. Though the work was not focused on the treatment of the coliforms and HE, the authors reported that the use of FeCl3 plus a given synthetic polymer removed the total of the HE present in the original wastewater samples (100%), not all the TC and FC (residual coliform values between 108 and 109 MPN/100 ml were observed after the coagulation-flocculation process).
Among the chemical processes currently used in treating, wastewater coagulation-flocculation has received considerable attention for obtaining a high level of contaminant removal [22]. Nowadays the most commonly used coagulants-flocculants are salts of Fe+3 and Al+3. However, their mechanisms are complex and depend on a big number of variables (cation concentration and pH) [23]. The effect of the interaction between the coagulant dose and pH on the removal of the SST and SVI is significant, while no such interaction has been seen between them and water recovery [24].
The use of biopolymers in the coagulation-flocculation process can produce more biodegradable sludge. Indeed, it is not necessary to adjust the pH in the wastewater to obtain good wastewater removals. Natural gums for industrial use are the starches, pectins, galactomannans and exudates such as natural rubber and natural gums (Arabic and mesquite are good examples), which are substances of plants widely used in food, pharmaceutical, textile, cosmetics and wine [25]. Carpinteyro-Urbán et al. [26], have reported the use of guar gum, locust bean gum, mesquite and Opuntia mucilage flocculants coagulants agents in the treatment of MWW. The adequate determination of coagulant and dose not only improve the characteristics of resulting water but also reduce the treatment cost.
Our research group has reported the use of natural coagulant-flocculant agents, such as guar and locust bean gums, Prosopis laevigata seed gum, Annona (muricata, diversiforme and cherimola) seed gum and Opuntia ficus-indica mucilage [27-30]. MWW and industrial wastewaters were successfully treated with reasonable COD and turbidity removals, low production of sludges and good sludge settleability properties.
Some authors [7,31] have reported the use of lime for the treatment of sewage sludges, by mixing the effect of pH adjust and high temperatures. As far as we know, there are no works of coagulation-flocculation being employed together with lime for disinfection purposes in wastewaters.
The aim of this work was to demonstrate the feasibility of treating MWW from Mexico City using coagulationflocculation process and a galactomannan-type polymer to reduce the organic load and the pathogens indicators (TC, FC and HE).
2. Materials and Methods
2.1. General Characterization of the MWW
The wastewaters employed in this work were collected from the inflow of the San Juan Ixhuatepec treatment plant, located in the State of Mexico. Wastewater was sampled twice (120 L each time) in the plant prior to the regular treatment applied to the MWW. The sampled wastewaters were stored at 4˚C. The general characterization of MWW was performed by measuring the physicochemical and microbiological parameters established in the Mexican Official Standard [32,33]. The relevant parameters were pH, COD, Turbidity, TC, FC and HE. The NOM-001-ECOL-1996 considers that FC and HE are the pathogenic contaminants to be determined in MWW. For the goal of this study these microbiology indicators were analyzed.
2.2. Determination of Sludge Volume and Z Potential
Additionally, sludge was measured using Imhoff cones by allowing 1 L samples to settle down for 1 hour. HE were measured according to the Mexican Norm [34]. The Z-potential was determined at the original pH of the sample using a commercial Z-meter (Malvern, USA). For the sake of completeness, the method for determining TC, FC and HE are described below.
2.3. Coliforms Quantification
The method is based on direct filtration of a sample or aliquot of the sample through a cellulose membrane which retains the organisms, either by placing the membrane in a selective culture medium m Endo agar LES (for growth of TC) or m FC agar (for FC growth). The membrane is incubated for 24 hours either at 35˚C - 37˚C for the detection of coliform organisms, or alternatively to 44.0˚C ± 1˚C for the presence of thermo-tolerant coliform organisms. A direct count of the colonies developed on the membrane prepared with a selective culture medium was performed [35].
2.4. Helminth Eggs Quantification
This method is based on the concentration and removal of HE by combining the principles of the method and the biphasic flotation method. A volume of 5 L was taken from wastewater. The sample was allowed to stand for 8 hours, the supernatant was aspirated by vacuum and discarded. The sediment was filtered in a sieve with 160 micron. Pore sieve is washed with 5 L of water and recovered wash water along with the sediment filter. The sample was allowed to stand overnight, aspirated supernatant carefully as possible. The sediment was deposited in the centrifuge containers rinsing 3 times the little jug perfectly with distilled water and placing it in containers for centrifugation. The tubes were centrifuged at 400 g by 5 min. The supernatant was decanted by vacuum ensuring that bottom of the tablet container was empty. The pellet was resuspended in 150 ml of solution of zinc sulfate (ZnSO4). All pellets were centrifuged at 400 g 5 min, and the supernatant was recovered by pouring it into a plastic container of 2000 ml. The sample was diluted in 1000 ml of distilled water and allowed to settle overnight. The maximum was aspirated by vacuum supernatant and the pellet resuspended with a little distilled water. The suspension was centrifuged at 480 g for 3 min. The supernatant was decanted and the pellet was resuspended with distilled water by centrifuging at 480 g for 3 min. The supernatant was decanted and the pellet was resuspended in 15 ml of the acid-alcohol solution using a shaker. The tubes were added to 10 ml of ethyl acetate and uncovering gently stirring occasionally to strip the gas. The sample was centrifuged at 660 g for 3 min and the supernatant aspirated as much as possible. Finally, less than 1 ml was left in the tube. Loss of the wafer was prevented. The pellet was homogenized and quantification proceeded directly by microscopy. A visual identification of structures finalized the HE determination [34].
2.5. Natural Gums Selection Process
Coagulation-flocculation process were developed using square jars with 2 L capacity with a speed of 100 rpm for 3 min (coagulation process) and then at 20 rpm for 15 min (flocculation process). Five natural gums were used as coagulant-flocculant: locust bean gum, guar (Drogueria Cosmolpolita, Mexico), Delonix regia (flamboyan) seed, Hydroxypropyl triammonium chloride guar gum HPTAC-guar (Conjunto Lar, S.A. de C.V, Mexico) and Prosopis laevigata (mezquite) seed gums. The FeCl3 (J.T. Baker, Mexico) as a pure chemical agent was used for comparison purposes.
In particular, Hidroxypropyl-triamonium chloride-guar (HPTAC-guar) is a cationic derivative of guar gum and it is sold as Cosmedia GuarTM. This product is frequently employ as a humidifier agent in shampoos and hair conditioner products. The structure of HPTAC-guar is presented at Figure 1. Four different concentrations (25, 50, 75 and 100 mg/L) of each coagulant-flocculant agent were assessed in order to choose the best one as well as its dose. Parameters such as COD and turbidity removal, pH, conductivity, sludge accumulation, TC, FC and HE removal were evaluated in selecting the best natural coagulant-flocculant.
Once the best natural coagulant-flocculant was selected, a second set of experiments with different concentrations of the elected gums (25 30, 40, 50 and 60 mg /L) was performed. This strategy was followed to optimize the gum dose (all these analyses were performed by triplicate). Once again, the parameters measured included COD removal, turbidity reduction, pH, conductivity, sludge formation and removal of biological indicators (TC, FC, and HE).
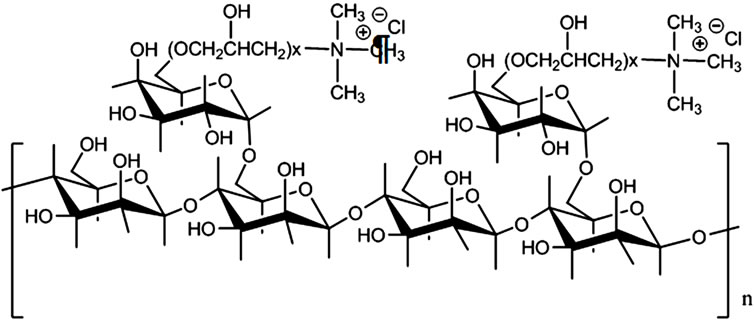
Figure 1. Structure of HPTAC-guar.
2.6. Mixed Biological/Chemical Coagulant for MWW Treatment
The initial pH of the MWW was changed using different concentrations of Ca(OH)2 (industrial grade) prepared with a concentration of 4%. This compound also serves as a coagulating agent. After the pH modification, the flocculation-coagulation process was done using the best dose concentration of the selected natural gum. The parameters analyzed in these tests were same considered in the previous studies. This process was also performed in triplicate.
3. Results and Discussion
3.1. Characterization of the MWW
The characterization of the physicochemical and microbiological MWW is shown in Table 2 (two water batches). Regarding the pH value, MMW is slightly acid. In this analysis the COD value for MWW is twice the COD value reported for others wastewaters from effluents generated in Mexico City [1]. Furthermore, FC, TC and HE are ten times the reported in the same study. In this sense, the wastewater samples are considered as highly contaminated. Regarding the MBAS concentration for MWW, the value is twice the value reported in the previous study.
Z-potential was measured for designing coagulatio nflocculation systems. The MWW has Z-potential positive (i.e., +15.73) at the fixed pH = 6.76. This implies that the net charge of the MWW is positive and should be treated by anionic flocculants. This characteristic is adequate to perform the flocculation-coagulation based on natural gums.
TC for the MWW sample were about 2.8 × 107 MNP/100 ml that is significative higher than the one obtained in previous studies. These values were obtained by measurements averaged from samples obtained in various days and various sampling times. On the counterpart, FC showed a value of 8.48 × 106.
The number of HE found in the first batch MMW (470 HE/L) must be highlighted. The concentration corresponds to the values reported in Mexican rural or periurban areas [4]. However, this value is higher than those

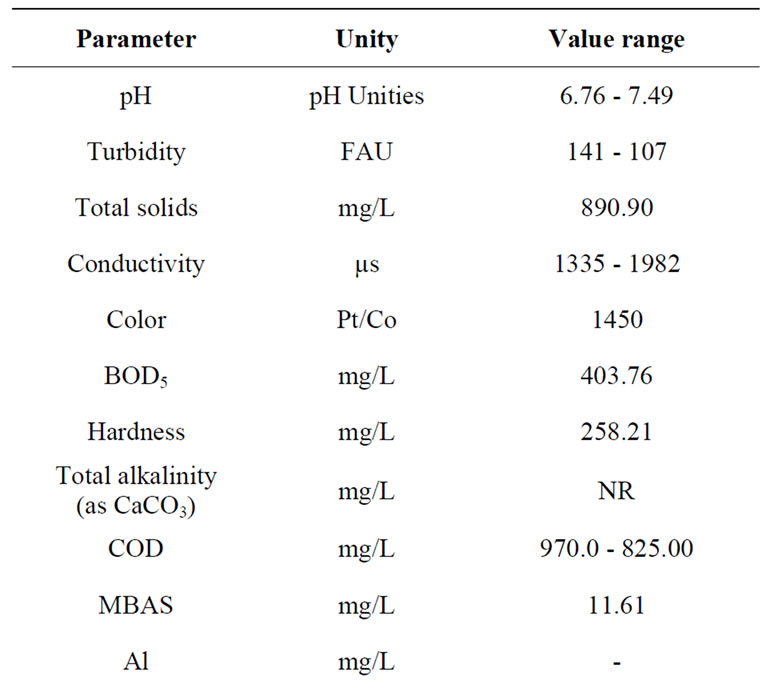
Table 2. Physico-chemical and microbiological characterization of the MWW.
obtained in other works. From this value, it can be assumed that the San Juan Ixhuatepec wastewater treatment plant could receive anonymously effluents from a hospital or slaughterhouse facilities (even when it is supposed to receive municipal wastewaters exclusively).
It is well known that HE as other biological contaminants present a variation along the days of the year and the hour of the day, when the sample was taken [1]. HE species found in the MWW were Diphyllobothrium latum, Ascaris lumbricoides, Hymenolepis nana and Toxocara canis. A brief description of every kind of HE found in MWW is given in the following paragraphs.
Diphyllobothrium latum: The HE have 67 to 71 microns in length and 40 to 51 um wide [36]. Fish in lakes are often heavily infected by this worm [37]. In humans, these organisms can lead to nonspecific abdominal symptoms and a small percentage of cases is developing macrocytic hypochromic anemia (Figure 2(a)) [36]. On the other hand The Ascaris lumbricoides: HE size is from 45 to 75 microns long and 35 - 50 microns wide [38]. The most common symptom in infected patients is vague abdominal pain, the larvae come to cause sensitization of the host, causing reactions such as pulmonary infiltrates,
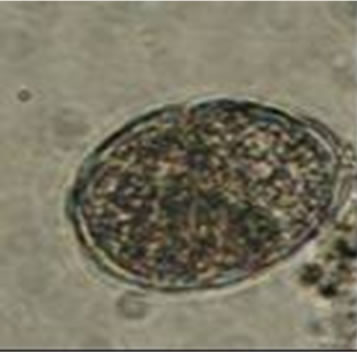 (a)
(a)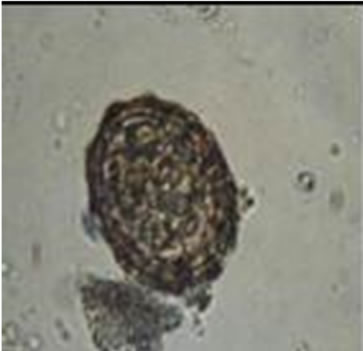 (b)
(b) (c)
(c)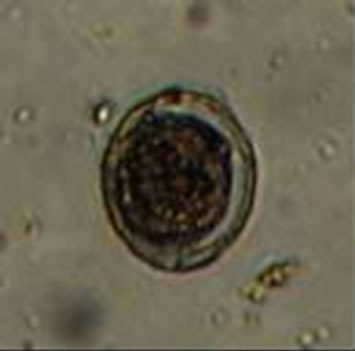 (d)
(d)
Figure 2. Microphotographies of helminth eggs found in the MWW sample.
asthma attacks and edema of the lips (Figure 2(b)) [37]. Hymenolepis nana: HE size of 30 to 44 microns [38]. This parasite has a cycle of internal autoinfection. Mild cases are usually asymptomatic, the larval forms destroy injures villi, adults irritate the intestinal wall, causing a mild enteritis: cases with moderate or massive parasitization abdominal pain, nausea, vomiting, weight loss, diarrhea, nervousness, headache, etc. (Figure 2(c)) [39]. Finally, Toxocara canis: HE measure 90 microns long and 75 microns wide [37]. Toxocariasis is a clinical syndrome that is caused by the invasion of human viscera by larvae. These parasites are roundworms of dogs and cats. In humans, the larvae released from ingested embryonated eggs, penetrate the intestinal mucous membrane and travels to the liver, lungs and other organs. The primary lesion is found in the liver and may have extensive necrosis of liver parenchyma. Lesions that contain the larvae of Toxocara found in liver, brain, eye, spinal cord, lungs, heart muscle, kidneys and lymph nodes. The disease occurs in children under 4 years of age (Figure 2(d)) [37].
Mijaylova et al. [1], reported the predominance of Ascaris eggs in the GC and DD in the range of 20 - 52 eggs/L, however Enterobius spp, Hymenolepis nana, Taenia spp, Necator spp, and Trichuris were also detected at concentrations of 0 - 8 eggs/L.
3.2. Selection of Natural Gum in the Process of Coagulation-Flocculation
The parameters analyzed for the selection of both natural gums and the doses were COD and turbidity. The natural gum showing higher COD removal (45.6%) and turbidity (19.3%) values was HPTAC-guar at the concentration of 50 mg/L. These removal values were higher than those obtained when using FeCl3 and all others natural gums.
Regarding the generation of sludges, it is quite noticeable that natural gums produced less sludge than FeCl3 (in the range of 0 - 0.5 mL/L while FeCl3 produced 1-3 mL/L). Microbiological removals obtained by using Prosopis laevigata (mesquite) seed gum and HPTAC-guar (both at a concentration of 100 mg/L) reached the full elimination of both TC and FC (Table 3), while FeCl3 at the 75 mg/L removed only the 100% of FC, but only 83.7% of TC.

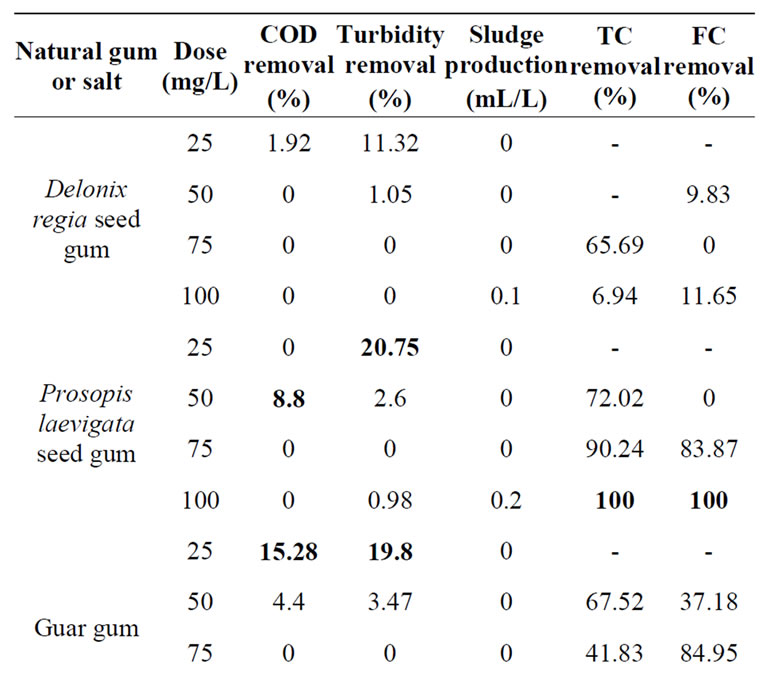
Table 3. Removal of chemical and microbiological parameters in the MWW after coagulation-flocculation process using different natural gums. Initial HE concentration was 470 HE/L.
Different natural gums concentrations (25, 50, 75 and 100 mg/L) were selected to determinate the optimal operating conditions in the coagulation-flocculation process. Nevertheless, this procedure also increases the COD by the aggregation of natural gums. The selected concentrations were fixed based on preliminary studies [26].
Galactomannans are the main forming element of all the natural gums. Among the tested gums, Prosopis laevigata seed gum and HPTAC-guar have the lowest ratio between manose and galactose. Hence, this ratio is used to select the most efficient gum using the COD reduction and sludge accumulation as key indicators. However, we have selected the HPTAC-guar because it can be obtained from commercial suppliers while the Prosopis laevigata should be extracted by a time consuming procedure.
The HPTAC-guar concentration was fixed to 25 mg/L. This selection was done by considering the after mentioned indicators. Even when the concentration of HPTACguar fixed to 50 mg/L is more efficient, the inclusion of the natural gum in the reactor also increases the COD. Therefore, when lower gum concentration is used, both COD reduction and sludge production (5.77% and 0 mL/L) are considered as acceptable for the second set of studies.
In this experiment, it was observed that beyond the optimum dose of coagulant-flocculant, the effect on the removal of physicochemical parameters is reversed. Indeed, instead of removing the evaluated parameters, all of them were increased. [26] reported that it is believed that biopolymers may provide sufficient organic load and instead of benefiting the process, they can increase the COD of the treated water.
The results showed that higher COD and turbidity removals were observed when HPTAC-guar was fixed to 50 mg/L. Complementary, the FC and TC removal at this concentration are greater than 40%. These results seem to be coincident with those presented by other studies. For example, Beltran et al. [40] have reported removal of coliforms between 96% and 94% using a natural gum extracted from Moringa oleifera. However, it is worth noticing that coliforms in their work reported was 400 to 800 CFU/100 mL, while those found in the MWW treated in this work were of 2.7 × 107 and 8.48 × 106 CFU/mL.
A summary of all results obtained for all the different gums is shown in Table 3. The results achieved in this set of experiments were used to select the most adequate gum to treat the MWW. In general, all the gums derived from galactomannans were more efficient to reduce all the considered indicators (physicochemical and microbiological). In particular, higher COD removals were preferred over all other indicators. This is the main reason to select a coagulant-flocculant concentration between 25 and 50 mg/L as the optimal one. Moreover, a higher gum concentration increases the COD content that is considered as a negative result.
3.3. Effect on the Physicochemical and Microbiological Removal by the HPTAC-Guar Concentration
Based on the results obtained in the last experiment, a second set of experiments was designed using 5 different doses of HPTAC-guar (that was selected as the most adequate natural coagulant-flocculant). These doses were 25, 30, 40, 50 and 60 mg/L.
All of them are within the range of those found as the more efficient ones in the previous experiment. This second concentration adjustment is proposed for obtaining a finest election of the HPTAC-guar concentration.
The best COD, turbidity, TC and FC removals were obtained by using natural rubber at a concentration of 50 and 60 mg/L. However, one of the best HE removal was obtained when 30 mg/L of natural gum was selected. This gum concentration also provided similar changes in the physicochemical parameters to those obtained when gum concentrations were 50 and 60 mg/L. The results obtained showed that removals obtained with natural rubber are higher than those obtained with FeCl3, Aguilar et al. [41] reported COD removal efficiencies of 86% using Iron salts at a dose of 100 mg/L. All these experiments were carried out by triplicate.
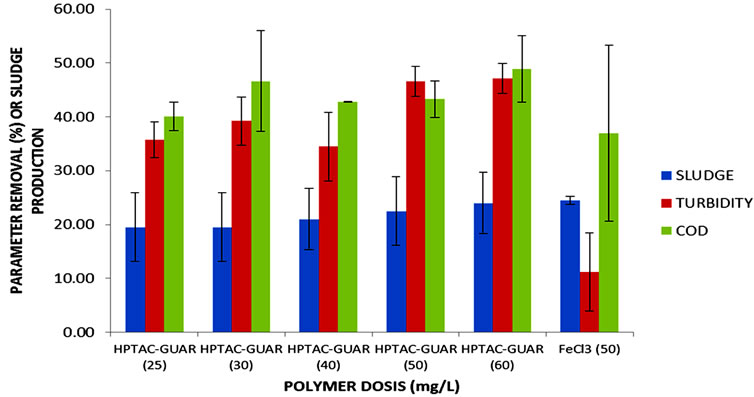
A simple variance analysis showed that no significant differences are observed when gum concentration is varied from 25 to 60 mg/L (Figure 3).
Particularly, COD removals are similar for all the cases considered in this experiment (Figure 3(a)). However, all microbiological indicators were efficiently removed when HPTAC-guar was fixed between 30 mg/L and 50 mg/L (Figure 3(b)). Indeed, HE was completely removed while TC and FC were eliminated above 80% in all these cases. Therefore, we have selected 30 mg/L as final HPTAC-guar concentration by two important criteria. The first one considers that when higher gum concentrations are in the reactor, more artificial COD is added to the experiment. The cost increase associated with the higher gum concentration was the second criterion considered here.
3.4. Effect on the Physicochemical and Microbiological Removals by Modifying the Initial pH of the MWW with Ca(OH)2
The adjustment of pH can be used to improve the coagulation-flocculation efficiency. This can be done using several chemical compounds. In particular, Ca(OH)2 has been reported as an efficient coagulant [42] and can modify the pH up to 12.5.
 (a)
(a) (b)
(b)
Figure 3. Removal of physicochemical (a) and microbeological (b) parameters in MWW after coagulation-flocculation process using HPTAC-guar at different concentrations.
The pH modification by Ca(OH)2 also alters the ideal condition for growing of TC, FC and HE (Table 4). Therefore, to add a controlled dose of Ca(OH)2 can improve the coagulation-flocculation efficiency and can serve to reduce the microbiological indicators. The following table shows the relationship between the volume of Ca(OH)2 added to the reactor and the modification of either microbiological and physicochemical parameters. In this experimental phase, the second batch of MWW was used.
When the initial pH of the MWW is used (no artificial pH adjustment), the COD removal is 1%. However when pH is adjusted to 12.5, this indicator is increased up to 80%. Results obtained in this part showed a direct relationship between the Ca(OH)2 volume and the COD removal.
Turbidity variation showed a similar behavior to the one gotten for COD (from 10% to 78% when pH is adjusted from 7.8 to 12.5). All the microbiological indicators were improved. Indeed, all of them (TC and FC) were completely eliminated in all the cases when the Ca(OH)2 was in the reaction. However, an important negative effect is the increase of sludge volume when Ca(OH)2 is in the reactor. The pH adjustment from 7.8 to 12.5 increased the sludge volume in almost 3100%.

Table 4. Removal of physicochemical and microbiological parameters in MWW, changing the pH value with Ca(OH)2.
3.5. Effect on the Physicochemical and Microbiological Removals by Coagulation Flocculation Using HPTAC-Guar and Modifying the MWW pH with Ca(OH)2
The complementary study that mixes the HPTAC-guar and Ca(OH)2 was used to understand if both the natural and the synthetic coagulant can improve the removal of either the physicochemical and microbiological indicators. Here, it is considered that Ca(OH)2 modifies the pH while HPTAC-guar combined with the hydroxile serve as coagulant/flocculant. This test used the Ca(OH)2 as a coagulant and HPTAC-guar as flocculant fixed the gum concentration at 25 mg/L.
The COD removal in the neutral pH (without pH modification) was 11.7%. However when pH is adjusted to 9.4, this removal is increased up to 50%. Results obtained in this part the study showed a direct an increasing relationship between the Ca(OH)2 volume and the COD removal. Therefore, the gum has no evident contribution to the removal efficiency of both physicochemical and microbiological indicators (Figure 4).
Turbidity variation showed a similar behavior to the one gotten for the COD (from 17% to 43% when pH is varied from 7.7 to 9.4). The addition of Ca(OH)2 increased all the monitored parameters compared to the cases when only the HPTAC-guar (COD 36% to 47%, Turbidity 37% to 42%) is used as coagulant/flocculant. However, the same important negative effect of Ca(OH)2 regarding the increase of sludge is observed (15 to 50 mL/L when pH is adjusted from its lowest value to the highest one) (Figure 4(a)). All the microbiological indicators were improved when both coagulants were in the reaction. Indeed, the pH showed a direct relationship in the removal of the TC (24% to 71.5%) and HE (100%) (Figure 4(b)).
It was observed that the higher removal in terms of the measured parameters is obtained by working at a pH of 9.4 (Figure 4(b)). Aguilar et al. [41], have reported that when working with iron and aluminum salts of modify-
 (a)
(a)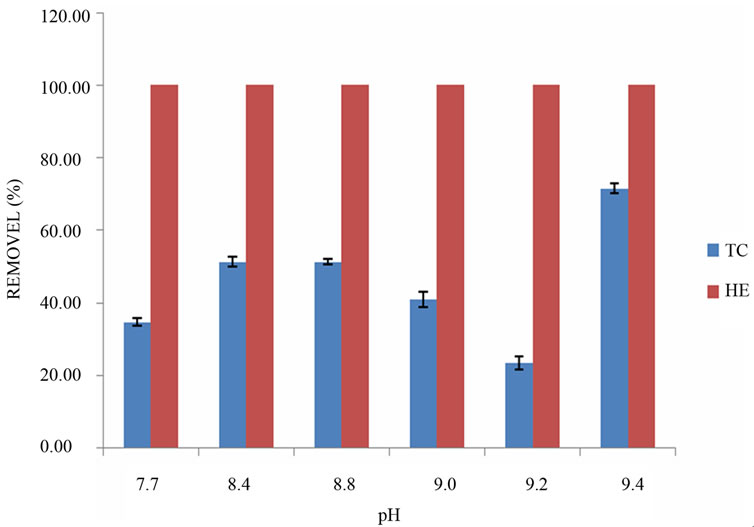 (b)
(b)
Figure 4. Removal of physicochemical (a) and microbiological (b) parameters in the MWW using Ca(OH)2 as coagulant and flocculant HPTAC-guar.
ing the pH in the process, reaches a maximum pH at which the improved process, in the case of this study was determined at pH 7, higher pH the COD removal was diminished.
4. Conclusion
The use of biopolymer HPTAC-guar as a coagulant-flocculant agent showed an efficient removal of parameters such as COD and turbidity. A remarkable decrease of biological indicators was observed, even greater than that obtained when using the FeCl3. Another parameter that should be highlighted is that the sludge using the biopolymer is less than those obtained when using the FeCl3. It was observed that the removal of microorganisms can be very efficient, but does not reach the allowable limits in the Mexican Official Standards, so it is advisable to fit the process to another, such as ozonation to help reaching permitted limits. The change of pH by Ca(OH)2 in the coagulation-flocculation process was shown to have a positive influence on the removal of chemical and biological parameters.
5. Acknowledgements
This work was partially supported by ICyT-DF Grant PICSO 10 - 8. We thank the San Juan Ixhuatepec Wastewater Treatment Plant the opportunity to use its raw wastewaters.
REFERENCES
- P. Mijaylova, L. Torres, E. Ramírez, S. López and L. Cardoso, “Characterization and Coagulation—Flocculation Treatability of Mexico City Wastewater Applying Ferric Chloride and Polymers,” Water Science Technology, Vol. 34, No. 3-4, 1996, pp. 235-247.
- M. T. Orta de Velázquez, N. Rojas, J. Martínez and I. Monje, “Destruction of Helminth Eggs (Ascaris suum) by Ozone,” Science & Engineering, Vol. 26, No. 4, 2004, pp. 359-366.
- S. Toze, “PCR and the Detection of Microbial Pathogens in Water and Wastewater,” Water Research, Vol. 33, No. 17, 1999, pp. 3545-3556. http://dx.doi.org/10.1016/S0043-1354(99)00071-8
- B. Jimenez, “Helminths (Worms) Eggs Control in Wastewater and Sludge,” International Symposium on New Directions in Urban Water Management, 2007.
- B. Jimenez and C. Maya-Rendon, “Helminths and Sanitation. Communicating Current Research and Educational Topics and Trends,” 2007.
- J. A. Barrios, B. Jimenez, O. Gonzalez, G. Salgado, L. Samaria and R. Iturbe, “Faecal Coliforms Destruction and Helmint Eggs in Physical-Chemical Sludges by Acid Means (In Spanish),” Proceedings of the XII FEMISCA National Congress, Morelia, 2000, pp. 683-692.
- S. Capizzi-Banas, M. Deloge, M. Remy and J. Schwartzbrod, “Liming as an Advanced Treatment for Sludge Sanitization: Helminth Eggs Eliminate Ion-Ascaris Eggs as Model,” Water Research, Vol. 38, 2004, pp. 3251-3258. http://dx.doi.org/10.1016/j.watres.2004.04.015
- B. Jimenez, “Helminth Ova Control in Wastewater and Sludge for Agricultural Reuse,” In: W. O. K. Grabow, Ed., Water Reuse New Paradigm towards Integrated Water Resources Management in Encyclopedia of Biological, Physiological and Health Sciences, Water and Health, II, Life Support System, EOLSS Publishers Co Ltd., UNESCO, 2008, pp. 429-449.
- S. Quinzaños, C. Dahl, R. Strube and R. Mujeriego, “Helmint Eggs Removal by Microscrening for Water Reclamation and Reuse,” Water Science and Technology, Vol. 57, No. 5, 2008, pp. 715-720. http://dx.doi.org/10.2166/wst.2008.171
- M. E. Sengupta, S. M. Thamsborg, T. J. Andersen, A. Olsen and A. Dastard, “Sedimentation of Helminth Eggs in Water,” Water Research, Vol. 45, No. 15, 2011, pp. 4651-4660.
- J. Nouri, A. H. Mahvi, R. Saeedi, K. Dindarloo and M. Rafiee and S. Dobaradaran, “Purification of and Removal Acaris and Fasciola Hepatica Eggs from Drinking Water Using Roughing Filters,” Korean Journal of Chemical Engineering, Vol. 25, No. 3, 2008, pp. 501-504. http://dx.doi.org/10.1007/s11814-008-0084-4
- N. Cabirol, M. Rojas and A. Noyola, “Removal of Helminth Eggs and Faecal Coliforms by Anaerobic Termophilic Sludge Digestion,” Water Science and Technology, Vol. 45, No. 10, 2002, pp. 269-274.
- M. von Sperling, V. H. Freire and C. A. de Lemos Chernacaro, “Performance Evaluation of a UASB-Activated Sludge System Treating Municipal Wastewater,” Water Science and Technology, Vol. 43, No. 11, 2001, pp. 323- 328.
- E. J. La Mota, E. Silva, A. Bustillos, H. Padron and J. Luque, “Combined Anaerobic/Aerobic Secondary Municipal Wastewater Treatment: Pilot Plant Desmostration of the UASB/Aerobic Solids Contact System,” Journal of Environmental Engineering-ASCE, Vol. 133, No. 4, 2007, pp. 397-403.
- J. Guo, J. Wang, D. Cui, L. Wang, F. Ma, C. Chang and J. Yang, “Application of Bioaugmentation in the Rapid Start-Up and Stable Operation of Biological Processes for Municipal Wastewater Treatment at Low Temperatures,” Bioresource Technology, Vol. 101, No. 17, 2010, pp. 6622-6629.
- R. Reinoso, L. A. Torres and E. Bécares, “Efficiency of Natural Systems for Removal of Bacteria and Pathogenic Parasites from Wastewater,” Science of the Total Environment, Vol. 395, No. 2-3, 2008, pp. 80-86.
- Y. Liu, T. Yang, D. Yuan and X. Wu, “Study of Municipal Wastewater Treatment with Oystert Shell as Biological Aerated Filter Médium,” Desalination, Vol. 254, No. 1-3, 2010, pp. 149-153.
- N. Rojas-Valencia, “Research on Ozone Application as Disinfectant and Actions Mechanisms on Wastewater Microorganisms. Science against Microbial Pathogens: Communicating Current Research and Technological Advances,” Microbiology Book Series-Number 3, Vol. 1, FORMATEX (A. Mendez-Vilas Edit), España, 2011, pp. 263- 271.
- C.-H. Lin, R.-F. Yu, W.-P. Cheng and C.-R. Liu, “Monitoring and Control of UV and UV-TiO2 Disinfections for Municipal Wastewater Reclamation Using Artificial Neural Networks,” Journal of Hazardous Materials, Vol. 209- 210, No. 1, 2012, pp. 348-354.
- Z. Bhatti, Q. Mahmood, I. Raja, A. Malik, N. Rachis and D. Wu, “Integrated Chemical Treatment of Municipal Wastewater Using Waste Hydrogen Peroxide and Ultraviolet Light,” Physics and Chemistry of the Earth, Vol. 36, No. 9-11, 2011, pp. 459-464.
- J. García, E. Bandala and B. Corona, “Inactivation of Helminth Eggs by Homogeneus Photocattalysis,” Aquaforum, Vol. 49, No. 1, 2008, pp. 14-18.
- L. Semerjian and G. Ayoub, “High-pH-Magnesium Coagulation-Flocculation in Wastewater Treatment,” Advances in Environmental Research, Vol. 7, No. 2, 2003, pp. 389-403.
- A. Bazer-Bachi, E. Puech-Coste, R. Ben Aim and J. L. Probst, “Modélisationmathé Matique du taux de Coagulant dans une Station de Traitementd_eau,” Revue des Sciences de l_Eau, Vol. 3, No. 4, 1990, pp. 377-397.
- A. Ahmad, S. Wong, T. Teng and A. Zuhairi, “Optimization of Coagulation-Flocculation Process for Pulp and Paper Mill Effluent by Response Surface Methodological Analysis,” Journal of Hazardous Materials, Vol. 145, No. 1-2, 2007, pp. 162-168.
- Y. López-Franco, F. Goycoolea, M. Valdez and A. Calderón de la Barca, “Goma de Mezquite: Una Alternativa al uso Industrial (Mesquite Gum: Analternativeto Industrial Use,” Interciencia, Vol. 31, No. 3, 2006, pp. 183- 188.
- S. Carpinteyro-Urbán, J. Yañez and L. Torres, “Coagulation Flocculation of Wastewaters Employing Guar, Locust Bean and Mesquite Gums, as Well as Opuntia Mucilage,” Proceedings of the 2nd IWA Mexico Young Water Professional Conference, Querétaro, 2010.
- L. G. Torres, S. Carpinteyro-Urbán and M. Vaca, “Use of Prosopis Laevigata Seed Gum and Opuntia Ficus-Indica Mucilage for Treatment of Municipal Wastewaters by Coagulation-Flocculation,” Natural Resources, in press, 2011.
- L. G. Torres, S. Carpinteyro-Urbán and L. J. Corzo-Rios, “Use of Annona Diversifolia and A. Muricata Seeds as Source of Natural Coagulant-Flocculant Aids for the Treatment of Wastewaters,” Submitted to Indian Journal of Biotechnology, 2012.
- S. Carpinteyro-Urbán, M. Vaca and L. Torres, “Can Vegetal Biopolymers Work as Coagulant-Flocculant Aids in the Treatment of High-Load Cosmetic Industrial Wastewaters? Submitted to Water, Air and Soil Pollution,” 2012.
- S. Carpinteyro-Urbán, M. Vaca and L. Torres, “Use of Response Surface Methodology in the Optimization of Coagulation-Flocculation of Wastewaters Employing Biopolymers. Submitted to International Journal of Environmental Research,” 2012.
- B. Bina, H. Movahedian and I. Kord, “The Effect of Lime Stabilization on the Microbiological Quality of Sewage Sludge,” Iranian Journal of Environmental Health and Science Engineering, Vol. 1, No. 1, 2004, pp. 34-38.
- SEMARNAT 1997 NOM-003-ECOL-1997, “Establishing Limits for Maximum Permissible Pollutants Treated Wastewater Who Reuse in Public Services. (In Spanish), México,” 1997.
- SEMARNAT 1996 NOM-001-ECOL-1996, “Establishing the Maximum Allowable Discharge of Pollutants in Sewage Water and Domestic Goods. (In Spanish), Mé- xico,” 1996.
- SECOFI 1999 NMX-AA-113-SCFI-1999, “Analysis of Water-Determination of Helminth Eggs-Test Method (in Spanish), México,” 1999.
- SECOFI 2006 NMX-AA-102-SCFI-2006, “Water Quality - Detection and Enumeration of Coliform Organisms, Thermotolerant Coliform Organisms and Presumptive Escherichia coli-Membrane Filtration Method. (in Spanish), México,” 2006.
- J. L. Soulsby, “Parasitology and Parasitarial Diseases in Domestic Animals,” 7th Edition (In Spanish), (Interamericana, ed). Mexico, 1987.
- H. W. Brown and F. A. Neva, “Clinical Parasitology,” 5th Edition (In Spanish), Editorial Interamericana, Mexico, 1985.
- WHO, “Integrated Guide to Sanitary Parasitology. (WHOEM/CEH/121/E Ed),” Amman-Jordan, 2005.
- R. Romero, “Human Microbiology and Parasitology. Ethiological Bases for the Infection Diseases. (In Spanish),” (Medica-Panamericana, ed). Mexico, 1993.
- J. Beltran and J. Sánches, “Improvement of Flocculation Process in Water Treatment by Using Moringa Oleífera Seeds Extract,” Proceedings or the Water & Industry 2011—IWA International Conference, Valladolid, 2011.
- M. I. Aguilar, J. Sáez, M. Lloréns, A. Soler, J. F. Ortuño, V. Messeguer and A. Fuentes, “Improvement of Coagulation—Flocculation Process Using Anionic Polyacrylamide as Coagulant Aid,” Chemosphere, Vol. 58, No. 1, 2005, pp. 47-65
- D. Georgiou, A. Aivazidis, J. Hatiras and K. Gimouhopoulos, “Treatment of Cotton Textile Wastewater Using Lime and Ferrous Sulfate,” Water Research, Vol. 37, No. 9, 2002, pp. 2248-2250.
NOTES
*Corresponding author.

